Abstract
The northeastern Anatolia region of Turkey is one of the richest plant biodiversity areas in Turkey. This study describes some important tree and fruit characteristics of 20 seed-propagated summer apple genotypes from northeastern Anatolia using morphological and biochemical markers. The genotypes were evaluated morphologically for tree growth habit, ripening date, fruit weight, fruit firmness, fruit shape, fruit color, fruit taste, juiciness and aroma. The biochemical characteristics measured were organic acids, SSC (soluble solid content), titratable acidity, vitamin C, total phenolic content and antioxidant capacity. We found significant differences for most of the morphological and biochemical characteristics. The genotypes exhibited quite diverse harvest dates ranging from 4 July to 16 August. Summer apple genotypes had fruit weight between 101.9 g and 133.5 g. The genotypes had diverse fruit shapes, including round, conic and oblate, and over fruit colours such as pink, red, yellow and green. Genotypes GUM7 and GUM13 stood out for fruit weight, SSC, total phenolic and antioxidant capacity. GUM7 and GUM13 present dissimilarity from the other genotypes. While the first showed a lower average fruit weight, SSC, total phenolic content and antioxidant capacity, the second revealed higher average of fruit weight, total phenolic content, antioxidant capacity and higher SSC. Due to their favorable characteristics, GUM13 can be used for further crosses with genotypes of other groups, which also present favorable characteristics.
1. Introduction
Anatolia is known as the origin center of several horticultural plants [,,,]. Among fruit crops, apples rank second in global fruit production with about 83 million tonnes and only bananas exceed this production quantity with approximately 113 million tonnes []. Since 2002, global apple production has significantly increased by a total of 30% and significant apple producers in the European Union such as Poland and Turkey have increased production, contributing to the overall supply to the European market. Other important producers, e.g., China, India, Chile, South Africa and Brazil, are also increasing their production and the USA, Italy and Argentina have maintained their production at a stable level. Apple production in Germany is stable, actually slightly increasing, and in Russia it is increasing, as fruit imports from Europe and Canada are banned [,].
Apple fruits attract consumers for their appearance and pleasant flavor and available market year around due to their high storability capacity [,].
Average yield increase and innovations in apple production systems are the reason for growing global apple production. In particular, the use of dwarf rootstocks, higher planting densities and new higher yielding cultivars contributed to this increase [].
The worldwide well-known 34 biodiversity hotspots that indicate high plant biodiversity areas are found in different continents. Located between Asia and Europe, Turkey has 3 out of 34 biodiversity hotspots. These plant biodiversity hotspots in Turkey are called the Caucasus, Irano-Anatolian and the Mediterranean []. The country lies at the nexus of Europe, the Middle East, Central Asia and Africa, and is characterized by quite diverse environment conditions. Geographically, the majority of land areas in the country include three biodiversity hotspots and encirclement by three seas (Mediterranean, Black Sea and Aegean Sea). The country is also well known for their high number of endemic plants, which estimated around 3000 [,,,]. In particular, northeastern Anatolia, including Gumushane, has one of the richest plant diversities in the country due to distinct topography and climatic conditions of the region. Gumushane province is placed between the eastern Black Sea region and eastern Anatolia region and shows climate characteristics of both regions.
Worldwide apple propagation is based on the vegetative method (grafting mostly) to maintain the valuable fruit properties, and evidence suggests that seed dispersal in the large-fruiting wild relatives of the apple in some areas in the world provides change seedlings and shows more biodiversity, but also had both positive and negative fruit traits, and these positive sides could be used in apple breeding.
Fruit biodiversity has an important cultural and commercial value for Gumushane province, in particular for rural people. The province’s people prefer to grow local fruit cultivars and seed-propagated genotypes belonging to mostly apples, walnuts, mulberries, apricots, etc. People living in the region highly preferred summer apples as nutrition but also an income source for villagers. The villagers sell summer apples in both fresh and dried form. Summer apple genotypes, an important local fruit in this province, are also considered important fruit genetic resources. The rich gene pool of seed-propagated summer apples in Turkey still keeps its importance due to considerable past and present contributions of farmers and rural communities, especially in the developing world, to the creation, conservation and availability of summer apple genetic resources [].
Determination of local fruit tree diversity and conservation of these trees is imperative as they are increasingly threatened. Genetic resource identification’s main objective is to reveal trait-specific germplasm such as early or late ripening, high fruit weight, low chilling, biotic and abiotic stress resistance and conservation for crop improvement utilization.
In the literature, characterization of summer apples by using morphological and biochemical traits is limited. Therefore, this research had the objective of characterizing of 20 seed summer apple genotypes in Gumushane province in Turkey by means of morphological and biochemical traits.
