Bambara Groundnut (Vigna subterranea L. Verdc): A Crop for the New Millennium, Its Genetic Diversity, and Improvements to Mitigate Future Food and Nutritional Challenges
Abstract
1. Introduction
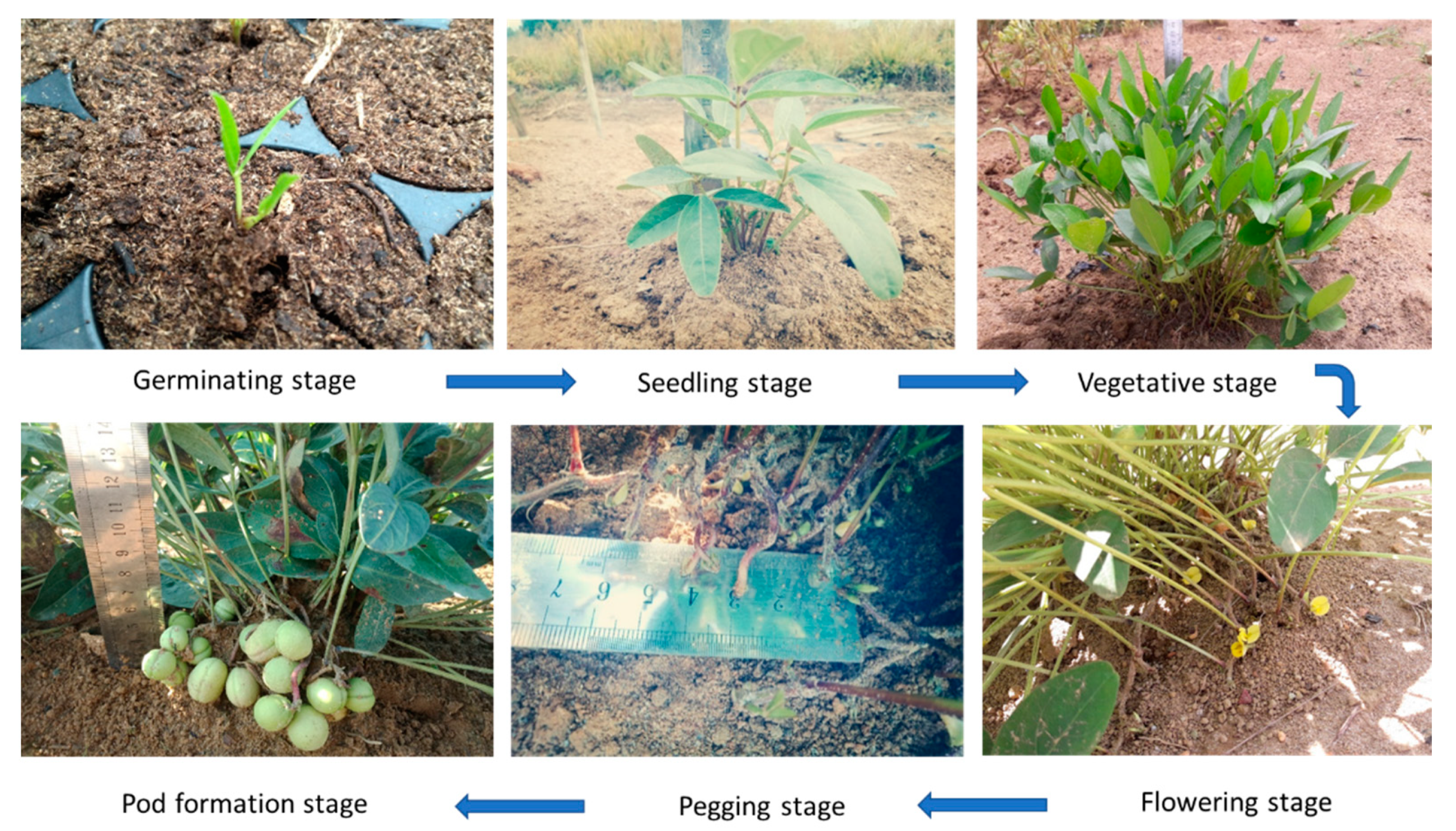
2. Botanical Description of Bambara Groundnut
Agronomic Attributes of Bambara Groundnut
3. Production Scenario of Bambara Groundnut
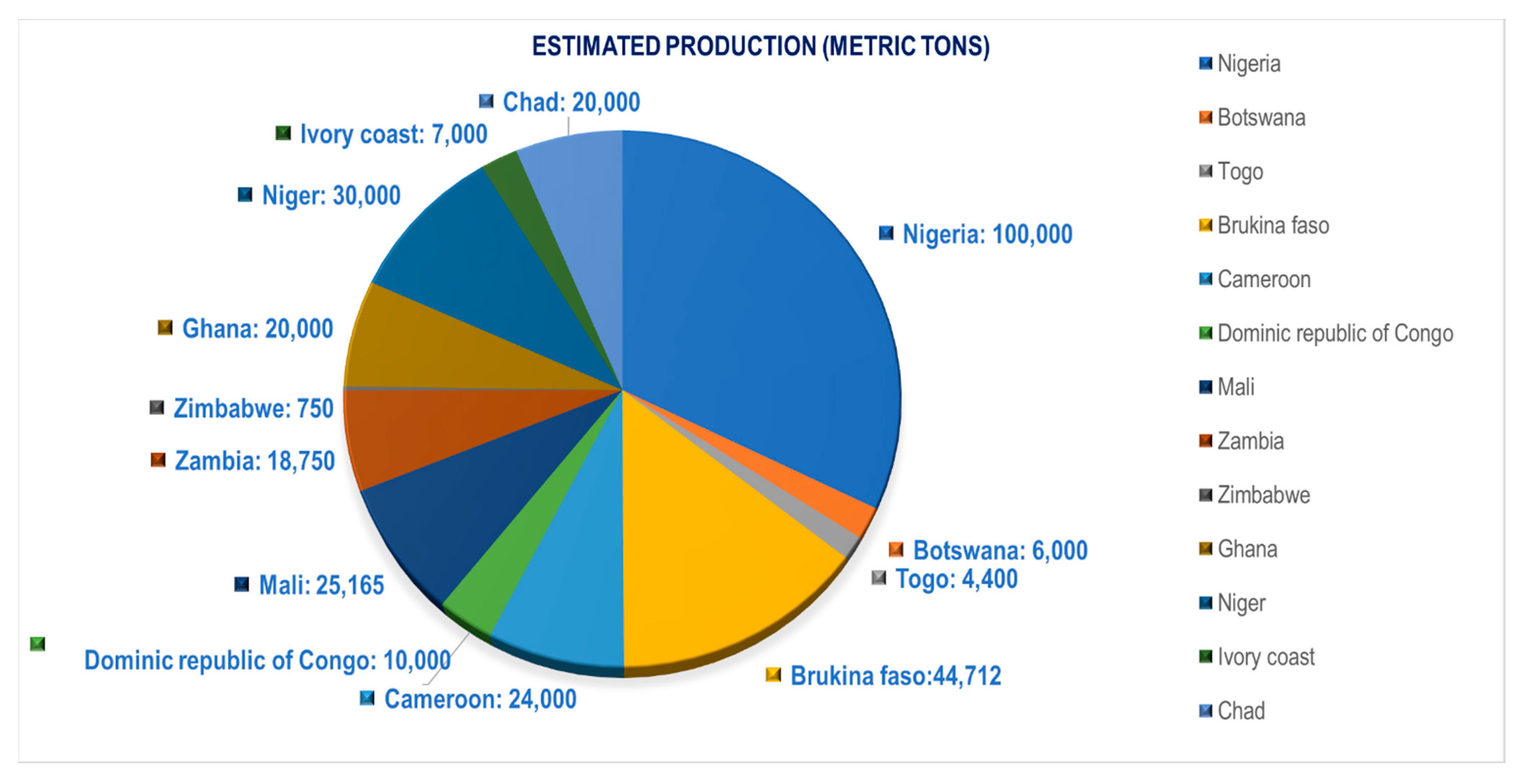
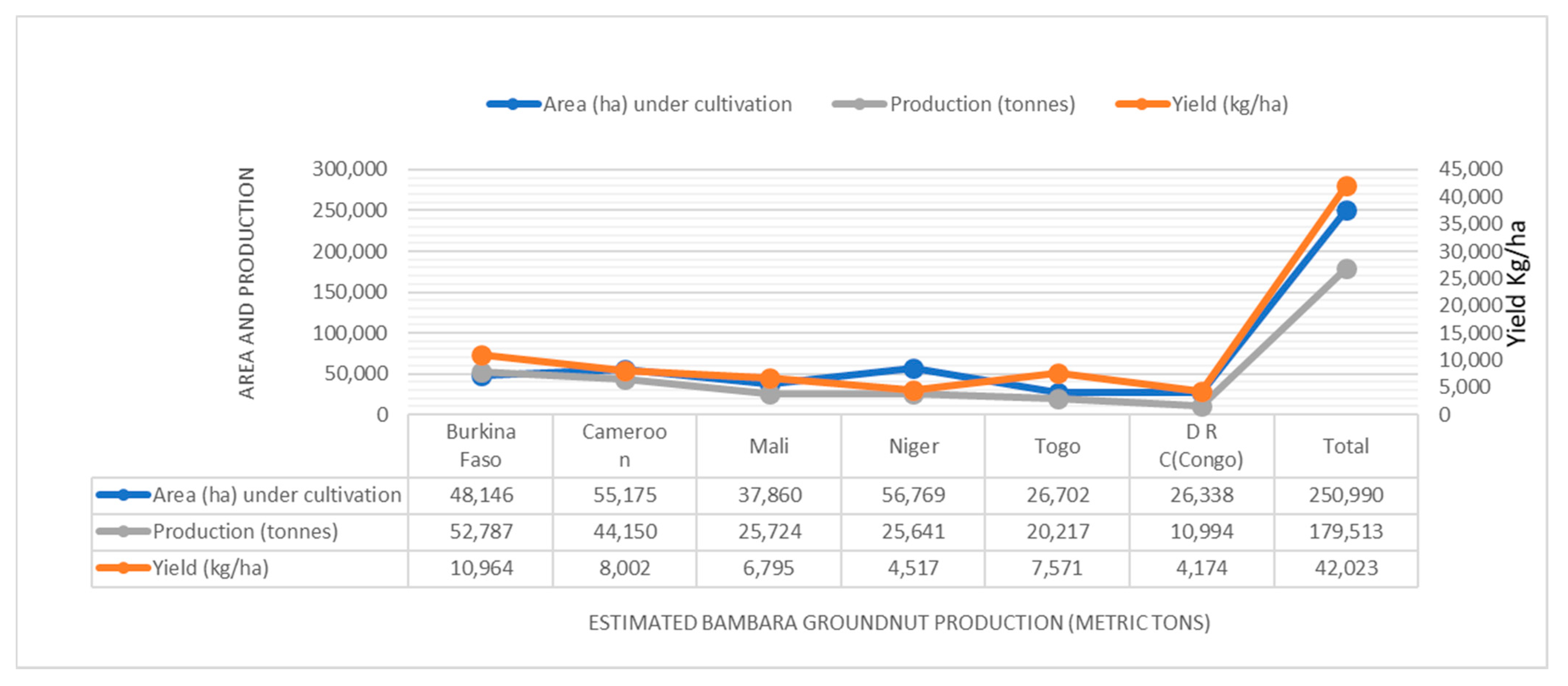
3.1. Major Production Areas
3.2. General Production Scenario in Bambara Groundnut
3.3. Center of Origin and Distribution of Bambara Groundnut
4. Potential Uses of Bambara Groundnut for Food Security
4.1. Health and Medicinal Significance of Bambara Groundnut
4.2. Role of Bambara Groundnut to Overcome Malnutrition and Nutritional Gap
