Geoenvironmental Implications and Biocenosis of Freshwater Lakes in the Arid Zone of East Kazakhstan
Abstract
1. Introduction
2. Study Area
2.1. Regional Setting
2.2. Site Location
2.3. The Sibe Lakes’ Hydrology and Hydrogeology
3. Aims, Material and Methods
- -
- species taxonomy composition;
- -
- total size of the biotic community;
- -
- total biomass;
- -
- biomass composition of dominant groups and species;
- -
- zonal quantitative and qualitative zooplankton distribution.
4. Results
4.1. Lake Hydrochemistry
4.2. Zooplankton Ecology
- Lake Sadyrkol
- Lake Tortkara
- Lake Shalkar
- Lake Korzhynkol
4.3. Macrozoobenthos Ecology
5. Résumé and Discussion
5.1. The Sibe Lakes’ Biodiversity and Ecology Status
5.2. The Sibe Lakes’ Sustainable Use
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chambers, P.A.; Lacoul, P.; Murphy, K.J.; Thomaz, S.M. Global diversity of aquatic macrophytes in freshwater. In Freshwater Animal Diversity Assessment. Developments in Hydrobiology; Balian, E.V., Lévêque, C., Segers, H., Martens, K., Eds.; Springer Nature: Cham, Switzerland, 2007; Volume 198, pp. 9–26. [Google Scholar]
- Bai, J.; Chen, X.; Li, J.; Yang, L.; Fang, H. Changes in the area of inland lakes in arid regions of central Asia during the past 30 years. Env. Monit Assess 2011, 178, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Jones, N.T.; Gilbert, B. Changing climate cues differentially alter zooplankton dormancy dynamics across latitude. J. Anim. Ecol. 2015, 85, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Asai, K.; Houki, A. Numerical estimation to organic pollution of flowing water by using the epilithic diatom assemblage diatom assemblage index (DAIpo). Sci. Total Environ. 1986, 55, 209–218. [Google Scholar] [CrossRef]
- Berthon, V.; Bouchez, A.; Rimet, F. Using diatom life-forms and ecological guilds toassess organic pollution and trophic level in rivers: A case study of rivers in south-eastern France. Hydrobiologia 2011, 673, 259–271. [Google Scholar] [CrossRef]
- Kókai, Z.; Bácsi, I.; Török, P.; Buczkó, K.; Balogh, C.; Tóthmérész, B. Halophilic diatom taxa are sensitive indicators of even short term changes in lowland lotic systems. Acta Bot. Croat 2015, 74, 287–302. [Google Scholar] [CrossRef]
- García-Chicote, J.; Armengol, X.; Rojo, C. Zooplankton species as indicators of trophic state in reservoirs from Mediterranean river basins. Inland Waters 2019, 9, 113–123. [Google Scholar] [CrossRef]
- Hall, C.M.; Page, S.J. The Geography of Tourism and Recreation: Environment, Place and Space; Routhledge: London, UK; New York, NY, USA, 1999; 309p. [Google Scholar]
- Wall, G. Implications of Global Climate Change for Tourism and Recreation in Wetland Areas. Clim. Chang. 1998, 40, 371–389. [Google Scholar] [CrossRef]
- Amirgaliev, N.A. Aral-Syrdarya Basin: Hydrochemistry, Problems of Aquatic Toxicology; Bastau: Almaty, Kazakhstan, 2007; 224p. [Google Scholar]
- Zhumagalieva, Z.M. Lake fund of Kazakhstan: Resumé. Doctoral Dissertation, Geographical Sciences, St. Petersburg, Russia, 2006; 27p. [Google Scholar]
- Dostaj, Z.D. Management of the Hydroecosystem of Lake Balkash; Institute of Geography: Almaty, Kazakhstan, 2009; 236p. [Google Scholar]
- Dmitriev, P.S.; Fomin, I.A.; Bekturganova, M.B. On the issue of the current state and significance of the lakes of the North Kazakhstan region. In Proceedings of the Kozybayev Readings 2013: Kazakhstan in the Cultural and Civilization Processes, Petropavlovsk, Kazakhstan, 15 November 2013; M. Kozybayev NKSU: Petropavlovsk, Kazakhstan, 2013; pp. 158–162. [Google Scholar]
