Current Knowledge of Medicinal Mushrooms Related to Anti-Oxidant Properties
Abstract
:1. Introduction
2. Material and Methods
3. Review
4. Current Knowledge
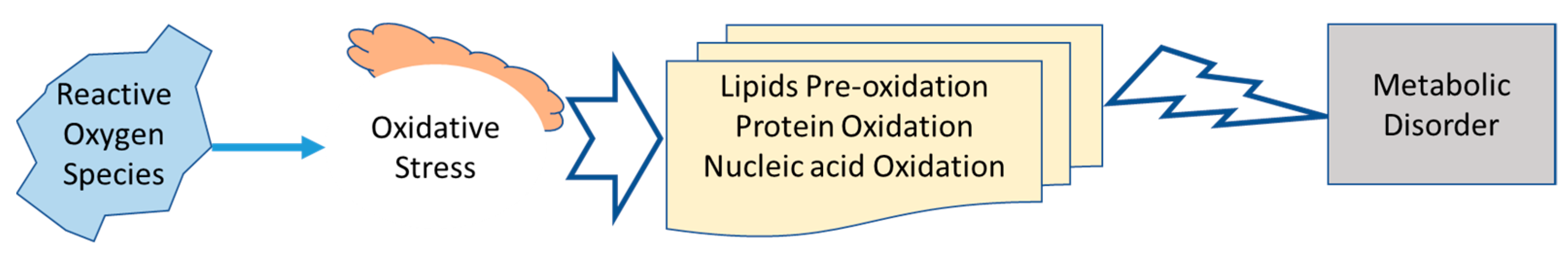
5. Antioxidant Potential
6. Metabolic Disorder
7. Results
8. Conclusions
9. Recommendation
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviation
| CCP | Cantharellus cibarius polysaccharide. |
| CHO | Cholesterol. |
| DLA | Dalton′s Lymphoma Ascites. |
| DPPH | 2: 2-diphenyl-1-picrylhydrazyl. |
| EAC | Ehrlich-Letter ascites carcinoma. |
| FRAP | Fluorescence recovery after photobleaching |
| FTIR | Fourier transform infrared spectroscopy. |
| NAFLD | Nonalcoholic fatty liver disorder. |
| TAG | Triglyceride levels. |
| GEE | Ganoderma lucidum ethanol extract |
| HMG-COA | β-Hydroxy β-methylglutaryl-CoA |
References
- Wasser, S.P. Current findings, future trends, and unsolved problems in studies of medicinal mushrooms. Appl. Microbiol. Biotechnol. 2011, 89, 1323–1332. [Google Scholar] [CrossRef]
- Dai, Y.-C.; Yang, Z.-L.; Cui, B.; Yu, C.-J.; Zhou, L.-W. Species Diversity and Utilization of Medicinal Mushrooms and Fungi in China. Int. J. Med. Mushrooms 2009, 11, 287–302. [Google Scholar] [CrossRef]
- Hobbs, C. Medicinal Mushrooms: An Exploration of Tradition, Healing, and Culture; Botanica Press: Summertown, TN, USA, 2002. [Google Scholar]
- Yang, Q.; Jong, C. Medicinal mushrooms in China. Mushroom Sci. 1989, 1, 631–643. [Google Scholar]
- Park, W.; Lee, H. Illustrated Book of Korean Medicinal Mushrooms; Kyo-Hak Publishing, Co.: Seoul, Korea, 1999. [Google Scholar]
- Lindequist, U.; Niedermeyer, T.; Jülich, W.-D. The Pharmacological Potential of Mushrooms. Evid. Based Complement. Altern. Med. 2005, 2, 285–299. [Google Scholar] [CrossRef] [Green Version]
- Ikekawa, T. Beneficial Effects of Edible and Medicinal Mushrooms on Health Care. Int. J. Med. Mushrooms 2001, 3, 3. [Google Scholar]
- Lee, P.C.; Bochner, B.R.; Ames, B.N. AppppA, heat-shock stress, and cell oxidation. Proc. Natl. Acad. Sci. USA 1983, 80, 7496–7500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pullen, J.; Saeed, K. An overview of biodiesel oxidation stability. Renew. Sustain. Energy Rev. 2012, 16, 5924–5950. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M. Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Moskovitz, J.; Bin Yim, M.; Chock, P. Free Radicals and Disease. Arch. Biochem. Biophys. 2002, 397, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free Radicals, Antioxidants in Disease and Health. Int. J. Biomed. Sci. IJBS 2008, 4, 89–96. [Google Scholar] [PubMed]
- Hogg, N. Free Radicals in Disease. Semin. Reprod. Med. 1998, 16, 241–248. [Google Scholar] [CrossRef]
- Liu, F.; Ooi, V.; Chang, S. Free radical scavenging activities of mushroom polysaccharide extracts. Life Sci. 1997, 60, 763–771. [Google Scholar] [CrossRef]
- Mau, J.-L.; Chao, G.-R.; Wu, K.-T. Antioxidant properties of methanolic extracts from several ear mushrooms. J. Agric. Food Chem. 2001, 49, 5461–5467. [Google Scholar] [CrossRef] [PubMed]
- Dinesh, R. The role of antioxidants and ROS scavenging machinery in wild mushrooms. In New and Future Developments in Microbial Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2021; pp. 245–251. [Google Scholar]
- Bamigboye, C.O.; Omomowo, I.O.; Alao, M.B.; Elegbede, J.A.; Adegoke, E. Free radical scavenging ability, mechanisms of action and health implications of oyster mushrooms (Pleurotus species): Pleurotus species (Oyster mushrooms): Free radical scavenging capacity. J. Microbiol. Biotechnol. Food Sci. 2021, 10, 636–647. [Google Scholar]
