Fish Oil and Fish Meal Production from Urban Fisheries Biomass in Japan
Abstract
1. Introduction
2. Materials and Methods
2.1. Fish Oil and Fish Meal from Urban Fisheries Biomass
2.2. Refinement Process of Fish Oil
2.3. Analysis of Fish Oil
2.3.1. Acid Value, Peroxide Value, p-Anisidine Value, and Total Oxidation Value
2.3.2. Thiobarbituric Acid Reactive Substances Value
2.3.3. Oxidative Stability
2.3.4. Fatty Acid Profile
2.4. Analysis of Fish Meal
2.5. Statistical Analysis
3. Results
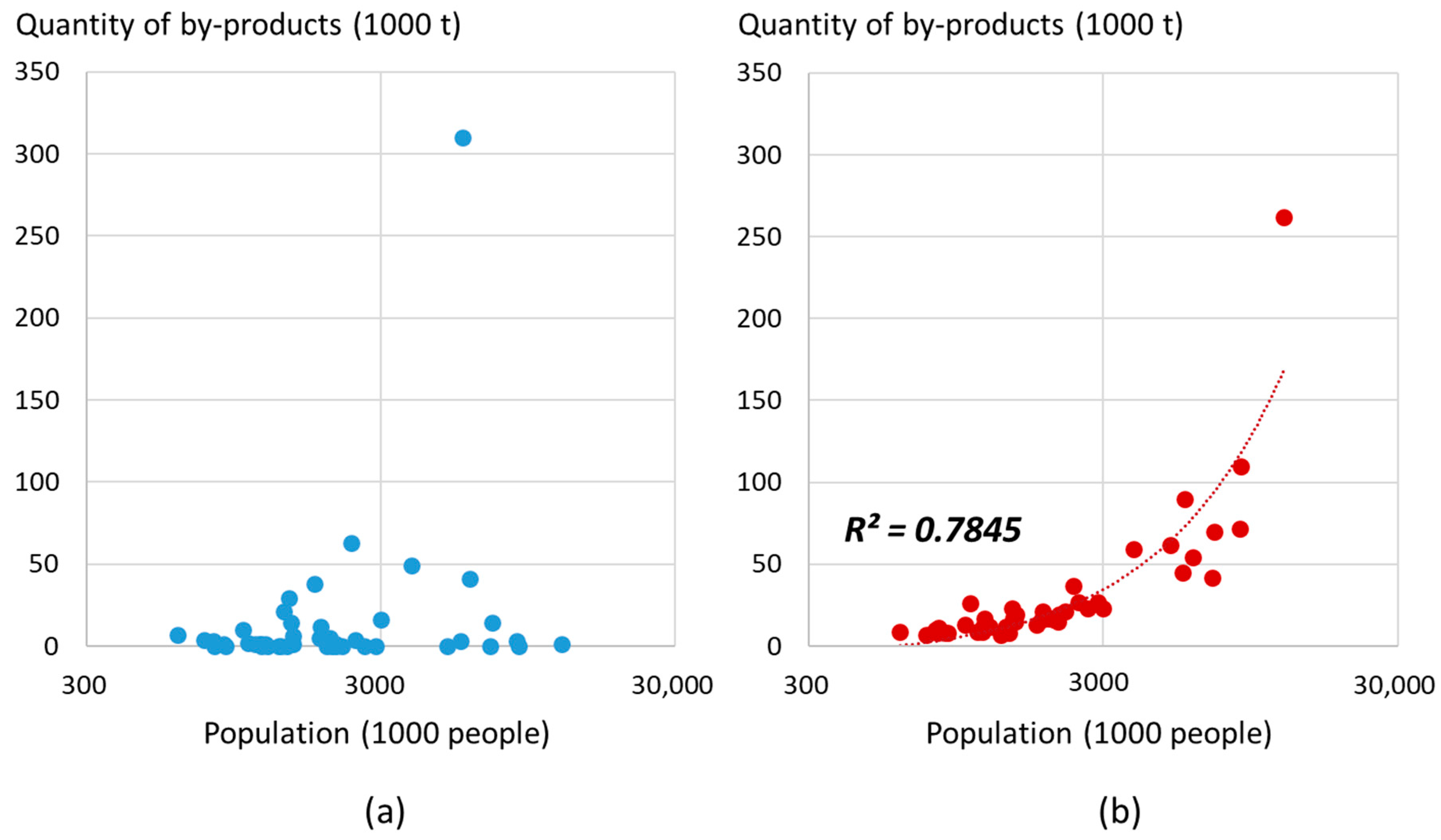
3.1. Urban Fisheries Biomass in Japan
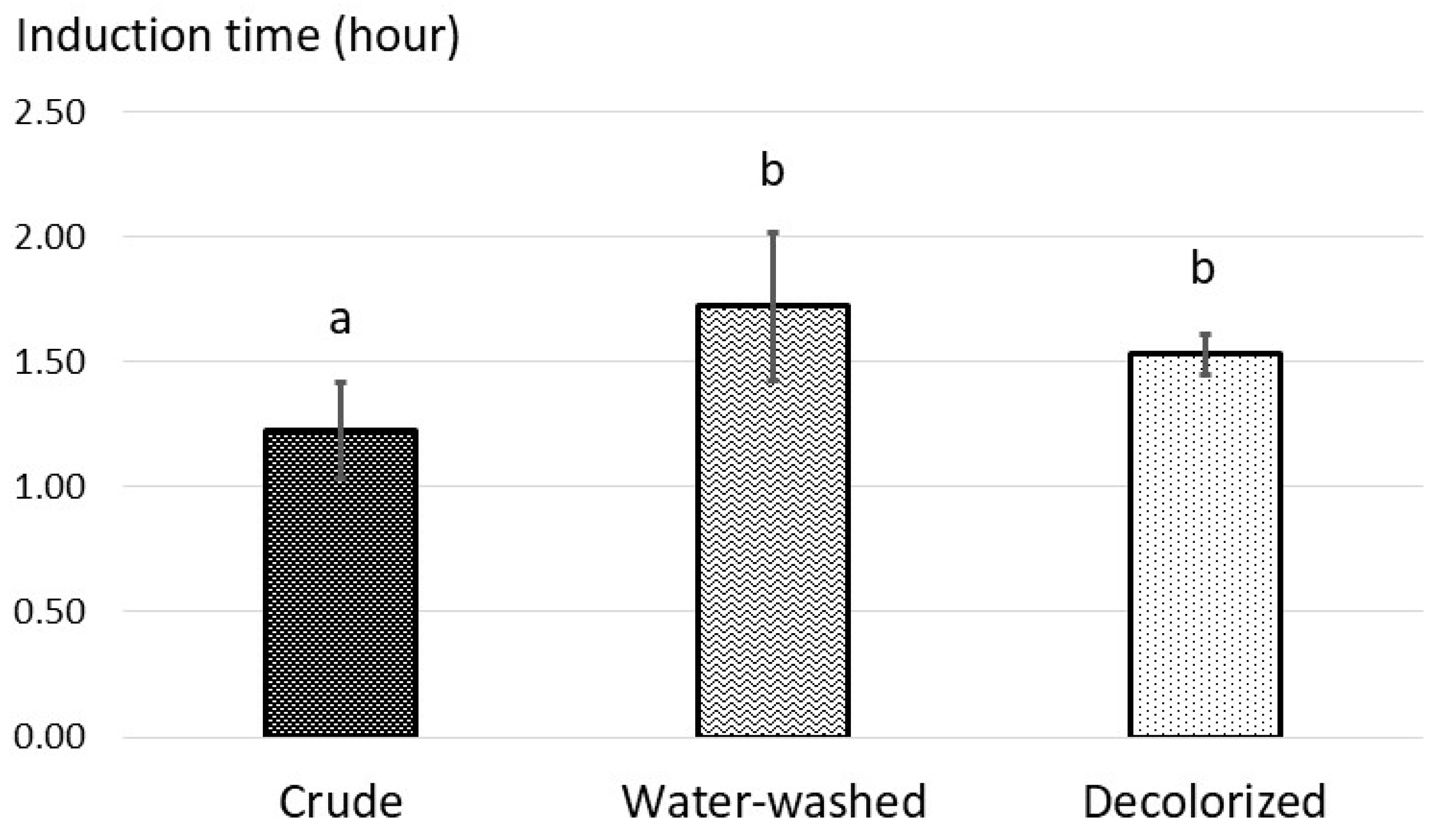
3.2. Quality of Fish Oil from Urban Fisheries Biomass
3.3. Quality of Fish Meal from Urban Fisheries Biomass
4. Discussion
4.1. Fish Oil and Fish Meal Produced from Fishery By-Products in Japan
4.2. Fish Oil and Fish Meal Produced from Fisheries By-Products in Japan
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Olsen, R.L.; Hasan, M.R. A limited supply of fishmeal: Impact on future increases in global aquaculture. Trends Food Sci. Technol. 2012, 27, 120–128. [Google Scholar] [CrossRef]
- Tacon, A.G.J.; Metian, M. Feed Matters: Satisfying the Feed Demand of Aquaculture. Rev. Fish. Sci. Aquac. 2015, 23, 1–10. [Google Scholar] [CrossRef]
- Alder, J.; Campbell, B.; Karpouzi, V.; Kaschner, K.; Pauly, D. Forage Fish: From Ecosystems to Markets. Annu. Rev. Environ. Resour. 2008, 33, 153–166. [Google Scholar] [CrossRef]
- Shepherd, J.; Bachis, E. Changing supply and demand for fish oil. Aquac. Econ. Manag. 2014, 18, 395–416. [Google Scholar] [CrossRef]
- Gatlin, D.M.; Barrows, F.T.; Brown, P.; Dabrowski, K.; Gaylord, T.G.; Hardy, R.W.; Herman, E.; Hu, G.; Krogdahl, Å.; Nelson, R.; et al. Expanding the utilization of sustainable plant products in aquafeeds: A review. Aquac. Res. 2007, 38, 551–579. [Google Scholar] [CrossRef]