- Ismailova, A.A.; Zhamankara, A.K.; Akbaeva, L.K.; Adamov, A.A.; Abakumov, A.I.; Tulegenov, S.A.; Muratov, R.M. Hydrochemical and hydrobiological indicators as characteristics of the ecological state of lakes (by the example of lakes Burabay and Ulken Shabakty). Kazn. Bull. Biol. Ser. 2013, 3, 504–507. [Google Scholar]
- Tursunova, A.A. Modern Assessment of Water Resources of the River. Ile Taking into Account the Circulation Processes in the Atmosphere: Author. Doctoral Dissertation, Geographical Sciences, Almaty, Kazakhstan, 2006; 16p. [Google Scholar]
- Tursunov, E.A.; Bazhieva, A.M. Small lakes of the Shchuchinsko-Borovsk resort area. Environmental problems and solutions. Hydrometeorol. Ecol. 2013, 2, 172–176. [Google Scholar]
- Tursunov, E.A.; Madibekov, A.S.; Kulebaev, K.M. Modern morphometric characteristics of the lakeBalkash. In Scientific Notes of the Russian State Hydrometeorological University; Russian State Hydrometeorological University: Saint-Petersburg, Russia, 2014; Volume 34, pp. 43–47. [Google Scholar]
- Barinova, S.S.; Bragina, T.M.; Nevo, E. Algal species diversity of arid region lakes in Kazakhstan and Izrael. Community Ecol. 2009, 10, 7–16. [Google Scholar] [CrossRef]
- Caisová, L.; Bašta, T.; Chlachula, J.; Komárek, J.; Husák, S. Taxonomic investigations of the crynobacterial and algal microflora from the Katon-KaragayNational Park (Altai, East Kazakhstan). Biodivers. Res. Conserv. 2010, 15, 45–54. [Google Scholar]
- Krupa, E.G. The structure of species dominance in the plankton of the Caspian Sea of Kazakhstan sector. In Several Aspects of Hydro-Ecological Problems of Kazakhstan; Malkovsky, I.M., Ed.; Kaganat: Almaty, Kazakhstan, 2011. [Google Scholar]
- Romanova, S.M.; Kazangapova, N.B. Quality of waters lakes Kazakhstan in contemporaneous period (for example of lake Kopa). Int. J. Biol. Chem. 2013, 6, 65–70. [Google Scholar]
- Barinova, S.; Krupa, E. Critical environmental factors for photosynthetic organisms of the Shardara Reservoir, Kazakhstan. Bull. Adv. Sci. Res. 2016, 2, 17–27. [Google Scholar]
- Klymiuk, V.; Barinova, S. Phytoplankton Cell Size in Saline Lakes. Research Journal of Pharmaceutical. Biol. Chem. Sci. 2016, 7, 1077–1085. [Google Scholar]
- Boros, E.; Jurecska, L.; Tatár, E.; Vörös, L.; Kolpakova, M. Chemical composition and trophic state of shallow saline steppe lakes in central Asia (North Kazakhstan). Environ. Monit Assess 2017, 189, 546. [Google Scholar] [CrossRef]
- Gaskova, O.; Kolpakova, M.; Naymushina, O.; Krivonogov, S. Geochemical processes controlling the water chemistry of saline lakes in the northern Kazakhstan region. In Proceedings of the 17th International Multidisciplinary Scientific GeoConference (SGEM 2017), Albena, Bulgaria, 29 June–5 July 2017; pp. 325–334. [Google Scholar]
- Jiyenbekov, A.; Barinova, S.; Bigaliev, A.; Nurashov, S.; Sametova, E.; Fahima, T. Algal comparative floristic of the Alakol lake Natural State Reserve and other lakes in Kazakhstan. Moj Ecol. Environ. Sci. 2018, 3, 252–258. [Google Scholar]
- Valeev, A.; Karatayev, M.; Abitbayeva, A.; Uxutbayeva, S.; Bektursinova, A.; Sharapkhanova, A. Monitoring Coastline Dynamics of Alakol Lake in Kazakhstan Using Remote Sensing Data. Geosciences 2019, 9, 404–413. [Google Scholar] [CrossRef]
- Akhmedenov, K.M. Tourist and recreational potential of the salt lakes of Western Kazakhstan. Geoj. Tour. Geosites 2020, 30, 782–787. [Google Scholar] [CrossRef]
