Digital Twin of COVID-19 Mass Vaccination Centers
Abstract
:1. Introduction
2. Literature Review
3. Problem Description
4. Materials and Methods
4.1. Simulation Model
4.2. Digital Technology
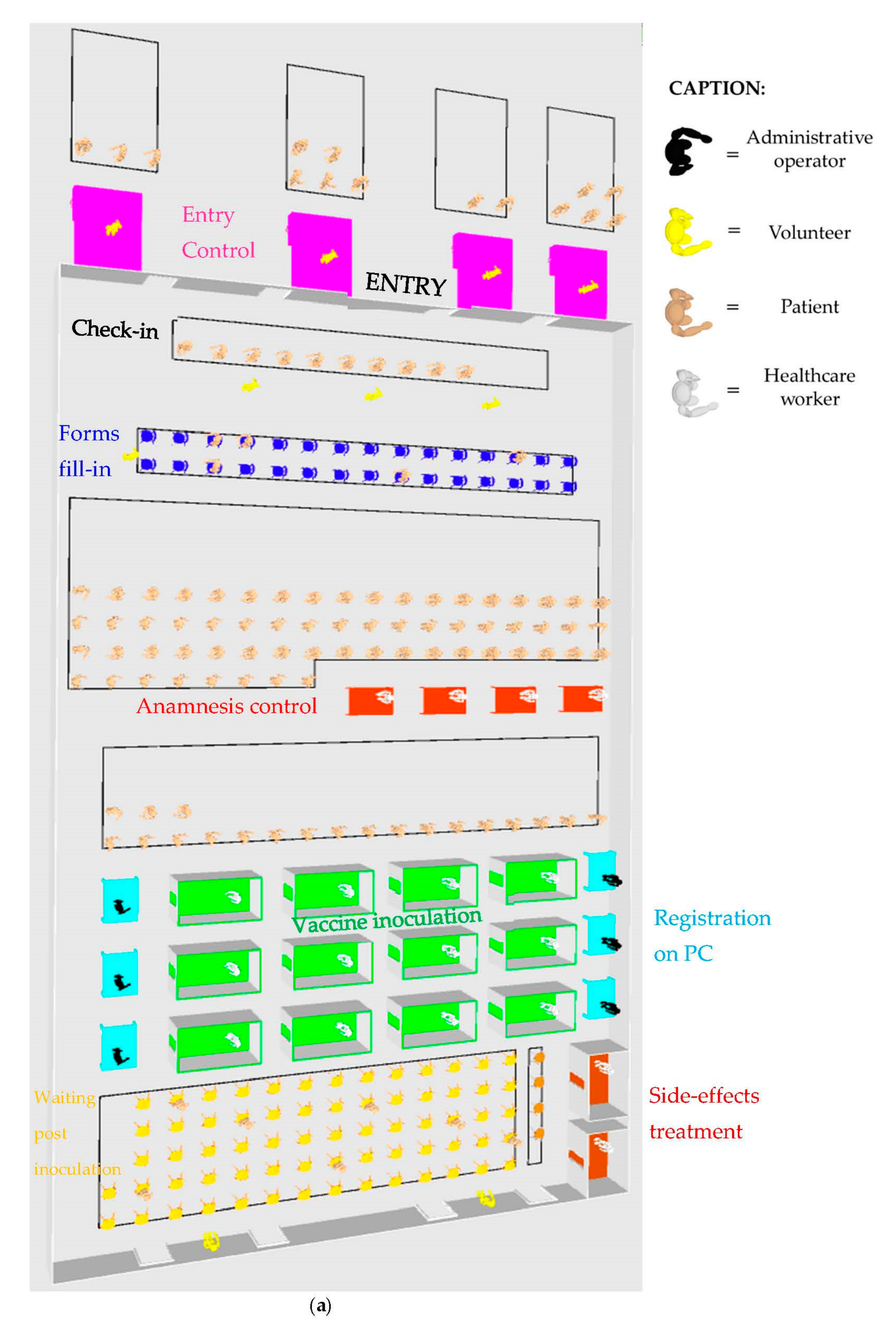