- Rao, L.G.; Kang, N.; Rao, A.V. Polyphenol antioxidants and bone health: A review. In Phytochemicals—A Global Perspective of Their Role in Nutrition and Health; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- Rizzo, G.; Goggi, S.; Giampieri, F.; Baroni, L. A review of mushrooms in human nutrition and health. Trends Food Sci. Technol. 2021. [Google Scholar] [CrossRef]
- Patocka, J.; Wu, R.; Nepovimova, E.; Valis, M.; Wu, W.; Kuca, K. Chemistry and Toxicology of Major Bioactive Substances in Inocybe Mushrooms. Int. J. Mol. Sci. 2021, 22, 2218. [Google Scholar] [CrossRef] [PubMed]
- Hetland, G.; Johnson, E.; Bernardshaw, S.V.; Grinde, B. Can medicinal mushrooms have prophylactic or therapeutic effect against COVID-19 and its pneumonic superinfection and complicating inflammation? Scand. J. Immunol. 2021, 93, e12937. [Google Scholar] [CrossRef]
- Uthan, E.T.; Senturk, H.; Uyanoglu, M.; Yamaç, M. First Report on the In Vivo Prebiotic, Biochemical, and Histological Effects of Crude Polysaccharide Fraction of Golden Chantharelle Mushroom, Cantharellus cibarius (Agaricomycetes). Int. J. Med. Mushrooms 2021, 23, 67–77. [Google Scholar] [CrossRef]
- Kozarski, M.; Klaus, A.; Niksic, M.; Jakovljević, D.; Helsper, J.P.; Van Griensven, L.J. Antioxidative and immunomodulating activities of polysaccharide extracts of the medicinal mushrooms Agaricus bisporus, Agaricus brasiliensis, Ganoderma lucidum and Phellinus linteus. Food Chem. 2011, 129, 1667–1675. [Google Scholar] [CrossRef]
- Badalyan, S.M. Edible and medicinal higher basidiomycetes mushrooms as a source of natural antioxidants. Int. J. Med. Mushrooms 2003, 5, 153–162. [Google Scholar] [CrossRef]
- Mau, J.-L.; Lin, H.-C.; Chen, C.-C. Antioxidant Properties of Several Medicinal Mushrooms. J. Agric. Food Chem. 2002, 50, 6072–6077. [Google Scholar] [CrossRef]
- Kim, M.-Y.; Seguin, P.; Ahn, J.-K.; Kim, J.-J.; Chun, S.-C.; Kim, E.-H.; Seo, S.-H.; Kang, E.-Y.; Kim, S.-L.; Park, Y.-J.; et al. Phenolic compound concentration and antioxidant activities of edible and medicinal mushrooms from Korea. J. Agric. Food Chem. 2008, 56, 7265–7270. [Google Scholar] [CrossRef]
- Yang, J.-H.; Lin, H.-C.; Mau, J.-L. Antioxidant properties of several commercial mushrooms. Food Chem. 2002, 77, 229–235. [Google Scholar] [CrossRef]
- Sulkowska-Ziaja, K.; Muszynska, B.; Motyl, P.; Pasko, P.; Ekiert, H. Phenolic compounds and antioxidant activity in some species of polyporoid mushrooms from Poland. Int. J. Med. Mushrooms 2012, 14, 385–393. [Google Scholar] [CrossRef]
- Turkoglu, A.; Duru, M.E.; Mercan, N.; Kivrak, I.; Gezer, K. Antioxidant and antimicrobial activities of Laetiporus sulphureus (Bull.) Murrill. Food Chem. 2007, 101, 267–273. [Google Scholar] [CrossRef]
- Aziz, T.; Mehmet, E.D.; Nazime, M.; Ibrahim, K.; Kudret, G. Antimicrobial and antioxidant activities of mycelia of 10 wild mushroom species. J. Med. Food 2010, 13, 415–419. [Google Scholar]
- Lee, I.-K.; Kim, Y.-S.; Jang, Y.-W.; Jung, J.-Y.; Yun, B.-S. New antioxidant polyphenols from the medicinal mushroom Inonotus obliquus. Bioorg. Med. Chem. Lett. 2007, 17, 6678–6681. [Google Scholar] [CrossRef]
- Song, X.; Cai, W.; Ren, Z.; Jia, L.; Zhang, J. Antioxidant and Hepatoprotective Effects of Acidic-Hydrolysis Residue Polysaccharides from Shiitake Culinary-Medicinal Mushroom Lentinus edodes (Agaricomycetes) in Mice. Int. J. Med. Mushrooms 2021, 23, 85–96. [Google Scholar] [CrossRef]