4.3. Anti-Nutritional Ingredients in Bambara Groundnut
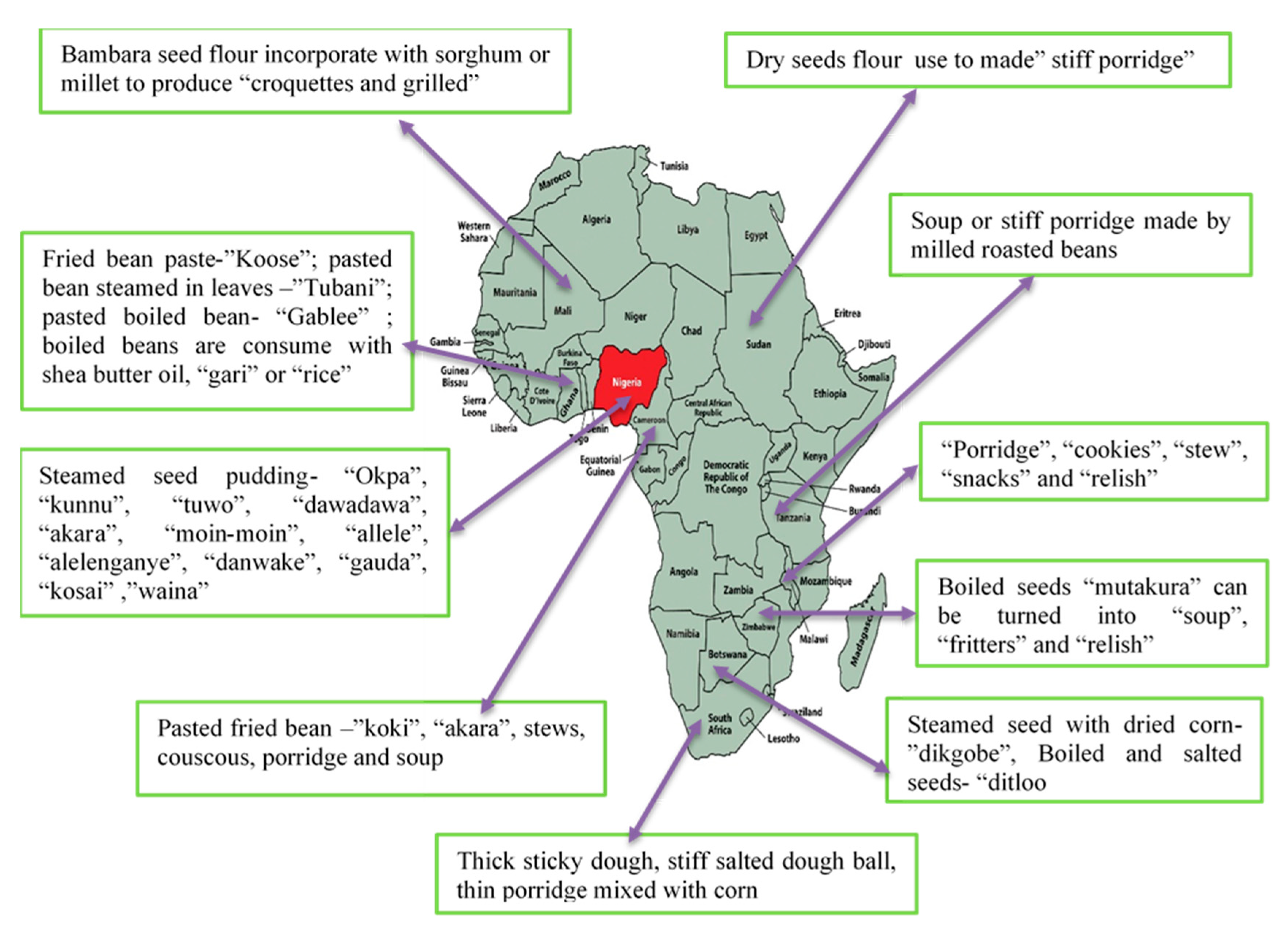
4.4. Processing of Bambara Groundnut
5. Bambara Groundnut Considered Underutilized Crops: Why?
- I.
- Certified food and nutrient values.
- II.
- The plant has been broadly grown in the past, or the plant is presently grown in a short geological region.
- III.
- Directly grown less than other comparable plant species.
- IV.
- The traditional or cultural heritage of their area of origin.
- V.
- Their cultivation and usage are poorly documented.
- VI.
- Adaptation to a certain agro-ecological zone.
- VII.
- Adaptation to marginal and poor soil conditions.
- VIII.
- Limited or no established seed supply scheme.