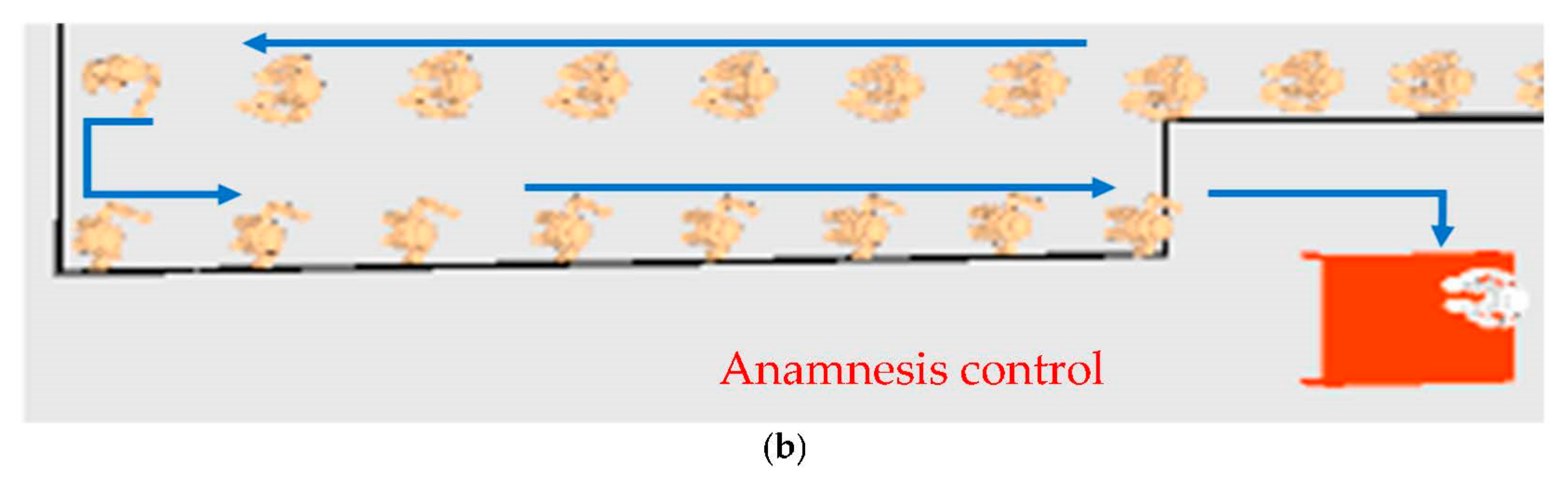
5. Case Study
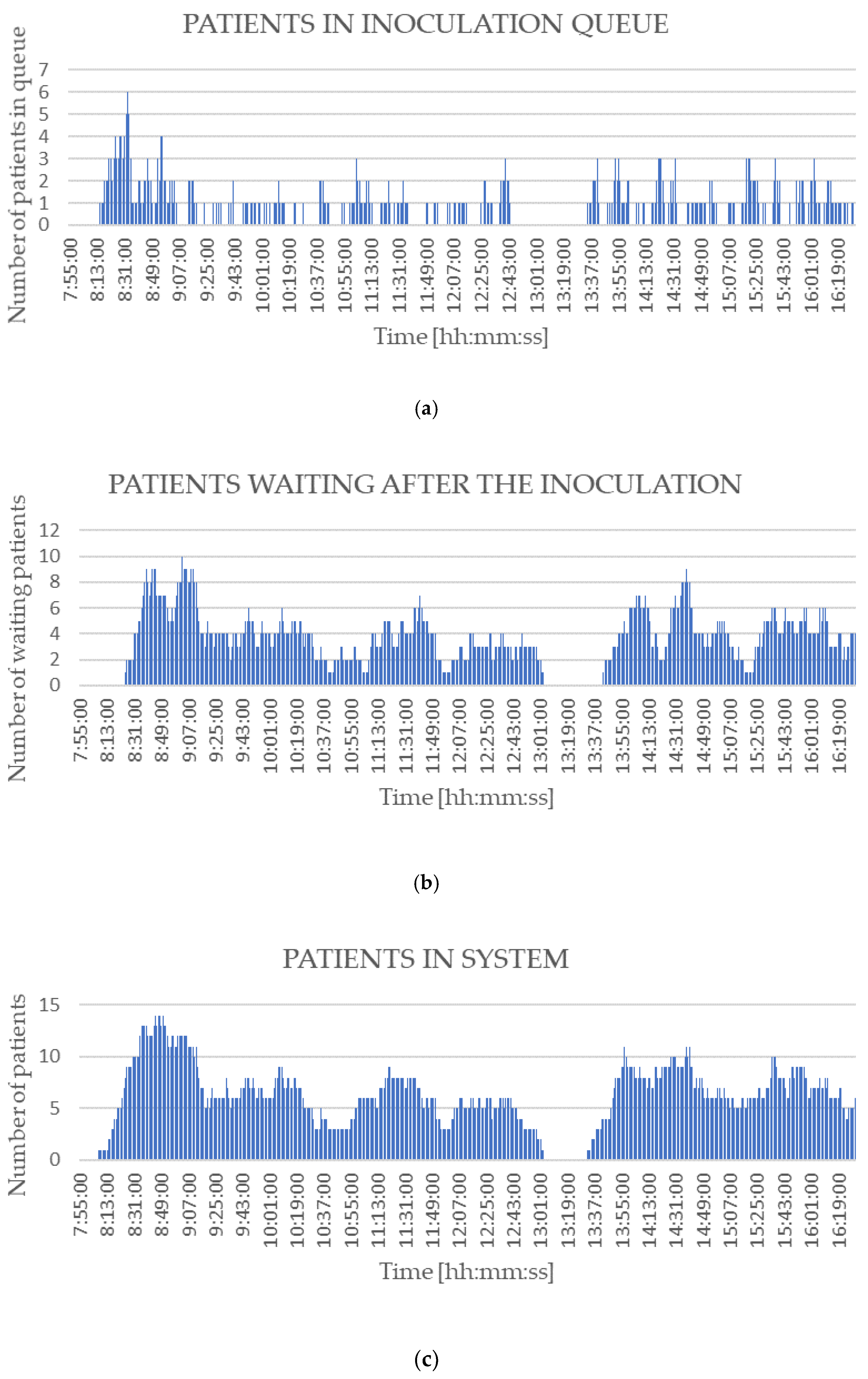
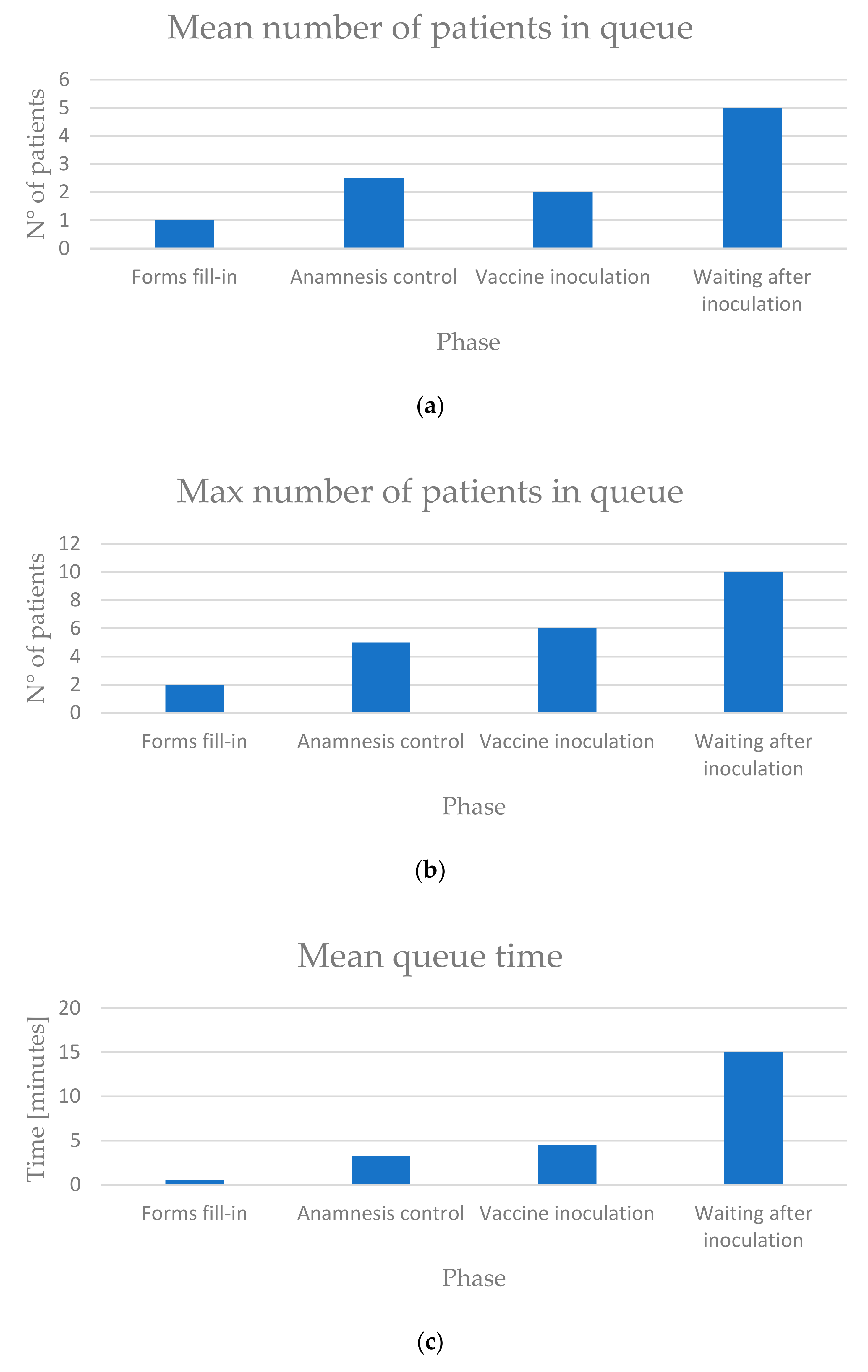
6. Results
- Duration of each phase for each patient and average durations.