- Chlachula, J.; Zhensikbayeva, N.Z.; Yegorina, A.V.; Kabdrakhmanova, N.K.; Czerniawska, J.; Kumarbekuly, S. Territorial Assessment of the East Kazakhstan Geo/Ecotourism: Sustainable Travel Prospects in the Southern Altai Area. Geosciences 2021, 11, 156. [Google Scholar] [CrossRef]
- Fránková, M.; Chlachula, J.; Czerniawska, J.; Myrzagaliyeva, A.B.; Doláková, N.; Jankovská, V. Diatom (Bacillariophyceae). Diversity of the Sibinskiye Lakes (East Kazakhstan). 2021, unpublished. [Google Scholar]
- Myrzagaliyeva, A.B.; Kozáková, M.; Chlachula, J. Algae diversity of the lakes Sibinskye, East Kazakhstan—International Symposium Microorganisms and the Biosphere Microbios. In Proceedings of the Materials of Symposium, Tashkent, Uzbekistan, 25–27 November 2015; pp. 16–17. [Google Scholar]
- Myrzagalieva, A.B.; Chlachula, J.; Czerniawska, J.; Doláková, N.; Jankovská, V. Vascular Flora and Aquatic Ecosystems of Sibinskye Lakes, East Kazakhstan. 2021, unpublished. [Google Scholar]
- Filonets, P.P. Essays on the Geography of Internal Waters of Central, Southern and Eastern Kazakhstan (Lakes, Reservoirs and Glaciers); Nauka: Alma-Ata, Kazakhstan, 1981; 292p. [Google Scholar]
- Zhensikbayeva, N.Z.; Saparov, K.T.; Chlachula, J.; Yegorina, A.V.; Atasoy, A.; Wendt, J.A. Natural potential of tourism development in southern Altai. Geoj. Tour. Geosites 2018, 21, 200–212. [Google Scholar]
- Chlachula, J. Geotourism perspectives in East Kazakhstan. Geogr. Environ. Sustain. 2019, 12, 29–43. [Google Scholar] [CrossRef]
- Chlachula, J. Biodiversity Protection of Southern Altai in the Context of Contemporary Environmental Transformations and Sustainable Development. Sector 2–3 (East Kazakhstan). In Final Report Field Studies; Irbis: Nerudova, Czech Republic, 2007; p. 223. [Google Scholar]
- Chlachula, J. Geoheritage of East Kazakhstan. Geoheritage 2020, 12, 91. [Google Scholar] [CrossRef]
- Grigoriev, A.A. (Ed.) Kazakhstan. General Physical and Geographical Characteristic; Nauka: Moscow, Russia; Leningrad, Russia, 1950; 492p. [Google Scholar]
- Prairie, Y.T.D.; Bird, D.F.; Coll, J.J. The summer metabolic balance in the epilimnion of southeastern Quebec lakes. Limnol. Oceanogr. 2002, 47, 316–321. [Google Scholar] [CrossRef]
- Lindström, E.S.; Bergström, A.K. Influence of inlet bacteria on bacterioplankton assemblage composition in lakes of different hydraulic retention time. Limnol. Oceanogr. 2004, 49, 125–136. [Google Scholar] [CrossRef]
- Liping, C.; Yin, Z.; Qigen, L.; Zhongjun, H.; Yuejuan, S.; Ziran, P.; Lijing, C. Spatial variations of macrozoobenthos and sediment nutrients in Lake Yangcheng: Emphasis on effect of pen culture of Chinese mitten crab. J. Environ. Sci. 2015, 37, 118–129. [Google Scholar]
- Zaharescu, D.G.; Burghelea, C.I.; Hooda, P.S.; Lester, R.N.; Palanca-Soler, A. Small lakes in big landscape: Multi-scale drivers of littoral ecosystem in alpine lakes. Sci. Total Environ. 2016, 551–552, 496–505. [Google Scholar] [CrossRef]
- Kivila, E.H.; Luoto, T.; Rantala, M.V.; Kiljuneri, M.; Rauio, M.; Nevalainien, L. Environmental controls on benthic food web functions and carbon resource use in subarctic lakes. Freshw. Biol. 2019, 64, 643–658. [Google Scholar] [CrossRef]
- Suturin, A. Biogeochemistry of Lake Baikal Stony Littoral. J. Geosci. Environ. Prot. 2019, 7, 72–79. [Google Scholar] [CrossRef][Green Version]
- Frerencz, B.; Toporowska, M.; Dawidek, J.; Sobolewski, W. Hydro-Chemical Conditions of Shaping the Water Quality of Shallow Łęczna-Włodawa Lakes (Eastern Poland). Clean Soil Air Water 2017. [Google Scholar] [CrossRef]
- Encyclopedia Britannica. 2008. Available online: https://www.britannica.com/ (accessed on 1 May 2021).
