A Review on the Efficient Catalysts for Algae Transesterification to Biodiesel
Abstract
1. Introduction
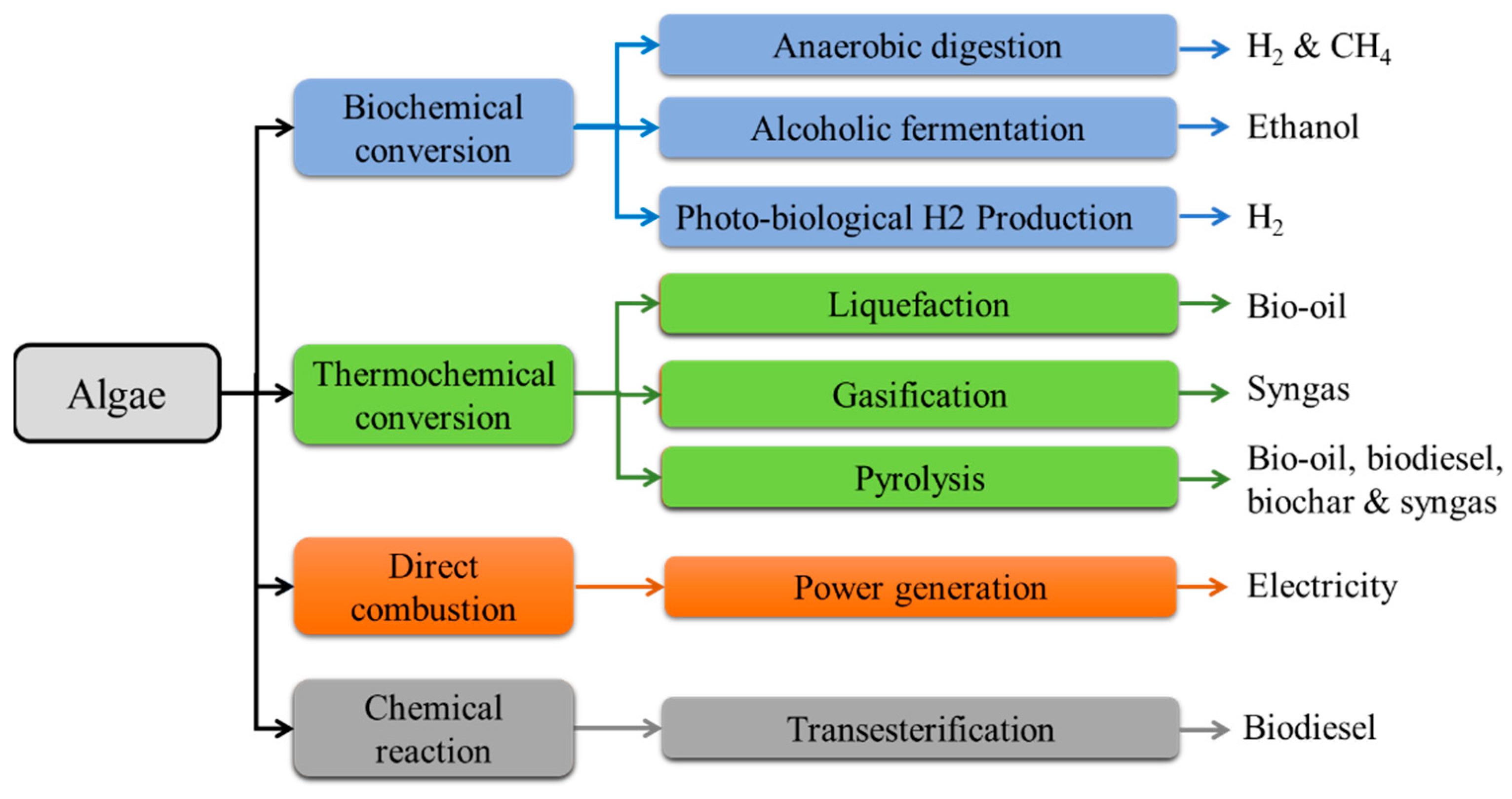
2. Energy Conversion Processes from Algae
- Biochemical conversion: a bioenergy resource is a bioethanol formed by the fermentation method. This method is a biochemical route in which sugars in algae are converted into bioethanol normally by yeast action. Hence, the presence and accessibility of sugars in algae are of crucial importance in bioethanol production. A mixture of methane and carbon dioxide as biogas besides ammonia is normally produced through the anaerobic digestion of algae. Methane is significant biogas in the aspect of being a bioenergy source. However, this method is considered an uneconomic and unsatisfactory process [21]. Hydrogen is one of the most important energy vectors which is used in various applications such as electricity and fuel cells. Moreover, hydrogen can be formed by photobiological and anaerobic digestion methods.
- Thermochemical conversion: thermochemical conversion is a process for the production of three different fuels, including bio-oil (gasoline, jet fuel, biodiesel), biogas, and biochar, and it normally happens at high temperatures with the absence of oxygen [22]. Upon pyrolysis process, two significant sources of energy, including hydrogen and bio-oil, are produced [23]. Furthermore, hydrogen can be produced by the gasification method. In the liquefaction process, wet biomass can be decomposed into smaller components and then repolymerized into large oily components or liquid biofuels [24].
- Direct combustion: In biopower plants, biomass is directly burned in excess air to heat the water in a boiler and produce high-pressure steam. The steam turns a turbine generator, and hence the electricity is generated.