- Timestamps of the beginning and the end of each phase.
- Number of patients in each queue in every minute.
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int (accessed on 21 April 2021).
- Holt, E. Slovakia to Test All Adults for SARS-CoV-2. Lancet 2020, 396, 1386–1387. [Google Scholar] [CrossRef]
- Kim, J.; Kwon, O. A Model for Rapid Selection and COVID-19 Prediction with Dynamic and Imbalanced Data. Sustainability 2021, 19, 3099. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control (ECDC). COVID-19 Vaccination and Prioritisation Strategies in the EU/EEA; Technical Report; ECDC: Solna Municipality, Sweden, 2020. [Google Scholar]
- CDC. Pfizer-BioNTech COVID-19 Vaccine Storage and Handling Summary; CDC: Atlanta, GA, USA, 2021.
- European Centre for Disease Prevention and Control (ECDC). Overview of the Implementation of COVID-19 Vaccination Strategies and Vaccine Deployment Plans in the EU/EEA; Technical Report; ECDC: Solna Municipality, Sweden, 2021. [Google Scholar]
- CDC. Moderna COVID-19 Vaccine: Storage and Handling Summary; CDC: Atlanta, GA, USA, 2021.
- Chiruţă, C.; Bulgariu, E.; Avsec, J.; Ferčec, B.; Mencinger, M. Comparison of the Evolution of the COVID-19 Disease between Romania and Italy. Appl. Syst. Innov. 2020, 3, 44. [Google Scholar] [CrossRef]
- Sandmann, F.G.; Davies, N.G.; Vassall, A.; Edmunds, W.J.; Jit, M.; Sun, F.Y.; Villabona-Arenas, C.J.; Nightingale, E.S.; Showering, A.; Knight, G.M.; et al. The Potential Health and Economic Value of SARS-CoV-2 Vaccination alongside Physical Distancing in the UK: A Transmission Model-Based Future Scenario Analysis and Economic Evaluation. Lancet Infect. Dis. 2021, S1473309921000797. [Google Scholar] [CrossRef]
- CDC. When Vaccine Is Limited, Who Gets Vaccinated First? Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/recommendations.html (accessed on 21 April 2021).
- SSE Arena to Be Mass Vaccination Centre. Available online: https://www.health-ni.gov.uk/news/sse-arena-be-mass-vaccination-centre (accessed on 21 April 2021).
- Rosen, B.; Waitzberg, R.; Israeli, A. Israel’s Rapid Rollout of Vaccinations for COVID-19. Isr. J. Health Policy Res. 2021, 10, 6. [Google Scholar] [CrossRef]
- Xu, L.D.; Xu, E.L.; Li, L. Industry 4.0: State of the Art and Future Trends. Int. J. Prod. Res. 2018, 56, 2941–2962. [Google Scholar] [CrossRef] [Green Version]
- Cohen, Y.; Faccio, M.; Galizia, F.G.; Mora, C.; Pilati, F. Assembly System Configuration through Industry 4.0 Principles: The Expected Change in the Actual Paradigms. IFAC-Pap. 2017, 50, 14958–14963. [Google Scholar] [CrossRef]
