Resource Recovery of Waste Nd–Fe–B Scrap: Effective Separation of Fe as High-Purity Hematite Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
2.1. Scrap Dissolution
2.2. Precipitation of Fe Impurity
2.3. Characterisation
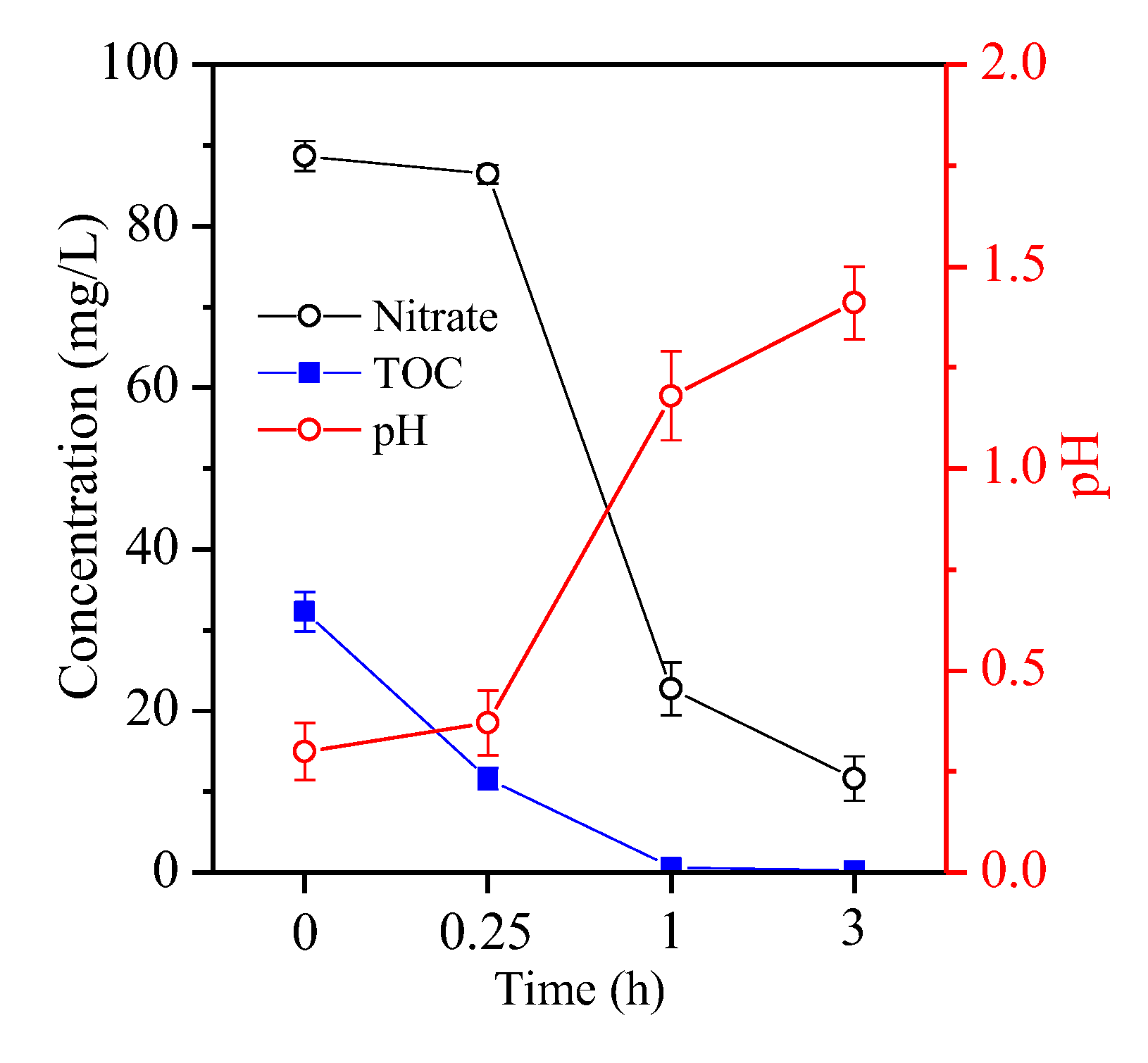
3. Results and Discussion
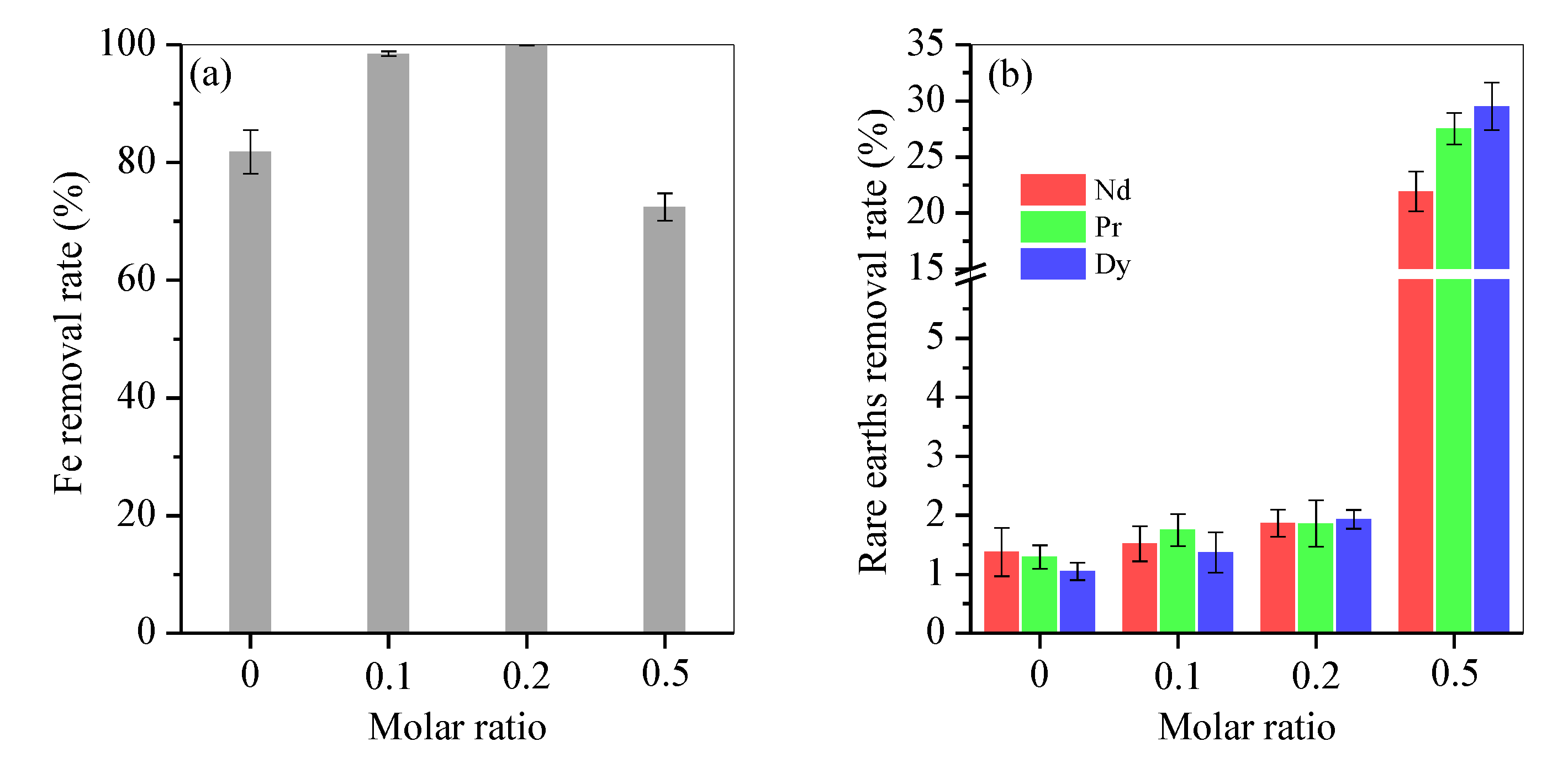
3.1. Fructose Dosage Optimisation
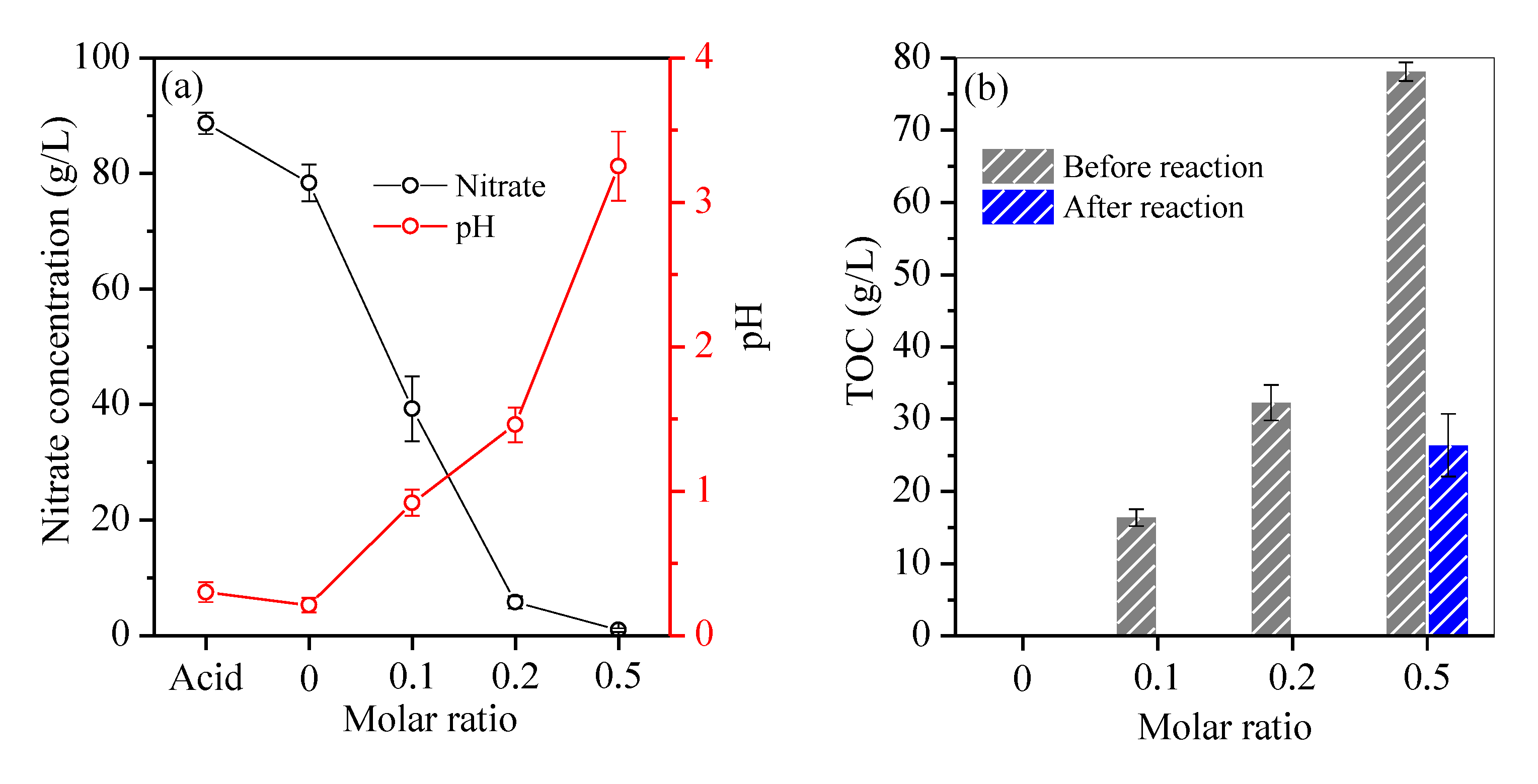
3.2. Composition of Residual Acid after Fe Removal
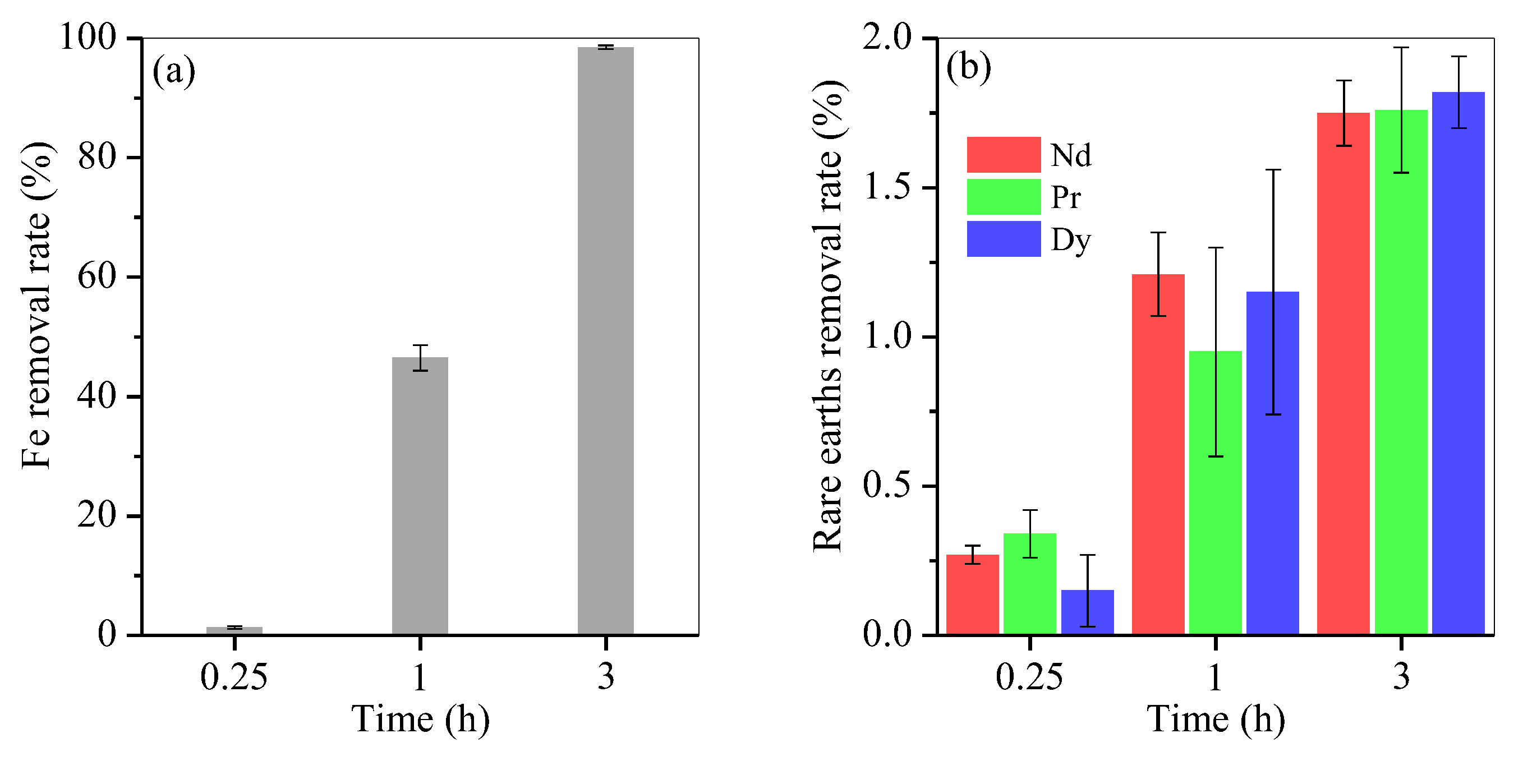
3.3. Hydrothermal Time Optimisation at the Optimal Fructose Dosage
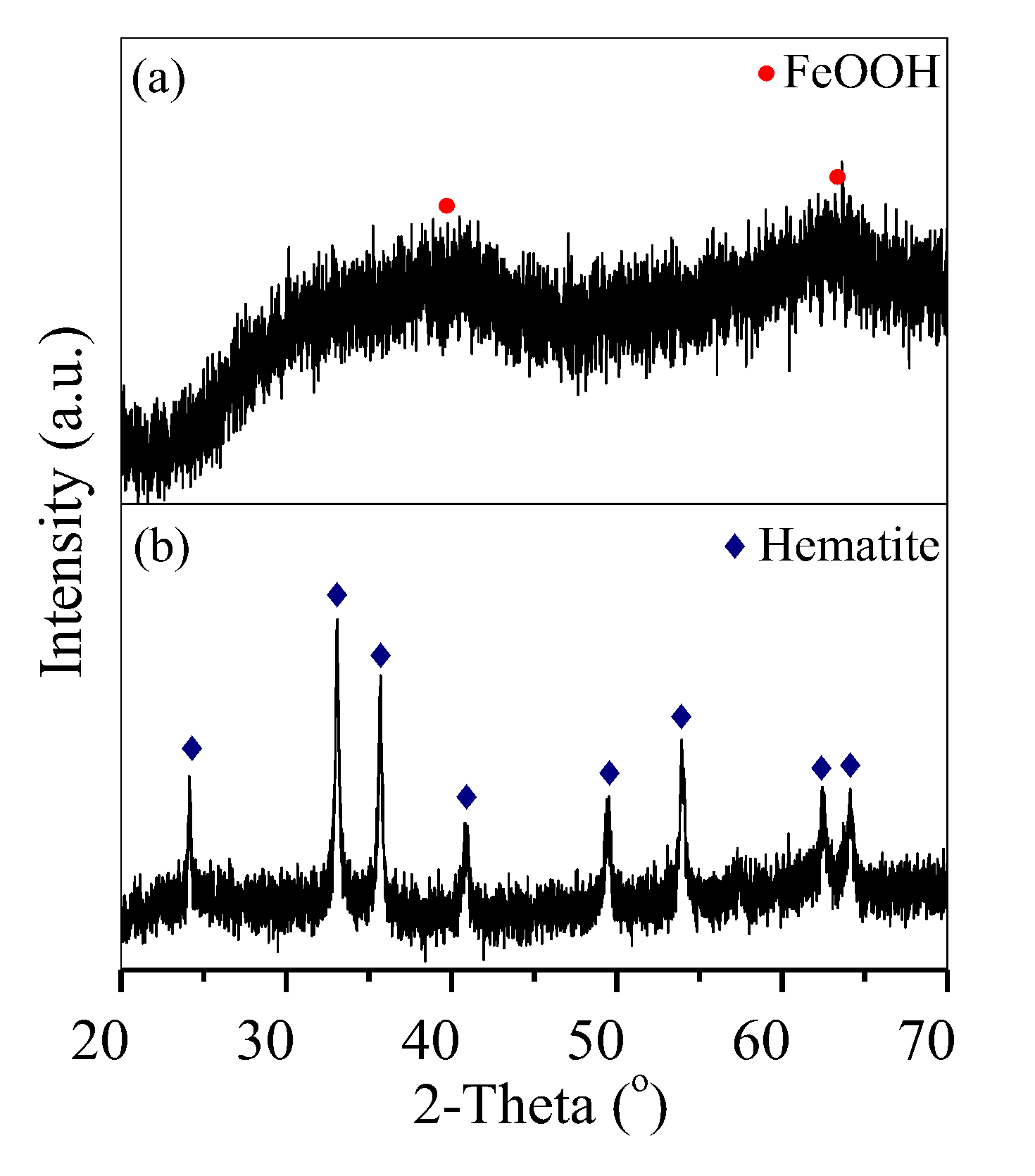
3.4. Hematite Precipitation Mechanism
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Firdaus, M.; Rhamdhani, M.A.; Durandet, Y.; Rankin, W.J.; McGregor, K. Review of high-temperature recovery of rare earth (Nd/Dy) from magnet waste. J. Sustain. Metall. 2016, 2, 276–295. [Google Scholar] [CrossRef]
- Önal, M.A.R.; Borra, C.R.; Guo, M.; Blanpain, B.; Van Gerven, T. Recycling of NdFeB Magnets Using Sulfation, Selective Roasting, and Water Leaching. J. Sustain. Metall. 2015, 1, 199–215. [Google Scholar] [CrossRef]