2. Materials and Methods
2.1. Plant Material
Twenty seed-propagated summer apple genotypes (named from GUM1 to GUM20, GUM representing the name of Gumushane province with 1–20 as the genotype number) were used. The altitude and coordinates of Gumushane is 1227 m and 40.4608° N, 39.4803° E. All 20 genotypes are change seedlings coming from seed and grown own roots. The ages of genotypes were between 25 and 30 years. The genotypes partly show alternate bearing and fruits harvested from genotypes in the years 2018 and 2019 at the commercial maturity stage. All genotypes were found as solitary trees in different hobby orchards in Gumushane. The pomological (fruit weight, fruit firmness, shape, color) and sensory characteristics (taste, juiciness and aroma) of the genotypes were determined based on 30 homogenous fruits per genotype. Harvested fruits were kept in refrigerator at −80 °C until their biochemical analyses were done. D.U.S guidelines were used for ripening time as: very early ripening (18 May–27 June), early ripening (28 June–17 July), early-mid-ripening (18 July–27 July) and mid ripening (28 July–6 August). A digital scale sensitive to 0.01 g (Scaltec SPB31) was used for fruit weight determination. A non-destructive acoustic firmness sensor (Aweta B.V., Pijnacker, The Netherlands) was used for fruit firmness that expressed as kg/cm2. Apple skin ground and over color were decided by observation and comparison to each other. Surface area of the over color was evaluated as a percentage. For fruit shape, index fruit diameters were used. Soluble solid content (SSC) was determined in juice by using a digital refractometer (Kyoto Electronics Manufacturing Co. Ltd., Kyoto, Japan, Model RA-250HE) at 22 °C. Vitamin C (Ascorbic acid) was quantified with the reflectometer set by using RQFlex (Merck Company, Darmstadt, Germany) and expressed as mg/100 g fresh weight base. Titratable acidity was determined by titrating 10 mL of 1:10 diluted apple pulp (10 g) with 0.1 M NaOH. For sensory characteristics (taste, aroma and juiciness), a trained panel including five experts established and evaluated apples’ sensory characteristics.
2.2. Analysis of Organic Acids
Organic acid analysis were done by Bevilacqua and Califano [] with HPLC method, and results are expressed as mg/100 mL.
2.3. Total Phenolic Content
Folin–Ciocalteu assay was used for determining total phenolic content. Gallic acid is used as a standard in the calculation [].
2.4. Antioxidant Capacity
DPPH method was used for antioxidant capacity determination. Samples were homogenized by centrifuge. A total of 950 μL 0.1 N DPPH (1,1-diphenyl-2-picrylhydrazyl) solution was added upon 50 μL supernatant. Then, it was read against the blank at 515 nm wavelength spectrophotometer []. Results were expressed as μmol of vitamin C equivalent/g fresh weight.
2.5. Statistical Analysis
For statistical analysis, SPSS software and procedures were used. Analysis of variance tables was constructed using the Least Significant Difference (LSD) method at p < 0.05. In order to find the main variation trends between fruit pomological and biochemical characters in the summer apple genotypes and to evaluate their correlation, data were processed according to principal component analysis (PCA) using SPSS for Windows Version 15.0, SPSS Inc. (Chicago, IL, USA).
3. Results
3.1. Phenological Traits
Harvest dates of summer apple genotypes sampled from Gumushane region are shown in Table 1. The genotypes exhibited variable harvest dates ranging from 4 July (GUM10) to 16 August (GUM14; Table 1). The majority of genotypes were harvested in July but only four genotypes, including GUM12, GUM13, GUM14 and GUM19, were harvested in August. Among these four genotypes, GUM14 was harvested on 16 August, and this indicates a 10–14-day-later harvest than GUM12, GUM13 and GUM19.

Table 1.
Phenological observation and pomological characteristic results of twenty genotypes.
3.2. Pomological Traits
Average fruit weights of 20 summer apple genotypes sampled from Gumushane are given in Table 1. We found significant differences among genotypes in terms of fruit weight (p < 0.05). Fruit weight varied from 101.9 g (GUM7) to 133.5 g (GUM13), respectively. All genotypes showed fruit weight over 100 g, and GUM13 and GUM1 had relatively bigger fruits than rest of the genotypes (Table 1).
The genotypes had equal number of fruit shape and six genotypes had round (GUM1, GUM7, GUM11, GUM15, GUM19 and GUM20), seven genotypes had conic (GUM2, GUM3, GUM4, GUM6, GUM10, GUM12 and GUM14) and seven genotypes had oblate fruit shape (GUM5, GUM8, GUM9, GUM13, GUM16, GUM17 and GUM18), respectively (Table 1).
Summer apple genotypes commonly included green and yellow ground skin color and green, red, purple, yellow and pink was found as over skin color. Over color coverage was found between 12% (GUM3) and 76% (GUM8; Table 1).
The genotypes showed fruit firmness between 4.16 kg/cm2 (GUM10) and 7.28 kg/cm2 (GUM19), indicating great diversity among genotypes (Table 1).
3.3. Sensory Analysis
Apple cultivars have diverse texture and taste characteristics that affect consumers because consumers have a high degree of sensitivity and can distinguish taste, juiciness and aroma, which form the sensory properties of apple fruit [].