- Abakumov, V.A. (Ed.) Guidelines for Methods of Hydrobiological Analysis of Surface Waters and Bottom Sediments; Gidrometeoizdat: Leningrad, Russia, 1983; 240p. [Google Scholar]
- Alipoor, V.; Rahimibashar, M.R.; Aliev, A. Temporal and Spatial Variation of Macrozoobenthos in the Chamkhale Estuary (South Caspian Sea). Middle-East J. Sci. Res. 2011, 10, 654–658. [Google Scholar]
- Kuzmetov, A.R.; Abdinazarov, X.X. Ecological-faunistic characteristics of reservoirs’ zooplankton in Uzbekistan. Eur. J. Chem. Nat. Sci. 2016, 1–2, 3–6. [Google Scholar] [CrossRef]
- Abdinazarov, X.; Madumarov, M.; Haydarov, S. Zooplankton of Sarikamish Lake (Uzbekistan). Open Access Libr. J. 2019, 6, 1–8. [Google Scholar] [CrossRef]
- Kozhova, O.M.; Izmesteva, L.R.; Erbaeva, E.A. A review of the hydrobiology of Lake Khubsugul (Mongolia). Hydrobiologia 1994, 291, 11–19. [Google Scholar] [CrossRef]
- Ermakov, V.V. Geochemical ecology and biogeochemical criteria for estimating the ecologic state of biospheric taxons. Geochem. Int. 2015, 53, 195–212. [Google Scholar] [CrossRef]
- Bazarova, B.B.; Tashlykova, N.A.; Afonina, E.Y.; Kuklin, A.P.; Matafonov, P.V.; Tsybekmitova, G.T.; Gorlacheva, E.-P.; Itigilova, M.T.; Afonin, A.V.; Butenko, M.N. Long-term fluctuations of the aquatic ecosystems in the Onon-Torey plain (Russia). Acta Ecol. Sin. 2019, 39, 157–165. [Google Scholar] [CrossRef]
- Mannig, B.; Müller, M.; Starke, E.; Merkenschlager, C.; Mao, W.; Zhi, X.; Podzun, R.; Jacob, D.; Paeth, H. Dynamical downscaling of climate change in Central Asia. Glob. Planet. Chang. 2013, 110, 26–39. [Google Scholar] [CrossRef]
- Velikovskaya, E.M. Development of the relief of the Southern Altai and Kalba and deep gold-bearing placers. Bull. MOIP Dep. Geol. 1946, 21, 57–77. [Google Scholar]
- Velikovskaya, E.M. Pliocene Quaternary deposits and the development of the Kalba relief. Sci. Notes Mosc. State Univ. Geol. 1947, 3, 58–63. [Google Scholar]
- Abulkabirova, M.A.; Stroeva, M.N. On the age of granite intrusions in Kalba. Geology 1955, 19, 40–50. [Google Scholar]
- Nekhoroshev, V.P. (Ed.) Geology of the USSR. Eastern Kazakhstan. Geology Description; Nedra: Moskow, Russia, 1967; 467p. [Google Scholar]
- Veselova, L.K. Morphostructure of the South-Eastern Kazakhstan Mountains. In Geography of Desertic and Mountain Region of Kazakhstan; Nauka: Moskow, Russia; Leningrad, Russia, 1970; pp. 38–48. [Google Scholar]
- Aubekerov, B.Z. Stratigraphy and Paleogeography of the Plain Zones of Kazakhstan during the Late Pleistocene and Holocene. Development of Landscape and Climate in Northern Asia in Late Pleistocene and Holocene; Nauka: Moskow, Russia, 1993; pp. 101–110. [Google Scholar]
- Galakhov, V.P.; Mukhametov, P.M. Glaciers of the Altai; Nauka: Novosibirsk, Russia, 1999; 136p. [Google Scholar]
- Baibatsha, A.B.; Aubekerov, B.J. Quaternary Geology of Kazakhstan; Gylym: Almaty, Kazakhstan, 2003; p. 147. [Google Scholar]
- Chlachula, J. Pleistocene climate change, natural environments and Palaeolithic occupation of East Kazakhstan. Quat. Int. 2010, 220, 64–87. [Google Scholar] [CrossRef]
- Obruchev, V.A. Selected Works on the Geography of Asia; Geografizdat: Moscow, Russia, 1951; Volume 2, 400p. [Google Scholar]
- Chupakhin, V. Physical Geographyof Kazakhstan; Mektep Press: Almaty, Kazakhstan, 1968; 260p. (In Russian) [Google Scholar]
- Dyachkov, B.A.; Mayorova, N.P.; Chernenko, Z.I. History of East Kazakhstan geological structures development in the Hercynian, Cimmerian and Alpine cycles of tectonic genesis, Part II. In Proceedings of the Ust-Kamenogorsk Kazakh Geographical Society; D. Serikbaev East Kazakhstan State Technical University Press: Ust-Kamenogorsk, Kazakhstan, 2014; pp. 42–48. (In Russian) [Google Scholar]
