Examining the Impacts of the Built Environment on Quality of Life in Cancer Patients Using Machine Learning
Abstract
1. Introduction
2. Methods
2.1. Survey Data Collection and Pre-Processing
2.1.1. Survey Design
2.1.2. Geocoding
2.1.3. Built Environment Measures
2.1.4. Perceived Built Environment and Accessibility
2.1.5. Quality of Life
2.1.6. Other Key Variables
2.2. Predictive Modeling for Quality of Life of Cancer Patients
3. Results
3.1. Descriptive Statistics and Factor Analysis
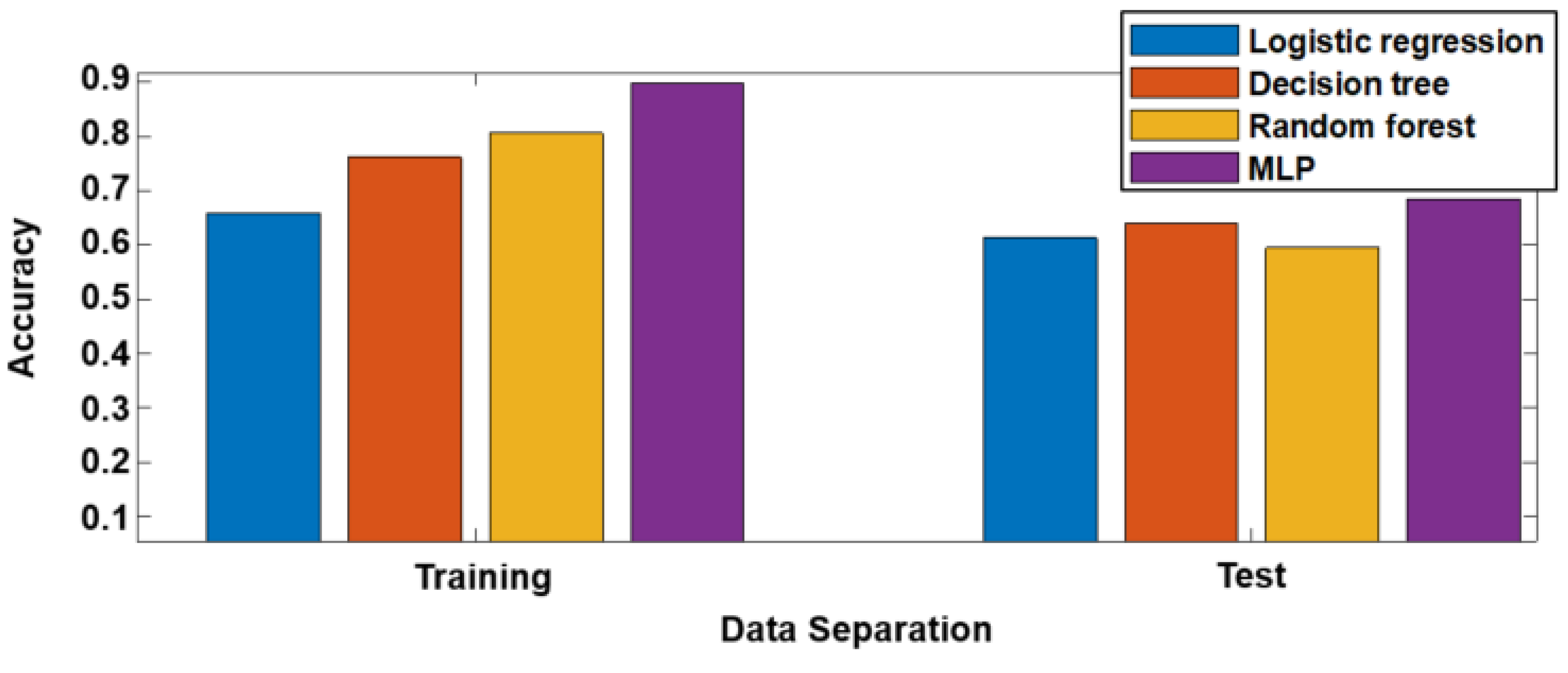
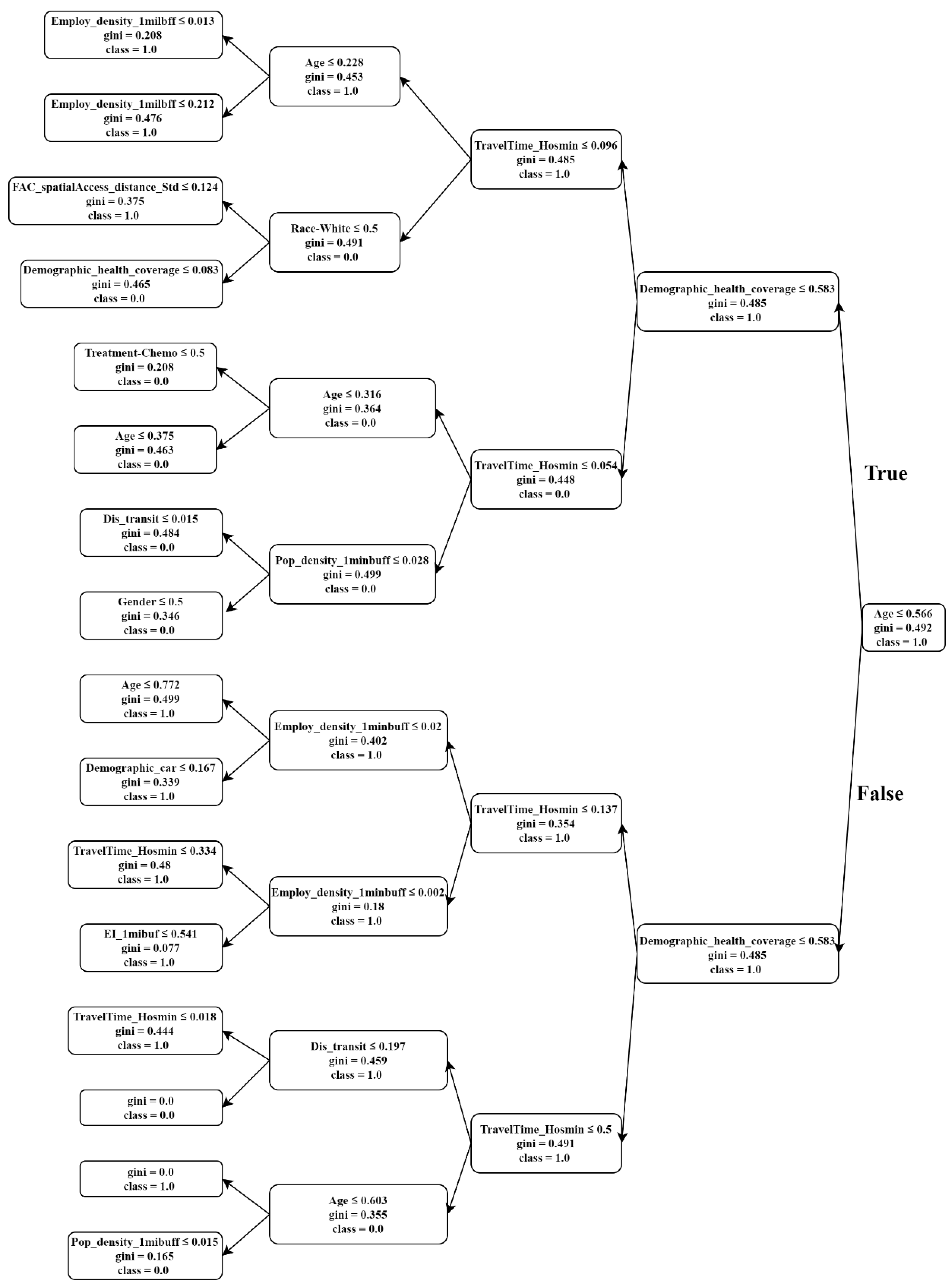
3.2. Predictive Modeling Results
4. Discussion
5. Conclusions
- Our findings regarding the effects of built environment features such as density and access to healthcare facilities on the QoL of cancer patients indicate that a supportive built environment can overcome the barriers in the outdoor environment, increase the likelihood of physical activity, and therefore improve perceived quality of life. These results point out that urban design and transportation planning need to become more friendly for this population group with particular needs and requirements.