- Yang, Y.; Walton, A.; Sheridan, R.; Güth, K.; Gauß, R.; Gutfleisch, O.; Buchert, M.; Steenari, B.-M.; Van Gerven, T.; Jones, P.T.; et al. REE Recovery from End-of-Life NdFeB Permanent Magnet Scrap: A Critical Review. J. Sustain. Metall. 2017, 3, 122–149. [Google Scholar] [CrossRef]
- Önal, M.A.R.; Borra, C.R.; Guo, M.; Blanpain, B.; Van Gerven, T. Hydrometallurgical recycling of NdFeB magnets: Complete leaching, iron removal and electrolysis. J. Rare Earths 2017, 35, 574–584. [Google Scholar] [CrossRef]
- Yang, F.; Deng, Z.; Wei, C.; Li, C.-X.; Li, X.-B. Iron-removal by hematite from leaching liquor of high iron sphalerite. Chin. J. Nonferrous Met. 2014, 24, 2387–2392. [Google Scholar]
- Lee, C.-H.; Chen, Y.-J.; Liao, C.-H.; Popuri, S.R.; Tsai, S.-L.; Hung, C.-E. Selective leaching process for neodymium recovery from scrap Nd-Fe-B magnet. Metall. Mater. Trans. A 2013, 44, 5825–5833. [Google Scholar] [CrossRef]
- Zhou, H.; Wang, Y.; Guo, X.; Dong, Y.; Su, X.; Sun, X. The recovery of rare earth by a novel extraction and precipitation strategy using functional ionic liquids. J. Mol. Liq. 2018, 254, 414–420. [Google Scholar] [CrossRef]
- Parhi, P.; Sethy, T.; Rout, P.; Sarangi, K. Separation and recovery of neodymium and praseodymium from permanent magnet scrap through the hydrometallurgical route. Sep. Sci. Technol. 2016, 51, 2232–2241. [Google Scholar] [CrossRef]