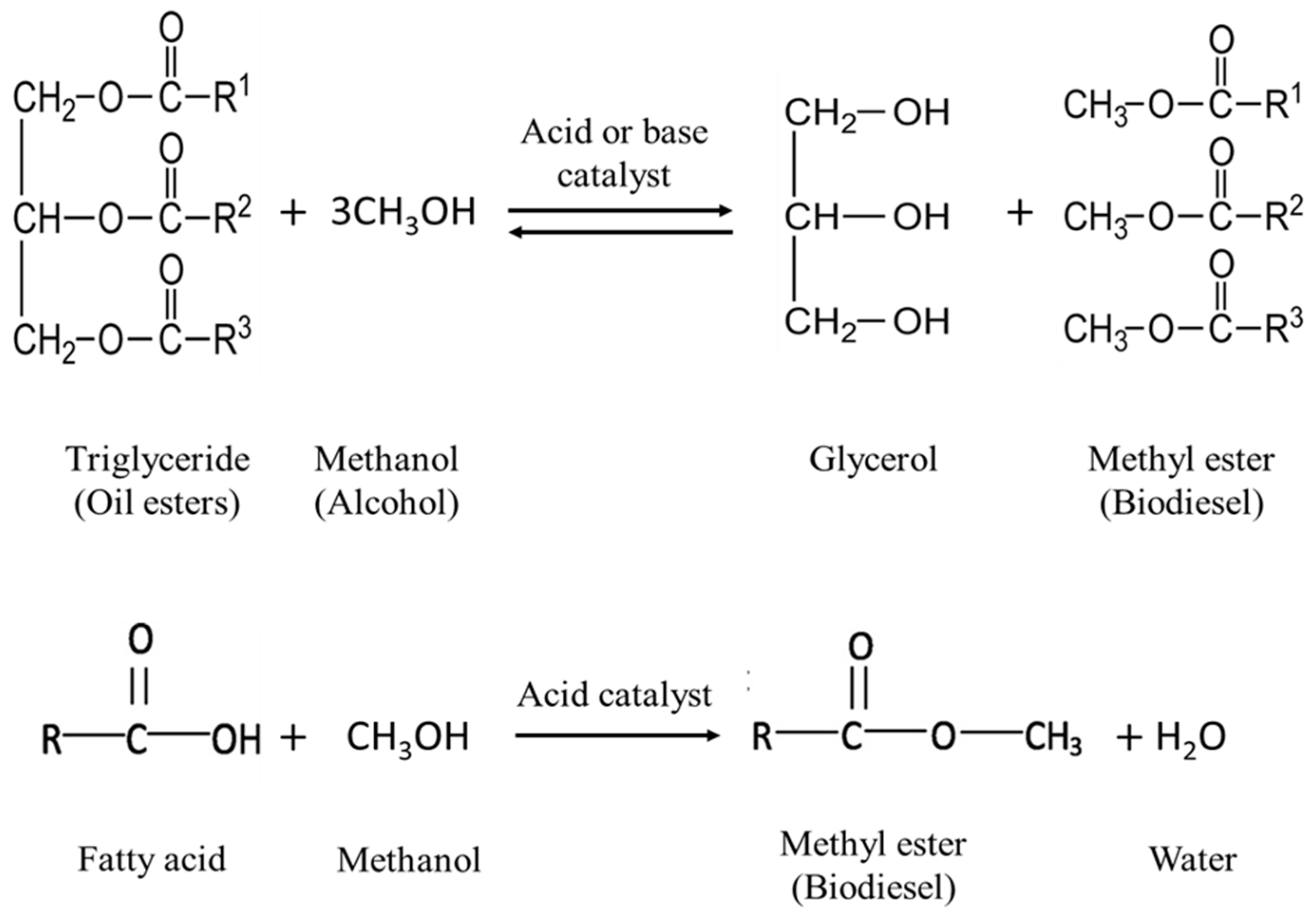
- Chemical reaction: among several bioenergy sources, biodiesel is considered as the transportation fuel especially suitable in diesel engines without modification. The presence of oils and lipids in the algae structure could be a renewed interest for making biodiesel with less polluting and more renewable nature. The transesterification process is the desired route in which algal extracted oil could be converted to biodiesel in the presence of alcohol and an acid or a base catalyst [14].
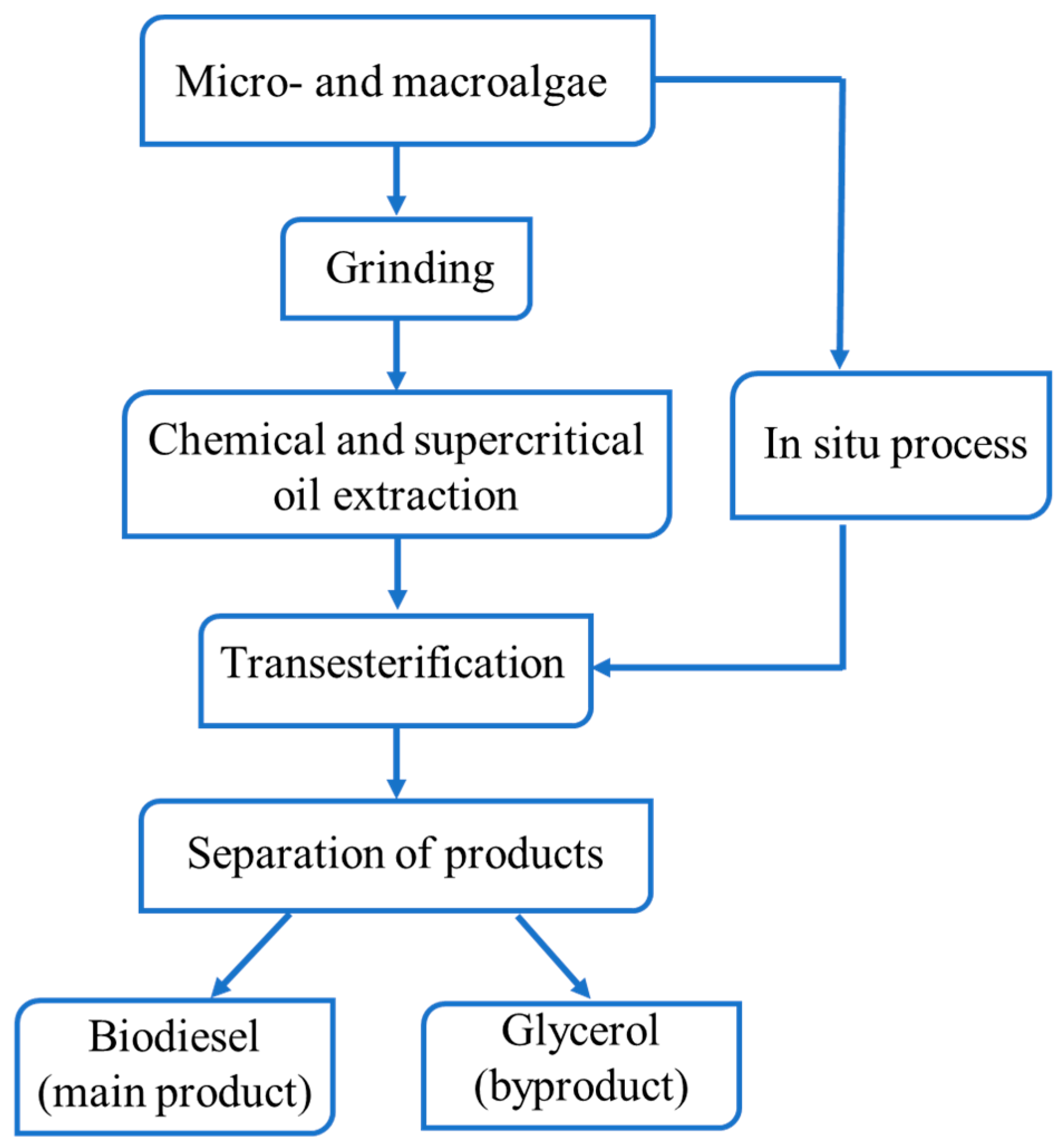
3. Transesterification Process
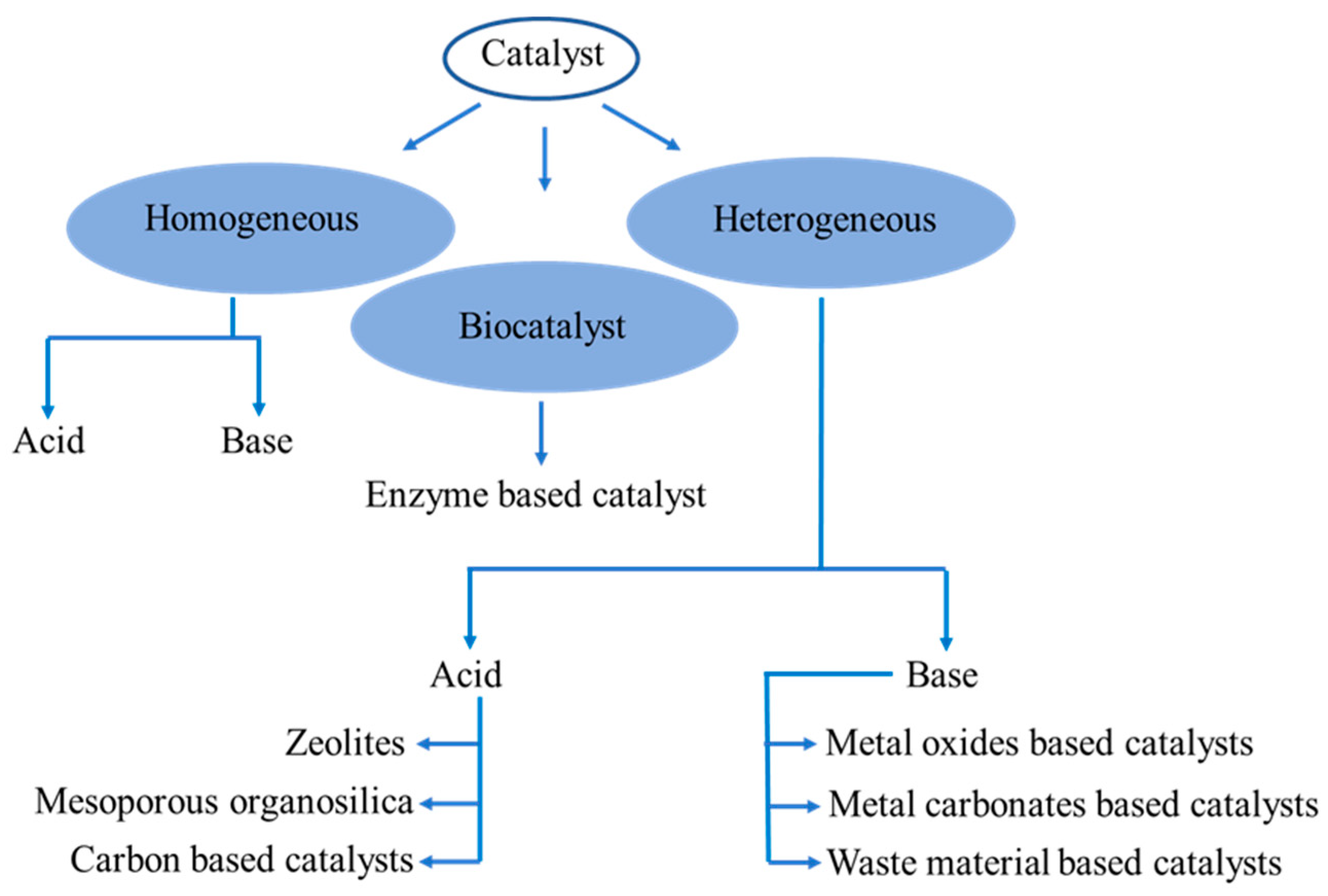
4. Efficient Transesterification Catalyst
4.1. Homogeneous Transesterification Catalysts
4.2. Heterogeneous Transesterification Catalysts
4.3. Biotransesterification Catalysts
4.4. Advantages and Disadvantages of Algae Transesterification Methods
- 1.
- Homogeneous transesterification catalysts:
- -
- Advantages: Since the homogeneous catalysts lead to the feasible mass transfer between lipids and catalysts, mild reaction conditions, including low temperature, low pressure, and short reaction time, are suitable for the transesterification process. This advantage is crucial in the economic aspect. In addition, the high reactivity of homogeneous catalyst, with the production of desirable yield and selectivity of biodiesel, is another prominent characteristic of this catalytic system [120].
- -