6. Genetic Assets of Bambara Groundnut and Its Improvement
6.1. Characterization and Conservation of Genetic Resources
6.2. Genetic Improvement of Existing Bambara Groundnut Germplasms
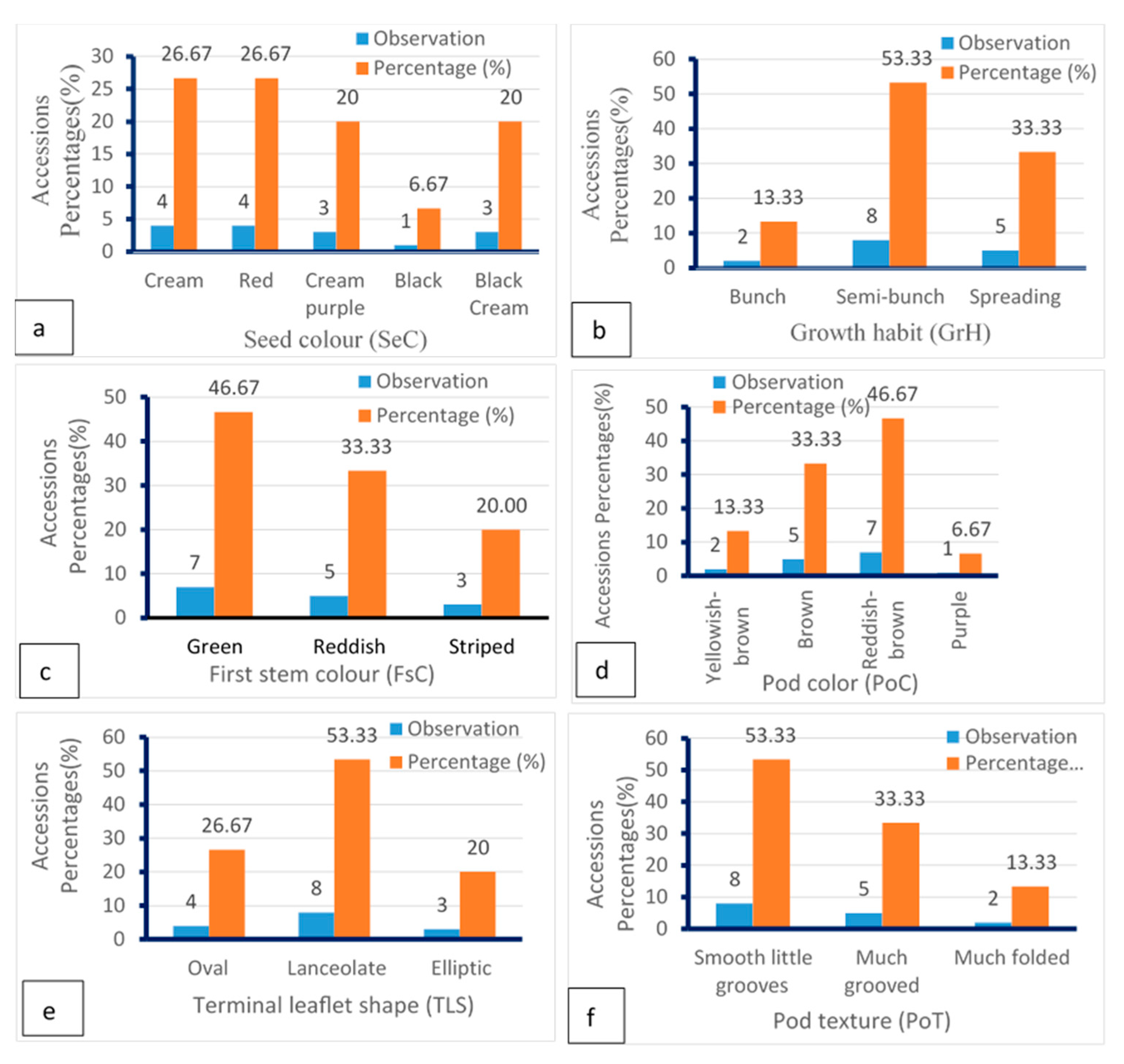
7. Qualitative and Quantitative Morphological Diversity Studied
8. Research on Hybridization for Bambara Groundnut Improvement
9. Specific Molecular “Toolbox” Used for Bambara Groundnut Improvement
10. Bambara Groundnut Improvement Research on Future Aspect
10.1. Advanced Genomics
10.2. Advanced Transcriptomics
10.3. TILLING and Eco-TILLING: Efficient and Rapid Approaches to Crop Improvement
10.4. Genetic Mapping and Recombinant Inbred Lines (RILs)
11. The Resulting Features Need to Be Acknowledged to Promote Research on This Underutilized Crop
- Heterosis vigor in a breeding scheme causes variety improvement. High levels of allelic diversity in the genotypes (between and within genotype) and across geographical origin are observed in the Bambara groundnut that causes high heterosis.
- From local landraces, pure lines should be developed after examining their stability in diverse climates (first in regulated climates such as greenhouses and then in open field environments).
- Multi-lines (a mixture of landraces) should be raised for a group of characteristics to adjust certain planting environments and other growers/consumers’ choices.
- Mega-environmental trials (METs) should be evaluated considering the genetic and agronomic attributes over a range of environments.
- To introduce additional beneficial traits, pure lines should be used in crossing programs.
- To discover important Quantitative Trait Loci (QTLs), genomic maps from F2, genetically engineered (GE), or transgenic and recombinant inbred lines should be generated.
- In underutilized Bambara groundnut research, the application of advanced techniques including MAS, TILLING, tissue culture, and transgenic techniques should be encouraged.
- Future research on Bambara groundnut should be focused on four aspects: i) increasing yield per unit area, ii) breeding for tolerance to biotic and abiotic stresses, iii) enhancing seed size and nutritional quality through bio-fortification, and iv) manufacturing for markets and value-added products.
- The involvement of financial and technical support to researchers and institutions is encouraged in Bambara groundnut research.
- A network or unique research platform should be created at different levels: national, regional, and international.
- Effective conferences, workshops, and training related to Bambara groundnut research and development should be organized.
12. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. World Food and Agriculture–Statistical Pocketbook; FAO: Rome, Italy, 2019. [Google Scholar]
- FAO; IFAD; UNICEF; WFP; WHO. The State of Food Security and Nutrition in the World; FAO; IFAD; UNICEF; WFP; WHO: Rome, Italy, 2020. [Google Scholar]
- FAO. The Future of Food and Agriculture–Alternative Pathways to Rome. 2018. Available online: http://www.fao.org/3/I8429EN/i8429en.pdf (accessed on 1 October 2018).
- EAT-Lancet Commission. EAT-Lancet Commission Summary Report. Available online: https://eatforum.org/content/uploads/2019/07/EATLancet_Commission_Summary_Report.pdf (accessed on 28 October 2019).
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Toulmin, C. Food security: The challenge of feeding 9 billion people. Science 2010, 327, 812–818. [Google Scholar] [CrossRef]
- Godfray, H.C.J.; Blacquiere, T.; Field, L.M.; Hails, R.S.; Petrokofsky, G.; Potts, S.G.; Raine, N.E.; Vanbergen, A.J.; McLean, A.R. A restatement of the natural science evidence base concerning neonicotinoid insecticides and insect pollinators. Proc. R. Soc. B Biol. Sci. 2014, 281, 20140558. [Google Scholar] [CrossRef]
- Khan, F.; Chai, H.H.; Ajmera, I.; Hodgman, C.; Mayes, S.; Lu, C. A transcriptomic comparison of two Bambara groundnut landraces under dehydration stress. Genes 2017, 8, 121. [Google Scholar] [CrossRef]
- Mayes, S.; Massawe, F.J.; Alderson, P.G.; Roberts, J.A.; Azam-Ali, S.N.; Hermann, M. The potential for underutilized crops to improve security of food production. J. Exp. Bot. 2012, 63, 1075–1079. [Google Scholar] [CrossRef] [PubMed]
- Chanyalew, S.; Ferede, S.; Damte, T.; Fikre, T.; Genet, Y.; Kebede, W.; Tolossa, K.; Tadele, Z.; Assefa, K. Significance and prospects of an orphan crop tef. Planta 2019. [Google Scholar] [CrossRef]
- Ho, W.K.; Chai, H.H.; Kendabie, P.; Ahmad, N.S.; Jani, J.; Massawe, F.; Kilian, A.; Mayes, S. Integrating genetic maps in Bambara groundnut [Vigna subterranea (L) Verdc.] and their syntenic relationships among closely related legumes. BMC Genom. 2017, 18. [Google Scholar] [CrossRef]
- Feldman, A.; Ho, W.K.; Massawe, F.; Mayes, S. Bambara Groundnut is a climate-resilient crop: How could a drought-tolerant and nutritious legume improve community resilience in the face of climate change? In Sustainable Solutions for Food Security; Springer: Cham, Switzerland, 2019; pp. 151–167. [Google Scholar]
- Frison, E.A.; Cherfas, J.; Hodgkin, T. Agricultural biodiversity Is essential for a sustainable improvement in food and nutrition security. Sustainability 2011, 3, 238–253. [Google Scholar] [CrossRef]
- Ntundu, W.H.; Shillah, S.A.; Marandu, W.Y.F.; Christiansen, J.L. Morphological diversity of Bambara groundnut [Vigna subterranea (L.) Verdc.] landraces in Tanzania. Genet. Resour. Crop Evol. 2006, 53, 367–378. [Google Scholar] [CrossRef]
- Ahmad, N.S. Bambara Groundnut, the Crop for the New Millennium: Molecular Techniques to Improve the Resiliency of Bambara Groundnut; Lambert Academic Publishers: Sunnyvale, CA, USA, 2012. [Google Scholar]
- Oyugi, M.; Nandi, O.M.J.; Amudavi, D.; Palapala, V. Influence of gender on farmers’ level of involvement in bambara production activities in Western Kenya. Asian J. Agril. Ext. Econ. Sociol. 2015, 4, 231–246. [Google Scholar] [CrossRef][Green Version]
- Halimi, A.R.; Mayes, S.; Barkla, B.; King, G. The potential of the underutilized pulse Bambara groundnut [Vigna subterranea (L.) Verdc.] for nutritional food security. J. Food Compos. Anal. 2019, 77, 47–59. [Google Scholar] [CrossRef]
- Chivenge, P.; Mabhaudhi, T.; Modi, A.T.; Mafongoya, P. The potential role of neglected and underutilised crop species as future crops under water scarce conditions in Sub-Saharan Africa. Int. J. Environ. Res. Public Health 2015, 12, 5685–5711. [Google Scholar] [CrossRef]
- Harouna, D.V.; Venkataramana, P.B.; Ndakidemi, P.A.; Matemu, A.O. Under-exploited wild Vigna species potentials in human and animal nutrition: A review. Glob. Food Secur. 2018, 18, 1–11. [Google Scholar] [CrossRef]
- Olayide, O.E.; Donkoh, S.A.; Ansah, I.G.K.; Adzawla, W.; O’Reilly, P.J.; Mayes, S.; Feldman, A.; Halimi, R.A.; Nyarko, G.; Ilori, C.O.; et al. Assessing socioeconomic factors influencing production and commercialization of Bambara groundnut as an indigenous climate resilient crop in Nigeria. In Handbook of Climate Change Resilience; Leal Filho, W., Ed.; Springer: Cham, Switzerland, 2018. [Google Scholar] [CrossRef]
- Paliwal, R.; Adegboyega, T.T.; Abberton, M.; Faloye, B.; Oyatomi, O. Potential of genomics for the improvement of underutilized legumes in sub-Saharan Africa. Legume Sci. 2021, 1–16. [Google Scholar] [CrossRef]
- Lin Tan, X.; Azam-Ali, S.; Goh, E.V.; Mustafa, M.A.; Chai, H.H.; Kuan Ho, W.; Mayes, S.; Mabhaudhi, T.; Azam-Ali, S.; Massawe, F. Bambara groundnut: An underutilized leguminous crop for global food security and nutrition. Front. Nutr. 2020, 7, 276. [Google Scholar] [CrossRef]
- Pabendon, M.B.; Azrai, M.; Kasim, F.; Mejaya, M.J. Prospek penggunaan markah molekuler dalam program pemuliaan jagung. Balitsereal 2007, 110–133. Available online: http://balitsereal.litbang.pertanian.go.id/wp-content/uploads/2016/11/delapan.pdf (accessed on 14 May 2021).