- To improve social equity, it is fundamental to design environments compatible with the needs of all community groups, including people who are struggling with chronic diseases that require ongoing medical attention or limit activities of daily living in the long term.
- Understanding the associations between built environment and health-related QoL can help in the development of intervention policies that aim to improve cancer patients’ wellbeing. Hence, there is a need for collaboration between transit agencies, MPOs, and community planners to target the living environment and mobility needs of people who are burdened with chronic disease. To this end, urban and transportation planners and practitioners should be more involved in this field and acquire more knowledge from other disciplines. Integrating transportation planning with public health and social studies could reinforce existing policies and strategies in transportation accessibility and equity and therefore increase wellbeing and QoL.
- In addition, there is an inherent need to develop a QoL measurement that comprehensively counts for subjective feelings as well as objective factors in terms of patients’ health condition, transportation, and built environment. This QoL measurement can be used as a policy tool by communities and local governments to evaluate the extent to which the mobility and built environment meet the needs of patients with chronic diseases.
- The inverse associations between population density and cancer patients’ QoL indicate that compact development strategies can be fulfilled when policymakers address the side effects of urban density, such as fear of crime, high noise, and traffic congestion. This compact development pattern should concentrate on strategies that increase robust transportation options and improve public health indicators such as air quality while creating safe and secure neighborhoods that preserve more open space.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| QoL | Quality of Life |
| IRB | Institutional Review Board |
| GIS | Geographic Information System |
| ACS | American Community Survey |
| EI | Entropy Index |
| KMO | Kaiser–Meyer–Olkin |
| MLP | Multi-layer Perceptron |
References
- Sallis, J.F.; Floyd, M.F.; Rodríguez, D.A.; Saelens, B.E. Role of Built Environments in Physical Activity, Obesity, and Cardiovascular Disease. Circulation 2012, 125, 729–737. [Google Scholar] [CrossRef]
- Saelens, B.E.; Handy, S.L. Built Environment Correlates of Walking: A Review. Med. Sci. Sports Exerc. 2008, 40 (Suppl. 7), S550–S566. [Google Scholar] [CrossRef]
- Brownson, R.C.; Hoehner, C.M.; Day, K.; Forsyth, A.; Sallis, J.F. Measuring the Built Environment for Physical Activity. Am. J. Prev. Med. 2009, 36, S99–S123.e12. [Google Scholar] [CrossRef]
- Handy, S.L.; Boarnet, M.G.; Ewing, R.; Killingsworth, R.E. How the built environment affects physical activity. Am. J. Prev. Med. 2002, 23, 64–73. [Google Scholar] [CrossRef]
- Van, H.V.; Deforche, B.; Van, C.J.; Goubert, L.; Maes, L.; Van de Weghe, N.; De Bourdeaudhuij, I. Relationship between the physical environment and different domains of physical activity in European adults: A systematic review. BMC Public Health 2012, 12, 807. [Google Scholar]
- Duncan, M. Would the replacement of park-and-ride facilities with transit-oriented development reduce vehicle kilometers traveled in an auto-oriented US region? Transp. Policy 2017, 81, 293–301. [Google Scholar] [CrossRef]
- Ewing, R.; Cervero, R. Travel and the Built Environment: A Meta-Analysis. J. Am. Plan. Assoc. 2010, 76, 265–294. [Google Scholar] [CrossRef]
- Lovasi, G.S.; Grady, S.; Rundle, A. Steps forward: Review and recommendations for research on walkability, physical activity and cardiovascular health. Public Health Rev. 2011, 33, 484. [Google Scholar] [CrossRef]
- Leal, C.; Chaix, B. The influence of geographic life environments on cardiometabolic risk factors: A systematic review, a methodological assessment and a research agenda. Obes. Rev. 2011, 12, 217–230. [Google Scholar] [CrossRef]
- Zapata-Diomedi, B.; Herrera, A.M.M.; Veerman, J.L. The effects of built environment attributes on physical activity-related health and health care costs outcomes in Australia. Health Place 2016, 42, 19–29. [Google Scholar] [CrossRef]
- Lynch, B.M.; Owen, N.; Hawkes, A.L.; Aitken, J.F. Perceived barriers to physical activity for colorectal cancer survivors. Support Care Cancer 2010, 18, 729–734. [Google Scholar] [CrossRef]
- Schootman, M.; Deshpande, A.D.; Pruitt, S.L.; Jeffe, D.B. Neighborhood foreclosures and self-rated health among breast cancer survivors. Qual. Life Res. 2012, 21, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Pruitt, S.L.; McQueen, A.; Deshpande, A.D.; Jeffe, D.B.; Schootman, M. Mediators of the effect of neighborhood poverty on physical functioning among breast cancer survivors: A longitudinal study. Cancer Causes Control 2012, 23, 1529–1540. [Google Scholar] [CrossRef]
- Keegan, T.H.M.; Shariff-Marco, S.; Sangaramoorthy, M.; Koo, J.; Hertz, A.; Schupp, C.W.; Yang, J.; John, E.M.; Gomez, S.L. Neighborhood influences on recreational physical activity and survival after breast cancer. Cancer Causes Control 2014, 25, 1295–1308. [Google Scholar] [CrossRef]
- Parsons, M.A.; Askland, K.D. Determinants of prostate cancer stage in northern New England: USA Franco-American contextual effects. Soc. Sci. Med. 2007, 65, 2018–2030. [Google Scholar] [CrossRef]
- Russell, E.; Kramer, M.R.; Cooper, H.L.F.; Thompson, W.W.; Arriola, K.R.J. Residential Racial Composition, Spatial Access to Care, and Breast Cancer Mortality among Women in Georgia. J. Urban Health 2011, 88, 1117–1129. [Google Scholar] [CrossRef] [PubMed]