- Disadvantages: One of the most important disadvantages of homogeneous catalysts is saponification, especially for basic systems. The difficulty of catalyst separation from the reaction mixture, its recovery and reusability, and also product purification are the challenges of this catalytic system. Moreover, acidic homogeneous catalysts are corrosive for the reaction system [121].
- 2.
- Heterogeneous transesterification catalysts:
- -
- Advantages: Easy catalyst separation from reaction products, reusability of the catalyst, and lower waste generation are the prominent advantages of heterogeneous catalytic systems. In addition, there are some solid catalysts that can handle both transesterifications of lipids and esterification of FFAs in the algal feedstock to biodiesels and increase the efficiency of the process.
- -
- Disadvantages: Weak, porous structure of heterogeneous catalyst causes the mass transfer limitation between algal lipids and catalyst active sites. Therefore, harsher reaction conditions are needed, which reduce the overall catalytic efficiency and increase the cost of the process [122].
- 3.
- Biotransesterification catalysts:
- -
- Advantages: enzymatic catalysts are highly efficient systems with the production of pure product, without side reaction and soap formation. In addition, this catalytic system needs less energy compared to other catalytic systems due to its high activity and recyclability properties.
- -
- Disadvantages: enzymatic catalysts are very expensive for being used at an industrial scale [123].