- Matsumiya, M.; Kikuchi, Y.; Yamada, T.; Kawakami, S. Extraction of rare earth ions by tri-n-butylphosphate/phosphonium ionic liquids and the feasibility of recovery by direct electrodeposition. Sep. Purif. Technol. 2014, 130, 91–101. [Google Scholar] [CrossRef]
- Ponou, J.; Garrouste, M.; Dodbiba, G.; Fujita, T.; Ahn, J.-W. Sulfation–Roasting–Leaching–Precipitation Processes for Selective Recovery of Erbium from Bottom Ash. Sustainability 2019, 11, 3461. [Google Scholar] [CrossRef]
- Jianmin, Z. Ferrihydrite: Surface structure and its effects on phase transformation. Clay. Clay. Miner. 1994, 42, 737–746. [Google Scholar] [CrossRef]
- Qu, Z.; Wu, Y.; Zhu, S.; Yu, Y.; Huo, M.; Zhang, L.; Yang, J.; Bian, D.; Wang, Y. Green Synthesis of Magnetic Adsorbent Using Groundwater Treatment Sludge for Tetracycline Adsorption. Engineering 2019, 5, 880–887. [Google Scholar] [CrossRef]
- Sun, M.; Yan, L.; Zhang, L.; Song, L.; Guo, J.; Zhang, H. New insights into the rapid formation of initial membrane fouling after in-situ cleaning in a membrane bioreactor. Process Biochem. 2019, 78, 108–113. [Google Scholar] [CrossRef]