- Andriolo, A.; Battini, D.; Calzavara, M.; Gamberi, M.; Peretti, U.; Persona, A.; Pilati, F.; Sgarbossa, F. New RFID Pick-to-Light System: Operating Characteristics and Future Potential. Int. J. RF Technol. 2016, 7, 43–63. [Google Scholar] [CrossRef]
- Madni, A.; Madni, C.; Lucero, S. Leveraging Digital Twin Technology in Model-Based Systems Engineering. Systems 2019, 7, 7. [Google Scholar] [CrossRef] [Green Version]
- Müller, J.M.; Kiel, D.; Voigt, K.-I. What Drives the Implementation of Industry 4.0? The Role of Opportunities and Challenges in the Context of Sustainability. Sustainability 2018, 10, 247. [Google Scholar] [CrossRef] [Green Version]
- Accorsi, R.; Bortolini, M.; Baruffaldi, G.; Pilati, F.; Ferrari, E. Internet-of-Things Paradigm in Food Supply Chains Control and Management. Procedia Manuf. 2017, 11, 889–895. [Google Scholar] [CrossRef]
- Lepore, D.; Micozzi, A.; Spigarelli, F. Industry 4.0 Accelerating Sustainable Manufacturing in the COVID-19 Era: Assessing the Readiness and Responsiveness of Italian Regions. Sustainability 2021, 13, 2670. [Google Scholar] [CrossRef]
- Vaccari, I.; Orani, V.; Paglialonga, A.; Cambiaso, E.; Mongelli, M. A Generative Adversarial Network (GAN) Technique for Internet of Medical Things Data. Sensors 2021, 21, 3726. [Google Scholar] [CrossRef]
- Ramallo-González, A.P.; González-Vidal, A.; Skarmeta, A.F. CIoTVID: Towards an Open IoT-Platform for Infective Pandemic Diseases Such as COVID-19. Sensors 2021, 21, 484. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.P.; Javaid, M.; Haleem, A.; Suman, R. Internet of Things (IoT) Applications to Fight against COVID-19 Pandemic. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 521–524. [Google Scholar] [CrossRef]
- Gunal, M.M. A Guide for Building Hospital Simulation Models. Health Syst. 2012, 1, 17–25. [Google Scholar] [CrossRef]
- Katsaliaki, K.; Mustafee, N. Applications of Simulation within the Healthcare Context. J. Oper. Res. Soc. 2011, 62, 1431–1451. [Google Scholar] [CrossRef]
- Arlegui, H.; Nachbaur, G.; Praet, N.; Bégaud, B.; Caro, J.J. Using Discretely Integrated Condition Event Simulation To Construct Quantitative Benefit–Risk Models: The Example of Rotavirus Vaccination in France. Clin. Ther. 2020, 42, 1983–1991.e2. [Google Scholar] [CrossRef]
- Ahmed, M.A.; Alkhamis, T.M. Simulation Optimization for an Emergency Department Healthcare Unit in Kuwait. Eur. J. Oper. Res. 2009, 198, 936–942. [Google Scholar] [CrossRef]