- Tacon, A.G.J.; Hasan, M.R.; Subasinghe, R.P. Use of Fishery Resources as Feed Inputs to Aquaculture Development: Trends and Policy Implications. In FAO Fisheries Circular; FAO: Rome, Italy, 2006; Volume 1018. [Google Scholar]
- Zhou, Q.-C.; Zhao, J.; Li, P.; Wang, H.-L.; Wang, L.-G. Evaluation of poultry by-product meal in commercial diets for juvenile cobia (Rachycentron canadum). Aquaculture 2011, 322–323, 122–127. [Google Scholar] [CrossRef]
- Kureshy, N.; Davis, D.A.; Arnold, C.R. Partial Replacement of Fish Meal with Meat-and-Bone Meal, Flash-Dried Poultry By-Product Meal, and Enzyme-Digested Poultry By-Product Meal in Practical Diets for Juvenile Red Drum. N. Am. J. Aquac. 2000, 62, 266–272. [Google Scholar] [CrossRef]
- Ido, A.; Iwai, T.; Ito, K.; Ohta, T.; Mizushige, T.; Kishida, T.; Miura, C.; Miura, T. Dietary effects of housefly (Musca domestica) (Diptera: Muscidae) pupae on the growth performance and the resistance against bacterial pathogen in red sea bream (Pagrus major) (Perciformes: Sparidae). Appl. Entomol. Zool. 2015, 50, 213–221. [Google Scholar] [CrossRef]
- Ido, A.; Hashizume, A.; Ohta, T.; Takahashi, T.; Miura, C.; Miura, T. Replacement of Fish Meal by Defatted Yellow Mealworm (Tenebrio molitor) Larvae in Diet Improves Growth Performance and Disease Resistance in Red Seabream (Pargus major). Animals 2019, 9, 100. [Google Scholar] [CrossRef]
- Hashizume, A.; Ido, A.; Ohta, T.; Thiaw, S.T.; Morita, R.; Nishikawa, M.; Takahashi, T.; Miura, C.; Miura, T. Housefly (Musca domestica) Larvae Preparations after Removing the Hydrophobic Fraction Are Effective Alternatives to Fish Meal in Aquaculture Feed for Red Seabream (Pagrus major). Fishes 2019, 4, 38. [Google Scholar] [CrossRef]