- Pavón, S.; Fortuny, A.; Coll, M.; Sastre, A. Neodymium recovery from NdFeB magnet wastes using Primene 81R· Cyanex 572 IL by solvent extraction. J. Environ. Manag. 2018, 222, 359–367. [Google Scholar] [CrossRef]
- Wieczorek-Ciurowa, K.; Kozak, A. The thermal decomposition of Fe (NO3)3·9H2O. J. Therm. Anal. Calorim. 1999, 58, 647–651. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, M.; Song, L.; Guo, J.; Zhang, L. Fate of NaClO and membrane foulants during in-situ cleaning of membrane bioreactors: Combined effect on thermodynamic properties of sludge. Biochem. Eng. J. 2019, 147, 146–152. [Google Scholar] [CrossRef]
- Zhu, S.; Fang, S.; Huo, M.; Yu, Y.; Chen, Y.; Yang, X.; Geng, Z.; Wang, Y.; Bian, D.; Huo, H. A novel conversion of the groundwater treatment sludge to magnetic particles for the adsorption of methylene blue. J. Hazard. Mater. 2015, 292, 173–179. [Google Scholar] [CrossRef]
- Geng, Z.; Yu, Y.; Zhu, S.; Yu, H.; Liu, J.; Bian, D.; Yang, X.; Huo, H.; Huo, M. Comparing polyethersulfone and polyurethane-immobilized cells of Comamonas testosteroni QYY in treatment of an accidental dye wastewater. Chem. Res. Chin. Univ. 2017, 33, 36–43. [Google Scholar] [CrossRef]