Table 2 shows fruit taste, aroma and juiciness properties of 20 seed-propagated summer apples. As indicated in Table 2, most genotypes had a sweet–sour taste (nine genotypes, GUM1, GUM2, GUM4, GUM7, GUM9, GUM13, GUM15, GUM18 and GUM20), and six genotypes had sweet taste (GUM3, GUM8, GUM11, GUM14, GUM17 and GUM19), and five genotypes (GUM5, GUM6, GUM10, GUM12 and GUM16) had a sour taste.

Table 2.
Sensory features of summer apple genotypes.
For considering aroma, eight genotypes had low (GUM3, GUM4, GUM6, GUM7, GUM11, GUM13, GUM15 and GUM17), six genotypes had moderate (GUM1, GUM2, GUM10, GUM12, GUM14 and GUM19) and six genotypes (GUM5, GUM8, GUM9, GUM16, GUM18 and GUM20) had high aroma. Among twenty genotypes, half of the genotypes had moderate juiciness (GUM1, GUM2, GUM5, GUM7, GUM8, GUM10, GUM11, GUM12, GUM17 and GUM20), eight genotypes had high juiciness (GUM3, GUM6, GUM9, GUM13, GUM14, GUM16, GUM18 and GUM19), and only two genotypes had low juiciness traits (GUM4 and GUM15; Table 1).
3.4. Organic Acids
Organic acid of 20 genotypes is given in Table 3. We found statistically significant differences among genotypes in terms of all organic acids (p < 0.05).

Table 3.
Organic acid content of summer apples (mg/100 mL).
3.5. Biochemical and Bioactive Content
Table 4 shows SSC, titratable acidity, vitamin C, total phenolic content and antioxidant capacity of seed-propagated apple genotypes. All of the above parameters were significantly affected by genotypes at 0.05 statistical level. The GUM13 genotype had the highest SSC content (12.35%), while the lowest value was obtained from the GUM7 genotype as 10.10%, respectively. From early-ripened genotypes to late-ripened genotypes, the SSC content was increased (Table 4).

Table 4.
Biochemical and bioactive characteristics of summer apples.
Another important inner quality criterion of apples along with SSC is acidity. The genotypes had titratable acidity values between 0.34 and 1.42% (Table 4).
Vitamin C content of 20 genotypes is given in Table 4. Apple fruits are well known for low vitamin C concentration. We also found low vitamin C content in 20 summer apple genotypes, ranging from 5.6 to 9.3 mg/100 g (Table 4).
Total phenolic content of the genotypes is given in Table 4. Genotypes differed from each other statistically for total phenolic content and the lowest total phenolic content was observed in GUM7 as 88.0 mg GAE/100 g, whereas the highest values are seen in the GUM13 genotype as 152.1 mg GAE/100 g, indicating a 2-fold difference between both genotypes (Table 4).
3.6. Principal Component Analysis (PCA)
A combination of all studied characteristics and genotypes was analyzed by principal components analysis (PCA), where grouping patterns were established. The dimensions of the model were found to be significant and explained 79.44% of the total variance (Figure 1). The first component (PCA1), accounting for 37.30% of the total variance, is dominated by fruit pomological characters, namely fruit weight and fruit firmness. The second component (PCA2), accounting for 29.28% of the total variance, is dominated by fruit biochemical characters, including total phenol and SSC.
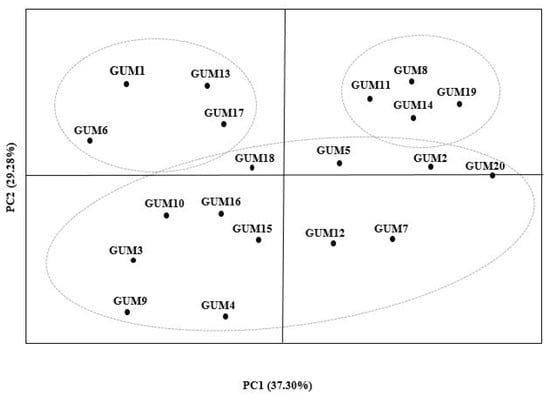
Figure 1.
Patterns of principal components analysis of fruits morphological and biochemical characteristics of studied genotypes.
4. Discussion
4.1. Phenological Traits
Summer apple genotypes sampled from Gumushane region exhibited variable harvest dates ranging from 4 July to 16 August. Considering the D.U.S scale, GUM1, GUM2, GUM5, GUM7, GUM9, GUM10, GUM14, GUM18 and GUM20 had early-ripening, GUM3, GUM4, GUM6, GUM11, GUM15 and GUM16 had early–mid-ripening and GUM8, GUM12, GUM13, GUM17 and GUM19 had mid-ripening traits. Previously based on the D.U.S scale, summer apples were classified as very early, early, mid-early and mid-ripening in Iran [].
In the temperate climate zone, summer apple cultivars are generally maturated in the mid-summer, but most cultivars are harvested in late summer. Summer apples are not suitable for preservation and must be consumed in a short time. They are also poorer in content than autumn and winter apples. For that reason, late season apples are sometimes called winter apples and are common in markets year around [,].