- Henry, M.; Gasco, L.; Piccolo, G.; Fountoulaki, E. Review on the use of insects in the diet of farmed fish: Past and future. Anim. Feed Sci. Technol. 2015, 203, 1–22. [Google Scholar] [CrossRef]
- Sprague, M.; Betancor, M.B.; Tocher, D.R. Microbial and genetically engineered oils as replacements for fish oil in aquaculture feeds. Biotechnol. Lett. 2017, 39, 1599–1609. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, T. A review of feed development for early life stages of marine finfish in Japan. Aquaculture 2001, 200, 203–222. [Google Scholar] [CrossRef]
- Kaneniwa, M. Efficient utilization of aquatic biomass resources. Nippon Suisan Gakkaishi 2009, 75, 93–95. [Google Scholar] [CrossRef]
- Stevens, J.R.; Newton, R.W.; Tlusty, M.; Little, D.C. The rise of aquaculture by-products: Increasing food production, value, and sustainability through strategic utilisation. Mar. Policy 2018, 90, 115–124. [Google Scholar] [CrossRef]
- Jackson, A.; Newton, R.W. Project to Model the Use of Fisheries By-Products in the Production of Marine Ingredients with Special Reference to Omega 3 Fatty Acids EPA and DHA; IFFO: London, UK, 2016; pp. 1–14. [Google Scholar]
- Ghaly, A.E.; Ramakrishnan, V.V.; Brooks, M.S.; Budge, S.M.; Dave, D. Fish processing wastes as a potential source of proteins, amino acids and oils: A critical review. J. Microb. Biochem. Technol. 2013, 5, 107–129. [Google Scholar]
- Ytrestøyl, T.; Aas, T.S.; Åsgård, T. Utilisation of feed resources in production of Atlantic salmon (Salmo salar) in Norway. Aquaculture 2015, 448, 365–374. [Google Scholar] [CrossRef]
- De Silva, S.S.; Turchini, G.M. Use of wild fish and other aquatic organisms as feed in aquaculture—A review of practices and implications in Asia-Pacific. In Fish as Feed Inputs for Aquaculture: Practices, Sustainability and Implications; Hasan, M.R., Halwart, M., Eds.; FAO: Rome, Italy, 2009; pp. 63–127. ISBN 978-92-5-106419-1. [Google Scholar]