- Zhu, S.; Lin, X.; Dong, G.; Yu, Y.; Yu, H.; Bian, D.; Zhang, L.; Yang, J.; Wang, X.; Huo, M. Valorization of manganese-containing groundwater treatment sludge by preparing magnetic adsorbent for Cu(II) adsorption. J. Environ. Manag. 2019, 236, 446–454. [Google Scholar] [CrossRef]
- Sandroni, V.; Smith, C.M.M. Microwave digestion of sludge, soil and sediment samples for metal analysis by inductively coupled plasma–atomic emission spectrometry. Anal. Chim. Acta 2002, 468, 335–344. [Google Scholar] [CrossRef]
- Liu, S.; Jian, S.; Huang, Z. Carbon spheres/activated carbon composite materials with high Cr(VI) adsorption capacity prepared by a hydrothermal method. J. Hazard. Mater. 2010, 173, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Heinen, A.W.; Peters, J.A.; van Bekkum, H. The oxidation of fructose on Pt/C catalysts. the formation of d-threo-hexo-2,5-diulose and the effect of additives. Carbohydr. Res. 1997, 304, 155–164. [Google Scholar] [CrossRef]
- Sheng, Q.; Shen, Y.; Zhang, H.; Zheng, J. Neodymium (III) hexacyanoferrate (II) nanoparticles induced by enzymatic reaction and their use in biosensing of glucose. Electrochim. Acta 2008, 53, 4687–4692. [Google Scholar] [CrossRef]