- Hernandez, I.; Ramirez-Marquez, J.E.; Starr, D.; McKay, R.; Guthartz, S.; Motherwell, M.; Barcellona, J. Optimal Staffing Strategies for Points of Dispensing. Comput. Ind. Eng. 2015, 83, 172–183. [Google Scholar] [CrossRef]
- Hupert, N.; Mushlin, A.I.; Callahan, M.A. Modeling the Public Health Response to Bioterrorism: Using Discrete Event Simulation to Design Antibiotic Distribution Centers. Med Decis. Mak. 2002, 22 (Suppl. S1), 17–25. [Google Scholar] [CrossRef]
- Aaby, K.; Herrmann, J.W.; Jordan, C.S.; Treadwell, M.; Wood, K. Montgomery County’s Public Health Service Uses Operations Research to Plan Emergency Mass Dispensing and Vaccination Clinics. Interfaces 2006, 36, 569–579. [Google Scholar] [CrossRef] [Green Version]
- Van de Kracht, T.; Heragu, S.S. Lessons from Modeling and Running the World’s Largest Drive-Through Mass Vaccination Clinic. Inf. J. Appl. Anal. 2021, 51, 91–105. [Google Scholar]
- Gupta, A.; Evans, G.W.; Heragu, S.S. Simulation and Optimization Modeling for Drive-through Mass Vaccination—A Generalized Approach. Simul. Model. Pract. Theory 2013, 37, 99–106. [Google Scholar] [CrossRef] [Green Version]
- Asgary, A.; Najafabadi, M.M.; Karsseboom, R.; Wu, J. A Drive-through Simulation Tool for Mass Vaccination during COVID-19 Pandemic. Healthcare 2020, 8, 469. [Google Scholar] [CrossRef]
- Grieves, M.; Vickers, J. Digital Twin: Mitigating Unpredictable, Undesirable Emergent Behavior in Complex Systems. In Transdisciplinary Perspectives on Complex Systems; Kahlen, F.-J., Flumerfelt, S., Alves, A., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 85–113. ISBN 978-3-319-38754-3. [Google Scholar]
- Balachandar, S.; Chinnaiyan, R. Reliable Digital Twin for Connected Footballer. In International Conference on Computer Networks and Communication Technologies; Smys, S., Bestak, R., Chen, J.I.-Z., Kotuliak, I., Eds.; Lecture Notes on Data Engineering and Communications Technologies; Springer: Singapore, 2019; Volume 15, pp. 185–191. ISBN 978-981-10-8680-9. [Google Scholar]
- Glaessgen, E.; Stargel, D. The Digital Twin Paradigm for Future NASA and U.S. Air Force Vehicles. In Proceedings of the 53rd AIAA/ASME/ASCE/AHS/ASC Structures, Structural Dynamics and Materials Conference & 20th AIAA/ASME/AHS Adaptive Structures Conference & 14th AIAA, Honolulu, HI, USA, 23–26 April 2012. [Google Scholar]