4.2. Pomological Traits
Average fruit weight of 20 summer apple genotypes sampled from Gumushane varied from 101.9 g to 133.5 g. According to the literature searched, abundant information is available on late season (autumn or winter) apples because they include over 7000 cultivars around the world and have higher fruit quality.
In the literature, pomological reports, including fruit weight, on summer apples from different parts of the world are scarce. Hajnajari et al. [] used found quite variable fruit weight (11.47–98.50 g) among summer apples in Iran. In another study, fruit weight was in range of 20.9 and 139.3 g in Turkey []. Balta [] found variable fruit weight (32.29 g to 138.25 g) in Turkey as well. Serdar et al. [] and Kaya et al. [] reported variable fruit weight from 54.3 g to 206.0 g and from 43 to 310 g among apple selections. Our fruit weight results were found within the limits of the above studies. Among the most important external quality indices of apple fruits are color and size.
The genotypes had round, conic and oblate fruit shape. Kaya et al. [] found diverse fruit shapes such as cylindiric, round, oblate and conic in apples in Turkey.
In this study, summer apple genotypes had a green and yellow ground color and over skin colors were more diverse, including purple, green, red, yellow and pink. Over color coverage was found between 12% and 76%. Ozrenk et al. [] and Kaya et al. [] reported that red, yellow and green over color and green and yellow ground color is common in apple fruits. Color preferences in apple is consumer-dependent. Color uniformity and intensity of blush and ground color (saturation of red) are main factors in this preference [].
Fruit firmness was between 4.16 kg/cm2 and 7.28 kg/cm2. Previously, Ozrenk et al. [], Karlidag and Esitken [] and Kalkisim et al. [] reported fruit firmness between 3.70–17.08 kg/cm2 in local apple cultivars. Most apple quality characteristics, including fruit firmness, are genetically controlled []. Consumers usually reject apples with a firmness of less than 4.5 kg/cm2 and, therefore, this is the minimum acceptable firmness level for many soft apple cultivars []. Skendrovic-Babojelic [] reported that among apple cultivars, ‘Pink Lady’ was the hardest cultivar with average value of 7.3 kg/cm2 fruit firmness, followed by ‘Granny Smith’ which had an average value of 6.4 kg/cm2, while ‘Idared’ had an average fruit firmness of 4.5 kg/cm2.
4.3. Sensory Analysis
Most genotypes had a sweet–sour taste (nine genotypes), and six genotypes had a sweet and five genotypes had a sour taste. For considering juiciness, genotypes showed nearly equal low, moderate and high juiciness traits. Among twenty genotypes, half of the genotypes had a moderate aroma and only two genotypes had low aroma. Skendrovic-Babojelic [] reported that apple cultivars differ from each other in terms of sensory characteristics in Croatia. Previously, apple cultivars were evaluated based on taste and juiciness characteristics, and it was found that most of the genotypes had a sour taste and low juiciness [,]. In Iran, a number of summer apple genotypes were evaluated, and it was concluded that genotypes had different aroma and taste characteristics []. In Turkey, Kalkisim et al. [] reported that apple genotypes had tart, sour and sweet tastes. Sugars, organic acids and phenolic compounds contribute to the aroma of apples [].
4.4. Organic Acids
All apple genotypes had malic acid as the highest portion, followed by citric acid, succinic acid, oxalic acid and tartaric acid. Previous studies also indicated that the abundance of malic acid in apple cultivars [,,,] and malic acid concentrations are between 2.06 and 4.62 mg/100 mL []. Organic acid and aromatic volatile compounds, and soluble sugars, are key components for fruit taste and strongly facilitate overall organoleptic quality and fruit stability [,].
4.5. Biochemical and Bioactive Content
SSC content was between 10.10% and 12.35% among 20 genotypes. Previously, apple genotypes/cultivars showed great variability in SSC, which varied from 10.0 to 17.11% [,,,,,]. Our SSC results are in general accordance with the above studies’ results. The high soluble solids content (SSC) indicates sweet taste (sugar content) of apples and is an accepted important index on fruit quality. Identification of apple fruit based on internal quality can also enhance the industry’s competitiveness and profitability and assure consumer satisfaction.
The genotypes had titratable acidity values between 0.34 and 1.42%. Cripps et al. [] reported titratable acidity in three apple cultivars between 0.32 and 0.90%. Kaya and Balta [] found that titratable acid ranged from 0.07 to 1.57% in apples. Serdar et al. [] also found titratable acidity in apples between 0.2 and 1.3%, which indicates similarities with our present findings. Titratable acidity strongly relates to the taste and maturity of apples, and consumers often have distinct preferences for acid- or sweet-tasting apples []. Nour et al. [] reported the average value of titratable acidity was 0.265% in Romania among 15 apple cultivars. To a high extent, the malic acid content in apples is correlated with titratable acidity [].