- Vander Hoogerstraete, T.; Blanpain, B.; Van Gerven, T.; Binnemans, K. From NdFeB magnets towards the rare-earth oxides: A recycling process consuming only oxalic acid. RSC Adv. 2014, 4, 64099–64111. [Google Scholar] [CrossRef]
- Min, L.; Wei, L.; Liu, S. Control of the morphology and chemical properties of carbon spheres prepared from glucose by a hydrothermal method. J. Mater. Res. 2012, 27, 1117–1123. [Google Scholar]
- Delgado-Andrade, C.; Seiquer, I.; Nieto, R.; Navarro, M.P. Effects of heated glucose–lysine and glucose–methionine model-systems on mineral solubility. Food Chem. 2004, 87, 329–337. [Google Scholar] [CrossRef]
- Qu, Z.; Dong, G.; Zhu, S.; Yu, Y.; Huo, M.; Xu, K.; Liu, M. Recycling of groundwater treatment sludge to prepare nano-rod erdite particles for tetracycline adsorption. J. Clean. Prod. 2020, 257, 120462. [Google Scholar] [CrossRef]
- Qu, Z.; Su, T.; Chen, Y.; Lin, X.; Yu, Y.; Zhu, S.; Xie, X.; Huo, M. Effective enrichment of Zn from smelting wastewater via an integrated Fe coagulation and hematite precipitation method. Front. Environ. Sci. Eng. 2019, 13, 94. [Google Scholar] [CrossRef]