- Ebrahimzadeh, M.A.; Nabavi, S.M.; Nabavi, S.F.; Eslami, S. Antioxidant and Free Radical Scavenging Activities of Culinary-Medicinal Mushrooms, Golden Chanterelle Cantharellus cibarius and Angel’s Wings Pleurotus porrigens. Int. J. Med. Mushrooms 2010, 12, 265–272. [Google Scholar] [CrossRef]
- Wei, S.; Van Griensven, L.J.L.D. Pro- and Antioxidative Properties of Medicinal Mushroom Extracts. Int. J. Med. Mushrooms 2008, 10, 315–324. [Google Scholar] [CrossRef]
- Woldegiorgis, A.Z.; Abate, D.; Haki, G.D.; Ziegler, G. Antioxidant property of edible mushrooms collected from Ethiopia. Food Chem. 2014, 157, 30–36. [Google Scholar] [CrossRef]
- Liang, C.-H.; Ho, K.-J.; Huang, L.-Y.; Tsai, C.-H.; Lin, S.-Y.; Mau, J.-L. Antioxidant properties of fruiting bodies, mycelia, and fermented products of the culinary-medicinal king oyster mushroom, Pleurotus eryngii (higher Basidiomycetes), with high ergothioneine content. Int. J. Med. Mushrooms 2013, 15, 267–275. [Google Scholar] [CrossRef]
- Khatua, S.; Paul, S. Acharya, K. Mushroom as the potential source of new generation of antioxidant: A review. Res. J. Pharm. Technol. 2013, 6, 3. [Google Scholar]
- Mohsin, M.; Negi, P.S.; Ahmed, Z. Determination of the antioxidant activity and polyphenol contents of wild Lingzhi or Reishi medicinal mushroom, Ganoderma lucidum (W. Curt. Fr.) P. Karst. (higher Basidiomycetes) from central Himalayan hills of India. Int. J. Med. Mushrooms 2011, 13, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Mathew, J.; Sudheesh, N.P.; Rony, K.A.; Smina, T.P.; Janardhanan, K.K. Antioxidant and antitumor activities of cultured mycelium of culinary-medicinal paddy straw mushroom Volvariella volvacea (Bull.: Fr.) singer (agaricomycetideae). Int. J. Med. Mushrooms 2008, 10, 139–147. [Google Scholar] [CrossRef]
- Nhi, N.; Hung, P. Nutritional composition and antioxidant capacity of several edible mushrooms grown in the Southern Vietnam. Int. Food Res. J. 2012, 19, 611. [Google Scholar]
- Tseng, Y.-H.; Yang, J.-H.; Mau, J.-L. Antioxidant properties of polysaccharides from Ganoderma tsugae. Food Chem. 2008, 107, 732–738. [Google Scholar] [CrossRef]
- Turkoglu, A.; Kivrak, I.; Mercan, N.; Duru, M.E.; Gezer, K.; Turkoglu, H. Antioxidant and antimicrobial activities of Morchella conica Pers. Afr. J. Biotechnol. 2006, 5, 1146–1150. [Google Scholar]
- Mau, J.-L.; Tsai, S.-Y.; Tseng, Y.-H.; Huang, S.-J. Antioxidant properties of methanolic extracts from Ganoderma tsugae. Food Chem. 2005, 93, 641–649. [Google Scholar] [CrossRef]
- Mau, J.-L.; Tsai, S.-Y.; Tseng, Y.-H.; Huang, S.-J. Antioxidant properties of hot water extracts from Ganoderma tsugae Murrill. LWT 2005, 38, 589–597. [Google Scholar] [CrossRef]
- Barros, L.; Baptista, P.; Estevinho, L.M.; Ferreira, I.C.F.R. Bioactive properties of the medicinal mushroom Leucopaxillus giganteus mycelium obtained in the presence of different nitrogen sources. Food Chem. 2007, 105, 179–186. [Google Scholar] [CrossRef]
- Tsai, S.-Y.; Huang, S.-J.; Lo, S.-H.; Wu, T.-P.; Lian, P.-Y.; Mau, J.-L. Flavour components and antioxidant properties of several cultivated mushrooms. Food Chem. 2009, 113, 578–584. [Google Scholar] [CrossRef]