- Japan Marine Oil Association. Marine Oil Statistical Yearbook 2018; Japan Marine Oil Association: Tokyo, Japan, 2019. [Google Scholar]
- Tarui, Y. Generation and collection of fisheries residue. In Advanced Utilization of Fsheries Resources; Sakaguchi, M., Hirata, T., Eds.; NTS Inc.: Tokyo, Japan, 2005; pp. 15–28. ISBN 4-86043-093-X. [Google Scholar]
- Šimat, V.; Vlahovic, J.; Soldo, B.; Skroza, D.; Ljubenkov, I.; Mekinic, I.G. Production and refinement of omega-3 rich oils from processing by-products of farmed fish species. Foods 2019, 8, 125. [Google Scholar] [CrossRef]
- Crexi, V.T.; Monte, M.L.; de Souza Soares, L.A.; Pinto, L.A.A. Production and refinement of oil from carp (Cyprinus carpio) viscera. Food Chem. 2010, 119, 945–950. [Google Scholar] [CrossRef]
- Japan Oil Chemists’ Society. Standard Methods for the Analysis of Fats, Oils and Related Materials: The JOCS Standard Methods for the Analysis of Fats, Oils and Related Materials; Japan Oil Chemists’ Society: Tokyo, Japan, 2018; ISBN 978-4-931-24906-6. [Google Scholar]
- The Pharmaceutical Society of Japan. Standard Methods of Analysis for Hygienic Chemists: With Commentary; KANEHARA & CO., LTD.: Tokyo, Japan, 2015; ISBN 978-4-307-47043-8. [Google Scholar]
- Läubli, M.W.; Bruttel, P.A. Determination of the oxidative stability of fats and oils: Comparison between the active oxygen method (AOCS Cd 12-57) and the rancimat method. J. Am. Oil Chem. Soc. 1986, 63, 792–795. [Google Scholar] [CrossRef]
- The Consumer Affairs Agency in Japan. Food Labelling. Available online: https://www.caa.go.jp/en/policy/food_labeling/ (accessed on 23 February 2020).