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Tang, X. Depletion of fossil fuels and anthropogenic climate change—A review. Energy Policy 2013, 52, 797–809. [Google Scholar] [CrossRef]
- Le Page, M. Our advanced civilisation is built on easily exploited coal, oil and gas, World without fossil fuels. New Sci. 2014, 224, 34–39. [Google Scholar] [CrossRef]
- Kumar, M.; Enamala, S.; Chavali, M.; Donepudi, J. Production of biofuels from microalgae—A review on cultivation, harvesting, lipid extraction, and numerous applications of microalgae. Renew. Sustain. Energy Rev. 2018, 94, 49–68. [Google Scholar] [CrossRef]
- Fallot, A.; Girard, P.; De Coopération, C.; Agronomique, R.; Cirad, D.; Dameron, V.; Griffon, M. The assessment of biofuel potentials on global and regional scales in the tropical world. Energy Sustain. Dev. 2006, 10, 80–91. [Google Scholar] [CrossRef]
- Fischer, G.; Prieler, S.; Van Velthuizen, H.; Berndes, G.; Londo, M.; De Wit, M. Biofuel production potentials in Europe : Sustainable use of cultivated land and pastures, Part II : Land use scenarios. Biomass Bioenergy 2010, 34, 173–187. [Google Scholar] [CrossRef]
- Schut, M.; Slingerland, M.; Locke, A. Biofuel developments in Mozambique. Update and analysis of policy, potential and reality. Energy Policy 2010, 38, 5151–5165. [Google Scholar] [CrossRef]
- Signoretto, M.; Taghavi, S.; Ghedini, E.; Menegazzo, F. Catalytic Production of Levulinic Acid (LA) from Actual Biomass. Molecules 2019, 24, 2760. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.A.; Davey, M.P.; Dennis, J.S.; Horst, I.; Howe, C.J.; Lea-smith, D.J.; Smith, A.G. Biodiesel from algae: Challenges and prospects. Curr. Opin. Biotechnol. 2010, 21, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Vassilev, S.V.; Vassileva, C.G. Composition, properties and challenges of algae biomass for biofuel application: An overview. Fuel 2016, 181, 1–33. [Google Scholar] [CrossRef]
- Benedetti, M.; Vecchi, V.; Barera, S.; Dall’Osto, L. Biomass from microalgae: The potential of domestication towards sustainable biofactories. Microb. Cell Fact. 2018, 17, 1–18. [Google Scholar] [CrossRef]
- Laurens, L.M. State of Technology Review—Algae Bioenergy. IEA Bioenergy Task 39. 2017. Available online: https://www.ieabioenergy.com/wp-content/uploads/2016/01/Laurens-Algae-Bioenergy-Report-IEA-webinar-170124-final-rev1.pdf (accessed on 10 September 2021).
- Khan, M.I.; Shin, J.H.; Kim, J.D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Fact. 2018, 17, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Wang, J.; Chen, P.; Ji, C.; Kang, Q.; Lu, B.; Li, K.; Liu, J.; Ruan, R. Bio-mitigation of carbon dioxide using microalgal systems: Advances and perspectives. Renew. Sustain. Energy Rev. 2017, 76, 1163–1175. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- McGinn, P.J.; Dickinson, K.E.; Bhatti, S.; Frigon, J.C.; Guiot, S.R.; O’Leary, S.J.B. Integration of microalgae cultivation with industrial waste remediation for biofuel and bioenergy production: Opportunities and limitations. Photosynth. Res. 2011, 109, 231–247. [Google Scholar] [CrossRef] [PubMed]
- Sayre, R. Microalgae: The potential for carbon capture. Bioscience 2010, 60, 722–727. [Google Scholar] [CrossRef]
- Ghasemi, Y.; Amini, S.R.; Naseri, A.T.; Najafabady, N.M.; Mobasher, M.A.; Dabbagh, F. Microalgae Biofuel Potentials (Review)1. Russ. Prikl. Biokhimiya i Mikrobiol. 2012, 48, 150–168. [Google Scholar] [CrossRef]
- Kojima, E.; Zhang, K.A.I. Growth and Hydrocarbon Production of Microalga Botryococcus braunii in Bubble Column Photobioreactors. J. Biosci. Bioeng. 1999, 87, 811–815. [Google Scholar] [CrossRef]
- Demirbas, A. Combustion efficiency impacts of biofuels. Energy Sources Part A Recover. Util. Environ. Eff. 2009, 31, 602–609. [Google Scholar] [CrossRef]
- Moroney, J.V.; Ynalvez, R.A. Algal Photosynthesis. Encycl. Life Sci. 2018. [Google Scholar] [CrossRef]
- Caliceti, M.; Argese, E.; Sfriso, A.; Pavoni, B. Heavy metal contamination in the seaweeds of the Venice lagoon. Chemosphere 2002, 47, 443–454. [Google Scholar] [CrossRef]
- Obidike, L.I.; Yoro, K.O. Effect of zeolitic nano-catalyst on biodiesel yield and biochar formation during the pyrolysis of tallow. Biofuels 2021. [Google Scholar] [CrossRef]
- Taghavi, S.; Norouzi, O.; Tavasoli, A.; Di, F.; Signoretto, M.; Menegazzo, F.; Di, A. ScienceDirect Catalytic conversion of Venice lagoon brown marine algae for producing hydrogen-rich gas and valuable biochemical using algal biochar and Ni/SBA-15 catalyst. Int. J. Hydrogen Energy 2018, 43, 19918–19929. [Google Scholar] [CrossRef]
- Demirbas, A. Mechanisms of liquefaction and pyrolysis reactions of biomass. Energy Convers. Manag. 2000, 41, 633–646. [Google Scholar] [CrossRef]
- Ohlrogge, J.B.; Jaworski, J.G. Regulation of fatty acid synthesis. Annu. Rev. Plant Biol. 1997, 48, 109–136. [Google Scholar] [CrossRef]
- Lu, X.; Vora, H.; Khosla, C. Overproduction of free fatty acids in E. coli: Implications for biodiesel production. Metab. Eng. 2008, 10, 333–339. [Google Scholar] [CrossRef]
- Voelker, T.A.; Worrell, A.C.; Anderson, L.; Bleibaum, J.; Fan, C.; Hawkins, D.J.; Radke, S.E.; Davies, H.M.; Voelker, T.A.; Worrell, A.C.; et al. Fatty Acid Biosynthesis Redirected to Medium Chains in Transgenic Oilseed Plants. Science 2016, 257, 72–74. [Google Scholar] [CrossRef] [PubMed]
- Voelker, T.A.; Davies, H.M. Alteration of the specificity and regulation of fatty acid synthesis of Escherichia coli by expression of a plant medium-chain acyl-acyl carrier protein thioesterase. J. Bacteriol. 1994, 176, 7320–7327. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.; Li, Y.; Wang, X.; Wan, Y.; Chen, Q.; Wang, C.; Lin, X.; Liu, Y.; Chen, P.; Ruan, R. Bioresource Technology Microwave-assisted pyrolysis of microalgae for biofuel production. Bioresour. Technol. 2011, 102, 4890–4896. [Google Scholar] [CrossRef] [PubMed]