Vitamin C content of 20 summer apple genotypes ranged from 5.6 to 9.3 mg/100 g. Previously, vitamin C content was reported between 5.2 and 17.2 mg/100 g [,,]. Loncaric and Prizota [] found amount of vitamin C between 4.75 and 8.42 mg/100 g in apple cultivars. Nour et al. (2010) reported low ascorbic acid contents (average 6.18 mg/100 g) with the exception of ‘Red Boskoop’ (18.7 mg/100 g) and ‘Idared’ (11.4 mg/100 g) among 15 apple cultivars in Romania. Ascorbic acid (Vitamin C) content of apples affected by genetic background, cultivation conditions and environmental effects [,].
Total phenolic content of summer apple genotypes was in the range of 88.0 mg GAE/100 g and 152.1 mg GAE/100 g, indicating a 2-fold difference between both genotypes. Total phenolic content is a frequently searched parameter in apples recently, and studies revealed that this phenomenon is quite variable among genotypes and strongly affected by genetic background, rootstocks, planting density, harvest time, location, storage condition, geographical condition, etc., and the amount of total phenolic content was found to be 46–212 mg GAE/100 g fresh weight base in different studies [,,,,,]. Our results in agreement with results of previous studies. In apple fruits, polyphenols are one of the main antioxidant ingredients [].
The antioxidant capacity of summer apples changed between 67 μmol of vitamin C equivalents/g and 135 μmol of vitamin C equivalents/g. Previously, the antioxidant capacity in apples was found to be between 110 and 491 []. These values indicate similarities to our results and reveal the importance of consuming summer apples as a health food. Previous studies are also indicating great biochemical diversity among fruit species [,,,,,,,,,,].
5. Conclusions
The number of summer apple cultivars throughout the world is very limited and a few studies conducted on summer apples for suitability to local climatic conditions. The results indicated that the region is particularly rich for summer apple genotypes and it can be accepted for ready breeding material. Summer apple genotypes such as GUM 13 had the highest fruit weight, total phenolic content, SSC and antioxidant activity, and GUM14 was found to be important for earliness.
Author Contributions
Conceptualization, N.K., S.E., M.R.B. and O.C.; data curation, N.K., S.E., M.R.B. and O.C.; formal analysis, N.K., S.E., M.R.B. and O.C.; methodology, N.K., S.E. and O.C.; visualization, N.K., S.E., O.C., T.N. and I.O. writing—original draft, N.K., S.E., O.C., T.N. and I.O.; writing—review and editing, N.K., S.E., O.C. and T.N. All authors have read and agreed to the published version of the manuscript.
Funding
The plant material was provided through activity no. 6.2.10 ref. 51834/2017-MZE-17253, subprogram “National Program of Conservation and Utilization of Plant Genetic Resources and Agrobiodiversity”, which is funded by the Ministry of Agriculture of the Czech Republic, and used the infrastructure acquired by the project CZ.02.1.01/0.0/0.0/16_017/0002334 Research Infrastructure for Young Scientists, which is co-financed by the Operational Program of Research, Development and Education.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All new research data were presented in this contribution.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Baser, K.H.C. Aromatic biodiversity among the flowering plant taxa of Turkey. Pure Appl. Chem. 2002, 74, 527–545. [Google Scholar] [CrossRef]
- Ercisli, S.; Esitken, A.; Cangi, R.; Sahin, F. Adventitious root formation of kiwifruit in relation to sampling date, IBA and Agrobacterium rubi inoculation. Plant Growth Regul. 2003, 41, 133–137. [Google Scholar] [CrossRef]
- Dogan, H.; Ercisli, S.; Jurikova, T.; Temim, E.; Leto, A.; Hadziabulic, A.; Tosun, M.; Narmanlioglu, H.K.; Zia-Ul-Haq, M. Physicochemical and antioxidant characteristics of fruits of cape gooseberry (Physalis peruviana L.) from Turkey. Oxid. Commun. 2014, 37, 1005–1014. [Google Scholar]
- Gecer, M.K.; Kan, T.; Gundogdu, M.; Ercisli, S.; Ilhan, G.; Sagbas, H.I. Physicochemical characteristics of wild and cultivated apricots (Prunus armeniaca L.) from Aras valley in Turkey. Genet. Resour. Crop Evol. 2020, 67, 935–945. [Google Scholar] [CrossRef]
- Food and Agricultural Organization. Statistical Database; FAO: Rome, Italy, 2021. [Google Scholar]