- Oladosu, Y.; Rafii, M.Y.; Abdullah, N.; Magaji, U.; Miah, G.; Hussin, G.; Ramli, A. Genotype× Environment interaction and stability analyses of yield and yield components of established and mutant rice genotypes tested in multiple locations in Malaysia. Acta Agric. Scand. B Soil Plant Sci. 2017, 67, 590–606. [Google Scholar] [CrossRef]
- Bamshaiye, O.M.; Adegbola, J.A.; Bamishaiye, E.I. Bambara groundnut: An under-utilized nut in Africa. Adv. Agril. Biotechnol. 2011, 1, 60–72. [Google Scholar]
- Linnemann, A.R.; Azam-Ali, S. Bambara nut (Vigna subterranean (L.) Verdc. In Underutilized Crops Series 2 Vegetables and Pulses; Chapman and Hall: London, UK, 1993; pp. 13–57. [Google Scholar]
- Toungos, M.D.; Sajo, A.A.; Gungula, D.T. Recommended fertilizer levels on Bambara groundnut (Vigna subterranea (L) Verde) in Yola Adamawa State, Nigeria. Agric. J. 2009, 4, 14–21. [Google Scholar]
- Bamnetwork. Bambara Groundnut. Crops. Available online: http://www.bambaragroundnut.org (accessed on 5 January 2017).
- Swanevelder, C.J. Bambara-Food for Africa; National Department of Agriculture of the ARC-Grain Crops Institute: Potchefstroom, South Africa, 1998; p. 16. [Google Scholar]
- Ojimelukwe, P.C.; Ayernor, G.S. Oligosaccharide composition and functional properties of flour and starch isolates from four cultivars of Bambarra groundnut seeds. J. Food Sci. Technol. 1992, 29, 319–321. [Google Scholar]
- Yakubu, H.; Kwari, J.D.; Sandabe, M.K. Effect of phosphorus fertilizer on nitrogen fixation by some grain legume varieties in Sudano–Sahelian zone of North Eastern Nigeria. Niger. J. Basic Appl. Sci. 2010, 18, 44–49. [Google Scholar] [CrossRef]
- Damfami, A.; Namo, O.A.T. Bambara groundnut (Vigna subterranea (L.) Verd.): A Review of its past, present and future role in human nutrition. J Agric For. Meteorol Res. 2020, 3, 274–281. [Google Scholar]
- Azam-Ali, S.N.; Sesay, A.; Karikari, S.K.; Massawe, F.; Aguilar-Manjarrez, J.; Bannayan, M.; Hampson, K.J. Assessing the potential of an underutilized crop–A case study using Bambara groundnut. Exp. Agric. 2001, 37, 433–472. [Google Scholar] [CrossRef]
- Kendabie, P.; Jorgensen, S.T.; Massawe, F.; Azam-Ali, S.; Mayes, S. Daylength effects on growth and seed production efficiency in Bambara groundnut (Vigna subterranea L.). In Proceedings of the 3rd International Conference on Neglected and Underutilized Species (NUS): For a Food-Secure Africa, Accra, Ghana, 25–27 September 2013; pp. 28–35. [Google Scholar]
- Brink, M.; Ramolemana, G.M.; Sibuga, K.P. Vigna subterranea (L.) Verdc. In Plant Resources of Tropical African Cereals and Pulses; Brink, M., Belay, G., Eds.; PROTA Foundation: Wageningen, The Netherlands, 2006; pp. 213–218. [Google Scholar]
- Baudoin, J.P.; Mergeai, G. Grain legumes in crop production in tropical Africa. Annu. Rep. 2001, 25, 313–317. [Google Scholar]
- Mabhaudhi, T.; Modi, A.T. Growth, phenological and yield responses of a Bambara groundnut (Vigna subterranea (L.) Verdc.) landrace to imposed water stress under field conditions. S. Afr. J. Plant Soil 2013, 30, 69–79. [Google Scholar] [CrossRef]
- Khan, M.M.H.; Rafii, M.Y.; Ramlee, S.I.; Jusoh, M.; Mamun, A. Genetic variability, heritability, and clustering pattern exploration of Bambara groundnut (Vigna subterranea L. Verdc) accessions for the perfection of yield and yield-related traits. BioMed Res. Int. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mabhaudhi, T.; Chimonyo, V.G.; Chibarabada, T.P.; Modi, A.T. Developing a roadmap for improving neglected and underutilized crops: A case study of South Africa. Front. Plant Sci. 2017, 8, 21–43. [Google Scholar] [CrossRef]
- Aliyu, S.; Massawe, F.; Mayes, S. Genetic diversity and population structure of Bambara groundnut [Vigna subterranea (L.) Verdc.]: Synopsis of the past two decades of analysis and implications for crop improvement programmes. Genet. Resour. Crop Evol. 2016, 63, 925–943. [Google Scholar] [CrossRef]
- Mabhaudhi, T.; Modi, A.T.; Beletse, Y.G. Growth, phenological and yield responses of a bambara groundnut (Vigna subterranea L. Verdc) landrace to imposed water stress: II. Rain shelter conditions. Afr. Crop Sci. J. 2013, 39, 191–198. [Google Scholar] [CrossRef]
- Azam-Ali, S.; Azam-Ali, S.N.; Aguilar-Manjarrez, J.; Bannayan-Avval, M. A Global Mapping System for Bambara Groundnut Production; FAO: Rome, Italy, 2001; Volume 1. [Google Scholar]
- Hillocks, R.J.; Bennett, C.; Mponda, O.M. Bambara nut: A review of utilisation, market potential and crop improvement. Afr. Crop Sci. J. 2012, 20, 1–16. [Google Scholar]
- FAOSTAT. 2017. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 5 March 2019).
- FAO. Forests for Improved Food Security and Nutrition Report; FAO: Rome, Italy, 2011; Available online: http://www.fao.org/docrep/014/i2011e/i2011e00.pdf (accessed on 15 February 2015).