- Topare, N.S.; Raut, S.J.; Renge, V.C.; Khedkar, S.V.; Chavan, Y.P.; Bhagat, S.L. Extraction of oil from algae by solvent extraction and oil expeller method. Int. J. Chem. Sci. 2011, 9, 1746–1750. [Google Scholar]
- Al hattab, M.; Ghaly, A. Microalgae Oil Extraction Pre-treatment Methods: Critical Review and Comparative Analysis Fundamentals of Renewable Energy and Applications. J. Fundam. Renew. Energy Appl. 2015, 5, 1000172. [Google Scholar] [CrossRef]
- Brown, T.M.; Duan, P.; Savage, P.E. Hydrothermal Liquefaction and Gasification of Nannochloropsis sp. Energy Fuel 2010, 24, 3639–3646. [Google Scholar] [CrossRef]
- Carrero, A.; Vicente, G.; Rodríguez, R.; Linares, M.; Peso, G.L. Hierarchical zeolites as catalysts for biodiesel production from Nannochloropsis microalga oil. Catal. Today 2011, 167, 148–153. [Google Scholar] [CrossRef]
- Ranjan, A.; Patil, C.; Moholkar, V.S. Mechanistic Assessment of Microalgal Lipid Extraction. Ind. Eng. Chem. Res. 2010, 49, 2979–2985. [Google Scholar] [CrossRef]
- Rawat, I.; Kumar, R.R.; Mutanda, T.; Bux, F. Biodiesel from microalgae : A critical evaluation from laboratory to large scale production. Appl. Energy 2013, 103, 444–467. [Google Scholar] [CrossRef]
- Li, W.; Zheng, P.; Guo, J.; Ji, J.; Zhang, M.; Zhang, Z.; Zhan, E. Bioresource Technology Characteristics of self-alkalization in high-rate denitrifying automatic circulation (DAC) reactor fed with methanol and sodium acetate. Bioresour. Technol. 2014, 154, 44–50. [Google Scholar] [CrossRef]
- Puri, M.; Sharma, D.; Barrow, C.J. Enzyme-assisted extraction of bioactives from plants. Trends Biotechnol. 2012, 30, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.; Schenk, P.M. Minireview Progress on lipid extraction from wet algal biomass for biodiesel production. Microb. Biotechnol. 2016, 9, 718–726. [Google Scholar] [CrossRef]
- Kumar, R.R.; Rao, P.H.; Arumugam, M. Lipid extraction methods from microalgae : A comprehensive review. Front. Energy Res. 2015, 2, 1–9. [Google Scholar] [CrossRef]
- Liu, X.; He, H.; Wang, Y.; Zhu, S.; Piao, X. Transesterification of soybean oil to biodiesel using CaO as a solid base catalyst. Fuel 2008, 87, 216–221. [Google Scholar] [CrossRef]
- Liu, X.; He, H.; Wang, Y.; Zhu, S. Transesterification of soybean oil to biodiesel using SrO as a solid base catalyst. Catal. Commun. 2007, 8, 1107–1111. [Google Scholar] [CrossRef]
- Anbessie, T.; Mamo, T.T.; Mekonnen, Y.S. Optimized Biodiesel Production from Waste Cooking Oil (WCO) using Calcium Oxide (CaO) Nano-catalyst. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef]
- Chouhan, A.P.S.; Sarma, A.K. Modern heterogeneous catalysts for biodiesel production: A comprehensive review. Renew. Sustain. Energy Rev. 2011, 15, 4378–4399. [Google Scholar] [CrossRef]
- Ma, H.; Li, S.; Wang, B.; Wang, R.; Tian, S. Transesterification of Rapeseed Oil for Synthesizing Biodiesel by K/KOH/γ-Al2O3 as Heterogeneous Base Catalyst. J. Oil Chem. Soc. 2008, 85, 263–270. [Google Scholar] [CrossRef]
- Kim, H.J.; Hilger, H.; Bae, S. NiSO4/SiO2 catalyst for biodiesel production from free fatty acids in brown grease. J. Energy Eng. 2013, 139, 35–40. [Google Scholar] [CrossRef]
- Han, F.Y.; Komiyama, M.; Uemura, Y.; Rabat, N.E. One-path catalytic supercritical methanothermal production of fatty acid methyl ester fractions from wet microalgae Chlorella vulgaris. Biomass Bioenergy 2020, 143, 105834. [Google Scholar] [CrossRef]
- Jafari, A.; Esmaeilzadeh, F.; Mowla, D.; Sadatshojaei, E.; Heidari, S.; Wood, D.A. New insights to direct conversion of wet microalgae impregnated with ethanol to biodiesel exploiting extraction with supercritical carbon dioxide. Fuel 2021, 285, 119199. [Google Scholar] [CrossRef]
- Ramachandran, K.; Suganya, T.; Gandhi, N.N.; Renganathan, S. Recent developments for biodiesel production by ultrasonic assist transesterification using different heterogeneous catalyst : A review. Renew. Sustain. Energy Rev. 2013, 22, 410–418. [Google Scholar] [CrossRef]
- Issariyakul, T.; Dalai, A.K. Comparative kinetics of transesterification for biodiesel production from palm oil and mustard oil. Can. J. Chem. Eng. 2012, 90, 342–350. [Google Scholar] [CrossRef]
- Sivaramakrishnan, R.; Incharoensakdi, A. Production of methyl ester from two microalgae by two-step transesterification and direct transesterification. Environ. Sci. Pollut. Res. 2017, 24, 4950–4963. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, H.; Godwin, J.J.; Sivamani, S. Data in Brief Data set for extraction and transesteri fi cation of bio-oil from Stoechospermum marginatum, a brown marine algae. Data Br. 2017, 14, 623–628. [Google Scholar] [CrossRef]
- Plata, V.; Kafarov, V.; Moreno, N. Optimization of Third Generation Biofuels Production: Biodiesel from Microalgae Oil by Homogeneous Transesterification. Chem. Eng. Trans. 2010, 21, 1201–1206. [Google Scholar] [CrossRef]
- Narula, V.; Thakur, A.; Uniyal, A.; Kalra, S.; Jain, S. Process parameter optimization of low temperature transesteri fi cation of algae-Jatropha Curcas oil blend. Energy 2017, 119, 983–988. [Google Scholar] [CrossRef]
- Dong, T.; Wang, J.; Miao, C.; Zheng, Y.; Chen, S. Biore source Tec hnology Two-step in situ biodiesel production from microalgae with high free fatty acid content. Bioresour. Technol. 2013, 136, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Kusdiana, D.; Saka, S. Effects of water on biodiesel fuel production by supercritical methanol treatment. Bioresour. Technol. 2004, 91, 289–295. [Google Scholar] [CrossRef]