- AOAC International. Official Methods of Analysis of AOAC International, 20th ed.; Latimer, W.G., Ed.; AOAC International: Gaithersburg, MD, USA, 2016; ISBN 0935584870. [Google Scholar]
- Tamase, K.; Kitada, Y.; Mizobuchi, M.; Sasaki, M. Simultaneous Determination of Histidine and Histamine in Fish by High Performance Liquid Chromatography. Food Hyg. Saf. Sci. (Shokuhin Eiseigaku Zasshi) 1984, 25, 525–529_1. [Google Scholar] [CrossRef][Green Version]
- The Ministry of Agriculture Foresty and Fisheries of Japan. Feed Analysis Standards. Available online: http://www.famic.go.jp/ffis/feed/bunseki/bunsekikijun.html (accessed on 23 February 2020).
- Ministry of Internal Affairs and Communications. System of Social and Demographic Statistics. Available online: https://www.e-stat.go.jp/ (accessed on 6 February 2020).
- Collette, B.; Chang, S.-K.; Di Natale, A.; Fox, W.; Juan Jorda, M.; Miyabe, N.; Nelson, R.; Uozumi, Y.; Wang, S. Thunnus Maccoyii. Available online: https://dx.doi.org/10.2305/IUCN.UK.2011-2.RLTS.T21858A9328286.en (accessed on 26 February 2020).
- Jacoby, D.; Gollock, M. Anguilla japonica. Available online: http://dx.doi.org/10.2305/IUCN.UK.2014-1.RLTS.T166184A1117791.en (accessed on 26 February 2020).
- The Ministry of Agriculture Foresty and Fisheries of Japan. Census of Fisheries. Available online: https://www.e-stat.go.jp/ (accessed on 6 February 2020).
- Ministry of Finance JAPAN. Trade Statistics of Japan. Available online: https://www.e-stat.go.jp/ (accessed on 6 February 2020).
- Japan Marine Oil Association. Marine Oil Statistical Yearbook 2015; Japan Marine Oil Association: Tokyo, Janpan, 2016. [Google Scholar]
- Nellemann, C.; MacDevette, M.; Manders, T.; Eickhout, B.; Svihus, B.; Prins, A.G.; Kaltenborn, B.P. The Environmental Food Crisis—The Environment’s Role in Averting Future Food Crises; UNEP/GRID-Arendal: Arendal, Norway, 2009; ISBN 9788277010540. [Google Scholar]
- Gustavsson, J.; Cederberg, C.; Sonesson, U.; van Otterdijk, R.; Meybeck, A. Global Food Losses and Food Waste—Extent, Causes and Prevention; FAO: Rome, Italy, 2011; ISBN 9789251072059. [Google Scholar]
- Belchior, C.; Boteler, B.; Jansen, H.; Piet, G. Seafood in Europe: A Food System Approach for Sustainability; European Environment Agency (EEA): Luxembourg, 2016. [Google Scholar]
- Parfitt, J.; Barthel, M.; MacNaughton, S. Food waste within food supply chains: Quantification and potential for change to 2050. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 3065–3081. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Funamoto, T.; Honda, S.; Yabuki, K.; Nishida, H.; Watanabe, C. Management of declining Japanese sardine, chub mackerel and walleye pollock fisheries in Japan. Fish. Res. 2009, 100, 68–77. [Google Scholar] [CrossRef]
- Codex Alimentarius Commision. Standard for Fish Oils; CODEX STAN: Rome, Italy, 2017. [Google Scholar]
- Huang, J.; Sathivel, S. Purifying salmon oil using adsorption, neutralization, and a combined neutralization and adsorption process. J. Food Eng. 2010, 96, 51–58. [Google Scholar] [CrossRef]