- Chlachula, J. Gemstones of eastern Kazakhstan. Geologos 2020, 26, 125–148. [Google Scholar] [CrossRef]
- Weather Atlas. 2021. Available online: https://www.weather-atlas.com/en/kazakhstan/ust-kamenogorsk-climate-rainfall (accessed on 1 May 2021).
- Utesheva, A.S. Climate of Kazakhstan; Gidrometeoizdat: Leningrad, Russia, 1959; 368p. [Google Scholar]
- Reference Book of Climate in Kazakhstan. Issue 10. East Kazakhstan Region; RSE “Kazhydromet”: Almaty, Kazakhstan, 2003; 77p.
- Yegorina, A.V. Physical Geography of East Kazakhstan; EHI Press: Ust‘-Kamenogorsk, Kazakhstan, 2002; 181p. (In Russian) [Google Scholar]
- Yegorina, A.V. The Climate of Southwest Altai; Textbook: Semey, Kazakhstan, 2015; 315p. [Google Scholar]
- Domanitskiy, A.P. The Rivers and Lakes of the USSR; Gidrometeoizdat: Almaty, Kazakhstan, 1971; 104p. (In Russian) [Google Scholar]
- Samarkhanov, T.N.; Yegorina, A.V.; Myrzagalieva, A.B.; Saparov, K.T. Climate factors as an evaluation componentof the recreation potential at the Sibinskiye lakes. Hydrometeorol. Ecol. 2019, 3, 7–21. (In Russian) [Google Scholar]
- Lurie, Y.Y. (Ed.) Unified Methods of Analysis of Waters, 2nd ed.; Chemistry: Moscow, Russia, 1973; 376p. [Google Scholar]
- Semenov, A.D. (Ed.) Manual to Chemical Analysis of Surface Waters of the Land; Gidrometeoizdat: Leningrad, Russia, 1977; 541p. [Google Scholar]
- Reznikov, A.A.; Kulikowska, E.P.; Sokolov, I.Y. Methods of Analysis of Natural Waters, 3rd ed.; Nedra: Moscow, Russia, 1970; 488p. [Google Scholar]
- Woodiwiss, F.S. Biotic index of the Trent River. Macroinvertebrates and Biological Studies. In Scientific Principles of Surface Water Quality Control by Hydrobiological Indices. Proceedings USSR-British Work; Gidrometeoizdat: Leningrad, Russia, 1977; pp. 132–161. [Google Scholar]
- Sharapova, L.I.; Falomeeva, A.P. Methodological Manual for Hydrobiological Fishery Studies of Water Bodies of Kazakhstan (Plankton, Zoobentos); Research Institute of Fisheries of the Republic of Kazakhstan: Almaty, Kazakhstan, 2006; 27p. [Google Scholar]
- Manuilova, E.F. Cladocera (Cladocera) of the Fauna of the USSR; Nauka: Moscow, Russia; Leningrad, Russia, 1964; 326p. [Google Scholar]
- Kutikova, L.A. Rotifers of the Fauna of the USSR (Rotatoria); Nauka: Leningrad, Russia, 1970; 744p. [Google Scholar]
- Kutikova, L.A.; Starobogatov, Y.I. (Eds.) Keys to Freshwater Invertebrates of the European Part of the USSR; Gidrometeoizdat: Leningrad, Russia, 1977; 512p. [Google Scholar]
- Ibrasheva, S.I.; Smirnova, V.A. Cladocera of Kazakhstan; Mektep: Alma-Ata, Kazakh, 1983; 135p. [Google Scholar]
- Balushkina, E.V.; Vinberg, G.G. The relationship between body weight and length in planktonic animals. In General Foundations of the Study of Aquatic Ecosystems; Vinberg, G.G., Ed.; Nauka: Leningrad, Russia, 1979; pp. 169–172. [Google Scholar]
- Shitikov, V.K. Quantitative Hydroecology: Methods, Criteria, Solutions; Book 1; Kriskunov, E.A., Ed.; Nauka: Moscow, Russia, 2005; 281p. [Google Scholar]
- Chernovsky, A.A. Keys to the Larvae of Mosquitoes of the Family Tendipedidae; USSR, Academy of Sciences: Moscow, Russia; Leningrad, Russia, 1949; 186p. [Google Scholar]
- Pankratova, V.Y. Larvae and Pupae of Mosquitoes of the Subfamily Podonominae and Tanypodinae of the USSR Fauna; Nauka: Leningrad, Russia, 1977; 154p. [Google Scholar]
- Pankratova, V.Y. Larvae and Pupae of Mosquitoes of the Subfamily Chironominae of the Fauna of the USSR; Nauka: Leningrad, Russia, 1983; 296p. [Google Scholar]