- Ibrahim, A.R.; Dansi, A.; Salifou, M.; Ousmane, A.; Alzouma, A.; Alou, W. Farmers’ practices, utilization, conservation and marketing of Bambara groundnut [Vigna subterranea (L.) Verdc.] in Dosso Region, Western Niger. Genet. Resour. Crop Evol. 2018, 65, 1907–1914. [Google Scholar] [CrossRef]
- Musa, M.; Massawe, F.; Mayes, S.; Alshareef, I.; Singh, A. Nitrogen fixation and N-balance studies on Bambara groundnut (Vigna subterranea L. Verdc) landraces grown on tropical acidic soils of Malaysia. Comm. Soil Sci. Plant Anal. 2016, 47, 533–542. [Google Scholar]
- Mayes, S.; Ho, W.K.; Chai, H.H.; Gao, X.; Kundy, A.C.; Mateva, K.I.; Zahrulakmal, M.; Hahiree, M.K.I.M.; Kendabie, P.; Licea, L.C.; et al. Bambara groundnut: An exemplar underutilised legume for resilience under climate change. Planta 2019, 250, 803–820. [Google Scholar] [CrossRef]
- FAO. Agricultural Statistics Database. Rome: Wold Agricultural. Information Center. 2013. Available online: http://faostat.fao.org/site/567/DesktopDefault.aspx (accessed on 10 February 2013).
- Shiyam, J.O.; Nkor, N.N.; Binang, W.B.; Effa, E.B. Yield response of Bambara groundnut (Voandzeia subterranea (L.) Thours.) varieties to organomineral fertilizer in the Coastal Forest of South-Eastern Nigeria. SCIREA J. Agril. 2016, 1, 91–106. [Google Scholar]
- Fatimah, S.; Ardiarini, N.R.; Kuswanto. Genetic diversity of Madurese Bambara groundnut [Vigna subterranea (L.) Verdc.] lines based on morphological and RAPD markers. SABRAO J. Breed. Genet. 2018, 50, 101–114. [Google Scholar]
- Bonny, B.S.; Dagou, S.E.K.A.; Ajoumani, K.; Koffi, K.G.; Kouonon, L.C.; Sie, R.S. Evaluation of the diversity in qualitative traits of Bambara groundnut germplasm (Vigna subterranea (L.) Verdc.) of Côte d’Ivoire. Afr. J. Biotechnol. 2019, 8, 23–36. [Google Scholar]
- Abejide, D.R.; Falusi, O.A.; Adebola, M.O.; Daudu, O.A.; Salihu, B.Z. Evaluation of seed yield of Nigerian Bambara groundnut [Vigna subterranea (L.) Verdc.] landraces under varying water conditions. Not. Sci. Biol. 2018, 10, 233–239. [Google Scholar] [CrossRef]
- Khan, M.M.H.; Rafii, M.Y.; Ramlee, S.I.; Jusoh, M.; Mamun, A. Genetic analysis and selection of Bambara groundnut (Vigna subterranean [L.] Verdc.) landraces for high yield revealed by qualitative and quantitative traits. Sci Rep. 2021, 11, 7597. [Google Scholar] [CrossRef]
- FAO. Regional Overview of Food Security and Nutrition in Africa 2016. The Challenges of Building Resilience to Shocks and Stresses; FAO: Rome, Italy, 2017; Available online: http://www.fao.org/3/a-i6813e.pdf (accessed on 20 September 2018).
- Holm, J.M.; Marloth, B.W. Bambarra groundnut or Njugobean. Farming S. Afr. Bull. 1940, 215, 195–198. [Google Scholar]
- Suwanprasert, J.; Toojinda, T.; Srinives, P.; Chanprame, S. Hybridization technique of Bambara groundnut (Vigna subterranea). Breed. Sci. 2006, 56, 125–129. [Google Scholar] [CrossRef]
- Chang, Y.; Liu, H.; Liu, M.; Liao, X.; Sahu, S.K.; Fu, Y.; Liu, X. The draft genomes of five agriculturally important African orphan crops. Giga Sci. 2019, 8. [Google Scholar] [CrossRef]
- Rungnoi, O.; Suwanprasert, J.; Somta, P.; Srinives, P. Molecular genetic diversity of Bambara groundnut (Vigna subterranea L. Verdc.) revealed by RAPD and ISSR marker analysis. SABRAO J. Breed. Genet. 2012, 44, 87–101. [Google Scholar]
- Mazahib, A.M.; Nuha, M.O.; Salawa, I.S.; Babiker, E.E. Some nutritional attributes of Bambara groundnut as influenced by domestic processing. Int. Food Res. J. 2013, 20, 1165–1171. [Google Scholar]
- Ndidi, U.S.; Ndidi, C.U.; Aimola, I.A.; Bassa, O.Y.; Mankilik, M.; Adamu, Z. Effects of processing (Boiling and Roasting) on the nutritional and antinutritional properties of Bambara groundnuts (Vigna subterranea [L.] Verdc.) from Southern Kaduna, Nigeria. J. Food Process. 2014, 2, 1–10. [Google Scholar] [CrossRef]
- Atoyebi, J.O.; Osilesi, O.; Abberton, M.; Adebawo, O.; Oyatomi, O. Quantification of selected anti-nutrients and bioactive compounds in African Bambara groundnut [Vigna subterranea (L.) Verdc.]. Am. J. Food Nutr. 2018, 6, 88–95. [Google Scholar] [CrossRef]
- Gbaguidi, A.A.; Dansi, A.; Dossou-Aminon, I.; Gbemavo, D.S.J.C.; Orobiyi, A.; Sanoussi, F.; Yedomonhan, H. Agromorphological diversity of local Bambara groundnut [Vigna subterranea (L.) Verdc.] collected in Benin. Genet. Resour. Crop Evol. 2018. [Google Scholar] [CrossRef]
- Gregory, P.J.; Mayes, S.; Hui, C.H.; Jahanshiri, E.; Julkifle, A.; Kuppusamy, G.; Kuan, H.W.; Lin, T.X.; Massawe, F.; Suhairi, T.A.S.T.M.; et al. Crops for the Future (CFF): An overview of research efforts in the adoption of underutilised species. Planta 2019, 250, 979–988. [Google Scholar] [CrossRef]
- Akpalu, M.M.; Atubilla, I.A.; Oppong-Sekyere, D. Assessing the level of cultivation and utilization of Bambara groundnut [Voandzeia subterrenea (L) Verdc.] in the sumbrungu community of Bolgatanga, Upper East Region, Ghana. Int. J. Plant Anim. Environ. Sci. (IJPAES). 2013, 3, 68–75. [Google Scholar]
- Okpuzor, J.; Ogbunugafor, H.A.; Okafor, U.; Sofidiya, M.O. Identification of protein types in Bambara nut seeds: Perspectives for dietary protein supply in developing countries. EXCLI J. 2010, 9, 17–28. [Google Scholar]
- Murevanhema, Y.Y.; Jideani, V.A. Potential of Bambara groundnut (Vigna subterranea (L.) Verdc) milk as a probiotic beverage—a review. Crit. Rev. Food Sci. Nutr. 2013, 53, 954–967. [Google Scholar] [CrossRef]
- Adebanke, B.M.; Kemisola, A.A.; Lola, K.F.; Mayowa, I. Effect of partial substitution of cow milk with Bambara groundnut milk on the chemical composition, acceptability and shelf life of yoghurt. Ann. Food Sci. Technol. 2017, 18, 92–99. [Google Scholar]
- Harris, T.; Jideani, V.; Le Roes-Hill, M. Flavonoids and tannin composition of Bambara groundnut (Vigna subterranea) of Mpumalanga, South Africa. Heliyon 2018, 4, e00833. [Google Scholar] [CrossRef]
- Hardy, Z.; Jideani, V.A. Functional characteristics and microbiological viability of foam-mat dried Bambara groundnut (Vigna subterranea) yogurt from reconstituted Bambara groundnut milk powder. Food Sci. Nutr. 2020, 8, 5238–5248. [Google Scholar] [CrossRef]
- Adeparusi, E.O.; Agbede, J.O. Evaluation of leucaena and gliricidia leaf protein concentrate as supplements to Bambara groundnut [Vigna subterranean (L.) verdc] in the diet of Oreochromis niloticus. Aquac. Nutr. 2005, 12, 335–342. [Google Scholar]
- Ijarotimi, O.S. Protein and hematological evaluations of infant formulated from cooking banana fruits (Musa spp, ABB genome) and fermented Bambara groundnut (Vigna subterranean L. Verdc) seeds. Nutr. Res. Pract. 2008, 2, 165–170. [Google Scholar] [CrossRef][Green Version]
- OECDFAO-UN. OECD and Food and Agriculture Organization of The United Nations-Meat; OECD-FAO Agricultural Outlook; OECD Publishing: Paris, France, 2010; pp. 147–158. [Google Scholar]
- Doku, E.V.; Karikari, S.K. Operational selection in wild Bambara groundnut. Ghana J. Sci. 1971, 11, 45–56. [Google Scholar]