- Freedman, B.; Pryde, E.H.; Mounts, T.L.; Regional, N. Variables Affecting the Yields of Fatty Esters from Transesterified Vegetable Oils. Jaocs 1984, 61, 1638–1643. [Google Scholar] [CrossRef]
- Ma, F.; Hanna, M.A. Biodiesel production: A review. Bioresour. Technol. 1999, 70, 1–15. [Google Scholar] [CrossRef]
- Suganya, T.; Kasirajan, R.; Renganathan, S. Bioresource Technology Ultrasound-enhanced rapid in situ transesterification of marine macroalgae Enteromorpha compressa for biodiesel production. Bioresour. Technol. 2014, 156, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Vieira, C.; Hachemi, I.; Pereira, S.; Mäki-arvela, P.; Aho, A.; Hemming, J.; Smeds, A.; Heinmaa, I.; Batista, F.; Cristina, D.; et al. A route to produce renewable diesel from algae: Synthesis and characterization of biodiesel via in situ transesterification of Chlorella alga and its catalytic deoxygenation to renewable diesel. Fuel 2015, 155, 144–154. [Google Scholar] [CrossRef]
- Mathimani, T.; Uma, L.; Prabaharan, D. Homogeneous acid catalysed transesteri fi cation of marine microalga Chlorella sp. BDUG 91771 lipid—An efficient biodiesel yield and its characterization. Renew. Energy 2015, 81, 523–533. [Google Scholar] [CrossRef]
- Chamola, R.; Khan, M.F.; Raj, A.; Verma, M.; Jain, S. Response surface methodology based optimization of in situ transesterification of dry algae with methanol, H2SO4 and NaOH. Fuel 2019, 239, 511–520. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, S.; Wang, Y.; Adhikari, S.; Dempster, T.A.; Wang, Y. Direct biodiesel production from wet microalgae assisted by radio frequency heating. Fuel 2019, 256, 115994. [Google Scholar] [CrossRef]
- Wahidin, S.; Idris, A.; Yusof, N.M.; Kamis, N.H.H.; Shaleh, S.R.M. Optimization of the ionic liquid-microwave assisted one-step biodiesel production process from wet microalgal biomass. Energy Convers. Manag. 2018, 171, 1397–1404. [Google Scholar] [CrossRef]
- Malekghasemi, S.; Kariminia, H.R.; Plechkova, N.K.; Ward, V.C.A. Direct transesterification of wet microalgae to biodiesel using phosphonium carboxylate ionic liquid catalysts. Biomass Bioenergy 2021, 150, 106126. [Google Scholar] [CrossRef]
- Chen, L.; Liu, T.; Zhang, W.; Chen, X.; Wang, J. Bioresource Technology Biodiesel production from algae oil high in free fatty acids by two-step catalytic conversion. Bioresour. Technol. 2012, 111, 208–214. [Google Scholar] [CrossRef] [PubMed]
- de Jesus, S.S.; Ferreira, G.F.; Moreira, L.S.; Filho, R.M. Biodiesel production from microalgae by direct transesterification using green solvents. Renew. Energy 2020, 160, 1283–1294. [Google Scholar] [CrossRef]
- Schenk, P.M.; Thomas-hall, S.R. Second Generation Biofuels: High-Efficiency Microalgae for Biodiesel Production. Bioenergy Res. 2008, 1, 20–43. [Google Scholar] [CrossRef]
- Mcneff, C.V.; Mcneff, L.C.; Yan, B.; Nowlan, D.T.; Rasmussen, M.; Gyberg, A.E.; Krohn, B.J.; Fedie, R.L.; Hoye, T.R. Continuous catalytic system for biodiesel production. Appl. Catal. A Gen. A 2008, 343, 39–48. [Google Scholar] [CrossRef]
- Krohn, B.J.; Mcneff, C.V.; Yan, B.; Nowlan, D. Bioresource Technology Production of algae-based biodiesel using the continuous catalytic Mcgyan Ò process. Bioresour. Technol. 2011, 102, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Umdu, E.S.; Tuncer, M.; Seker, E. Bioresource Technology Transesterification of Nannochloropsis oculata microalga’s lipid to biodiesel on Al2O3 supported CaO and MgO catalysts. Bioresour. Technol. 2009, 100, 2828–2831. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A. Valorization of harmful algae E. compressa for biodiesel production in presence of chicken waste derived catalyst. Renew. Energy 2018, 129, 132–140. [Google Scholar] [CrossRef]
- Hwa, S.; Islam, A.; Taufiq-yap, Y.H. Chemical Engineering Research and Design Algae derived biodiesel using nanocatalytic transesterification process. Chem. Eng. Res. Des. 2016, 111, 362–370. [Google Scholar] [CrossRef]
- Lotero, E.; Liu, Y.; Lopez, D.E.; Suwannakarn, K.; Bruce, D.A.; Goodwin, J.G. Synthesis of Biodiesel via Acid Catalysis. Ind. Eng. Chem. Res. 2005, 44, 5353–5363. [Google Scholar] [CrossRef]
- Environ, E.; Shimizu, K.; Satsuma, A. Environmental Science Toward a rational control of solid acid catalysis for green synthesis and biomass conversion. Energy Environ. Sci. 2011, 4, 3140–3153. [Google Scholar] [CrossRef]
- Misono, M.; Ono, I.; Koyano, G.; Aoshima, A. Heteropolyacids. Versatile green catalysts usable in a variety of reaction media *. Pure Appl. Chem. 2000, 72, 1305–1311. [Google Scholar] [CrossRef]
- Borges, M.E.; Díaz, L. Recent developments on heterogeneous catalysts for biodiesel production by oil esterification and transesterification reactions: A review. Renew. Sustain. Energy Rev. 2012, 16, 2839–2849. [Google Scholar] [CrossRef]
- Okuhara, T. Water-Tolerant Solid Acid Catalysts. Chem. Rev. 2002, 102, 3641–3666. [Google Scholar] [CrossRef] [PubMed]
- Kiss, A.A.; Dimian, A.C.; Rothenberg, G. Solid Acid Catalysts for Biodiesel Production—Towards Sustainable Energy. Adv. Synth. Catal. 2006, 384, 75–81. [Google Scholar] [CrossRef]
- Suppes, G.J.; Dasari, M.A.; Doskocil, E.J.; Mankidy, P.J.; Goff, M.J. Transesterification of soybean oil with zeolite and metal catalysts. Appl. Chem. A Gen. 2004, 257, 213–223. [Google Scholar] [CrossRef]
- Macario, A.; Giordano, G.; Onida, B.; Cocina, D.; Tagarelli, A.; Maria, A. Applied Catalysis A: General Biodiesel production process by homogeneous/heterogeneous catalytic system using an acid-base catalyst. Appl. Chem. A Gen. 2010, 378, 160–168. [Google Scholar] [CrossRef]