- Majcherek, D.; Weresa, M.A.; Ciecierski, C. Understanding Regional Risk Factors for Cancer: A Cluster Analysis of Lifestyle, Environment and Socio-Economic Status in Poland. Sustainability 2020, 12, 9080. [Google Scholar] [CrossRef]
- Wray, A.J.D.; Minaker, L.M. Is cancer prevention influenced by the built environment? A multidisciplinary scoping review. Cancer 2019, 125, 3299–3311. [Google Scholar] [CrossRef]
- Pitchforth, E.; Russell, E.; Van der Pol, M. Access to specialist cancer care: Is it equitable? Br. J. Cancer 2002, 87, 1221–1226. [Google Scholar] [CrossRef]
- Jordan, H. The Index of Multiple Deprivation 2000 and accessibility effects on health. J. Epidemiol. Community Health 2004, 58, 250–257. [Google Scholar] [CrossRef]
- Campbell, N.C.; Elliott, A.M.; Sharp, L.; Ritchie, L.D.; Cassidy, J.; Little, J. Rural and urban differences in stage at diagnosis of colorectal and lung cancers. Br. J. Cancer 2001, 84, 910–914. [Google Scholar] [CrossRef]
- Flytkjær Virgilsen, L.; Møller, H.; Vedsted, P. Cancer diagnostic delays and travel distance to health services: A nationwide cohort study in Denmark. Cancer Epidemiol. 2019, 59, 115–122. [Google Scholar] [CrossRef]
- Ringstrom, M.J.; Christian, J.; Bush, M.L.; Levy, J.E.; Huang, B.; Gal, T.J. Travel distance: Impact on stage of presentation and treatment choices in head and neck cancer. Am. J. Otolaryngol. 2018, 39, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Shariff-Marco, S.; Von Behren, J.; Reynolds, P.; Keegan, T.H.M.; Hertz, A.; Kwan, M.L.; Roh, J.M.; Thomsen, C.; Kroenke, C.H.; Ambrosone, C.; et al. Impact of Social and Built Environment Factors on Body Size among Breast Cancer Survivors: The Pathways Study. Cancer Epidemiol. Biomark. Prev. 2017, 26, 505–515. [Google Scholar] [CrossRef]
- Conroy, S.M.; Shariff-Marco, S.; Koo, J.; Yang, J.; Keegan, T.H.M.; Sangaramoorthy, M.; Hertz, A.; Nelson, D.O.; Cockburn, M.; Satariano, W.A.; et al. Racial/Ethnic Differences in the Impact of Neighborhood Social and Built Environment on Breast Cancer Risk: The Neighborhoods and Breast Cancer Study. Cancer Epidemiol. Biomark. Prev. 2017, 26, 541–552. [Google Scholar] [CrossRef]
- Chaix, B.; Rosvall, M.; Lynch, J.; Merlo, J. Disentangling contextual effects on cause-specific mortality in a longitudinal 23-year follow-up study: Impact of population density or socioeconomic environment? Int. J. Epidemiol. 2006, 35, 633–643. [Google Scholar] [CrossRef]
- Freedman, V.A.; Grafova, I.B.; Rogowski, J. Neighborhoods and Chronic Disease Onset in Later Life. Am. J. Public Health 2011, 101, 79–86. [Google Scholar] [CrossRef]
- Rantakokko, M.; Iwarsson, S.; Kauppinen, M.; Leinonen, R.; Heikkinen, E.; Rantanen, T. Quality of Life and Barriers in the Urban Outdoor Environment in Old Age: Quality of Life and Outdoor Environment. J. Am. Geriatr. Soc. 2010, 58, 2154–2159. [Google Scholar] [CrossRef]
- Engel, L.; Chudyk, A.M.; Ashe, M.C.; McKay, H.A.; Whitehurst, D.G.T.; Bryan, S. Older adults’ quality of life–Exploring the role of the built environment and social cohesion in community-dwelling seniors on low income. Soc. Sci. Med. 2016, 164, 1–11. [Google Scholar] [CrossRef]
- WHOQOL. Measuring Quality of Life: The World Health Organization Quality of Life Instruments (the WHOQOL-100 and the WHOQOL-BRIEF); Division of Mental Health and Prevention of Substance Abuse: Geneva, Switzerland, 1997. [Google Scholar]
- Fonseca, V.; Caeiro, J.; Nogueira, F. Social Model—Innovation and Behavioural Intervention as a Public Policy of Action within an Oncology and Loneliness Scope. Sustainability 2021, 13, 1544. [Google Scholar] [CrossRef]
- Kim, D.; Subramanian, S.V.; Kawachi, I. Social capital and physical health. In Social Capital and Health; Springer: Berlin/Heidelberg, Germany, 2008; pp. 139–190. [Google Scholar]
- Gao, M.; Ahern, J.; Koshland, C.P. Perceived built environment and health-related quality of life in four types of neighborhoods in Xi’an, China. Health Place 2016, 39, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Heydarnejad, M.S.; Hassanpour, D.A.; Solati, D.K. Factors affecting quality of life in cancer patients undergoing chemotherapy. Afr. Health Sci. 2011, 11, 266–270. [Google Scholar] [PubMed]
- Payne, R.; Mathias, S.D.; Pasta, D.J.; Wanke, L.A.; Williams, R.; Mahmoud, R. Quality of life and cancer pain: Satisfaction and side effects with transdermal fentanyl versus oral morphine. J. Clin. Oncol. 1998, 16, 1588–1593. [Google Scholar] [CrossRef] [PubMed]
- Parker, P.A.; Baile, W.F.; de Moor, C.; Cohen, L. Psychosocial and demographic predictors of quality of life in a large sample of cancer patients. Psychooncology 2003, 12, 183–193. [Google Scholar] [CrossRef]
- Costa, A.L.S.; Heitkemper, M.M.; Alencar, G.P.; Damiani, L.P.; da Silva, R.M.; Jarrett, M.E. Social support is a predictor of lower stress and higher quality of life and resilience in Brazilian patients with colorectal cancer. Cancer Nurs. 2017, 40, 352–360. [Google Scholar] [CrossRef]
- Astrup, G.L.; Rustøen, T.; Hofsø, K.; Gran, J.M.; Bjordal, K. Symptom burden and patient characteristics: Association with quality of life in patients with head and neck cancer undergoing radiotherapy. Head Neck 2017, 39, 2114–2126. [Google Scholar] [CrossRef]
- Sarna, L.; Padilla, G.; Holmes, C.; Tashkin, D.; Brecht, M.L.; Evangelista, L. Quality of life of long-term survivors of non–small-cell lung cancer. J. Clin. Oncol. 2002, 20, 2920–2929. [Google Scholar] [CrossRef]