- Chen, P.; Yong, Y.; Gu, Y.; Wang, Z.; Zhang, S.; Lu, L. Comparison of antioxidant and antiproliferation activities of polysaccharides from eight species of medicinal mushrooms. Int. J. Med. Mushrooms 2015, 17, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.-H.; Xiao, D.-M.; Chen, D.-X.; Xiao, Y.; Liang, Z.-Q.; Zhong, J.-J. Polysaccharides from the Medicinal MushroomCordyceps taiiShow Antioxidant and Immunoenhancing Activities in aD-Galactose-Induced Aging Mouse Model. Evid. Based Complement. Altern. Med. 2012, 2012, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Wong, J.Y.; Chye, F.Y. Antioxidant properties of selected tropical wild edible mushrooms. J. Food Compos. Anal. 2009, 22, 269–277. [Google Scholar] [CrossRef]
- Ferreira, I.C.; Barros, L.; Abreu, R. Antioxidants in wild mushrooms. Curr. Med. Chem. 2009, 16, 1543–1560. [Google Scholar] [CrossRef] [Green Version]
- Elisashvili, V. Submerged cultivation of medicinal mushrooms: Bioprocesses and products. Int. J. Med. Mushrooms 2012, 14, 211–239. [Google Scholar] [CrossRef] [PubMed]
- Sujata, W.; Devi, S.N.; Mandal, S.C. Phytochemicals and Investigations on Traditionally Used Medicinal Mushrooms. In Evidence Based Validation of Traditional Medicines; Springer: Berlin/Heidelberg, Germany, 2021; pp. 965–984. [Google Scholar]
- Jain, A.; Ganeshpurkar, A.; Rai, G. Medicinal mushrooms: Towards a new horizon. Pharmacogn. Rev. 2010, 4, 127–135. [Google Scholar] [CrossRef] [Green Version]
- Ajith, T.A.; Janardhanan, K.K. Indian medicinal mushrooms as a source of antioxidant and antitumor agents. J. Clin. Biochem. Nutr. 2007, 40, 157–162. [Google Scholar] [CrossRef] [Green Version]
- De Silva, D.D.; Rapior, S.; Hyde, K.D.; Bahkali, A. Medicinal mushrooms in prevention and control of diabetes mellitus. Fungal Divers. 2012, 56, 1–29. [Google Scholar] [CrossRef]
- Adeyi, A.O.; Awosanya, S.A.; Adeyi, O.E.; James, A.S.; Adenipekun, C.O. Ganoderma lucidum ethanol extract abrogates metabolic syndrome in rats: In vivo evaluation of hypoglycemic, hypolipidemic, hypotensive and antioxidant properties. Obes. Med. 2021, 22, 100320. [Google Scholar] [CrossRef]
- Lotfy, T.M.R.; El-Hendy, H.A.R.; Shawir, S.M.S. Effect of Mushroom, Oat and Their Mixtures as Functional Foods on Antioxidant Activities, Hematological Parameters of Obese Rats and its Sensory Evaluation. Alex. Sci. Exch. J. 2021, 42, 21–36. [Google Scholar]
- Al-Faqeeh, L.A.S.; Naser, R.; Kagne, S.R.; Khan, S.W. Activity of mushrooms against diabetic and inflammation: A review. GSC Biol. Pharm. Sci. 2021, 14, 37–44. [Google Scholar] [CrossRef]
- Martinez-Medina, G.A.; Chávez-González, M.L.; Verma, D.K.; Prado-Barragán, L.A.; Martínez-Hernández, J.L.; Flores-Gallegos, A.C.; Thakur, M.; Srivastav, P.P.; Aguilar, C.N. Bio-funcional components in mushrooms, a health opportunity: Ergothionine and huitlacohe as recent trends. J. Funct. Foods 2021, 77, 104326. [Google Scholar] [CrossRef]
- Rani, A.; Saini, K.; Bast, F.; Mehariya, S.; Bhatia, S.; Lavecchia, R.; Zuorro, A. Microorganisms: A Potential Source of Bioactive Molecules for Antioxidant Applications. Molecules 2021, 26, 1142. [Google Scholar] [CrossRef]
- Wachtel-Galor, S.; Tomlinson, B.; Benzie, I.F.F. Ganoderma lucidum (‘Lingzhi’), a Chinese medicinal mushroom: Biomarker responses in a controlled human supplementation study. Br. J. Nutr. 2004, 91, 263–269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lo, H.-C. Bioactive components and possible activities of medicinal mushrooms in alleviating the pathogenesis of nonalcoholic fatty liver disease. Int. J. Med. Mushrooms 2021, 23, 29–41. [Google Scholar] [CrossRef]