- Coelho, F.; Relvas, S.; Barbosa-Póvoa, A.P. Simulation-Based Decision Support Tool for in-House Logistics: The Basis for a Digital Twin. Comput. Ind. Eng. 2021, 153, 107094. [Google Scholar] [CrossRef]
- Korth, B.; Schwede, C.; Zajac, M. Simulation-Ready Digital Twin for Realtime Management of Logistics Systems. In Proceedings of the 2018 IEEE International Conference on Big Data (Big Data), Seattle, WA, USA, 10–13 December 2018; pp. 4194–4201. [Google Scholar]
- Jimenez, J.I.; Jahankhani, H.; Kendzierskyj, S. Health Care in the Cyberspace: Medical Cyber-Physical System and Digital Twin Challenges. In Digital Twin Technologies and Smart Cities; Farsi, M., Daneshkhah, A., Hosseinian-Far, A., Jahankhani, H., Eds.; Internet of Things; Springer International Publishing: Cham, Switzerland, 2020; pp. 79–92. ISBN 978-3-030-18731-6. [Google Scholar]
- Liu, Y.; Zhang, L.; Yang, Y.; Zhou, L.; Ren, L.; Wang, F.; Liu, R.; Pang, Z.; Deen, M.J. A Novel Cloud-Based Framework for the Elderly Healthcare Services Using Digital Twin. IEEE Access 2019, 7, 49088–49101. [Google Scholar] [CrossRef]
- Hashim, U.N.; Salahuddin, L.; Ikram, R.R.R.; Hashim, U.R.; Choon, N.H.; Mohayat, M.H.N. The Design and Implementation of Mobile Heart Monitoring Applications Using Wearable Heart Rate Sensor. Int. J. Adv. Comput. Sci. Appl. 2021, 12, 6. [Google Scholar]
- Karakra, A.; Fontanili, F.; Lamine, E.; Lamothe, J.; Taweel, A. Pervasive Computing Integrated Discrete Event Simulation for a Hospital Digital Twin. In Proceedings of the 2018 IEEE/ACS 15th International Conference on Computer Systems and Applications (AICCSA), Aqaba, Jordan, 28 October–1 November 2018; pp. 1–6. [Google Scholar]
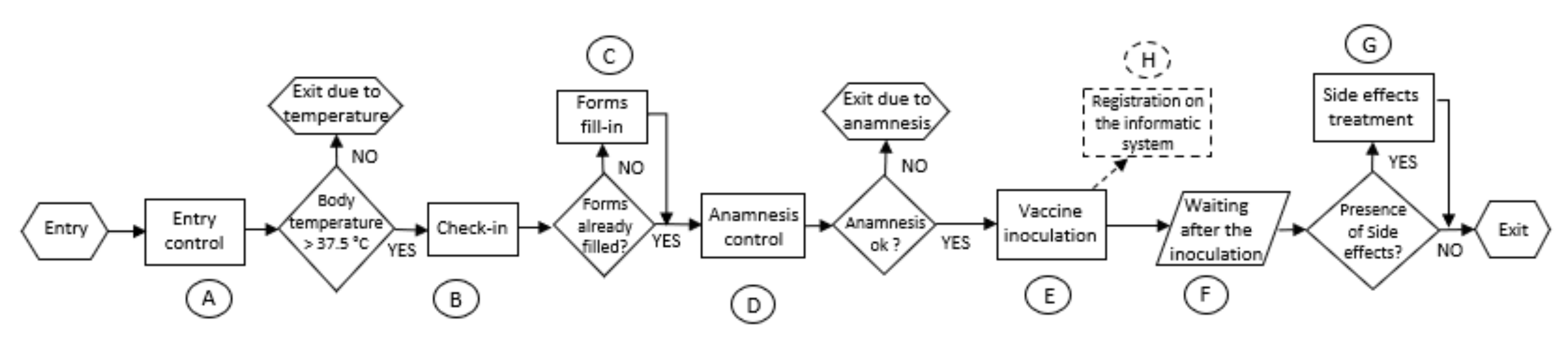
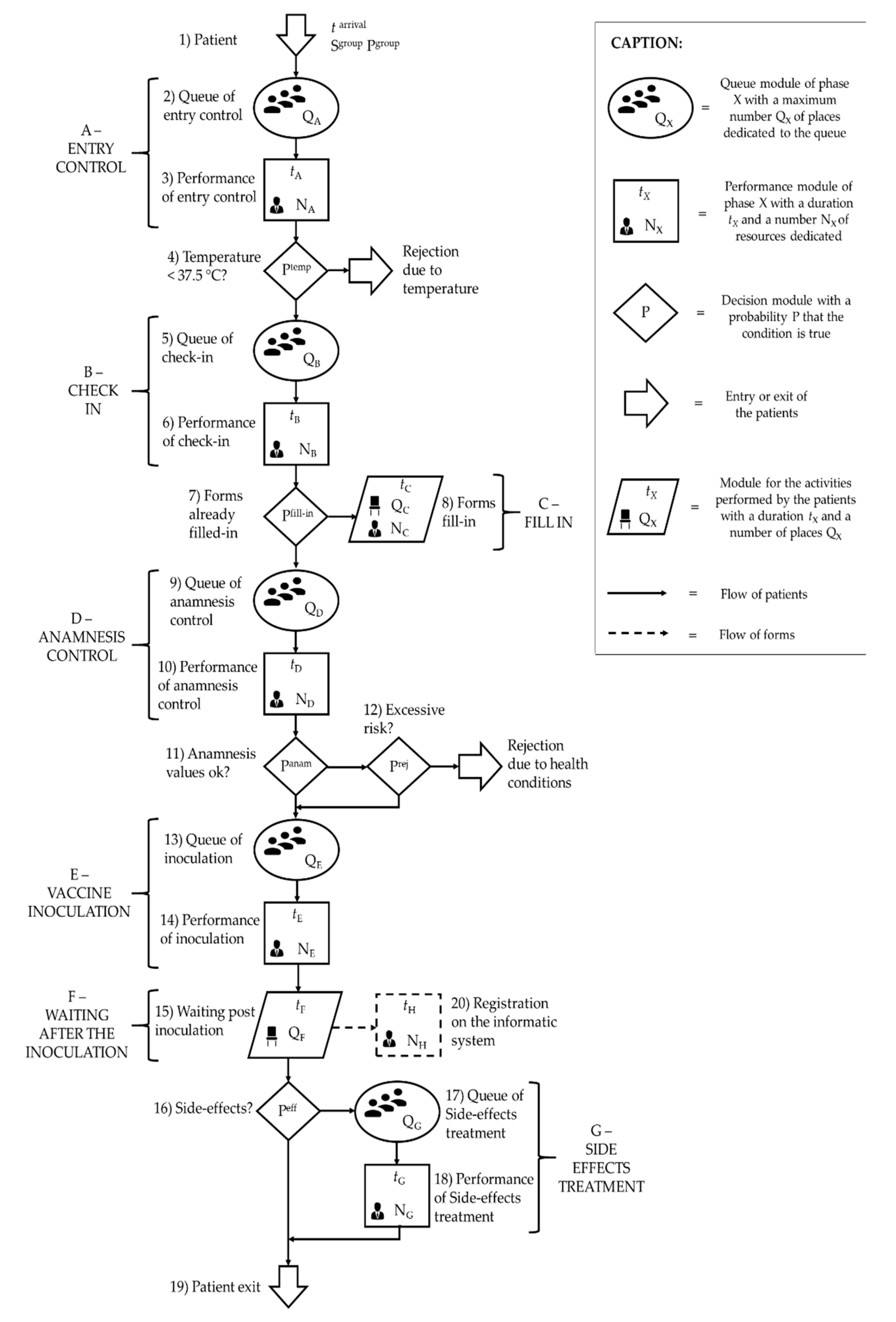
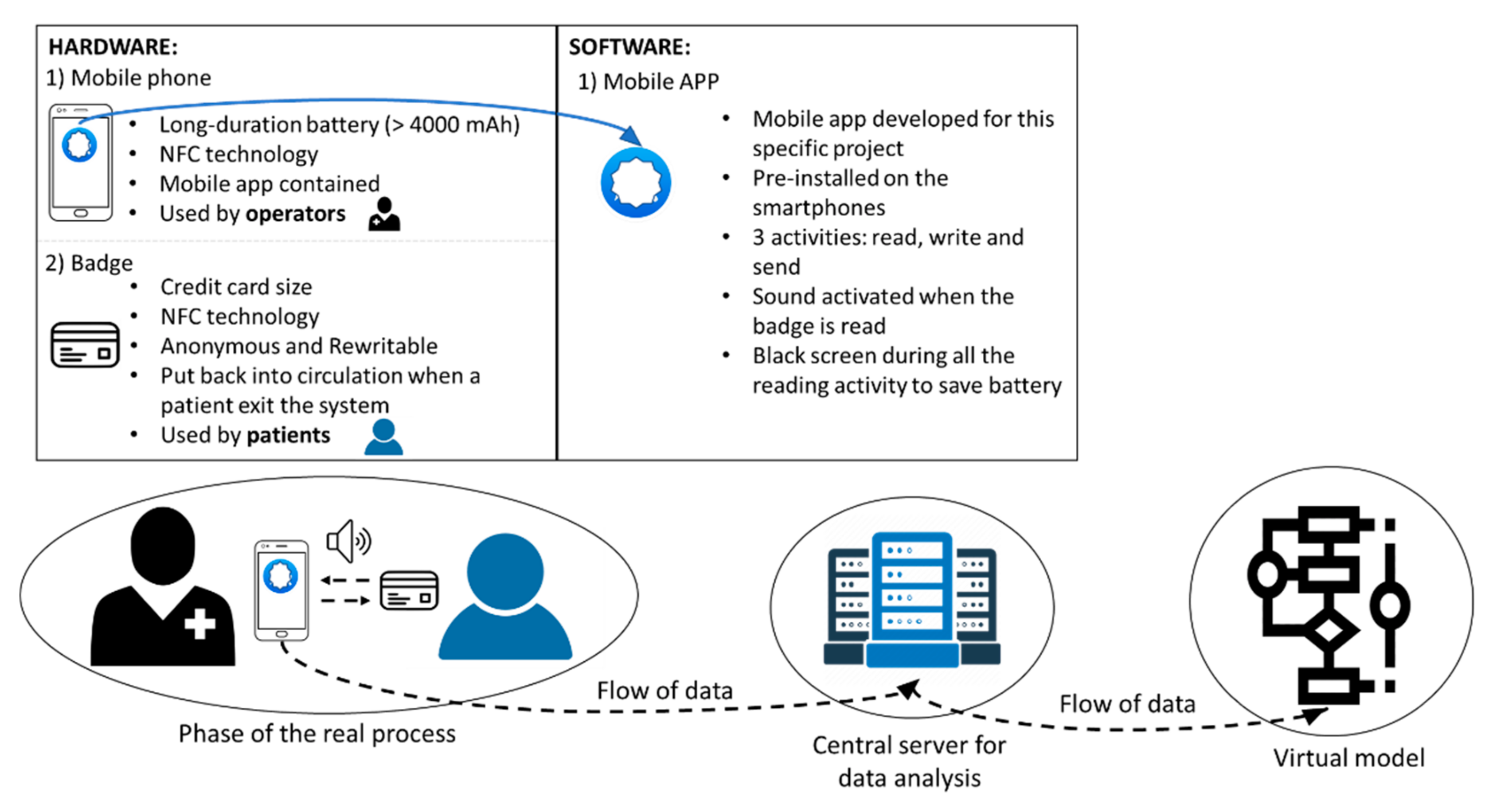


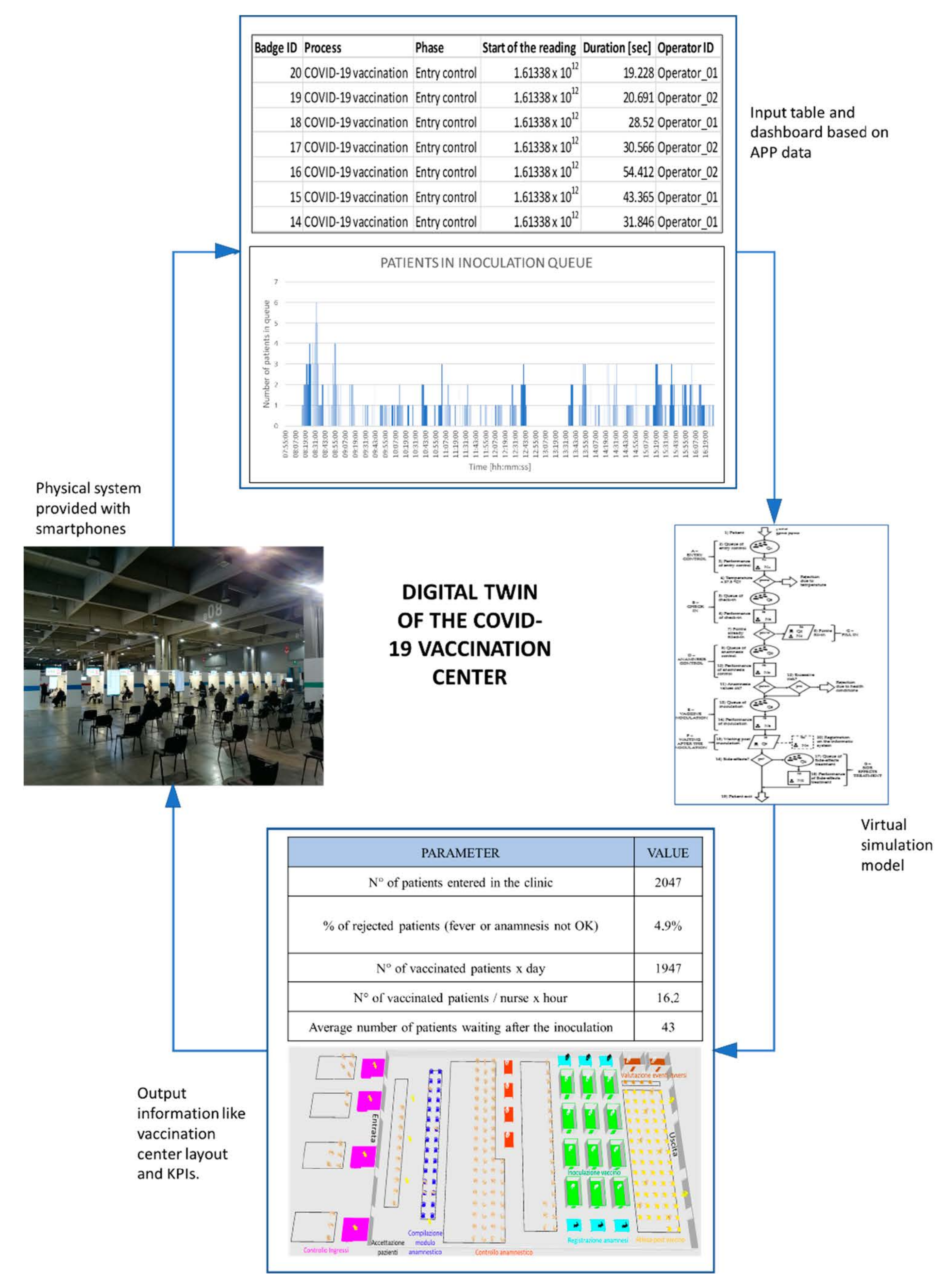


| Name | Symbol | Units of Measure |
|---|---|---|
| Group size | Sgroup | patients |
| Probability of a specific group size | Pgroup | % |
| Inter-arrival time | tarrival | sec |
| Probability of having a body temperature lower than 37.5 °C | Ptemp | % |
| Probability of having already filled-in the forms at home | Pfill-in | % |
| Probability that the anamnesis is completely ok | Panam | % |
| Probability of being rejected because of the anamnesis | Prej | % |
| Probability of experiencing side effects | Peff | % |
| Working time of a specific phase i | ti i = A, …, H | sec |
| Number of resources needed in a specific phase i | Ni i = A, …, H | resources |