- Cimprich, B.; Ronis, D.L.; Martinez-Ramos, G. Age at Diagnosis and Quality of Life in Breast Cancer Survivors. Cancer Pract. 2002, 10, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Drury, A.; Payne, S.; Brady, A.-M. Identifying associations between quality of life outcomes and healthcare-related variables among colorectal cancer survivors: A cross-sectional survey study. Int. J. Nurs. Stud. 2020, 101, 103434. [Google Scholar] [CrossRef]
- Wenzel, L.; DeAlba, I.; Habbal, R.; Kluhsman, B.C.; Fairclough, D.; Krebs, L.U.; Anton-Culver, H.; Berkowitz, R.; Aziz, N. Quality of life in long-term cervical cancer survivors. Gynecol. Oncol. 2005, 97, 310–317. [Google Scholar] [CrossRef]
- Green, C.; Hart-Johnson, T. Neighborhood SES and chronic pain: Impact on quality of life of cancer survivors. J. Pain. 2011, 12, 84. [Google Scholar] [CrossRef]
- Oh, Y.J.; Kim, C.-M.; Lee, Y.J.; Yoon, J.; Kim, M.S.; Kim, S.J.; Shin, H.H. Predictive Factors for Quality of Life and Impact of Physical Activity in Korean Breast Cancer Survivors. Korean J. Fam. Pract. 2018, 8, 380–385. [Google Scholar] [CrossRef]
- Mishra, S.I.; Scherer, R.W.; Geigle, P.M.; Berlanstein, D.R.; Topaloglu, O.; Gotay, C.C. Exercise Interventions on Health-Related Quality of Life for Cancer Survivors; Cochrane Gynaecological, Neuro-Oncology and Orphan Cancer Group, Ed.; Cochrane Database of Systematic Reviews; Wiley: Hoboken, NJ, USA, 2012; Available online: http://doi.wiley.com/10.1002/14651858.CD007566.pub2 (accessed on 17 April 2020).
- Ho, K.Y.; Li, W.H.C.; Lam, K.W.K.; Wei, X.; Chiu, S.Y.; Chan, C.-F.G.; Chung, O.K.J. Relationships among fatigue, physical activity, depressive symptoms, and quality of life in Chinese children and adolescents surviving cancer. Eur. J. Oncol. Nurs. 2019, 38, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishna, A.; Longo, T.A.; Fantony, J.J.; Harrison, M.R.; Inman, B.A. Physical activity patterns and associations with health-related quality of life in bladder cancer survivors. Urol. Oncol. Semin. Orig. Investig. 2017, 35, 540.e1–540.e6. [Google Scholar] [CrossRef] [PubMed]
- Schootman, M.; Perez, M.; Schootman, J.C.; Fu, Q.; McVay, A.; Margenthaler, J.; Colditz, G.A.; Kreuter, M.W.; Jeffe, D.B. Influence of built environment on quality of life changes in African-American patients with non-metastatic breast cancer. Health Place 2020, 63, 102333. [Google Scholar] [CrossRef] [PubMed]
- Zebrack, B.J. Cancer Survivor Identity and Quality of Life. Cancer Pract. 2000, 8, 238–242. [Google Scholar] [CrossRef]
- Marans, R.W. Quality of urban life & environmental sustainability studies: Future linkage opportunities. Habitat Int. 2015, 45, 47–52. [Google Scholar]
- Del Mar Martínez-Bravo, M.; Martínez-del-Río, J.; Antolín-López, R. Trade-offs among urban sustainability, pollution and livability in European cities. J. Clean. Prod. 2019, 224, 651–660. [Google Scholar] [CrossRef]
- Nipp, R.D.; El-Jawahri, A.; Fishbein, J.N.; Eusebio, J.; Stagl, J.M.; Gallagher, E.R.; Park, E.R.; Jackson, V.A.; Pirl, W.F.; Greer, J.A.; et al. The relationship between coping strategies, quality of life, and mood in patients with incurable cancer: Coping in Patients with Incurable Cancer. Cancer 2016, 122, 2110–2116. [Google Scholar] [CrossRef]
- Vieira, V.M.; Villanueva, C.; Chang, J.; Ziogas, A.; Bristow, R.E. Impact of community disadvantage and air pollution burden on geographic disparities of ovarian cancer survival in California. Environ. Res. 2017, 156, 388–393. [Google Scholar] [CrossRef]
- Robert, S.A.; Strombom, I.; Trentham-Dietz, A.; Hampton, J.M.; McElroy, J.A.; Newcomb, P.A.; Remington, P.L. Socioeconomic risk factors for breast cancer: Distinguishing individual-and community-level effects. Epidemiology 2004, 15, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, S.; Ewing, R. A longitudinal study of changes in urban sprawl between 2000 and 2010 in the United States. Landsc. Urban Plan. 2014, 128, 72–82. [Google Scholar] [CrossRef]
- Ma, L.; Dill, J. Associations between the objective and perceived built environment and bicycling for transportation. J. Transp. Health 2015, 2, 248–255. [Google Scholar] [CrossRef]
- Nilsson, C.J.; Avlund, K.; Lund, R. Social Inequality in Onset of Mobility Disability among Older Danes: The Mediation Effect of Social Relations. J. Aging Health 2010, 22, 522–541. [Google Scholar] [CrossRef]
- Sallis, J.F.; Saelens, B.E.; Frank, L.D.; Conway, T.L.; Slymen, D.J.; Cain, K.L.; Chapman, J.E.; Kerr, J. Neighborhood built environment and income: Examining multiple health outcomes. Soc. Sci. Med. 2009, 68, 1285–1293. [Google Scholar] [CrossRef]
- Chen, M.; Hao, Y.; Hwang, K.; Wang, L.; Wang, L. Disease Prediction by Machine Learning Over Big Data from Healthcare Communities. IEEE Access 2017, 5, 8869–8879. [Google Scholar] [CrossRef]
- Charleonnan, A.; Fufaung, T.; Niyomwong, T.; Chokchueypattanakit, W.; Suwannawach, S.; Ninchawee, N. Predictive analytics for chronic kidney disease using machine learning techniques. In Proceedings of the 2016 Management and Innovation Technology International Conference (MITicon), Bang-San, Thailand, 12–14 October 2016; pp. MIT-80–MIT-83. [Google Scholar]
- Ding, C.; Cao, X.; Næss, P. Applying gradient boosting decision trees to examine non-linear effects of the built environment on driving distance in Oslo. Transp. Res. Part A Policy Pract. 2018, 110, 107–117. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Z.; Razavian, N. Deep EHR: Chronic Disease Prediction Using Medical Notes. arXiv 2018, arXiv:180804928. Available online: http://arxiv.org/abs/1808.04928 (accessed on 25 October 2020).