- Lu, J.; He, R.; Sun, P.; Zhang, F.; Linhardt, R.J.; Zhang, A. Molecular mechanisms of bioactive polysaccharides from Ganoderma lucidum (Lingzhi), a review. Int. J. Biol. Macromol. 2020, 150, 765–774. [Google Scholar] [CrossRef]
- Rahman, M.A.; Al Masud, A.; Lira, N.Y.; Shakil, S. Proximate analysis, phtochemical screening and antioxidant activity of different strains of Ganoderma lucidum (Reishi mushroom). Open J. Biol. Sci. 2020, 5, 24–27. [Google Scholar]
- Ryu, D.H.; Cho, J.Y.; Bin Sadiq, N.; Kim, J.-C.; Lee, B.; Hamayun, M.; Lee, T.S.; Kim, H.S.; Park, S.H.; Nho, C.W.; et al. Optimization of antioxidant, anti-diabetic, and anti-inflammatory activities and ganoderic acid content of differentially dried Ganoderma lucidum using response surface methodology. Food Chem. 2021, 335, 127645. [Google Scholar] [CrossRef]
- Al-Faqeeh, L.A.S.; Naser, R.; Kagne, S.R.; Khan, S.W. Nutritional values, ethno-medicinal uses and antioxidant activity of mushrooms: A review. Eur. J. Biomed. 2021, 8, 292–300. [Google Scholar]
- Sun, J.; He, H.; Xie, B.J. Novel antioxidant peptides from fermented mushroom Ganoderma lucidum. J. Agric. Food Chem. 2004, 52, 6646–6652. [Google Scholar] [CrossRef] [PubMed]
- Radhika, R.; Sundar, S.K.; Anuradha, R.; Radhika, R.; Sundar, S.K.; Anuradha, R. Antioxidant Property of Fruiting Bodies of Ganoderma Lucidum. Int. J. Mod. Agric. 2021, 10, 3508–3514. [Google Scholar]
- Bhat, Z.A.; Wani, A.H.; War, J.M.; Bhat, M.Y. Major bioactive properties of ganoderma polysaccharides: A review. Asian J. Pharm. Clin. Res. 2021, 14, 11–24. [Google Scholar] [CrossRef]
| Model | Mushroom | Effect | Active Constituents | Mechanism | Mushroom Product | Year, Country, Reference |
|---|---|---|---|---|---|---|
| In vivo | Agaricus blazei Murill, Hericium erinaceus, Grifola frondosa | Immuno modulatory, Prophylactic for covid 19 | Polysaccharides | Protect against infection and decrease proinflmatory cytokines | Ab-M water extract | 2020, Norway, [21] |
| In vivo | Cantharellus cibarius polysaccharide | Raises prebiotic index | Polysaccharides | Raises the lactobacillus and decreases the clostridium count | Polysaccharide extract | 2020, Turkey, [22] |
| In vitro | Ganoderma tsugae, Ganoderma lucidum, Coriolus versicolor | Antioxidant, Immuno stimulant | Triterpenoids, Proteins, Polysaccharides | Scavenges free radicals, cytokine expression via TLR4 signaling pathway | Methanolic extract | 2002, China, [25] |
| In vitro | Pleurotus ostreatus, Agaricus bisporus, Flammulina velutipes, Pleurotus eryngii, Lentinus edodes | Antioxidant | Galactomannan, Linoleic, Palmitic, Stearic acids, Polysaccharides, P-coumaric acid, Ellagic acid, Chlorogenic acid, Caffeic acid, Ferulic acid, Qercetin, Sterols | Fight against free-electron species | Dried freeze, stock solution with DSMO | 2008, Korea, [26] |
| In vitro & In vivo | Pleurotus pulmonarius, Phellinus rimosus, Ganoderma lucidum, Pleurotus florida | Antimutagenic, Antioxidant | Amino acids, Polysaccharides, Phenol, Flavonoids, Enzymes, Steroids, Nucleotides, Mineral, Vitamins | Inhibit DLA and EAC cell lines, Fight against free radicals, Improve mitochondrial energy status | Ethyl acetate, Hot water, Aqueous extract, Methanolic extract | 2006, India, [54] |
| In vitro | Ganoderma applanatum, Gnoderma lucidum, Lentinus edodes, Trametes versicolor | Antioxidant, Anti-inflammatory | Polyphenols, Alpha and Beta Glucans, Uronic acid | Increase pro-inflamatory cytokines production, Inhibit lipid peroxidation | Polysaccharide extract | 2012, Netherlands, [23] |