- Oyeyinka, S.A.; Tijani, T.S.; Oyeyinka, A.T.; Arise, A.K.; Balogun, M.A.; Kolawole, F.L.; Obalowu, M.A.; Joseph, J.K. Value added snacks produced from Bambara groundnut (Vigna subterranea) paste or flour. LWT 2018, 88, 126–131. [Google Scholar] [CrossRef]
- Jideani, V.A.; Diedericks, C.F. Nutritional, therapeutic, and prophylactic properties of Vigna subterranea and Moringa oleifera. Antioxid. Antidiabetic Agents Hum. Health 2014, 187–207. [Google Scholar] [CrossRef]
- Dansi, A.; Vodouhè, R.; Azokpota, P.; Yedomonhan, H.; Assogba, P.; Adjatin, A.; Loko, Y.L.; Dossou-Aminon, I.; Akpagana, K. Diversity of the neglected and underutilized crop species of importance in Benin. Sci. World J. 2012. [Google Scholar] [CrossRef]
- Buta, M.B.; Emire, S.A.; Posten, C.; Andrée, S.; Greiner, R. Reduction of β-ODAP and IP6 contents in Lathyrus sativus L. seed by high hydrostatic pressure. Food Res. Int. 2019, 120, 73–82. [Google Scholar] [CrossRef]
- Mubaiwa, J.; Fogliano, V.; Chidewe, C.; Bakker, E.J.; Linnemann, A.R. Utilization of Bambara groundnut (Vigna subterranea (L.) Verdc.) for sustainable food and nutrition security in semi-arid regions of Zimbabwe. PLoS ONE 2018, 13, e0204817. [Google Scholar] [CrossRef]
- Ijarotimi, O.S.; Esho, T.R. Comparison of nutritional composition and anti-nutrient status of fermented, germinated and roasted Bambara groundnut seeds (Vigna subterranea). Br. Food J. 2009, 111, 376–386. [Google Scholar] [CrossRef]
- Alain, M.M.M.; Israël, M.L.; René, M.S. Improving the nutritional quality of cowpea and Bambara bean flours for use in infant feeding. Pak. J. Nutr. 2007, 6, 660–664. [Google Scholar]
- Gruere, G.; Giuliani, A.; Smale, M. Marketing underutilized plant species for the benefit of the poor: A conceptual framework. Int. Food Policy Res. Inst. 2006, 154, 1–36. [Google Scholar] [CrossRef]
- Tadele, Z.; Bartels, D. Promoting orphan crops research and development. Planta 2019, 250, 675–676. [Google Scholar] [CrossRef]
- Abu, H.; Buah, S. Characterization of Bambara groundnut landraces and their evaluation by farmers in the upper West Region of Ghana. J. Dev. Sustain. Agric. 2011, 6, 64–74. [Google Scholar]
- Cullis, C.; Kunert, K.J. Unlocking the potential of orphan legumes. J. Expt. Bot. 2017, 68, 1895–1903. [Google Scholar] [CrossRef]
- Ibny, F.Y.; Jaiswal, S.K.; Mohammed, M.; Dakora, F.D. Symbiotic effectiveness and ecologically adaptive traits of native rhizobial symbionts of Bambara groundnut [Vigna subterranea (L.) Verdc.] in Africa and their relationship with phylogeny. Sci. Rep. 2019, 9, 1–17. [Google Scholar] [CrossRef]
- Mubaiwa, J. Managing the hard-to-cook (HTC) phenomenon in Bambara groundnut (Vigna subterranea (L.) Verdc.) processing for resource limited communities in Zimbabwe. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2018. [Google Scholar]
- Khan, F.; Azman, R.; Chai, H.H.; Mayes, S.; Lu, C. Genomic and transcriptomic approaches towards the genetic improvement of an underutilised crops: The case of Bambara groundnut. Afr. Crop Sci. J. 2016, 24, 429–458. [Google Scholar] [CrossRef]
- Molosiwa, W.P.M.; Massawe, F.J.; Wayah, S.B.; Sani, J.M. Ribosomal DNA variation in landraces of Bambara groundnut. Afr. J. Biotechnol. 2013, 12. [Google Scholar] [CrossRef][Green Version]
- Molosiwa, O.O.; Aliyu, S.; Stadler, F.; Mayes, K.; Massawe, F.; Kilian, A.; Mayes, S. SSR marker development, genetic diversity and population structure analysis of Bambara groundnut [Vigna subterranea (L.) Verdc.] landraces. Genet. Resour. Crop Evol. 2015, 62, 1225–1243. [Google Scholar] [CrossRef]
- Massawe, F.J.; Mwale, S.S.; Azam-Ali, S.N.; Roberts, J.A. Breeding in Bambara groundnut (Vigna subterranea (L.) Verdc.): Strategic considerations. Afr. J. Biotechnol. 2005, 4, 463–471. [Google Scholar]
- Esfeld, K.; Uauy, C.; Tadele, Z. Application of TILLING for orphan crop improvement. In Biotechnology of Neglected and Underutilized Crops; Jain, S., Dutta Gupta, S., Eds.; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar] [CrossRef]
- Adzawla, W.; Donkoh, S.A.; Nyarko, G.; O’Reilly, P.J.; Olayide, O.; Mayes, S.; Feldman, A.B.; Halimi, A.R. Adoption of Bambara groundnut production and its effects on farmers’ welfare in North Ghana. Afr. J. Agric. Res. 2016, 11, 583–594. [Google Scholar] [CrossRef]
- Muhammad, I.; Rafii, M.Y.; Ramlee, S.I.; Nazli, M.H.; Harun, A.R.; Oladosu, Y.; Musa, I.; Arolu, F.; Chukwu, S.C.; Haliru, B.S.; et al. Exploration of Bambara hroundnut (Vigna subterranea (L.) verdc.), an underutilized crop, to aid global food security: Varietal improvement, genetic diversity and processing. Agronomy 2020, 10, 766. [Google Scholar] [CrossRef]
- Aliyu, S.; Massawe, F.J. Microsatellites based marker molecular analysis of Ghanaian Bambara groundnut [Vigna subterranean (L.) Verdc.] landraces alongside morphological characterization. Genet. Resour. Crop Evol. 2013, 60, 777–787. [Google Scholar] [CrossRef]
- Olukolu, B.A.; Mayes, S.; Stadler, F.; Ng, N.Q.; Fawole, I.; Dominique, D.; Azam-Ali, S.N.; Abbott, A.G.; Kole, C. Genetic diversity in Bambara groundnut (Vigna subterranea (L.) Verdc.) As revealed by phenotypic descriptors and DArT marker analysis. Genet. Resour. Crop Evol. 2012, 59, 347–358. [Google Scholar] [CrossRef]
- Aliyu, S.; Massawe, F.; Mayes, S. Beyond landraces: Developing improved germplasm resources for underutilized species–A case for Bambara groundnut. Biotechnol. Gen. Eng. Revs. 2014, 30, 127–141. [Google Scholar] [CrossRef]
- Mayes, S.; Ho, W.K.; Kendabie, P.; Chai, H.H.; Aliyu, S.; Feldman, A.R.Y.O.; Halimi, R.A.; Massawe, F.E.S.T.O.; Azam-Ali, S.A.Y.E.D. Applying molecular genetics to underutilised species–Problems and opportunities. Malays. Appl. Biol. 2015, 44, 1–9. [Google Scholar]
- Molosiwa, O.O. Genetic Diversity and Population Structure Analysis of Bambara groundnut [Vigna subterranea (L.) Verdc.] Landraces Using Morpho-Agronomic and SSR Markers. Ph.D. Thesis, University of Nottingham, Nottingham, UK, 2012. [Google Scholar]
- Bonny, B.S.; Adjoumani, K.; Seka, D.; Koffi, K.G.; Kouonon, L.C.; Koffi, K.K.; Bi, I.A.Z. Agromorphological divergence among four agro-ecological populations of Bambara groundnut [Vigna subterranea (L) Verdc.] in Côte d’Ivoire. Ann. Agric. Sci. 2019, 64, 103–111. [Google Scholar] [CrossRef]
- Unigwe Unigwe, A.E.; Gerrano, A.S.; Adebola, P.; Pillay, M.; Monrovia, L. Morphological variation in selected accessions of Bambara groundnut (Vigna subterranea L. Verdc) in South Africa. J. Agric. Sci. 2016, 8, 69–80. [Google Scholar] [CrossRef]
- IPGRI; IITA; BAMNET. Descriptors for Bambara Groundnut (Vigna Subterranea); International Plant Genetic Resources Institute: Rome, Italy; International Institute of Tropical Agriculture: Ibadan, Nigeria; The International Bambara Groundnut Network: Hamburg, Germany, 2000. [Google Scholar]
- Hendre, P.S.; Muthemba, S.; Kariba, R.; Muchugi, A.; Fu, Y.; Chang, Y.; Song, B.; Liu, H.; Liu, M.; Liao, X.; et al. African Orphan Crops Consortium (AOCC): Status of developing genomic resources for African orphan crops. Planta 2019, 250, 989–1003. [Google Scholar] [CrossRef] [PubMed]