- Kan, C.; Yang, H.; Kumara, S. Parallel computing and network analytics for fast Industrial Internet-of-Things (IIoT) machine information processing and condition monitoring. J. Manuf. Syst. 2018, 46, 282–293. [Google Scholar] [CrossRef]
- Ye, Z.; Liu, C.; Tian, W.; Kan, C. A Deep Learning Approach for the Identification of Small Process Shifts in Additive Manufacturing using 3D Point Clouds. Procedia Manuf. 2020, 48, 770–775. [Google Scholar] [CrossRef]
- Hu, C.; Liu, Z.; Jiang, Y.; Shi, O.; Zhang, X.; Xu, K.; Suo, C.; Wang, Q.; Song, Y.; Yu, K.; et al. Early prediction of mortality risk among patients with severe COVID-19, using machine learning. Int. J. Epidemiol. 2020, 49, 1918–1929. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7543461/ (accessed on 25 October 2020). [CrossRef] [PubMed]
- Jahangiri, A.; Rakha, H.A. Applying Machine Learning Techniques to Transportation Mode Recognition Using Mobile Phone Sensor Data. IEEE Trans. Intell. Transp. Syst. 2015, 16, 2406–2417. [Google Scholar] [CrossRef]
- Yang, H.; Kan, C.; Liu, G.; Chen, Y. Spatiotemporal Differentiation of Myocardial Infarctions. IEEE Trans. Automat. Sci. Eng. 2013, 10, 938–947. [Google Scholar] [CrossRef]
- Cheng, C.; Kan, C.; Yang, H. Heterogeneous recurrence analysis of heartbeat dynamics for the identification of sleep apnea events. Comput. Biol. Med. 2016, 75, 10–18. [Google Scholar] [CrossRef]
- Hastie, T.; Tibshirani, R.; Friedman, J. The Elements of Statistical Learning: Data Mining, Inference, and Prediction; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Raileanu, L.E.; Stoffel, K. Theoretical Comparison between the Gini Index and Information Gain Criteria. Ann. Math. Artif. Intell. 2004, 41, 77–93. [Google Scholar] [CrossRef]
- Mofrad, R.B.; Schoonenboom, N.S.M.; Tijms, B.M.; Scheltens, P.; Visser, P.J.; van der Flier, W.M.; Teunissen, C.E. Decision tree supports the interpretation of CSF biomarkers in Alzheimer’s disease. Alzheimer’s Dement. Diagn. Assess. Dis. Monit. 2019, 11, 1–9. [Google Scholar]
- Cheng, L.; De Vos, J.; Zhao, P.; Yang, M.; Witlox, F. Examining non-linear built environment effects on elderly’s walking: A random forest approach. Transp. Res. Part D Transp. Environ. 2020, 88, 102552. [Google Scholar] [CrossRef]
- Kaklauskas, A.; Dzemyda, G.; Tupenaite, L.; Voitau, I.; Kurasova, O.; Naimaviciene, J.; Rassokha, Y.; Kanapeckiene, L. Artificial Neural Network-Based Decision Support System for Development of an Energy-Efficient Built Environment. Energies 2018, 11, 1994. [Google Scholar] [CrossRef]
- Etminani-Ghasrodashti, R.; Kan, C.; Mozaffarian, L. Investigating the Role of Transportation Barriers in Cancer Patients’ Decision Making Regarding the Treatment Process. Transp. Res. Rec. 2021. [Google Scholar] [CrossRef]
- Kan, C.; Chen, Y.; Yang, H. Multiscale Quality Control of Telemedicine ECG Signal Acquisition, Telehealthcare Computing and Engineering: Principles and Design; CRC: Boca Raton, FL, USA, 2012. [Google Scholar]
- Thomas, A.A.; Gallagher, P.; O’Céilleachair, A.; Pearce, A.; Sharp, L.; Molcho, M. Distance from treating hospital and colorectal cancer survivors’ quality of life: A gendered analysis. Support Care Cancer 2015, 23, 741–751. [Google Scholar] [CrossRef]
- Spees, L.P.; Wheeler, S.B.; Varia, M.; Weinberger, M.; Baggett, C.D.; Zhou, X.; Petermann, V.M.; Brewster, W.R. Evaluating the urban-rural paradox: The complicated relationship between distance and the receipt of guideline-concordant care among cervical cancer patients. Gynecol. Oncol. 2019, 152, 112–118. [Google Scholar] [CrossRef]
- Silver, D.; Blustein, J.; Weitzman, B.C. Transportation to Clinic: Findings from a Pilot Clinic-Based Survey of Low-Income Suburbanites. J. Immigr. Minority Health 2012, 14, 350–355. [Google Scholar] [CrossRef]
- Salloum, R.G.; Smith, T.J.; Jensen, G.A.; Lafata, J.E. Factors associated with adherence to chemotherapy guidelines in patients with non-small cell lung cancer. Lung Cancer 2012, 75, 255–260. [Google Scholar] [CrossRef]
- Zullig, L.L.; Jackson, G.L.; Provenzale, D.; Griffin, J.M.; Phelan, S.; van Ryn, M. Transportation—A Vehicle or Roadblock to Cancer Care for VA Patients with Colorectal Cancer? Clin. Colorectal Cancer 2012, 11, 60–65. [Google Scholar] [CrossRef]