| In vivo Nicotinamide and streptozocin-induced | Agaricus bisporus, Agaricus campestris, Auricularia auricula-judae, Coprinus comatus, Cordyceps spp., Cordycepsmilitaris, Cordyceps sinensis, Ganoderma lucidum, Sensu lato, Grifola frondosa, Inonotus obliquus, Lentinula edodes, Phellinus spp., Tremella fuciformis, Wolfiporia extensa, Hericium erinaceus | Antidiabetic | Protein, Fats, Fibers, Sterol, Meroterpenoid, Vitamins, Minerals | Maintain blood glucose level, Activate insulin | Crude extract | 2012, Thailand, [55] |
| In vitro | Lentinula edodes, Pleurotus ostreatus | Antioxidant, Chelating power, Reducing power | Vitamin B complex, Polysaccharides, Fibers. | Inhibit 1, 3-diethyl-2-thiobarbituric acid | Methanolic extract | 2002, Brazil, [27] |
| In vitro | Inonotus obliquus | Antioxidant | Triterpenes | Activity against DPPH, ABTS | Methanolic extract | 2007, Japan, [31] |
| In vivo and In vitro | Poria cocos wolf, Gomphidius rutilus, Frondosa | Antihypertriglyceridemic, Antihyperglycemic, Antiinflammatory, Antioxidant | Triterpene acids, Polyacetylene, Glucoside, Phenolic compounds | Lipolysis, Gut microbiota modulation, Fight against reactive oxygen species | Dried fruit | 2021, Taiwan, [62] |
| In vivo | Ganoderma lucidum | Antioxidant | Polysaccharides, Ergosterol | Reduces blood glucose, cholesterol and triglyceride level | Ethanolic extract | 2021, Nigeria, [56] |
| In vitro, Case study | Auricular, Fomes fomentarius, Ganoderma lucidum, Coprinus comatus, Hericium erinaceus, Lentinula edodes, Monascus purpureus, Piptoporus betulinu, Trametes versicolor, Poria cocos | Hepatoprotective, Antiinflammatory, Lignin biosynthesis, Antihypertensive, Antioxidant | Cholorogenic acid, Ergosterol, Lentinan, Lovastatin, Ergothioneine, Monacolins, Calcium, Potassium, Fibers, Polysaccharides | Lower cholesterol by inhibiting HMG-coA reductase, the rate-limiting step for cholesterol synthesis in the liver; reduce oxidation stress | Dried form | 2021, India, [52] |
| In vivo | Lentinus edodes | Hepatoprotective, Antioxidant | Stereoisomers, Linoleic acid | Mitigate LDL oxidation | Acid hydrolysis | 2021, China, [32] |
| In vivo | Pleurotus ostreatus, Avena sativa | Metabolic syndrome amelioration | Vitamins, Minerals, Polysaccharides | Reduce weight and lowering blood glucose level | Dried form | 2021, Egypt, [57] |
| In vivo | Agaricus sylvaticus, Pleurotus pulmonarius, Astraeus hygometricus | Antihyperglycemic | Polysaccharides, Minerals | Contains glyceraldehyde-3-phosphate dehydrogenase-like protein which lowers glucose level | Ethanolic extract, Methanolic extract, Aqueous extract | 2021, India, [58] |
| In vivo | Tylopilus ballouii, Pleurotus sajor-caju, Ganoderma lucidum | Anti-inflammatory | Cordycepin, Adenosine | Reduce inflammatory cytokines | Chloroform, Ethanolic, Methanolic, Aqueous extract | 2021, India, [58] |
| In vitro | Albatrellus confluens, Lactarius deliciosus, Auricularia judae | Immuno modulator, Boost up immunity, Antioxidant, | Scutigera, Carbohydrate, Gallic acid | Inhibit oxaliplatin development in ganglion, Reduce oxidation stress | Dried form | 2021, Mexico, [59] |
| In vitro | Boletus edulis, P. salmoneostramineus, Aspergillus brasiliensis | Antioxidant, Antibacterial, Anti-inflammatory | Hispidin, Illudane, Sesquiterpenoids | Inhibit cellular damage | Methanolic extract | 2021, India, [60] |
| In vitro | Wild mushrooms | Antioxidant | Ascorbic acid, Tocopherol, Polyphenol | Inhibit oxidative stress | Methanolic extract | 2009, Portugal, [50] |