- Fouad, N.E.; Ismail, H.M.; Zaki, M.I. Recovery of red iron oxide pigmentary powders from chemically-modified steel-pickling chemical waste. J. Mater. Sci. Lett. 1998, 17, 27–29. [Google Scholar] [CrossRef]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, S.; Su, T.; Chen, Y.; Qu, Z.; Lin, X.; Lu, Y.; Huo, M. Resource Recovery of Waste Nd–Fe–B Scrap: Effective Separation of Fe as High-Purity Hematite Nanoparticles. Sustainability 2020, 12, 2624. https://doi.org/10.3390/su12072624
Zhu S, Su T, Chen Y, Qu Z, Lin X, Lu Y, Huo M. Resource Recovery of Waste Nd–Fe–B Scrap: Effective Separation of Fe as High-Purity Hematite Nanoparticles. Sustainability. 2020; 12(7):2624. https://doi.org/10.3390/su12072624
Chicago/Turabian StyleZhu, Suiyi, Ting Su, Yu Chen, Zhan Qu, Xue Lin, Ying Lu, and Mingxin Huo. 2020. "Resource Recovery of Waste Nd–Fe–B Scrap: Effective Separation of Fe as High-Purity Hematite Nanoparticles" Sustainability 12, no. 7: 2624. https://doi.org/10.3390/su12072624
APA StyleZhu, S., Su, T., Chen, Y., Qu, Z., Lin, X., Lu, Y., & Huo, M. (2020). Resource Recovery of Waste Nd–Fe–B Scrap: Effective Separation of Fe as High-Purity Hematite Nanoparticles. Sustainability, 12(7), 2624. https://doi.org/10.3390/su12072624