- Way, R.D.; Aldwinckle, H.S.; Lamb, R.C.; Rejman, A.; Sansavini, S.; Shen, T.; Watkins, R.; Westwood, M.N.; Yoshida, Y. Apples (Malus). In Genetic Resources of Temprate Fruit and Nut Crops 1; Moore, J.N., Ballington, J.R., Eds.; Acta Horticultural Science: Wageningen, The Netherlands, 1990. [Google Scholar]
- Butiuc-Keul, A.; Coste, A.; Farkas, A.; Cristea, V.; Isac, V.; Halmagyi, A. Molecular characterization of apple (Malus × domestica Borkh.) genotypes originating from three complementary conservation strategies. Turk. J. Agric. For. 2019, 43, 464–477. [Google Scholar] [CrossRef]
- Asif Ali, M.; Raza, H.; Azam, K.; Manzoor, H. Effect of different periods of ambient storage on chemical composition of apple fruit. Int. J. Agric. Biol. 2004, 6, 568–571. [Google Scholar]
- Skendrovic Babojelic, M.; Ivancic, K.; Druzic, J.; Kovac, A.; Voca, S. Chemical and sensory characteristics of three apple cultivars (Malus×domestica Borkh.). Agric. Consp. Sci. 2007, 72, 317–322. [Google Scholar]
- Milosevic, T.; Milosevic, N.; Mladenovic, N. Tree vigor, yield, fruit quality, and antioxidant capacity of apple (Malus × domestica Borkh.) influenced by different fertilization regimes: Preliminary results. Turk. J. Agric. For. 2019, 43, 48–57. [Google Scholar] [CrossRef]
- Mittermeier, R.A.; Gil, P.R.; Hoffman, M.; Pilgrim, J.; Brooks, T.; Mittermeier, J.C.; Lamoreux, J.; da Fonseca, G.A.B. Hotspots Revisited: Earth’s Biologically Richest and Most Endangered Terrestrial Ecoregions; Amsterdam University Press: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Gonlugur-Demirci, G. Crustacean fauna of the Turkish Black Sea coasts: A check list. Crustaceana 2006, 79, 1129–1139. [Google Scholar] [CrossRef]
- Sezen, I.; Ercisli, S.; Cakir, O.; Koc, A.; Temim, E.; Hadziabulic, A. Biodiversity and landscape use of sea buckthorn (Hippophae rhamnoides L.) in the Coruh Valley of Turkey. Erwerb-Oobstbau 2015, 57, 23–28. [Google Scholar] [CrossRef]
- Ekim, T. Türkiye’nin bitkileri. In Türkiye’nin Önemli Doga Alanlar; Eken, G., Bozdogan, M., Isfendiyaroglu, S., Kilic, D.T., Lise, Y., Eds.; Doga Dernegi: Ankara, Turkey, 2006; pp. 47–48. (In Turkish) [Google Scholar]
- Atalay, I. The effects of mountainous areas on biodiversity: A case study from the northern Anatolian Mountains and the Taurus Mountains. In Proceedings of the 8th International Symposium on High Mountain Remote Sensing Cartography, La Paz, Bolivia, 21–27 March 2005; Volume 41, pp. 17–26. [Google Scholar]
- Kalkisim, O.; Ozdes, D.; Okcu, Z.; Karabulut, B.; Senturk, H.B. Determination of pomological and morphological characteristics and chemical compositions of local apple varieties grown in Gumushane, Turkey. Erwerbs-Obstbau 2016, 58, 41–48. [Google Scholar] [CrossRef]
- Bevilacqua, A.E.; Califano, A.N. Determination of organic acids in dairy products by high performance liquid chromatography. J. Food Sci. 1989, 54, 1076–1079. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Calorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagent. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Hajnajari, H.; Kohneshine Leily, H.; Bakhshi, D. Selection of promising early ripening progenies and assessment of earliness heritability in the breeding program of apple. Agric. Conspec. Sci. 2019, 84, 245–256. [Google Scholar]
- Balta, M.F.; Kaya, T. Morphological and pomological characteristics of ‘Cebegirmez’ and ‘Bey’ apple varieties. In Proceedings of the V. National Horticulture Congress, Erzurum, Turkey, 4–7 September 2007; pp. 687–691. [Google Scholar]
- Kaya, T.; Balta, F.; Sensoy, S. Fruit quality parameters and molecular analysis of apple germplasm resources from Van Lake Basin, Turkey. Turk. J. Agric. For. 2015, 39, 864–875. [Google Scholar] [CrossRef]
- Ozrenk, K.; Gundogdu, M.; Kaya, T.; Kan, T. Pomological traits of local apples grown in Çatak and Tatvan districts. Yuz. Yil Univ. J. Agric. 2011, 21, 57–63. [Google Scholar]
- Serdar, U.; Ersoy, B.; Ozturk, A.; Demirsoy, H. Local apple cultivars from Camili region. In Proceedings of the V. National Horticulture Congress, Erzurum, Turkey, 4–7 September 2007; pp. 575–579. [Google Scholar]
- Dobrzanski, B.; Rybczynski, R. Colour change of apple as a result of storage, shelf-life, and bruising. Int. Agrophys. 2002, 16, 261–268. [Google Scholar]