- Keskitalo, J.; Salonen, K. Manual for Integrated Monitoring. Subprogramme Hydrobiology of Lakes; Publications of the Water and Environment Administration: Helsinki, Finland, 1994; Volume 16, pp. 26–33. [Google Scholar]
- Kitaev, S.P. Ecological Bases of Biological Productivity of Lakes in Different Natural Zones; Nauka: Moscow, Russia, 1984; 207p. [Google Scholar]
- Sládeček, V. System of Water Quality from the Biological Point of View. Arch. Hidrobiol. Ergeb. Limnol. 1973, 7, 1–218. [Google Scholar]
- Alimov, A.F. Regularities of changes in the structural and functional characteristics of communities of aquatic organisms. Hydrobiol. J. 1995, 31, 3–11. [Google Scholar]
- Bakanov, A.I. Using zoobenthos for monitoring freshwater reservoirs (review). Biol. Intern. Waters 2000, 1, 68–82. [Google Scholar]
- Macioszczyk, A.; Dobrzyński, D. Hydrogeochemistry. Zone of Active Groundwater Exchange; PWN: Warszawa, Poland, 2007; 448p. (In Polish) [Google Scholar]
- Konstantinov, A.S. General Hydrobiology; Nauka: Moskow, Russia, 1986; 472p. [Google Scholar]
- Shitikov, V.K.; Zinchenko, T.D.; Golovatyuk, L.V. Assessing Surface Water Quality Based on Indicator Zoobenthos Species. Water Resour. 2004, 31, 323–332. [Google Scholar] [CrossRef]
- Perus, J.; Bonsdorff, E.; Baeck, S.; Lax, H.-G.; Villnaes, A.; Westberg, V. Zoobenthos as Indicators of Ecological Status in Coastal Brackish Waters: A Comparative Study from the Baltic Sea. Ambio A J. Hum. Environ. 2007, 36, 250–256. [Google Scholar] [CrossRef]
- Pešić, V.; Gadawski, P.; Gligorović, B.; Glöer, P.; Grabowski, M.; Kováče, T.; Murány, D.; Płóciennik, M.; Šundić, D. The Diversity of the zoobenthos communities of the Lake Skadar/Shkodra Basin. In The Skadar/Shkodra Lake Environment, The Handbook of Environmental Chemistry; Pešić, V., Karaman, G., Kostianoy, A., Eds.; Springer: Cham, Switzerland, 2018; Volume 80, pp. 255–294. [Google Scholar] [CrossRef]
- Kurmangaliyeva, S.G. Seasonal dynamics of zooplankton in the Lower Kolsay Lakes. Biol. Sci. 1974, 7, 87–91. [Google Scholar]
- Krupa, E.G. Zooplankton in Lothic and Limnetic Ecosystems of Kazakhstan. Structure, Patterns Offormation; Palmarium Academic Publishing: Saarbrucken, Germany, 2012; 346p. [Google Scholar]
- Smirnova, D.A. State of zooplankton of the Middle and Lower Kolsay Lakes (Chilik river watershed) during the beginning of their recreational use. Kazakh Natl. Univ. Bull. Biol. Ser. 2000, 4, 54–60. [Google Scholar]
- Krupa, E.G.; Barinova, S.M.; Romanova, S.M.; Malybekov, A.B. Hydrobiologicalassessment of the high mountain Kolsay Lakes (Kungey Alatau, Southeastern Kazakhstan) ecosystems in climatic gradient. Br. J. Environ. Clim. Chang. 2016, 6, 259–278. [Google Scholar] [CrossRef]
- Krupa, E.G.; Barinova, S.S.; Romanova, S.M. Zooplankton size structure in the Kolsay Mountain Lakes (Kungei Alatau, southeastern Kazakhstan) and its relationships with environmental Factors. Water Resour. 2019, 46, 403–414. [Google Scholar] [CrossRef]
- Bolduc, P.; Bertolo, A.; Pinel-Alloul, B. Does submerged aquatic vegetation shape zooplankton community structure and functional diversity? A test with a shallow fluvial lake system. Hydrobiologia 2016, 778, 151–165. [Google Scholar] [CrossRef]
- Yapiyev, V.; Samarkhanov, K.; Tulegenova, N.; Jumassultanova, S.; Verhoef, A.; Saidaliyeva, Z.; Umirov, N.; Sagintayev, Z.; Namazbayeva, A. Estimation of water storage changes in small endorheic lakes in Northern Kazakhstan. J. Arid Environ. 2019, 160, 42–55. [Google Scholar] [CrossRef]