- Wen, M.; Hawkley, L.C.; Cacioppo, J.T. Objective and perceived neighborhood environment, individual SES and psychosocial factors, and self-rated health: An analysis of older adults in Cook County, Illinois. Soc. Sci. Med. 2006, 63, 2575–2590. [Google Scholar] [CrossRef]
- Cao, X. How does neighborhood design affect life satisfaction? Evidence from Twin Cities. Travel Behav. Soc. 2016, 5, 68–76. [Google Scholar] [CrossRef]
- Mouratidis, K. Compact city, urban sprawl, and subjective well-being. Cities 2019, 92, 261–272. [Google Scholar] [CrossRef]
- Ewing, R.; Schmid, T.; Killingsworth, R.; Zlot, A.; Raudenbush, S. Relationship between Urban Sprawl and Physical Activity, Obesity, and Morbidity. Am. J. Health Promot. 2003, 18, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, M.; Thompson, J.; de Sá, T.H.; Ewing, R.; Mohan, D.; McClure, R.; Roberts, I.; Tiwari, G.; Giles-Corti, B.; Sun, X.; et al. Land use, transport, and population health: Estimating the health benefits of compact cities. Lancet 2016, 388, 2925–2935. [Google Scholar] [CrossRef]
- Cramer, V.; Torgersen, S.; Kringlen, E. Quality of Life in a City: The Effect of Population Density. Soc. Indic. Res. 2004, 69, 103–116. [Google Scholar] [CrossRef]
- Ngom, R.; Gosselin, P.; Blais, C.; Rochette, L. Type and Proximity of Green Spaces Are Important for Preventing Cardiovascular Morbidity and Diabetes—A Cross-Sectional Study for Quebec, Canada. IJERPH 2016, 13, 423. [Google Scholar] [CrossRef] [PubMed]
- Glazier, R.H.; Creatore, M.I.; Weyman, J.T.; Fazli, G.; Matheson, F.I.; Gozdyra, P.; Moineddin, R.; Shriqui, V.K.; Booth, G.L. Density, Destinations or Both? A Comparison of Measures of Walkability in Relation to Transportation Behaviors, Obesity and Diabetes in Toronto, Canada. PLoS ONE 2014, 9, e85295. [Google Scholar] [CrossRef]
- Carrus, G.; Scopelliti, M.; Lafortezza, R.; Colangelo, G.; Ferrini, F.; Salbitano, F.; Agrimi, M.; Portoghesi, L.; Semenzato, P.; Sanesi, G. Go greener, feel better? The positive effects of biodiversity on the well-being of individuals visiting urban and peri-urban green areas. Landsc. Urban Plan. 2015, 134, 221–228. [Google Scholar] [CrossRef]
- Lederbogen, F.; Kirsch, P.; Haddad, L.; Streit, F.; Tost, H.; Schuch, P.; Wüst, S.; Pruessner, J.C.; Rietschel, M.; Deuschle, M.; et al. City living and urban upbringing affect neural social stress processing in humans. Nature 2011, 474, 498–501. [Google Scholar] [CrossRef]
- Ewing, R.H.; Hamidi, S. Measuring Sprawl 2014; Smart Growth America: Washington, DC, USA, 2014. [Google Scholar]
- Bantema-Joppe, E.J.; de Bock, G.H.; Woltman-van Iersel, M.; Busz, D.M.; Ranchor, A.V.; Langendijk, J.A.; Maduro, J.H.; van den Heuvel, E.R. The impact of age on changes in quality of life among breast cancer survivors treated with breast-conserving surgery and radiotherapy. Br. J. Cancer 2015, 112, 636–643. [Google Scholar] [CrossRef]
- Arndt, V.; Merx, H.; Stürmer, T.; Stegmaier, C.; Ziegler, H.; Brenner, H. Age-specific detriments to quality of life among breast cancer patients one year after diagnosis. Eur. J. Cancer 2004, 40, 673–680. [Google Scholar] [CrossRef]
- Champion, V.L.; Wagner, L.I.; Monahan, P.O.; Daggy, J.; Smith, L.; Cohee, A.; Ziner, K.W.; Haase, J.E.; Miller, K.D.; Pradhan, K.; et al. Comparison of younger and older breast cancer survivors and age-matched controls on specific and overall quality of life domains: Comparison of Breast Cancer Survivors. Cancer 2014, 120, 2237–2246. [Google Scholar] [CrossRef]
- Penson, D.F.; Stoddard, M.L.; Pasta, D.J.; Lubeck, D.P.; Flanders, S.C.; Litwin, M.S. The association between socioeconomic status, health insurance coverage, and quality of life in men with prostate cancer. J. Clin. Epidemiol. 2001, 54, 350–358. [Google Scholar] [CrossRef]
- Conlisk, E.A.; Lengerich, E.J.; Demark-Wahnefried, W.; Schildkraut, J.M.; Aldrich, T.E. Prostate cancer: Demographic and behavioral correlates of stage at diagnosis among blacks and whites in North Carolina. Urology 1999, 53, 1194–1199. [Google Scholar] [CrossRef]
- Pan, J.; Lei, X.; Liu, G.G. Health insurance and health status: Exploring the causal effect from a policy intervention. Health Econ. 2016, 25, 1389–1402. [Google Scholar] [CrossRef]