- Karlidag, H.; Esitken, A. Some pomological properties of apple and pear cultivars grown in upper Coruh valley. Yuz. Yil Univ. J. Agric. 2006, 16, 93–96. [Google Scholar]
- Malenfant, D. Enquête détaillants: La qualité a sa place. In Proceedings of the Clinique Pomme, Journée Horticoles Régionales, Saint-Rémi, QC, Canada, 7 September 1988; pp. 11–14. [Google Scholar]
- Prange, R.K.; Meheriuk, M.; Lougheed, E.C.; Lidster, P.D. Harvest and storage. In Producing Apples in Eastern and Central Canada; Embree, C.G., Ed.; Agriculture Canada: Kentville, NS, Canada, 1993; pp. 64–69. [Google Scholar]
- Mendoza, F.; Lu, R.; Cen, H. Grading of apples based on firmness and soluble solids content using Vis/SWNIR spectroscopy and spectral scattering techniques. J. Food Eng. 2014, 125, 59–68. [Google Scholar] [CrossRef]
- Gundogdu, M.; Canan, I.; Okatan, V. Bioactive contents and some horticultural characteristics of local apple genotypes from Turkey. J. Anim. Plant Sci. 2018, 28, 865–874. [Google Scholar]
- Mikulic Petkovsek, M.M.; Stampar, F.; Veberic, R. Parameters of inner quality of the apple scab resistant and susceptible apple cultivars (Malus domestica Borkh.). Sci. Hortic. 2007, 114, 37–44. [Google Scholar] [CrossRef]
- Wu, J.; Gao, H.; Zhao, L.; Liao, X.; Chen, F.; Wang, Z.; Hu, X. Chemical compositional characterization of some apple cultivars. Food Chem. 2007, 103, 88–93. [Google Scholar] [CrossRef]
- Ma, B.; Yuan, Y.; Gao, M.; Li, C.; Ogutu, C.; Li, M.; Ma, F. Determination of predominant organic acid components in Malus species: Correlation with apple domestication. Metabolites 2018, 8, 74. [Google Scholar] [CrossRef] [Green Version]
- Borsani, J.; Budde, C.O.; Porrini, L.; Lauxmann, M.A.; Lombardo, V.A.; Murray, R.; Andreo, C.S.; Drincovich, M.F.; Lara, M.V. Carbon metabolism of peach fruit after harvest: Changes in enzymes involved in organic acid and sugar level modifications. J. Exp. Bot. 2009, 60, 1823–1837. [Google Scholar] [CrossRef] [Green Version]
- Chen, F.X.; Liu, X.H.; Chen, L.S. Developmental changes in pulp organic acid concentration and activities of acid-metabolising enzymes during the fruit development of two loquat (Eriobotrya japonica Lindl.) cultivars differing in fruit acidity. Food Chem. 2009, 114, 657–664. [Google Scholar] [CrossRef]
- Balta, F.; Uca, O. Some morphological and pomological characteristics of local summer apples grown in Igdir region in Turkey. Yuz. Yil Univ. J. Agric. 1996, 6, 87–95. [Google Scholar]
- Karadeniz, T.; Gokalp, G.; Kabay, T. Morphological and pomological characterization of local apples from Ulus and Maden districts in Turkey. Yuz. Yil Univ. J. Agric. 1996, 6, 115–125. [Google Scholar]
- Corumlu, M.S. Some Phonological and Pomological Characteristics of Local Apple Cultivars from Iskilip Districts in Corum Province in Turkey. Master’s Thesis, Ordu University, Ordu, Turkey, 2010; p. 92. [Google Scholar]
- Polat, A.A.; Caliskan, O. Performances of some apple cultivars in Kirikhan conditions. In Proceedings of the V. National Horticulture Congress, Erzurum, Turkey, 4–7 September 2007; pp. 638–641. [Google Scholar]
- Cripps, S.; Richards, L.A. ‘Pink Lady’ Apple. HortScience 1993, 28, 1057. [Google Scholar] [CrossRef] [Green Version]
- Kaya, T.; Balta, F. Apple selection in Van province. J. Agric. Sci. 2013, 2, 91–98. [Google Scholar]
- Daillant-Spinnler, B.; MacFie, H.J.H.; Beyts, P.K.; Hedderley, D. Relationships between perceived sensory properties and major preference directions of 12 varieties of apples from the southern hemisphere. Food Qual. Prefer. 1996, 7, 113–126. [Google Scholar] [CrossRef]
- Nour, V.; Trandafir, I.; Ionica, M.E. Compositional characteristics of fruits of several apple (Malus domestica Borkh.) cultivars. Not. Bot. Hort. Agrobot. Cluj-Napoca 2010, 38, 228–233. [Google Scholar]
- Markowskı, J.; Baron, A.; Mıeszczakowska, M.; Płocharskı1, W. Chemical composition of French and Polish cloudy apple juices. J. Hortic. Sci. 2009, 84, 68–74. [Google Scholar] [CrossRef]
- Abacı, Z.T.; Sevindik, E. Ardahan bölgesinde yetiştirilen elma çeşitlerinin biyoaktif bileşiklerinin ve toplam antioksidan kapasitesinin belirlenmesi. Yuz. Yil Univ. Tar. Bil. Derg. 2014, 24, 175–184. [Google Scholar]