| Number of places to dedicate in queue for a specific phase i | Qi i = A, …, H | places |
| Name | Symbol | Units of Measure |
|---|---|---|
| Number of patients vaccinated per hour per nurse | Npat/nurse | Patients/nurse × hour |
| Number of patients vaccinated per day | Npat | Patients/day |
| Average time spent in the system by a patient | Tsys-avg | min |
| Maximum time spent in the system by a patient | Tsys-max | min |
| Average time spent in queue by a patient | Twait-avg | min |
| Maximum time spent in queue by a patient | Twait-max | min |
| Resource utilization for each phase i | Ui i = A, …, H | % |
| Time-Related Parameter | Statistical Distribution [s] |
|---|---|
| tarrival = time between 2 consecutive arrivals | TRIA(51,120,510) |
| tA = working time of entry control phase | 7 + WEIB(17.2, 1.03) |
| tB = working time of check-in phase | 6.5 + GAMM(6.27, 2.22) |
| tC = working time of forms fill-in phase | 54 + WEIB(73.6, 1.15) |
| tD = working time of anamnesis control phase | OK: 24.5 + WEIB(19, 1.54) NOT OK: 32 + WEIB(24.4, 1.51) |
| tE = working time of vaccine inoculation phase | 67 + GAMM(21.7, 2.12) |
| tF = waiting time after the inoculation | TRIA(480,780,960) |
| tG = working time of side effects treatment phase | REAL EFFECTS: TRIA(240,420,600) FEAR: TRIA(60,120,180) |
| tH = working time of registration phase | TRIA(42.5,75.5,122) |
| Probability Parameter | Value [%] |
|---|---|
| Ptemp = Probability of having a body temperature lower than 37.5 °C | 99 |
| Pfill-in = Probability of having already filled-in the forms at home | 60 |
| Panam = Probability that the anamnesis is completely ok | 30 |
| Prej = Probability of being rejected because of the anamnesis | 5 |
| Peff = Probability of experiencing side effects | 5 |
| PARAMETERS | SCENARIOS | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | ||
| Triangular coefficients for interarrival time [s] | A | 54 | 51 | 51 | 48 | 42 |
| B | 127 | 120 | 120 | 113 | 99 | |
| C | 540 | 510 | 510 | 480 | 420 | |
| Number of resources (Ni) i = A, …, G or i = “Exit control” | A- Entry control | 4 | 4 | 4 | 4 | 4 |
| B- Check-in | 3 | 3 | 3 | 3 | 3 | |
| D- Anamnesis Control | 4 | 4 | 4 | 4 | 4 | |
| E- Vaccine Inoculation | 12 | 13 | 12 | 12 | 13 | |
| H- Registration on PC | 6 | 6 | 6 | 6 | 6 | |
| G- Side effects treatment | 2 | 2 | 2 | 2 | 2 | |
| Exit control | 2 | 2 | 2 | 2 | 2 | |
| Num tot medical resources | 18 | 19 | 18 | 18 | 19 | |
| Num tot non-medical resources | 15 | 15 | 15 | 15 | 15 | |