| In vivo | Lepista personata, Trametes versicolor, Pleurotus ostreatus, C. micaceus, Hypholoma fasciculare, Pholiota alnicola, Marasmius oreades, Coprinus comatus, Schizophyllum commune, Lentinus edodes, Suillus luteus, Volvariella bombycine | Antioxidant | Phenolic compounds (Catechin, Ferulic, Gallic acid, Vanillic acid) | Presence of phenolic components balances and reduces oxidation stress | Methanolic extract | 2003, Armenia, [24] |
| In vivo | Hypsizygus | Anticarcinogenic | EA6 Polysaccharide | Inhibition against tumor production | Aqueous extract | 2001, Japan, [7] |
| In vitro | Auricularia auricular | Antioxidant | Retinol, Ascorbic acid, Polyphenolic bioactive components | Inhibit oxidation | Methanolic extract | 2001, Taiwan, [15] |
| In vitro | Laetiporus sulphureus | Antioxidant, Antimicrobial | Bioactive phenolic, Flavonoids components | Inhibits growth of bacteria and scavenges free radicals | Ethanolic extract | 2007, Turkey, [29] |
| In vitro | Cantharellus cibarius, Pleurotus porrigens | Antioxidant | Polysaccharides | Fight against free radicals | Methanolic and ethyl acetate | 2010, Iran, [33] |
| In vitro | Agaricus campestris, Antrodia, Auricularia american, Coprinus comatus, Cordyceps subgen, Hericium clathroides, Grifola frondosa, Ganoderma lucidum, Lentinus fuscus, Phellinus rickii, Trametes gibbosa | Antioxidant | Polyphenolic Components, Polysaccharides | Fight against free radicals | Hot water extract | 2008, Netherlands, [34] |
| Submerged cultivation | Basidiomycetes | Metabolic diversity | Steroids, Alkaloids, Nucleotide, Polyphenol, Polysaccharide, Cytokines | Na | Na | 2012, Georgia, [51] |
| Intervention study | Ganoderma lucidum | Immuno modulator, Antioxidant, Lipid lowering | Polysaccharides, Triterpenoids, Proteins, Enzymes, Steroids, Sterols, Nucleotides, Minerals, Enzymes | Associated with cholesterol lowering capacity | Fresh mushroom | 2004, Hong Kong, [61] |
| In vitro and In vivo | Cordyceps taii | Antioxidant, Boost up immune system | Cordycepic acid, Glutamic acid, Polyamines, Amino acids, Cyclic dipeptides, Saccharides, Sugar derivatives, Sterols | Polysaccharides increase production of antioxidant enzymes and inhibit lipid per oxidation | Aqueous extract | 2012, China, [48] |
| In vitro | Morchella hortensis, Meripilus giganteus, Armillaria mellea, Morchella costata, Morchella elata, Morchella rotunda, Paxillus involutus, Morchella esculenta var. Vulgaris, Pleurotus eryngii, Pleurotus ostreatus | Antibacterial, Antioxidant | Polyphenols, Flavonoids | Reduce oxidation stress and inhibit growth of bacteria | Ethanolic extract | 2010, Turkey, [30] |
| In vitro | Pleurotus porrigens, Hygrocybe conica | Antioxidant | Polyphenolic components | Fights against free radicals | Methanolic extract | 2009, Malaysia, [49] |
| In vitro | Phellinus rimosus, Lentinula edodes, Ganoderma, Agaricus bisporus | Antioxidant, Immuno modulator, Antimicrobial, Antiprotozoal | Protein, Peptide, Glycoprotein, Polysaccharide, Lentinan, Vitamin C, Vitamin D, Folate, Thiamine | Lentinan causes tumor regression | Ethanolic extract | 2010, India, [53] |
| In vitro | Pleurotus eryngii | Antioxidant | Ergothioneine | Scavenges free radicals | Ethanolic, Hot water extract | 2013, Taiwan, [36] |
| In vitro | P. ostreatus, L. edodes, A. campestris, L. sulphureus, T. clypeatus, T. letestui, T. microcarpus | Antioxidant | Phenolic component, Flavonoids | Fight against free radicals | Methanolic extract | 2014, Ethiopia, [35] |