- Kuo, C.H.; Liao, H.Z.; Wang, Y.H.; Wang, H.M.D.; Shieh, C.J.; Tseng, C.Y. Highly efficient extraction of EPA/DHA-enriched oil from cobia liver using homogenization plus sonication. Eur. J. Lipid Sci. Technol. 2017, 119, 1–8. [Google Scholar] [CrossRef]
- Aidos, I.; Van der Padt, A.; Boom, R.M.; Luten, J.B. Upgrading of maatjes herring byproducts: Production of crude fish oil. J. Agric. Food Chem. 2001, 49, 3697–3704. [Google Scholar] [CrossRef]
- Aidos, I.; Van Der Padt, A.; Boom, R.M.; Luten, J.B. Quality of crude fish oil extracted from herring byproducts of varying states of freshness. J. Food Sci. 2003, 68, 458–465. [Google Scholar] [CrossRef]
- Nazir, N.; Diana, A.; Sayuti, K. Physicochemical and Fatty Acid Profile of Fish Oil from Head of Tuna (Thunnus albacares) Extracted from Various Extraction Method. Int. J. Adv. Sci. Eng. Inf. Technol. 2017, 7, 709–715. [Google Scholar] [CrossRef]
- Kobayashi, T.; Takeya, H. Fish Discard Collection System: The Profitability and Complementary Role of the Public Sector in Food Recycling. Agric. Mark. J. Japan 2003, 12, 21–31. [Google Scholar]
- Jeon, G.H.; Kim, H.S.; Myung, S.H.; Cho, S.H. The effect of the dietary substitution of fishmeal with tuna by-product meal on growth, body composition, plasma chemistry and amino acid profiles of juvenile Korean rockfish (Sebastes schlegeli). Aquac. Nutr. 2014, 20, 753–761. [Google Scholar] [CrossRef]
- Li, P.; Wang, X.; Hardy, R.W.; Gatlin, D.M. Nutritional value of fisheries by-catch and by-product meals in the diet of red drum (Sciaenops ocellatus). Aquaculture 2004, 236, 485–496. [Google Scholar] [CrossRef]
- FAO. Technical Guidelines on Aquaculture Certification; FAO: Rome, Italy, 2011; ISBN 9789250069128. [Google Scholar]
- Huntington, T.C.; Hasan, M.R. Fish as feed inputs for aquaculture: Practices, sustainability and implications: A global synthesis. In Fish as Feed Inputs for Aquaculture: Practices, Sustainability and Implications; Hasan, M.R., Halwart, M., Eds.; FAO Fisheries and Aquaculture Technical Paper No. 518; FAO: Rome, Italy, 2009; pp. 1–61. [Google Scholar]
- Jędrejek, D.; Levic, J.; Wallace, J.; Oleszek, W. Animal by-products for feed: Characteristics, European regulatory framework, and potential impacts on human and animal health and the environment. J. Anim. Feed Sci. 2016, 25, 189–202. [Google Scholar] [CrossRef]
- Pramod, G.; Pitcher, T.J. Estimates of illegal and unreported seafood imports to Japan. Mar. Policy 2017, 84, 42–51. [Google Scholar] [CrossRef]

| Total Amount of Fish 1 | Southern Bluefin Tuna (Thunnus maccoyii) | Japanese Eel (Anguilla japonica) | Contamination Risk (%) | |
|---|---|---|---|---|
| Wild-catch production (t) | 2,715,220 | 4072 | 71 | 0.15 |
| Aquaculture production (t) | 284,472 | 0 | 20,979 | 7.37 |
| Import of live fish and fishery products (t) | 3,765,865 | 10,362 | 6816 | 0.46 |
| Total production (t) | 6,765,557 | 14,434 | 27,866 | 0.63 |
| Oil Samples | AV (mg KOH/g) | POV (meq of O2/kg) | p-AV | TOTOX Value | TBARs (μmol/g) |
|---|---|---|---|---|---|