- Kale, H.P.; Carroll, N.V. Self-reported financial burden of cancer care and its effect on physical and mental health-related quality of life among US cancer survivors: Cancer-Related Financial Burden and HRQOL. Cancer 2016, 122, 283–289. [Google Scholar] [CrossRef]
- Arcury, T.A.; Preisser, J.S.; Gesler, W.M.; Powers, J.M. Access to Transportation and Health Care Utilization in a Rural Region. J. Rural Health 2005, 21, 31–38. [Google Scholar] [CrossRef]
- Pucher, J.; Renne, J.L. Rural mobility and mode choice: Evidence from the 2001 National Household Travel Survey. Transportation 2005, 32, 165–186. [Google Scholar] [CrossRef]
- Guidry, J.J.; Aday, L.A.; Zhang, D.; Winn, R.J. Transportation as a barrier to cancer treatment. Cancer Pract. 1997, 5, 361–366. [Google Scholar] [PubMed]
- Coughlin, S.S.; King, J. Breast and cervical cancer screening among women in metropolitan areas of the United States by county-level commuting time to work and use of public transportation, 2004 and 2006. BMC Public Health 2010, 10, 146. [Google Scholar] [CrossRef]
- Ross, C.E.; Willigen, M.V. Education and the Subjective Quality of Life. J. Health Soc. Behav. 1997, 38, 275. [Google Scholar] [CrossRef]
- Berglund, G.; Bolund, C.; Fornander, T.; Rutqvist, L.E.; Sjödén, P.-O. Late effects of adjuvant chemotherapy and postoperative radiotherapy on quality of life among breast cancer patients. Eur. J. Cancer Clin. Oncol. 1991, 27, 1075–1081. [Google Scholar] [CrossRef]
- Prigerson, H.G.; Bao, Y.; Shah, M.A.; Paulk, M.E.; LeBlanc, T.W.; Schneider, B.J.; Garrido, M.M.; Reid, M.C.; Berlin, D.A.; Adelson, K.B.; et al. Chemotherapy Use, Performance Status, and Quality of Life at the End of Life. JAMA Oncol. 2015, 1, 778. [Google Scholar] [CrossRef]
- Morrow, P.K.; Broxson, A.C.; Munsell, M.F.; Basen-Enquist, K.; Rosenblum, C.K.; Schover, L.R.; Nguyen, L.H.; Hsu, L.; Castillo, L.; Hahn, K.M.; et al. Effect of Age and Race on Quality of Life in Young Breast Cancer Survivors. Clin. Breast Cancer 2014, 14, e21–e31. [Google Scholar] [CrossRef][Green Version]
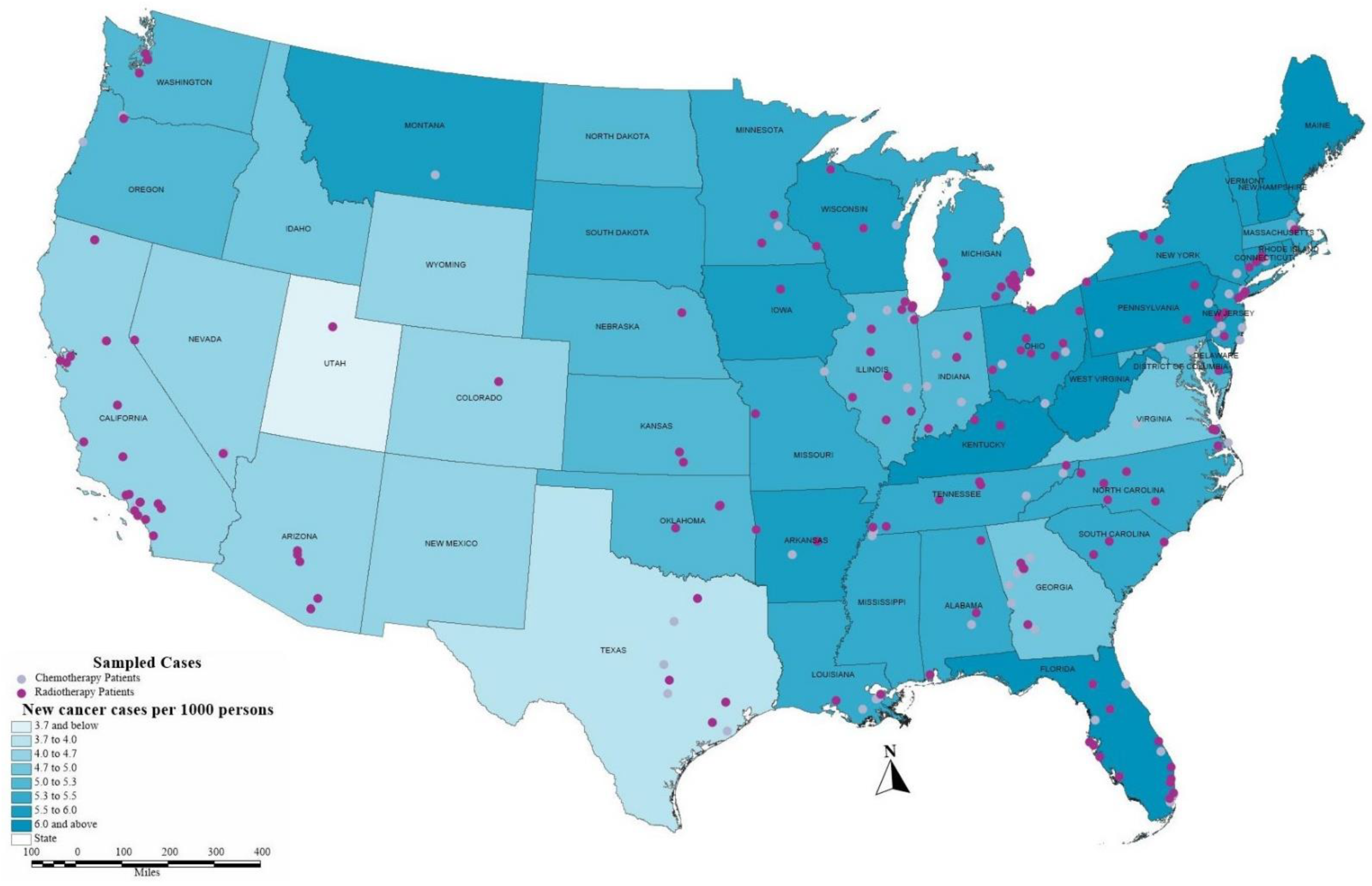
| Variables | Description | ||||
|---|---|---|---|---|---|
| Count | Percent | Mean | S.D. | ||
| Socio-demographic attributes | |||||
| Gender | Female | 292 | 49.6 | ||
| Male | 297 | 50.4 | |||
| Race | White | 510 | 86.6 | ||
| Non-white | 79 | 13.4 | |||
| Education | Well-educated (bachelor and above) | 316 | 53.7 | ||
| Less-educated (below bachelor) | 256 | 43.5 | |||
| Missing | 17 | 2.9 | |||
| Employment status | Employee | 220 | 37.4 | ||
| Not-employee | 364 | 61.8 | |||
| Missing | 5 | 0.8 | |||
| Residential status | Owner | 219 | 37.2 | ||
| Not-owner | 366 | 62.1 | |||
| Missing | 4 | 0.7 | |||
| Number of cars in the household | 0 | 45 | 7.6 | ||
| 1 | 243 | 41.3 | |||
| 2 | 217 | 36.8 | |||
| 3 or more | 84 | 14.3 | |||
| Health insurance | Medicaid | 96 | 16.3 | ||
| Medicare | 208 | 35.3 | |||
| Affordable Care Act | 21 | 3.6 | |||
| Employer-paid insurance | 142 | 24.1 | |||
| Private health insurance | 54 | 9.2 | |||
| Uninsured | 30 | 5.1 | |||
| Other insurance | 37 | 6.3 | |||
| Missing | 1 | 0.2 | |||
| Income | 50,872 | 28,132 | |||
| Age | 53 | 15.58 | |||
| Household Size | 2.55 | 1.36 | |||
| Built environment characteristics | |||||
| Population density | 3714 | 6761 | |||
| Entropy index | 0.66 | 0.04 | |||
| Intersection density | 172 | 96 | |||
| Transit stop density | 12 | 20 | |||
| Distance to transit (min) | 27.29 | 95.23 | |||
| Travel distance to the closest large hospital (min) | 12 | 206 | |||
| Perceptions | |||||
| Perceived built environment | 99.72 | 25.33 | |||
| Perceived accessibility | 92.99 | 15.16 | |||
| Health-related variables | |||||
| Cancer type (diagnosis) | Easy | 285 | 48.4 | ||
| Intermediate | 203 | 34.5 | |||
| Hard | 72 | 12.2 | |||
| Unknown | 29 | 4.9 | |||
| Cancer treatments | |||||
| Radiotherapy | 1 = having radiotherapy | 266 | 45.2 | ||
| 0 = not having radiotherapy | 323 | 54.8 | |||
| Chemotherapy | 1 = having chemotherapy | 273 | 46.3 | ||
| 0 = not having radiotherapy | 316 | 53.7 | |||
| Other | 1 = having other treatment | 261 | 44.3 | ||
| 0 = not having radiotherapy | 328 | 55.7 | |||
| Quality of life | |||||
| Overall quality of life | Terrible | 17 | 2.9 | ||
| Poor | 67 | 11.4 | |||
| Average | 168 | 28.5 | |||
| Good | 219 | 37.2 | |||