| NUM TOT RESOURCES | 33 | 34 | 33 | 33 | 34 | |
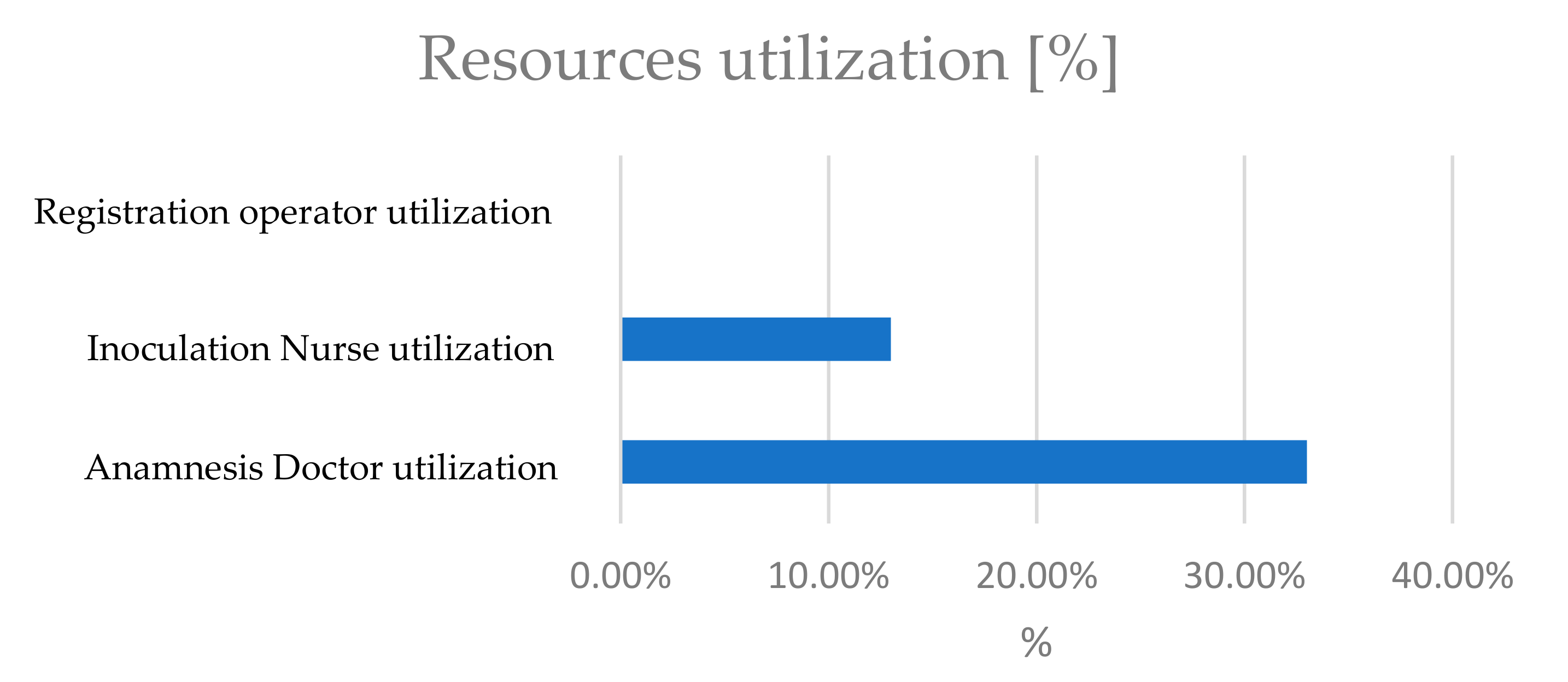
| Resource utilization (Ui) i = A, …, G | A- Entry control | 33.8% | 36.5% | 36.3% | 38.1% | 44.0% |
| B- Check-in | 38.1% | 41.2% | 40.8% | 42.9% | 49.5% | |
| D- Anamnesis Control | 71.6% | 77.4% | 76.6% | 80.4% | 91.8% | |
| E- Vaccine Inoculation | 75.0% | 74.7% | 80.2% | 84.1% | 88.4% | |
| H- Registration on PC | 71.6% | 77.4% | 76.5% | 80.5% | 91.4% | |
| G- Side effects treatment | 19.4% | 22.6% | 21.9% | 23.2% | 25.4% | |
| Time [min] | Tsys-avg | 23.7 | 23.9 | 25.1 | 26.4 | 32.8 |
| Tsys-max | 52.7 | 59.3 | 60.9 | 64.1 | 97.3 | |
| Twait-avg | 4 | 3.4 | 5.4 | 5.9 | 12.4 | |
| Twait-max | 38.4 | 44.3 | 48 | 56 | 83.4 | |
| Number of places to dedicate in the layout (Qi) i = A, …, G | A- Entry control | 26 | 32 | 25 | 27 | 43 |
| B- Check-in | 29 | 30 | 30 | 29 | 33 | |
| D- Anamnesis Control | 101 | 136 | 116 | 143 | 320 | |
| E- Vaccine Inoculation | 43 | 17 | 64 | 74 | 20 | |
| F- Waiting post Inoculation | 66 | 68 | 67 | 67 | 70 | |
| G- Side effects treatment | 3 | 4 | 4 | 3 | 3 | |
| Num tot places in the layout | 268 | 287 | 306 | 343 | 489 | |
| Npat | 2026 | 2192 | 2164 | 2270 | 2567 | |
| Npat/nurse | 16.9 | 16.9 | 18.0 | 18.9 | 19.8 | |
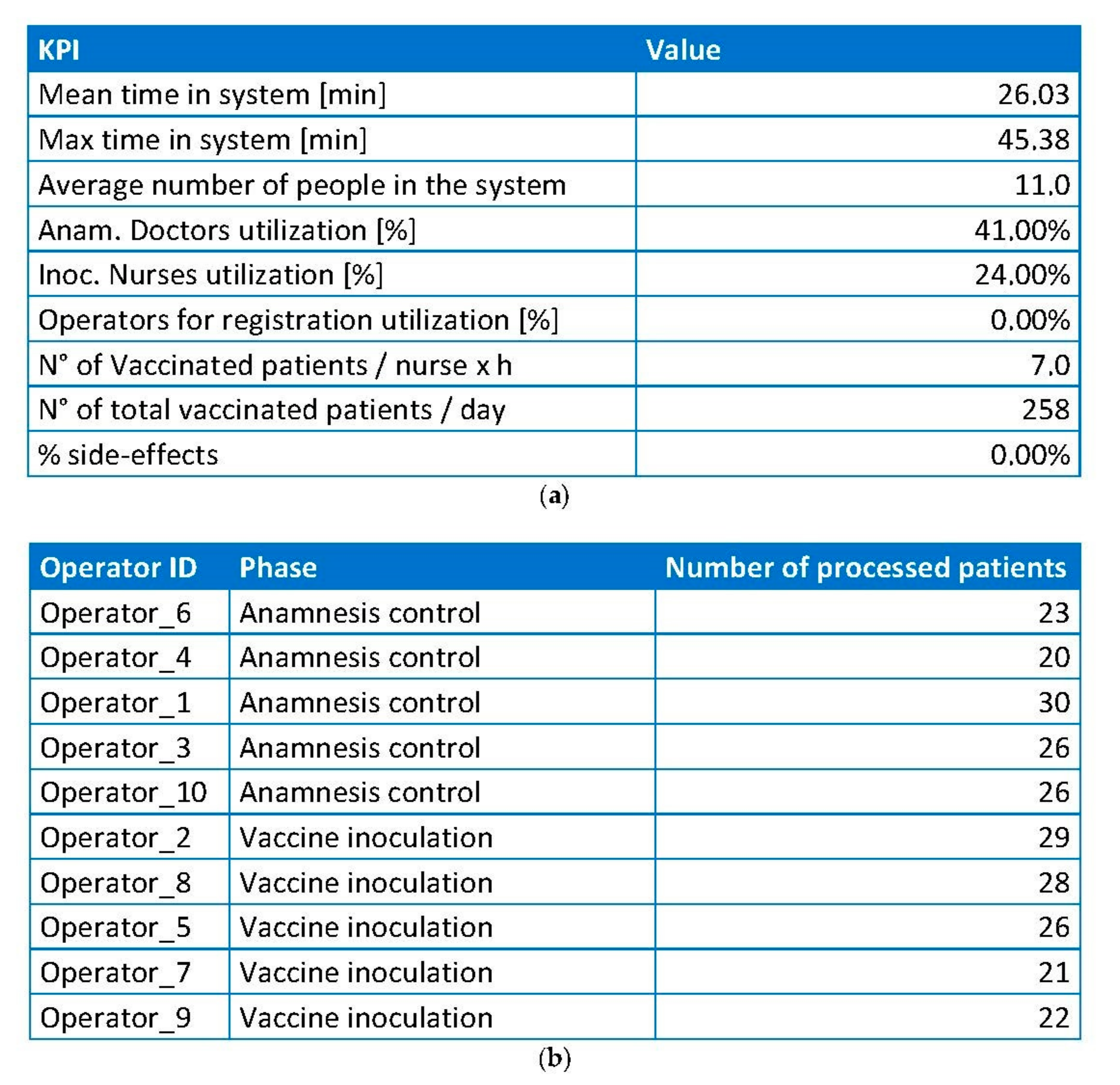
| Output Parameter | Value | Units of Measures |
|---|---|---|
| Npat/nurse | 18.03 | Patients/nurse × hour |
| Npat | 2164 | Patients/day |
| Tsys-avg | 25.1 | min |
| Tsys-max | 60.9 | min |
| Twait-avg | 5.4 | min |
| Twait-max | 48 | min |
| UA | 36.3 | % |
| UB | 40.8 | % |
| UD | 76.6 | % |
| UE | 80.2 | % |
| UG | 21.9 | % |
| UH | 76.5 | % |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pilati, F.; Tronconi, R.; Nollo, G.; Heragu, S.S.; Zerzer, F. Digital Twin of COVID-19 Mass Vaccination Centers. Sustainability 2021, 13, 7396. https://doi.org/10.3390/su13137396
Pilati F, Tronconi R, Nollo G, Heragu SS, Zerzer F. Digital Twin of COVID-19 Mass Vaccination Centers. Sustainability. 2021; 13(13):7396. https://doi.org/10.3390/su13137396
Chicago/Turabian StylePilati, Francesco, Riccardo Tronconi, Giandomenico Nollo, Sunderesh S. Heragu, and Florian Zerzer. 2021. "Digital Twin of COVID-19 Mass Vaccination Centers" Sustainability 13, no. 13: 7396. https://doi.org/10.3390/su13137396
APA StylePilati, F., Tronconi, R., Nollo, G., Heragu, S. S., & Zerzer, F. (2021). Digital Twin of COVID-19 Mass Vaccination Centers. Sustainability, 13(13), 7396. https://doi.org/10.3390/su13137396