| Crude | 4.25 ± 0.39 a | 5.89 ± 0.94 a | 24.99 ± 1.80 a | 36.77 ± 2.58 a | 1.09 ± 0.09 a |
| Deoxidized | 0.12 ± 0.09 b | 2.56 ± 0.81 b | 27.12 ± 5.19 a | 32.23 ± 4.25 a | 0.77 ± 0.14 b |
| Water-washed | 0.17 ± 0.06 b | 1.56 ± 0.36 b | 15.62 ± 3.25 b | 18.73 ± 2.76 b | 0.69 ± 0.08 b |
| Decolorized | 0.35 ± 0.09 b | 1.04 ± 0.13 c | 18.16 ± 1.78 b | 20.25 ± 1.85 b | 0.60 ± 0.07 b |
| Cod liver oil | 0.49 | 7.09 | 25.19 | 39.37 | 1.38 |
| Components (% Fatty Acids) | Refined Oil from UFB | Cod Liver Oil | |
|---|---|---|---|
| Saturated fatty acid | |||
| 17:0 | 0.5 | ±0.0 | 0.1 |
| 18:0 | 4.4 | ±0.1 | 2.9 |
| 20:0 | 0.2 | ±0.0 | 0.1 |
| Monounsaturated fatty acid | |||
| 16:1 | 5.5 | ±0.1 | 8.9 |
| 16:2 | 0.9 | ±0.1 | 1.0 |
| Polyunsaturated fatty acid | |||
| ω-9 | |||
| 18:1n-9 | 21.7 | ±0.2 | 16.9 |
| 20:1n-9 | 2.7 | ±0.0 | 4.5 |
| 22:1n-9 | 0.5 | ±0.0 | 1.5 |
| 24:1n-9 | 0.7 | ±0.0 | 0.9 |
| ω-7 | |||
| 18:1n-7 | 3.3 | ±0.0 | 6.1 |
| ω-6 | |||
| 18:2n-6 | 4.9 | ±0.2 | 1.6 |
| 18:3n-6 | 0.1 | ±0.0 | - |
| 20:2n-6 | 0.4 | ±0.0 | 0.3 |
| 20:3n-6 | 0.1 | ±0.0 | - |
| 20:4n-6 | 1.1 | ±0.0 | 0.6 |
| 22:4n-6 | 0.2 | ±0.0 | - |
| 22:5n-6 | 0.5 | ±0.1 | 0.1 |
| ω-3 | |||
| 18:3n-3 | 1.6 | ±0.0 | 1.0 |
| 18:4n-3 | 1.7 | ±0.0 | 2.8 |
| 20:3n-3 | 0.2 | ±0.1 | - |
| 20:4n-3 | 0.9 | ±0.1 | 0.9 |
| 20:5n-3 | 7.9 | ±0.1 | 15.0 |
| 21:5n-3 | 0.4 | ±0.0 | 0.7 |
| 22:5n-3 | 2.3 | ±0.0 | 1.6 |
| 22:6n-3 | 14.2 | ±0.1 | 10.3 |
| FM from UFB 1 | FM from Anchoveta 2 | ||
|---|---|---|---|
| Proximate Composition (% dry basis) | |||
| Crude protein | 67.5 | ±0.9 | 71.3 |
| Crude fat | 8.0 | ±0.5 | 9.8 |
| Ash | 20.3 | ±1.3 | 16.4 |
| Amino Acids (% dry basis) | |||
| Ala | 5.2 | ±0.1 | 4.9 |
| Arg | 4.5 | ±0.1 | 4.3 |
| Asp | 6.1 | ±0.1 | 7.0 |
| Cys | 0.6 | ±0.0 | 0.7 |
| Glu | 8.9 | ±0.1 | 9.9 |
| Gly | 7.1 | ±0.2 | 4.9 |
| His | 2.4 | ±0.1 | 2.7 |
| Ile | 2.6 | ±0.0 | 3.1 |
| Leu | 4.7 | ±0.1 | 5.6 |
| Lys | 5.0 | ±0.1 | 5.9 |
| Met | 1.9 | ±0.0 | 2.0 |
| Phe | 2.6 | ±0.0 | 3.1 |
| Pro | 4.1 | ±0.1 | 3.1 |
| Ser | 3.0 | ±0.0 | 2.9 |
| Thr | 3.0 | ±0.0 | 3.3 |
| Trp | 0.7 | ±0.0 | 0.9 |
| Tyr | 2.0 | ±0.0 | 3.3 |
| Val | 3.2 | ±0.0 | 3.8 |
| FM from UFB 1 | FM from Anchoveta 2 | ||
|---|---|---|---|
| Histamine (mg/100 g) | 45.5 | ±32.6 | 23.0 |
| VBN (mg/100 g) | 112.5 | ±33.4 | 90 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ido, A.; Kaneta, M. Fish Oil and Fish Meal Production from Urban Fisheries Biomass in Japan. Sustainability 2020, 12, 3345. https://doi.org/10.3390/su12083345
Ido A, Kaneta M. Fish Oil and Fish Meal Production from Urban Fisheries Biomass in Japan. Sustainability. 2020; 12(8):3345. https://doi.org/10.3390/su12083345
Chicago/Turabian StyleIdo, Atsushi, and Mika Kaneta. 2020. "Fish Oil and Fish Meal Production from Urban Fisheries Biomass in Japan" Sustainability 12, no. 8: 3345. https://doi.org/10.3390/su12083345
APA StyleIdo, A., & Kaneta, M. (2020). Fish Oil and Fish Meal Production from Urban Fisheries Biomass in Japan. Sustainability, 12(8), 3345. https://doi.org/10.3390/su12083345