- Wu, J.; Liu, W.; Zeng, H.; Ma, L.; Bai, R. Water quantity and quality of xix lakes in the arid Xinjiang region, NW China. Environ. Process. 2014, 1, 115–125. [Google Scholar] [CrossRef]
- Kamp, J.; Koshkin, M.A.; Hrabina, T.M.; Katzner, T.N.; Milner-Gulland, J.E.; Schreiber, D.; Sheldon, R.; Schmalenko, A.; Smelansky, I.; Terraube, J.; et al. Persistent and novel threats to the biodiversity of Kazakhstan’s steppes and semi-deserts. Biodivers. Conserv. 2016, 25, 2521–2541. [Google Scholar] [CrossRef]
- Zhaglovskaya, A.; Chlachula, J.; Thevs, N.; Myrzagaliyeva, A.; Aidosova, S. Natural regeneration potential of the black saxaul shrub-forests in semi-deserts of Central Asia–the Ili River delta area, SE Kazakhstan. Pol. J. Ecol. 2017, 3, 352–368. [Google Scholar] [CrossRef]
- Bayandinova, S.; Mamutov, Z.; Ispanova, G. Man-Made Ecology of East Kazakhstan; Environmental Science and Engineering; Springer Nature Press: Singapore, 2018; 136p. [Google Scholar]






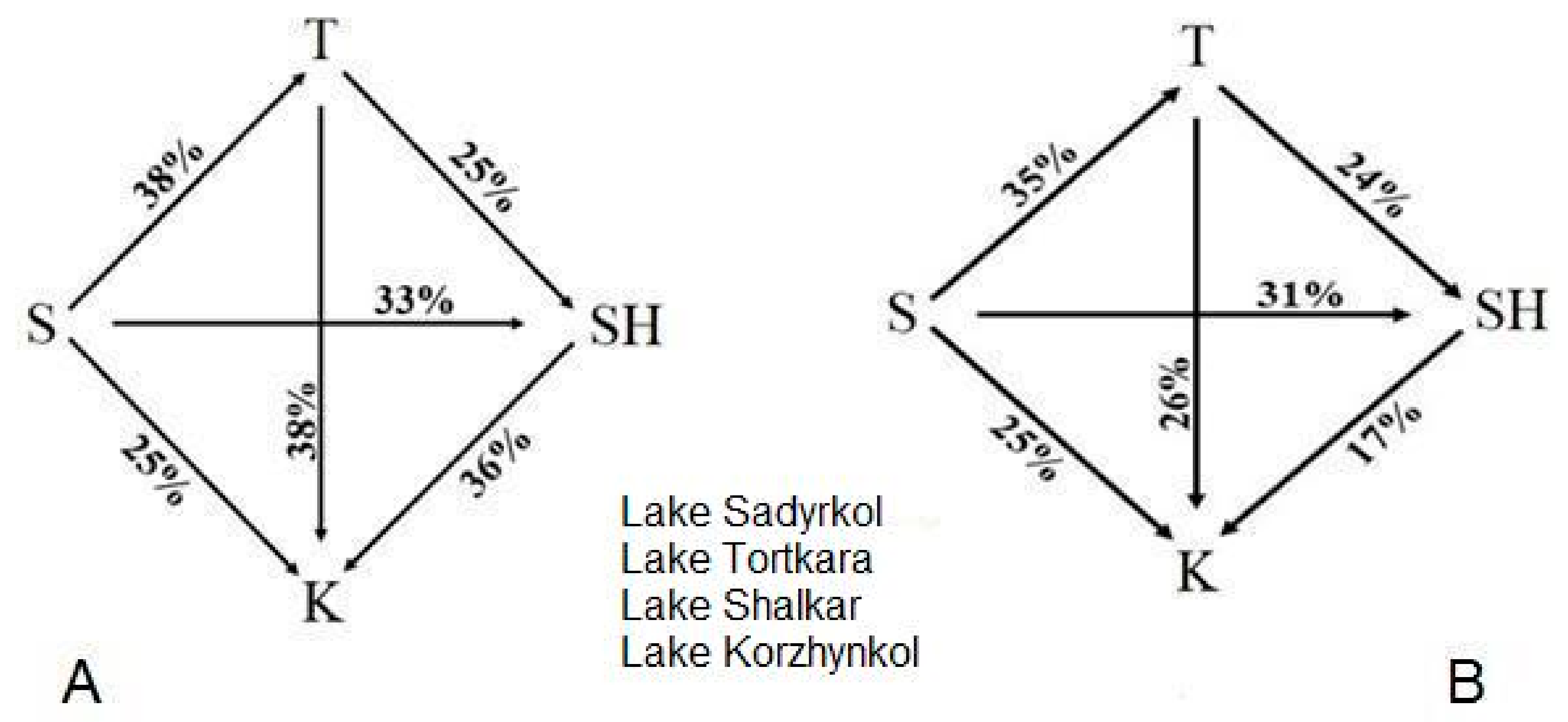
| LAKES | O2− * | pH | NH4+ * | NO3− * | NO2− * | *HCO3− | Hardness ** | K+ * | Ca2+ * | CO32 * | Mg2+ * | Na+ * | Permanganate Number *** | SO₄2− * | Residue * | PO43− * | Cl− * |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S | 10.93 | 9.13 | 0.297 | ˂0.1 | ˂0.007 | 137.5 | 1.20 | 2.40 | 14.8 | 15.0 | 5.4 | 5.99 | 2.8 | 25.2 | 85.0 | 0.039 | 5.33 |
| T | 11.20 | 9.16 | 0.274 | ˂0.1 | ˂0.007 | 129.6 | 1.16 | 2.18 | 14.2M | 12.0 | 5.4 | 5.68 | 2.72 | 22.6 | 83.6 | 0.03 | 5.10 |
| Sh | 12.47 | 9.08 | 0.375 | ˂0.1 | ˂0.007 | 127.3 | 1.27 | 3.66 | 16.5 | 12.0 | 5.3 | 8.15 | 3.61 | 30.5 | 92.8 | 0.055 | 5.10 |
| K | 11.22 | 9.07 | 0.327 | ˂0.1 | ˂0.007 | 125.8 | 1.21 | 2.85 | 17.8 | 9.0 | 3.8 | 7.82 | 2.94 | 27.5 | 90.0 | 0.044 | 3.94 |
| Taxon | D/s | Frequency of Occurrence (%) | |||
|---|---|---|---|---|---|
| Sadyrkol | Tortkara | Shalkar | Korzhynkol | ||
| Rotifera | |||||
| Keratella cochlearis (Gosse) | β-o | - | - | - | 50 |
| Copepoda | |||||
| Eudiaptomus sp. | - | - | 50 | - | - |
| Acanthocyclops sp. | - | - | 50 | - | 100 |
| Cyclops vicinus (Uljanine) | β | - | 50 | - | 100 |
| Mesocyclops leuckarti (Claus) | o | 100 | 50 | 50 | 50 |
| Cladocera | |||||
| Diaphanosoma brachyurum (Lievin) | o-β | - | 50 | 50 | 100 |
| Ceriodaphnia quadrangula (Muller) | β | 100 | 50 | - | 50 |
| Bosmina longirostris (Muller) | o-β | - | - | 50 | 50 |
| Bosmina longispina (Leydig) | β | - | - | - | 50 |
| Daphnia longispina (Muller) | β | 50 | - | 100 | 50 |
| Daphnia cucullata (Sars) | β | - | - | 50 | 50 |
| Pleuroxus sp. | - | - | - | - | 50 |
| Chydorus sphaericus(Muller) | β | - | - | 50 | - |
| Alona rectangular (Sars) | o | - | - | 50 | - |
| Simocephalus vetulus (Muller) | o-β | 50 | 50 | 50 | - |
| Total nubmers/species richness | 4 | 7 | 8 | 11 | |
| Zooplankton Category/Zone | Sadyrkol | Tortkara | Shalkar | Korzhynkol | ||||
|---|---|---|---|---|---|---|---|---|
| Littoral | Pelagial | Littoral | Pelagial | Littoral | Pelagial | Littoral | Pelagial | |
| Rotifera | - | - | - | - | - | - | 4.8 | - |
| Copepoda | 27.3 | 3.5 | 1.19 | 2.31 | - | 3.74 | 73.2 | 11.01- |
| Cladocera | 21.7 | 1.6 | - | 1.11 | 9.15 | 14.33 | 17.1 | 7.96 |
| Total | 49.0 | 5.1 | 1.19 | 3.42 | 9.15 | 18.07 | 95.1 | 18.97 |
| Zooplankton Category/Zone | Sadyrkol | Tortkara | Shalkar | Korzhynkol | ||||
|---|---|---|---|---|---|---|---|---|
| Littoral | Pelagial | Littoral | Pelagial | Littoral | Pelagial | Littoral | Pelagial | |
| Rotifera | - | - | - | - | - | - | 6 | - |
| Copepoda | 274 | 123 | 119 | 62 | - | 62 | 915 | 180 |