- Melero, J.A.; Bautista, L.F.; Morales, G.; Iglesias, J.; Briones, D. Biodiesel Production with Heterogeneous Sulfonic Acid-Functionalized Mesostructured Catalysts. Energy Fuel 2009, 23, 539–547. [Google Scholar] [CrossRef]
- Serrano, D.P.; Escola, M.; Rodrı, M.; Juan, R.; Uni, C. Hierarchical Zeolites with Enhanced Textural and Catalytic Properties Synthesized from Organofunctionalized Seeds. Chem. Mater. 2006, 18, 2462–2464. [Google Scholar] [CrossRef]
- Macario, A.; Giordano, G. Catalytic Conversion of Renewable Sources for Biodiesel Production: A Comparison between Biocatalysts and Inorganic Catalysts. Catal. Lett. 2013, 143, 159–168. [Google Scholar] [CrossRef]
- Qu, S.; Chen, C.; Guo, M.; Jiang, W.; Lu, J.; Yi, W.; Ding, J. Microwave-assisted in-situ transesterification of Spirulina platensis to biodiesel using PEG/MgO/ZSM-5 magnetic catalyst. J. Clean. Prod. 2021, 311, 127490. [Google Scholar] [CrossRef]
- Bala, D.D.; Misra, M.; Chidambaram, D. Single step esterification of algae oil using mesoporous solid acid catalyst. Fuel 2014, 117, 1093–1095. [Google Scholar] [CrossRef]
- Rana, A.; Alghazal, M.S.M.; Alsaeedi, M.M.; Bakdash, R.S.; Basheer, C.; Al-saadi, A.A. Preparation and Characterization of Biomass Carbon—Based Solid Acid Catalysts for the Esterification of Marine Algae for Biodiesel Production. BioEnergy Res. 2019, 12, 433–442. [Google Scholar] [CrossRef]
- Gurusamy, S.; Kulanthaisamy, M.R.; Hari, D.G.; Veleeswaran, A.; Thulasinathan, B.; Muthuramalingam, J.B.; Balasubramani, R.; Chang, S.W.; Arasu, M.V.; Al-Dhabi, N.A.; et al. Environmental friendly synthesis of TiO2-ZnO nanocomposite catalyst and silver nanomaterilas for the enhanced production of biodiesel from Ulva lactuca seaweed and potential antimicrobial properties against the microbial pathogens. J. Photochem. Photobiol. B Biol. 2019, 193, 118–130. [Google Scholar] [CrossRef]
- Sala, J.G. Ethyl Octyl Ether Synthesis from 1-Octanol and Ethanol or Diethyl Carbonate; University of Baecelona: Barcelona, Spain, 2013. [Google Scholar]
- Karinen, R. Etherification of some C8-alkenes to fuel ethers. In Industrial Chemistry Publication Series; Helsinki University of Technology: Espoo, Finland, 2002. [Google Scholar]
- Lopez, D.E.; Jr, J.G.G.; Bruce, D.A.; Lotero, E. Transesterification of triacetin with methanol on solid acid and base catalysts. Appl. Catal. A Gen. 2005, 295, 97–105. [Google Scholar] [CrossRef]
- Ravenelle, R.M.; Schü, F.; Amico, A.D.; Danilina, N.; Van Bokhoven, J.A.; Lercher, J.A.; Jones, C.W.; Sievers, C. Stability of Zeolites in Hot Liquid Water. J. Phys. Chem. 2010, 114, 19582–19595. [Google Scholar] [CrossRef]
- Laitinen, A.; Harlin, A. Production of p-Cymene from Crude Sulphate Turpentine with Commercial Zeolite Catalyst Using a Continuous Fixed Bed Reactor. Org. Process. Res. Dev. 2014, 18, 1468–1475. [Google Scholar] [CrossRef]
- Cseri, T.; Bkassy, S.; Figueras, F.; De Menorval, L.; Dutartre, R. Characterization of clay-based K catalysts and their application in Friedel-Crafts alkylation of aromatics. Appl. Catal. A Gen. 1995, 132, 141–155. [Google Scholar] [CrossRef]
- Asikainen, M.; Munter, T.; Linnekoski, J. Conversion of polar and non-polar algae oil lipids to fatty acid methyl esters with solid acid catalysts—A model compound study. Bioresour. Technol. 2015, 191, 300–305. [Google Scholar] [CrossRef]
- Sánchez, D.A.; Tonetto, G.M.; Ferreira, M.L. Burkholderia cepacia lipase: A versatile catalyst in synthesis reactions. Biotechnology 2017, 115, 6–24. [Google Scholar] [CrossRef]
- Nielsen, P.M.; Brask, J.; Fjerbaek, L. Review Article Enzymatic biodiesel production: Technical and economical considerations. Eur. J. Lipid Sci. Technol. 2008, 110, 692–700. [Google Scholar] [CrossRef]
- Mukherjee, J.; Nath, M. Dual bioimprinting of Thermomyces lanuginosus lipase for synthesis of biodiesel. Biotechnol. Rep. 2016, 10, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, J.; Canet, A.; Rivera, I.; Osório, N.M.; Sandoval, G.; Valero, F.; Ferreira-dias, S. Bioresource Technology Biodiesel production from crude Jatropha oil catalyzed by non-commercial immobilized heterologous Rhizopus oryzae and Carica papaya lipases. Bioresour. Technol. 2020, 213, 88–95. [Google Scholar] [CrossRef]
- Mohamad, N.R.; Haziqah, N.; Marzuki, C.; Buang, N.A.; Wahab, R.A. An overview of technologies for immobilization of enzymes and surface analysis techniques for immobilized enzymes. Biotechnol. Biotechnol. Equip. 2015, 29, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Kensingh, P.; Chulalaksananukul, W.; Charuchinda, S. Lipase immobilization on Scirpus grossus L.f. fiber support by glutaraldehyde-crosslinked technique for biodiesel synthesis. J. Biotechnol. 2010, 150, 163. [Google Scholar] [CrossRef]
- Yan, J.; Yan, Y.; Liu, S.; Hu, J.; Wang, G. Preparation of cross-linked lipase-coated micro-crystals for biodiesel production from waste cooking oil. Bioresour. Technol. 2011, 102, 4755–4758. [Google Scholar] [CrossRef] [PubMed]
- Kumari, V.; Shah, S.; Gupta, M.N. Preparation of biodiesel by lipase-catalyzed transesterification of high free fatty acid containing oil from Madhuca indica. Energy Fuels 2007, 21, 368–372. [Google Scholar] [CrossRef]
- Nematian, T.; Salehi, Z.; Shakeri, A. Conversion of bio-oil extracted from Chlorella vulgaris micro algae to biodiesel via modified superparamagnetic nano-biocatalyst. Renew. Energy 2020, 146, 1796–1804. [Google Scholar] [CrossRef]
- Chen, J.Z.; Wang, S.; Zhou, B.; Dai, L.; Liu, D.; Du, W. A robust process for lipase-mediated biodiesel production from microalgae lipid. RSC Adv. 2016, 6, 48515–48522. [Google Scholar] [CrossRef]