- Lee, K.W.; Kim, Y.J.; Kim, D.; Lee, H.J.; Lee, C.J. Major phenolics in apple and their contribution to the total antioxidant capacity. J. Agric. Food Chem. 2003, 51, 6516–6520. [Google Scholar] [CrossRef]
- Loncacic, A.; Pilizota, V. Effect of variety, growing season and storage on polyphenol profile and antioxidant activity of apple peels. Sci. J. Nutr. Diet 2014, 3, 96–105. [Google Scholar]
- Vrhovsek, U.; Rigo, A.; Tonon, D.; Mattivi, F. Quantitation of polyphenols in different apple varieties. J. Agric. Food Chem. 2004, 52, 6532–6538. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, K.L.; Liu, R.H. Apple peels as a value-added food ingredient. J. Agric. Food Chem. 2003, 51, 1676–1683. [Google Scholar] [CrossRef] [PubMed]
- Kschonsek, J.; Wolfram, T.; Stöckl, A.; Böhm, V. Polyphenolic compounds analysis of old and new apple cultivars and contribution of polyphenolic profile to the in vitro antioxidant capacity. Antioxidants 2018, 7, 20. [Google Scholar] [CrossRef] [Green Version]
- Guyot, S.; Marnet, N.; Djamel, L.; Sanoner, P.; Drilleau, J.F. Reversed-phase HPLC following thiolysis for quantitative estimation and characterization of the four main classes of phenolic compounds in different tissue zones of a French cider apple variety (Malus domestica Var. Kermerrien). J. Agric. Food Chem. 1988, 46, 1698–1705. [Google Scholar] [CrossRef]
- Tsao, R.; Yang, R.; Young, J.C.; Zhu, H. Polyphenolic profiles in eight apple cultivars using High-Performance Liquid Chromatography (HPLC). J. Agric. Food. Chem. 2003, 51, 6347–6353. [Google Scholar] [CrossRef] [PubMed]
- Kevers, C.; Pincemail, J.; Tabart, J.; Defraigne, J.O.; Dommes, J. Influence of cultivar, harvest time, storage conditions, and peeling on the antioxidant capacity and phenolic and ascorbic acid contents of apples and pears. J. Agric. Food Chem. 2011, 59, 6165–6171. [Google Scholar] [CrossRef]
- Serce, S.; Ozgen, M.; Torun, A.A.; Ercisli, S. Chemical composition, antioxidant activities and total phenolic content of Arbutus andrachne L. (Fam. Ericaceae) (the Greek strawberry tree) fruits from Turkey. J. Food Compost. Anal. 2010, 23, 619–623. [Google Scholar] [CrossRef]
- Dogan, H.; Ercisli, S.; Temim, E.; Hadziabulic, A.; Tosun, M.; Yilmaz, S.O.; Zia-Ul-Haq, M. Diversity of chemical content and biological activity in flower buds of a wide number of wild grown caper (Capparis ovate Desf.) genotypes from Turkey. C. R. Acad. Bulg. Sci. 2014, 67, 1593–1600. [Google Scholar]
- Gundogdu, M.; Ozrenk, K.; Ercisli, S.; Kan, T.; Kodad, O.; Hegedus, A. Organic acids, sugars, vitamin C content and some pomological characteristics of eleven hawthorn species (Crataegus spp.) from Turkey. Biol. Res. 2014, 47, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozkan, G. Phenolic compounds, organic acids, vitamin C and antioxidant capacity in Prunus spinose. C. R. Acad. Bulg. Sci. 2019, 72, 267–273. [Google Scholar]
- Engin, S.P.; Mert, C. The effects of harvesting time on the physicochemical components of aronia berry. Turk. J. Agric. For. 2020, 44, 361–370. [Google Scholar] [CrossRef]
- Gecer, M.K.; Ozkan, G.; Sagbas, H.I.; Ilhan, G.; Gundogdu, M.; Ercisli, S. Some important horticultural properties of summer apple genotypes from Coruh valley in Turkey. Int. J. Fruit Sci. 2020, 20, S1406–S1416. [Google Scholar] [CrossRef]
- Karatas, N.; Sengul, M. Some important physicochemical and bioactive characteristics of the main apricot cultivars from Turkey. Turk. J. Agric. For. 2020, 44, 651–661. [Google Scholar] [CrossRef]
- Ozkan, G.; Ercisli, S.; Sagbas, H.I.; Ilhan, G. Diversity on fruits of wild grown European cranberrybush from Coruh valley in Turkey. Erwerbs-Obstbau 2020, 62, 275–279. [Google Scholar] [CrossRef]
- Bujdosó, G.; Cseke, K. The Persian (English) walnut (Juglans regia L.) assortment of Hungary: Nut characteristics and origin. Sci. Hortic. 2021, 283, 110035. [Google Scholar] [CrossRef]
- Bujdosó, G.; Illés, B.; Varjas, V.; Cseke, K. Is “Esterhazy II”, an old walnut variety in the Hungarian gene bank, the original genotype? Plants 2021, 10, 854. [Google Scholar] [CrossRef] [PubMed]
- Rampáčková, E.; Göttingerová, M.; Gála, P.; Kiss, T.; Ercişli, S.; Nečas, T. Evaluation of Protein and Antioxidant Content in Apricot Kernels as a Sustainable Additional Source of Nutrition. Sustainability 2021, 13, 4742. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).