- Ribaut, J.M.; Ragot, M. Modernising breeding for orphan crops: Tools, methodologies, and beyond. Planta 2019, 250, 971–977. [Google Scholar] [CrossRef]
- Mohammed, S.M. Pre-Breeding of Bambara groundnut (Vigna subterranea [L.] Verdc.). Ph.D. Thesis, University of KwaZulu-Natal, Pietermaritzburg, South Africa, 2014. [Google Scholar]
- Madamba, R. Breeding Bambara groundnut varieties suitable for Zimbabwean conditions. In Workshop on Conservation and Improvement of Bambara Groundnuts (Vigna subterranea L. Verdc.); Department of Research & Specialist Services: Harare, Zimbabwe, 1995; pp. 14–16. [Google Scholar]
- Kesawat, M.S.; Kumar, B.D. Molecular markers: It’s application in crop improvement. J. Crop Sci. Biotechnol. 2009, 12, 169–181. [Google Scholar] [CrossRef]
- Somta, P.; Chankaew, S.; Rungnoi, O.; Srinives, P. Genetic diversity of the Bambara groundnut (Vigna subterranea (L.) Verdc.) as assessed by SSR markers. Genome 2011, 54, 898–910. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.S.; Redjeki, E.S.; Ho, W.K.; Aliyu, S.; Mayes, K.; Massawe, F.; Kilian, A.; Mayes, S. Construction of a genetic linkage map and QTL analysis in Bambara groundnut [Vigna subterranea (L.) Verdc.]. Genome 2016. [Google Scholar] [CrossRef]
- Chai, H.H.; Ho, W.K.; Graham, N.; May, S.; Massawe, F.; Mayes, S. A cross-species gene expression marker-based genetic map and QTL analysis in Bambara groundnut. Genes 2017, 8, 84. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.M.; Roberts, J.A.; Azam-Ali, S.N.; Mayes, S. Development of microsatellite markers for Bambara groundnut (Vigna subterranean (L.) Verdc.)—An underutilized African legume crop species. Mol. Ecol. Notes 2007. [Google Scholar] [CrossRef]
- Beena, R.; Sheshshayee, M.S.; Madhura, J.N.; Prasad, T.G.; Udayakumar, M. Development of SSR markers and genetic variability in physiological traits in Bambara groundnut [Vigna subterranea (L) Verdc]. In Prospects in Bioscience: Addressing the Issues; Springer India: New Delhi, India, 2012; pp. 229–242. [Google Scholar]
- Ntundu, W.H.; Bach, I.C.; Christiansen, J.L.; Andersen, S.B. Analysis of genetic diversity in Bambara groundnut [Vigna subterranea (L.) Verdc] landraces using amplified fragment length polymorphism (AFLP) markers. Afr. J. Biotechnol. 2004, 3, 220–225. [Google Scholar]
- Massawe, F.; Roberts, J.; Azam-Ali, S.; Davey, M.R. Genetic diversity in Bambara groundnut (Vigna subterranea (L.) Verdc) landraces assessed by random amplified polymorphic DNA (RAPD) markers. Genet. Resour. Crop Evol. 2003, 50, 737–741. [Google Scholar] [CrossRef]
- Mukakalisa, C.; Kandawa-Schulz, M.; Mapaure, I. Genetic diversity in landraces of Bambara groundnut found in Namibia using RAPD markers. In Proceedings of the II International Symposium on Underutilized Plant Species: Crops for the Future-Beyond Food Security, Kuala Lumpur, Malaysia, 27 June 2017; pp. 683–687. [Google Scholar]
- Stadler, F. Analysis of Differential Gene Expression under Water-Deficit Stress and Genetic Diversity in Bambara groundnut [Vigna subterranea (L.) Verdc.] Using Novel High-Throughput Technologies. Ph.D. Thesis, Technische Universitat Munchen, Munchen, Germany, 2009. [Google Scholar]
- Abberton, M.; Batley, J.; Bentley, A.; Bryant, J.; Cai, H.; Cockram, J.; Costa de Oliveira, A.; Cseke, L.J.; Dempewolf, H.; De Pace, C.; et al. Global agricultural intensification during climate change: A role for genomics. Plant Biotechnol. J. 2016, 14, 1095–1098. [Google Scholar] [CrossRef]
- Chang, Y.; Liu, H.; Liu, M.; Liao, X.; Sahu, S.K.; Fu, Y.; Song, B.; Cheng, S.; Kariba, R.; Muthemba, S.; et al. Genomic data of the Bambara groundnut [Vigna subterranea (L.) Verdc.]. Giga Sci. 2018. [Google Scholar] [CrossRef]
- Barilli, E.; Cobos, M.J.; Carrillo, E.; Kilian, A.; Carling, J.; Rubiales, D. A high-density integrated DArTseq SNP-based genetic map of Pisum fulvum and identification of QTLs controlling rust resistance. Front. Plant Sci. 2018. [Google Scholar] [CrossRef]
- Akpinar, B.A.; Lucas, S.J.; Budak, H.; Akpýnar, B.A. Genomics approaches for crop improvement against abiotic stress. Sci. World J. 2013, 361921. [Google Scholar] [CrossRef]
- Dhanapal, A.P. Genomics of crop plant genetic resources. Adv. Bio-Sci. Biotechnol. 2012, 3, 378–385. [Google Scholar] [CrossRef][Green Version]
- Armstead, I.; Donnison, I.; Aubry, S.; Harper, J.; Hörtensteiner, S.; James, C.; Mani, J.; Moffet, M.; Ougham, H.; Roberts, L.; et al. Cross-species identification of Mendel’s I locus. Science 2007, 315, 73. [Google Scholar] [CrossRef]
- Cannon, S.B.; Mitra, A.; Baumgarten, A.; Young, N.D.; May, G. The roles of segmental and tandem gene duplication in the evolution of large gene families in Arabidopsis thaliana. BMC Plant Biol. 2004, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.B. Understanding crop genetic diversity under modern plant breeding. Theor. Appl. Genet. 2015, 128, 2131–2142. [Google Scholar] [CrossRef]
- Basu, S.M.; Massawe, F.; Azam-Ali, S.; Graham, N.; Broadley, M.; May, S.; Mayes, S. Developing Xspecies approaches for genomics and transcriptomics-using resources developed in major species for research in Bambara groundnut. In Proceedings of the II International Symposium on Underutilized Plant Species: Crops for the Future-Beyond Food Security, Kuala Lumpur, Malaysia, 27 June–1 July 2011; pp. 773–778. [Google Scholar]
- Chai, H.H.; Massawe, F.; Mayes, S. Assessment of a segregating population for the improvement of drought tolerance in Bambara groundnut. In Proceedings of the XXIX International Horticultural Congress on Horticulture: Sustaining Lives, Livelihoods and Landscapes (IHC2014), Brisbane, Australia, 17–22 August 2014; pp. 339–346. [Google Scholar]
- Bonthala, V.S.; Mayes, K.; Moreton, J.; Blythe, M.; Wright, V.; May, S.T.; Massawe, F.; Mayes, S.; Twycross, J. Identification of gene modules associated with low temperatures response in Bambara groundnut by network-based analysis. PLoS ONE 2016, 11, e0148771. [Google Scholar] [CrossRef]
- McCallum, C.M.; Comai, L.; Greene, E.A.; Henikoff, S. Targeted screening for induced mutations. Nat. Biotechnol. 2000, 18, 455–457. [Google Scholar] [CrossRef]
- Comai, L.; Henikoff, S. TILLING: Practical single-nucleotide mutation discovery. Plant J. 2006, 45, 684–694. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.L.; Till, B.J.; Laport, R.G.; Darlow, M.C.; Kleffner, J.M.; Jamai, A.; El-Mellouki, T.; Liu, S.; Ritchie, R.; Nielsen, N.; et al. TILLING to detect induced mutations in soybean. BMC Plant Biol. 2008, 8, 1–10. [Google Scholar] [CrossRef]
- Triques, K.; Sturbois, B.; Gallais, S.; Dalmais, M.; Chauvin, S.; Clepet, C.; Aubourg, S.; Rameau, C.; Caboche, M.; Bendahmane, A. Characterization of Arabidopsis thaliana mismatch specific endonucleases: Application to mutation discovery by TILLING in pea. Plant J. 2007, 51, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Till, B.J.; Jankowicz-Cieslak, J.; Sági, L.; Huynh, O.A.; Utsushi, H.; Swennen, R.; Terauchi, R.; Mba, C. Discovery of nucleotide polymorphisms in the Musa gene pool by Ecotilling. Theor. Appl. Genet. 2010, 121, 1381–1389. [Google Scholar] [CrossRef] [PubMed]