- Guldhe, A.; Singh, B.; Rawat, I.; Permaul, K.; Bux, F. Biocatalytic conversion of lipids from microalgae Scenedesmus obliquus to biodiesel using Pseudomonas fluorescens lipase. Fuel 2015, 147, 117–124. [Google Scholar] [CrossRef]
- Bharathiraja, B.; Kumar, R.R.; Praveenkumar, R.; Chakravarthy, M.; Yogendran, D.; Jayamuthunagai, J. Bioresource Technology Biodiesel production from different algal oil using immobilized pure lipase and tailor made r Pichia pastoris with Cal A and Cal B genes. Bioresour. Technol. 2016, 213, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Guldhe, A.; Singh, P.; Kumari, S.; Rawat, I.; Permaul, K.; Bux, F. Biodiesel synthesis from microalgae using immobilized Aspergillus niger whole cell lipase biocatalyst. Renew. Energy 2016, 85, 1002–1010. [Google Scholar] [CrossRef]
- López, E.N.; Medina, A.R.; Antonio, P.; Moreno, G.; Cerdán, L.E.; Valverde, L.M.; Grima, E.M. Bioresource Technology Biodiesel production from Nannochloropsis gaditana lipids through transesterification catalyzed by Rhizopus oryzae lipase. Bioresour. Technol. 2016, 203, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Kyung, O.; Hyun, Y.; Na, J.; Oh, Y.; Yeol, E. Bioresource Technology Highly efficient extraction and lipase-catalyzed transesterification of triglycerides from Chlorella sp. KR-1 for production of biodiesel. Bioresour. Technol. 2013, 147, 240–245. [Google Scholar] [CrossRef]
- Veillette, M.; Chamoumi, M.; Nikiema, J.; Faucheux, N.; Heitz, M. Production of Biodiesel from Microalgae. Adv. Chem. Eng. 2012, 10, 245–260. [Google Scholar] [CrossRef]
- Prommuak, C.; Pavasant, P.; Quitain, A.T.; Goto, M.; Shotipruk, A. Microalgal lipid extraction and evaluation of single-step biodiesel production. Eng. J. 2012, 16, 157–166. [Google Scholar] [CrossRef]
- Martinez-Guerra, E.; Gude, V.G.; Mondala, A.; Holmes, W.; Hernandez, R. Extractive-transesterification of algal lipids under microwave irradiation with hexane as solvent. Bioresour. Technol. 2014, 156, 240–247. [Google Scholar] [CrossRef]
- Fasaei, F.; Bitter, J.H.; Slegers, P.M.; van Boxtel, A.J.B. Techno-economic evaluation of microalgae harvesting and dewatering systems. Algal Res. 2018, 31, 347–362. [Google Scholar] [CrossRef]
- Lee, Y.L.; Chuang, Y.C.; Su, H.M.; Wu, F.S. Freeze-dried microalgae of Nannochloropsis oculata improve soybean oil’s oxidative stability. Appl. Microbiol. Biotechnol. 2013, 97, 9675–9683. [Google Scholar] [CrossRef]
- Chen, C.L.; Chang, J.S.; Lee, D.J. Dewatering and Drying Methods for Microalgae. Dry. Technol. 2015, 33, 443–454. [Google Scholar] [CrossRef]
- Melgar, B.; Dias, M.I.; Barros, L.; Ferreira, I.C.F.R.; Rodriguez-Lopez, A.D.; Garcia-Castello, E.M. Ultrasound and microwave assisted extraction of Opuntia fruit peels biocompounds: Optimization and comparison using RSM-CCD. Molecules 2019, 24, 3618. [Google Scholar] [CrossRef] [PubMed]
- Kapoore, R.V.; Butler, T.O.; Pandhal, J.; Vaidyanathan, S. Microwave-assisted extraction for microalgae: From biofuels to biorefinery. Biology 2018, 7, 18. [Google Scholar] [CrossRef]
- Gim, G.H.; Kim, S.W. Optimization of Cell Disruption and Transesterification of Lipids from Botryococcus braunii LB572. Biotechnol. Bioprocess Eng. 2018, 23, 550–556. [Google Scholar] [CrossRef]
- Miao, X.; Li, P.; Li, R.; Zhong, J. In situ biodiesel production from fast-growing and high oil content chlorella pyrenoidosa in rice straw hydrolysate. J. Biomed. Biotechnol. 2011, 2011, 141207. [Google Scholar] [CrossRef]
- Polshettiwar, V.; Varma, R.S. Green chemistry by nano-catalysis. Green Chem. 2010, 12, 743–775. [Google Scholar] [CrossRef]
- Jiang, C.; Jia, J.; Zhai, S. Mechanistic understanding of toxicity from nanocatalysts. Int. J. Mol. Sci. 2014, 15, 13967–13992. [Google Scholar] [CrossRef]
- Mohammed, N.I.; Kabbashi, N.A.; Alade, A.O.; Sulaiman, S. Advancement in the Utilization of Biomass-Derived Heterogeneous Catalysts in Biodiesel Production. Green Sustain. Chem. 2018, 8, 74–91. [Google Scholar] [CrossRef]
- Ranganathan, S.V.; Narasimhan, S.L.; Muthukumar, K. An overview of enzymatic production of biodiesel. Bioresour. Technol. 2008, 99, 3975–3981. [Google Scholar] [CrossRef]
- Amin Umar, A. Feasibility Study of Algae Biodiesel Production in the Cambois Peninsular (UK). Int. J. Energy Environ. Res. 2014, 2, 21–54. [Google Scholar]
| Catalyst | Surface Area (m2·g−1) | Acid Site Concentration (μmol·g−1) | Substrates with Higher Yields mol% of Methyl Palmitate Product a |
|---|---|---|---|
| Amberlyst 15 | 42 | 4700 | 1, 5, 3 |
| Amberlyst 46 | 57 | 870 | 1, 5, 3 |
| Smopex 101 | 0.15 | 4000 | 1, 5, 3 |
| Nafion NR50 | Very low | 900 | 1, 5, 3, 2 |
| Zeolite CBV8014 | 425 | 395 | 1, 3 |
| Zeolite CBV600 | 660 | 712 | 1, 3, 5 (totally low) |
| Montmorillonite K-10 | 250 | 650 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghedini, E.; Taghavi, S.; Menegazzo, F.; Signoretto, M. A Review on the Efficient Catalysts for Algae Transesterification to Biodiesel. Sustainability 2021, 13, 10479. https://doi.org/10.3390/su131810479
Ghedini E, Taghavi S, Menegazzo F, Signoretto M. A Review on the Efficient Catalysts for Algae Transesterification to Biodiesel. Sustainability. 2021; 13(18):10479. https://doi.org/10.3390/su131810479
Chicago/Turabian StyleGhedini, Elena, Somayeh Taghavi, Federica Menegazzo, and Michela Signoretto. 2021. "A Review on the Efficient Catalysts for Algae Transesterification to Biodiesel" Sustainability 13, no. 18: 10479. https://doi.org/10.3390/su131810479
APA StyleGhedini, E., Taghavi, S., Menegazzo, F., & Signoretto, M. (2021). A Review on the Efficient Catalysts for Algae Transesterification to Biodiesel. Sustainability, 13(18), 10479. https://doi.org/10.3390/su131810479