| Cladocera | 627 | 102 | - | 66 | 722 | 1531 | 610 | 496 |
| Total | 901 | 225 | 119 | 128 | 722 | 1593 | 1531 | 676 |
| Sibe Lakes | Shannon Index | Uniformity Index | Biotic Littoral Index | Water Quality Class |
|---|---|---|---|---|
| Lake Sadyrkol | 2.61 ± 0.11 | 0.52 ± 0.13 | 9 | II (clean) |
| Lake Tortkara | 2.38 ± 0.19 | 0.43 ± 0.02 | 7 | II (clean) |
| Lake Shalkar | 2.09 ± 0.22 | 0.40 ± 0.04 | 6 | III (slightly polluted) |
| Lake Korzhynkol | 1.94 ± 0.05 | 0.58 ± 0.14 | 8 | II (clean) |
| Benthos Group | Lake Sadyrkol | Lake Tortkara | Lake Shalkar | Lake Korzhynkol | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Littoral | Pelagial | Littoral | Pelagial | Littoral | Pelagial | Littoral | Pelagial | |||||||||
| A | B | A | B | A | B | A | B | A | B | A | B | P | B | P | B | |
| Mollusca | 32.0 | 10.02 | 40 | 0.32 | 440 | 20.6 | 40 | 0.14 | 280 | 738 | 280 | 8.52 | 520 | 3.98 | 200 | 1.52 |
| Larvae Odonata | 40 | 0.20 | - | - | - | - | 80 | 9.64 | - | - | - | - | 40 | 0.60 | - | - |
| Larvae Ephemeroptera | 280 | 0.90 | - | - | - | - | 80 | 0.12 | - | - | - | - | 200 | 0.25 | - | - |
| Larvae Trichoptera | - | - | - | - | 40 | 0.16 | - | - | - | - | 40 | 0.17 | - | - | ||
| Larvae Coleoptera | 120 | 0.54 | - | - | - | - | 80 | 0.27 | - | - | - | - | 80 | 0.25 | - | - |
| Gammaridae | 560 | 1.84 | - | - | - | - | - | - | - | - | 480 | 2.48 | 7120 | 22.21 | - | - |
| Larvae Ceratopogonidae | - | - | - | - | 40 | 0.19 | - | - | - | - | - | - | - | - | ||
| Larvae Chironomidae | 1400 | 8.24 | 200 | 1.28 | 120 | 0.96 | 280 | 0.53 | 520 | 16.36 | 480 | 3.36 | 160 | 0.44 | 40 | 0.52 |
| Oligochaeta | 200 | 0.04 | - | - | - | - | 40 | 0.04 | 80 | 0.24 | - | - | - | - | - | - |
| Hirudinea | 40 | 0.16 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Sialidae | 40 | 1.28 | - | - | - | - | - | - | 40 | 0.28 | - | - | - | - | - | - |
| Hydracarina | 200 | 0.28 | - | - | 40 | 0.14 | - | - | - | - | 40 | 0.08 | - | - | ||
| Total | 3200 | 20.5 | 240 | 1.60 | 560 | 21.56 | 720 | 11.23 | 920 | 24.24 | 1240 | 14.37 | 8200 | 27.98 | 240 | 2.04 |
| Trophic class | high | low | high | increased | high | increased | high | low | ||||||||
| Lake average | A–1720 B–11.05 | A–640 B–16.40 | A–1080 B–19.31 | A–4220 B–15.01 | ||||||||||||
| Trophic class | increased | increased | increased | increased | ||||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samarkhanov, T.N.; Myrzagaliyeva, A.B.; Chlachula, J.; Kushnikova, L.B.; Czerniawska, J.; Nigmetzhanov, S.B. Geoenvironmental Implications and Biocenosis of Freshwater Lakes in the Arid Zone of East Kazakhstan. Sustainability 2021, 13, 5756. https://doi.org/10.3390/su13105756
Samarkhanov TN, Myrzagaliyeva AB, Chlachula J, Kushnikova LB, Czerniawska J, Nigmetzhanov SB. Geoenvironmental Implications and Biocenosis of Freshwater Lakes in the Arid Zone of East Kazakhstan. Sustainability. 2021; 13(10):5756. https://doi.org/10.3390/su13105756
Chicago/Turabian StyleSamarkhanov, Talant N., Anar B. Myrzagaliyeva, Jiri Chlachula, Ludmila B. Kushnikova, Jolanta Czerniawska, and Sayan B. Nigmetzhanov. 2021. "Geoenvironmental Implications and Biocenosis of Freshwater Lakes in the Arid Zone of East Kazakhstan" Sustainability 13, no. 10: 5756. https://doi.org/10.3390/su13105756
APA StyleSamarkhanov, T. N., Myrzagaliyeva, A. B., Chlachula, J., Kushnikova, L. B., Czerniawska, J., & Nigmetzhanov, S. B. (2021). Geoenvironmental Implications and Biocenosis of Freshwater Lakes in the Arid Zone of East Kazakhstan. Sustainability, 13(10), 5756. https://doi.org/10.3390/su13105756