| Excellent | 118 | 20 | |||
| Please Indicate How Well Your Residence and Its Location Meet the Following Characteristics | Loadings | |
| Perceived built environment | Easy access to your health provider | 0.788 |
| Easy access to drugstores | 0.797 | |
| Closeness to work/school | 0.772 | |
| Closeness to family members who can take care of me when I need them | 0.730 | |
| Affordable neighborhood according to income and treatment costs | 0.805 | |
| Quiet, safe, and secure neighborhood according to mental and physical condition | 0.735 | |
| Please indicate the approximate travel distance (in minutes) from your current residence to the following errands | Loadings | |
| Perceived accessibility | Closest public transit station | 0.517 |
| Closest gas station | 0.846 | |
| Closest restaurant/fast-food place | 0.905 | |
| Closest drugstore | 0.896 | |
| Closest grocery store | 0.889 | |
| Patients’ primary health provider | 0.584 |
| Logistic Regression | Decision Tree | Random Forest | MLP | |
|---|---|---|---|---|
| F-score | 0.71 | 0.70 | 0.69 | 0.72 |
| AUROC | 0.64 | 0.67 | 0.63 | 0.66 |
| Feature Name | Importance Score | |
|---|---|---|
| 1 | Age | 0.14198 |
| 2 | Travel distance to closest large hospital | 0.12031 |
| 3 | Perceived accessibility | 0.11206 |
| 4 | Distance to transit (min) | 0.10921 |
| 5 | Population density | 0.09243 |
| 6 | Health insurance | 0.08123 |
| 7 | Entropy index | 0.07142 |
| 8 | Education (well-educated) | 0.03246 |
| 9 | Number of cars in the household | 0.02834 |
| 10 | Transit stop density | 0.02341 |
| 11 | Cancer treatments (chemotherapy) | 0.02240 |
| 12 | Employment status (employee) | 0.01907 |
| 13 | Cancer type (diagnosis) | 0.01650 |
| 14 | Gender | 0.01106 |
| 15 | Cancer treatments (radiotherapy) | 0.00730 |
| 16 | Race (white) | 0.00659 |
| 17 | Cancer treatments (other) | 0.00292 |
| Variables | Coef | St. Error | Z | p-Values |
|---|---|---|---|---|
| Socio-demographic attributes | ||||
| Gender (female) | 0.2621 | 0.230 | 1.141 | 0.254 |
| Race (white) | −0.0448 | 0.315 | −0.142 | 0.887 |
| Education (well-educated) | 0.6215 | 00.213 | 2.922 | 0.003 *** |
| Employment status (employee) | 0.2489 | 0.231 | 1.078 | 0.281 |
| Number of cars in the household | 0.2589 | 0.394 | 0.657 | 0.511 |
| Health insurance | −0.7557 | 0.381 | −1.984 | 0.047 *** |
| Age | 1.8632 | 0.523 | 3.565 | 0.000 *** |
| Built environment characteristics | ||||
| Population density | −14.1817 | 7.153 | −1.983 | 0.047 *** |
| Entropy index | −0.2651 | 0.629 | −0.421 | 0.673 |
| Transit stop density | −0.4187 | 0.706 | −0.593 | 0.553 |
| Distance to transit (min) | −2.2074 | 1.367 | −1. 614 | 0.106 |
| Travel distance to closest large hospital | 1.6386 | 1.127 | 1.453 | 0.146 |
| Perceptions | ||||
| Perceived accessibility | −1.1933 | 0.774 | −1.543 | 0.123 |
| Health-related variables | ||||
| Cancer type (diagnosis) | 0.1053 | 0.308 | 0.342 | 0.732 |
| Cancer treatments (radiotherapy) | 0.0180 | 0.251 | 0.072 | 0.943 |
| Cancer treatments (chemotherapy) | −0.7943 | 0.263 | −3.021 | 0.003 *** |
| Cancer treatments (other) | −0.3485 | 0.299 | −1.164 | 0.244 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Etminani-Ghasrodashti, R.; Kan, C.; Arif Qaisrani, M.; Mogultay, O.; Zhou, H. Examining the Impacts of the Built Environment on Quality of Life in Cancer Patients Using Machine Learning. Sustainability 2021, 13, 5438. https://doi.org/10.3390/su13105438
Etminani-Ghasrodashti R, Kan C, Arif Qaisrani M, Mogultay O, Zhou H. Examining the Impacts of the Built Environment on Quality of Life in Cancer Patients Using Machine Learning. Sustainability. 2021; 13(10):5438. https://doi.org/10.3390/su13105438
Chicago/Turabian StyleEtminani-Ghasrodashti, Roya, Chen Kan, Muhammad Arif Qaisrani, Omer Mogultay, and Houliang Zhou. 2021. "Examining the Impacts of the Built Environment on Quality of Life in Cancer Patients Using Machine Learning" Sustainability 13, no. 10: 5438. https://doi.org/10.3390/su13105438
APA StyleEtminani-Ghasrodashti, R., Kan, C., Arif Qaisrani, M., Mogultay, O., & Zhou, H. (2021). Examining the Impacts of the Built Environment on Quality of Life in Cancer Patients Using Machine Learning. Sustainability, 13(10), 5438. https://doi.org/10.3390/su13105438