- Tadele, Z. New approaches to plant breeding of orphan crops in Africa. In Proceedings of the International Conference, Bern, Switzerland, 19–21 September 2007; Stämpli AG: Bern, Switzerland, 2009; pp. 19–21. [Google Scholar]




| Common Names (Major Legumes) | Scientific Name | Production/Year (Million tons) | Production (ton/ha) |
|---|---|---|---|
| Cowpea | Vigna unguiculata (L.) Walp | 4.9 | 0.49 |
| Dry bean | Phaseolus vulgaris | 3.8 | 0.66 |
| Faba Bean | Vicia faba | 0.6 | 1.22 |
| Chickpea | Cicer arietinum | 0.3 | 0.94 |
| Lentil | Lens culinaris | 0.1 | 1.10 |
| Groundnut | Arachis hypogaea | 9.0 | 0.91 |
| Soybean | Glycine max | 1.4 | 1.22 |
| Pigeon bean | Cajanus cajan | 0.4 | 0.77 |
| Dry bean | Phaseolus vulgaris | 3.8 | 0.66 |
| Macronutrient Depending on Genetic and Environmental Factors, Stage of Maturation, and Method of Analysis | |
|---|---|
| Carbohydrate | 64.4% of total dry seed weight |
| Polysaccharides and oligosaccharides complex | 22 to 49.5% of total dry seed weight |
| Amylose | 19.6–35.1% |
| Amylopectin | 1–2% |
| Protein | 9.6–40% (average 23.6%) Vicilin (7s) and Legumin (11S) reported as major elements of protein |
| Bambara groundnut protein isolate (BGPI) | Varied from 81.4 to 92.8% |
| Invitro protein digestibility (IVPD) | 70–81% raw BG |
| 82–87.5% cooked BG | |
| Lipids | 1.4–9.7% |
| Fatty acids | Major: oleic and linoleic acid (omega 6); third most: palmitic acid and linolenic acid exist in small amounts |
| Micronutrients | |
| K, Ca, Mg, Fe, P, and Zn | Abundant: the handiness of micronutrients was poorly affected by anti-nutritional factors in seeds. The density amount and availability of K, Ca, Mg, Fe, P, and Zn in Bambara grains were affected by a storage system, duration, approach of processing, and the position of trace nutrients (seed coat, hilum, testa, seed leaf, or cotyledon). A red-coated seed contains more Fe than cream- and black-coated seeds. |
| Phytochemicals | Flavonoids and tannins (generally in seed coat; majorly in dark or red-colored seeds) |
| Flavonoids | Epicatechin major: in raw red seed |
| Catechin major: in cooked red seed | |
| Proanthocyanins | Polymers of epicatechin and catechin also have neuroprotective, antitumor, cardioprotective, and antioxidant properties (abundant in brown and red seeds) |
| Fiber | 1.4 to 10.3% |
| Anti-nutrient factors | |
| Tannins condensed | 0.0011–18.61 mg/g |
| Phytic acid | 1.10–15.11 mg/g |
| Inhibitor trypsin | 0.06–73.40 TI mg/g |
| Pectin | Bind the micronutrients (Ca, Zn, and Fe) |
| Raffinose and stachyose, flatus causing–alphaoilgosacharides, Saponins, Oxalate, Hydrogen cyanide | Trace amount present in Bambara groundnut |
| Genotype Type | |||||||
|---|---|---|---|---|---|---|---|
| Institutions | Accession Number | Percentage (%) | Wild Species | Landraces | Breeding Lines | Advanced Cultivars | Others |
| International Institute of Tropical Agriculture, Nigeria | 2031 | 33 | less than 1 | 100 | x | x | x |
| ORSTMONTP, France | 1416 | 23 | x | 100 | x | x | x |
| Department of Agricultural Research, Botswana | 338 | 6 | x | 2 | x | x | 98 |
| Plant Genetic Resources Research Institute, Ghana | 296 | 5 | x | - | x | x | 100 |
| National Plant Genetic resources Center, Tanzania | 283 | 5 | less than 1 | 81 | x | x | 18 |
| SADC Plant Genetic Resources Centre, Zambia | 232 | 4 | x | 100 | x | x | x |
| Others (26) | 1549 | 25 | 1 | 59 | 9 | 1 | 29 |
| 6145 | 100 | ||||||
| Sl. N0. | Qualitative Traits (Descriptors) | Scale | References |
|---|---|---|---|
| 1 | Growth habit (GrH) | 1. Bunch type * 2. Semi-bunch type 3. Spreading type | Abu and Buah [83]; Aliyu and Massawe [94]; Molosiwa et al. [89]; Ntundu et al. [13]; Olukolu et al. [95]; Aliyu et al. [39], Khan et al. [37] |
| 2 | Stem Hairiness (StH) | 0. Absent; 1. Sparse; 2. Dense | Gbaguidi et al. [62] |
| 3 | First stem Color (FSC) | 1. Green; 2. Reddish; 3. Striped | Gbaguidi et al. [62] |
| 4 | Terminal Leaflet shape (TLS) | 1. Oval *; 2. Round; 3. Lanceolate; 4. Elliptic | Molosiwa et al. [89]; Ntundu et al. [13]; Aliyu et al. [96]; Gbaguidi et al. [62]; Mayes et al. [97] |
| 5 | Terminal Leaflet Colour (TLC) | 1. Red; 2. Green *; 3. Purple | Molosiwa, [98] |
| 6 | Pigment of petiole (PetP) | 1. Reddish green; 2. Green * | Aliyu and Massawe [94]; Aliyu et al. [96] |
| 7 | Pod shape (PoS) | 1. End in a point with hook another side 2. End in a point with round another side * 3. Without a point | Molosiwa et al. [89]; Ntundu et al. [13] |
| 8 | Pod color (PoC) | 1. Reddish-brown *; 2. Yellowish-brown *; 3. Brown*; 4. purple *; 5. Black | Ntundu et al. [13]; Molosiwa [98]; Aliyu et al. [96] |
| 9 | Pod texture (PoT) | 1.Much grooved; 2. Smooth few grooved. 3. Abundant folded | Molosiwa et al. [89]; Ntundu et al. [13] |
| 10 | Seed shape (SeS) | 1. Oval *; 2. Round | Ntundu et al. [13]; Gbaguidi et al. [62] |
| 11 | Color of seed (SeC) | 1. Cream *; 2. Red *; 3. Cream purple 4. Black; 5. Black bream | Gbaguidi et al. [62] |
| 12 | Eye Colour (EyC) | 0. Absent 1. Black | Aliyu et al. [39] |
| 13 | Testa pattern (TeP) | 1. Dotted 2. Marbled striped with the entire line 3. Slight rhomboid spot in one side of hilum only 4. Slight rhomboid spot on both side of the hilum 5. Abundant rhomboid spot in both side of the hilum 6. Holstein pattern 7. No pattern * | Aliyu et al. [39] |
| 14 | Fully expanded terminal leaflet color | 1. Purple 2. Green * 3. Red | Aliyu et al. [39]; Molosiwa [98] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.M.H.; Rafii, M.Y.; Ramlee, S.I.; Jusoh, M.; Al-Mamun, M. Bambara Groundnut (Vigna subterranea L. Verdc): A Crop for the New Millennium, Its Genetic Diversity, and Improvements to Mitigate Future Food and Nutritional Challenges. Sustainability 2021, 13, 5530. https://doi.org/10.3390/su13105530
Khan MMH, Rafii MY, Ramlee SI, Jusoh M, Al-Mamun M. Bambara Groundnut (Vigna subterranea L. Verdc): A Crop for the New Millennium, Its Genetic Diversity, and Improvements to Mitigate Future Food and Nutritional Challenges. Sustainability. 2021; 13(10):5530. https://doi.org/10.3390/su13105530
Chicago/Turabian StyleKhan, Md Mahmudul Hasan, Mohd Y. Rafii, Shairul Izan Ramlee, Mashitah Jusoh, and Md Al-Mamun. 2021. "Bambara Groundnut (Vigna subterranea L. Verdc): A Crop for the New Millennium, Its Genetic Diversity, and Improvements to Mitigate Future Food and Nutritional Challenges" Sustainability 13, no. 10: 5530. https://doi.org/10.3390/su13105530
APA StyleKhan, M. M. H., Rafii, M. Y., Ramlee, S. I., Jusoh, M., & Al-Mamun, M. (2021). Bambara Groundnut (Vigna subterranea L. Verdc): A Crop for the New Millennium, Its Genetic Diversity, and Improvements to Mitigate Future Food and Nutritional Challenges. Sustainability, 13(10), 5530. https://doi.org/10.3390/su13105530