| In vitro | Daedaleopsis confragosa, Fomitopsis pinicola, Gloeophyllum sepiarium, Laetiporus sulphureus, Piptoporus betulinus | Antioxidant | Protocatechuic, p-Hydroxybenzoic, Vanillic acids | Fight against free radicals | Methanolic extract | 2012, Poland, [28] |
| In vitro | Fistulina hepatica, Pleorotus squarrosulus, Polyporus grammocephalus, Phellinus linteus, Austreus hygrometricus, Macrocybe gigantean | Antioxidant | Tannic, Gallic, Protocatechuic, Gentisic acids | Fight against free radicals | Ethanoli, Methanolic, Ethyl acetate, Hot water extract | 2013, Calcutta, [37] |
| In vitro | Ganoderma lucidum | Antioxidant | Polyphenol components | Shows Antioxidant potential | Methanolic extract | 2011, India, [38] |
| In vitro and In vivo | Volvariella volvacea | Antioxidant, Antitumor | Na | Na | Ethanolic aqueous extract | 2008, India, [39] |
| In vitro | Pleurotus ostreatus, Volvariella volvacea, Lentinula edodes, Auricularia polytricha Ganoderma lucidum | Nutritional value, Antioxidant | Phenolic compounds | Inhibit free radical oxygen species | Powder | 2012, Vietnam, [40] |
| In vitro | Ganoderma lucidum | Antioxidant | G. Lucidum Polysaccharide (glp) | Potential source of antioxidants and anti-tumor | Ethanolic and hot water extract | 2008, China, [41] |
| In vitro | Morchella conica pers | Antioxidant | Phenolic compounds | Fights against oxidation stress | Ethanolic | 2006, Turkey, [42] |
| In vitro | Ganoderma tsugae Murrill | Antioxidant | Ascorbic acid, Beta carotene, Phenolic components | Beneficial for antioxidant system of body | Methanolic extract | 2005, Taiwan, [43] |
| In vitro | Ganoderma tsugae Murrill | Antioxidant | Phenolic components | Fight against oxidation damage | Hot water extract | 2005, Taiwan, [44] |
| In vitro | Leucopaxillus | Antioxidant, Antibacterial | Flavonoids and phenols | Responsible for their antioxidant capacity | Methanolic extract | 2007, Portugal, [45] |
| In vitro | Pleurotus ferulae, Clitocybe maxima, Pleurotus ostreatus gray | Antioxidant | Phenolic components | Scavenger against free radicals | Ethanolic and hot water extract | 2009, Taiwan, [46] |
| In vitro | Ganoderma lucidum, Hypsizygus marmoreus, P. ostreatus, P. nebrodensis, Lentinus edodes, Pleurotus eryngii, Flammulina velutipes, Hericium erinaceus | Antioxidant, Antitumor | Polysaccharides | Correlate with antioxidant potential and anti-proliferation | Methanolic extract | 2015, China, [47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shaffique, S.; Kang, S.-M.; Kim, A.-Y.; Imran, M.; Aaqil Khan, M.; Lee, I.-J. Current Knowledge of Medicinal Mushrooms Related to Anti-Oxidant Properties. Sustainability 2021, 13, 7948. https://doi.org/10.3390/su13147948
Shaffique S, Kang S-M, Kim A-Y, Imran M, Aaqil Khan M, Lee I-J. Current Knowledge of Medicinal Mushrooms Related to Anti-Oxidant Properties. Sustainability. 2021; 13(14):7948. https://doi.org/10.3390/su13147948
Chicago/Turabian StyleShaffique, Shifa, Sang-Mo Kang, Ah-Yeong Kim, Muhammad Imran, Muhammad Aaqil Khan, and In-Jung Lee. 2021. "Current Knowledge of Medicinal Mushrooms Related to Anti-Oxidant Properties" Sustainability 13, no. 14: 7948. https://doi.org/10.3390/su13147948
APA StyleShaffique, S., Kang, S.-M., Kim, A.-Y., Imran, M., Aaqil Khan, M., & Lee, I.-J. (2021). Current Knowledge of Medicinal Mushrooms Related to Anti-Oxidant Properties. Sustainability, 13(14), 7948. https://doi.org/10.3390/su13147948








