RETRACTED: In-Situ Yeast Fermentation to Enhance Bioconversion of Coconut Endosperm Waste into Larval Biomass of Hermetia illucens: Statistical Augmentation of Larval Lipid Content
Abstract
1. Introduction
2. Materials and Methods
2.1. Coconut Endosperm Waste
2.2. Black Soldier Fly Larvae (BSFL)
2.3. Rearing of BSFL Using CEW Inoculated with Yeast
2.4. Growth Performance of BSFL
2.5. Treatment of CEW Via Reduction by BSFL
2.6. Biochemical Analysis
2.6.1. Lipid Solvent Extraction
2.6.2. Transesterification and FAME Analysis
3. Results and Discussion
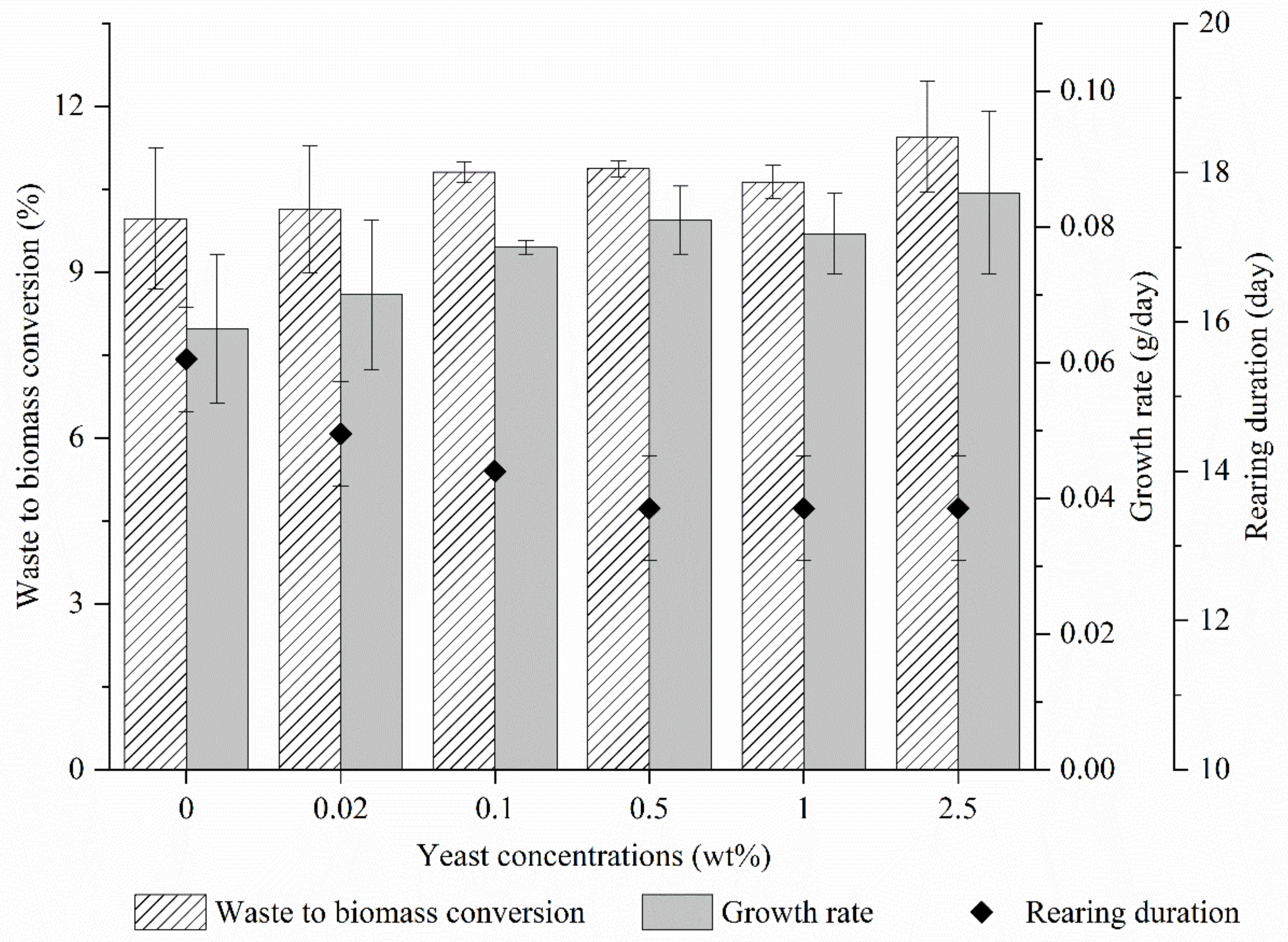
3.1. BSFL Growth through Rearing at Different Yeast Concentrations
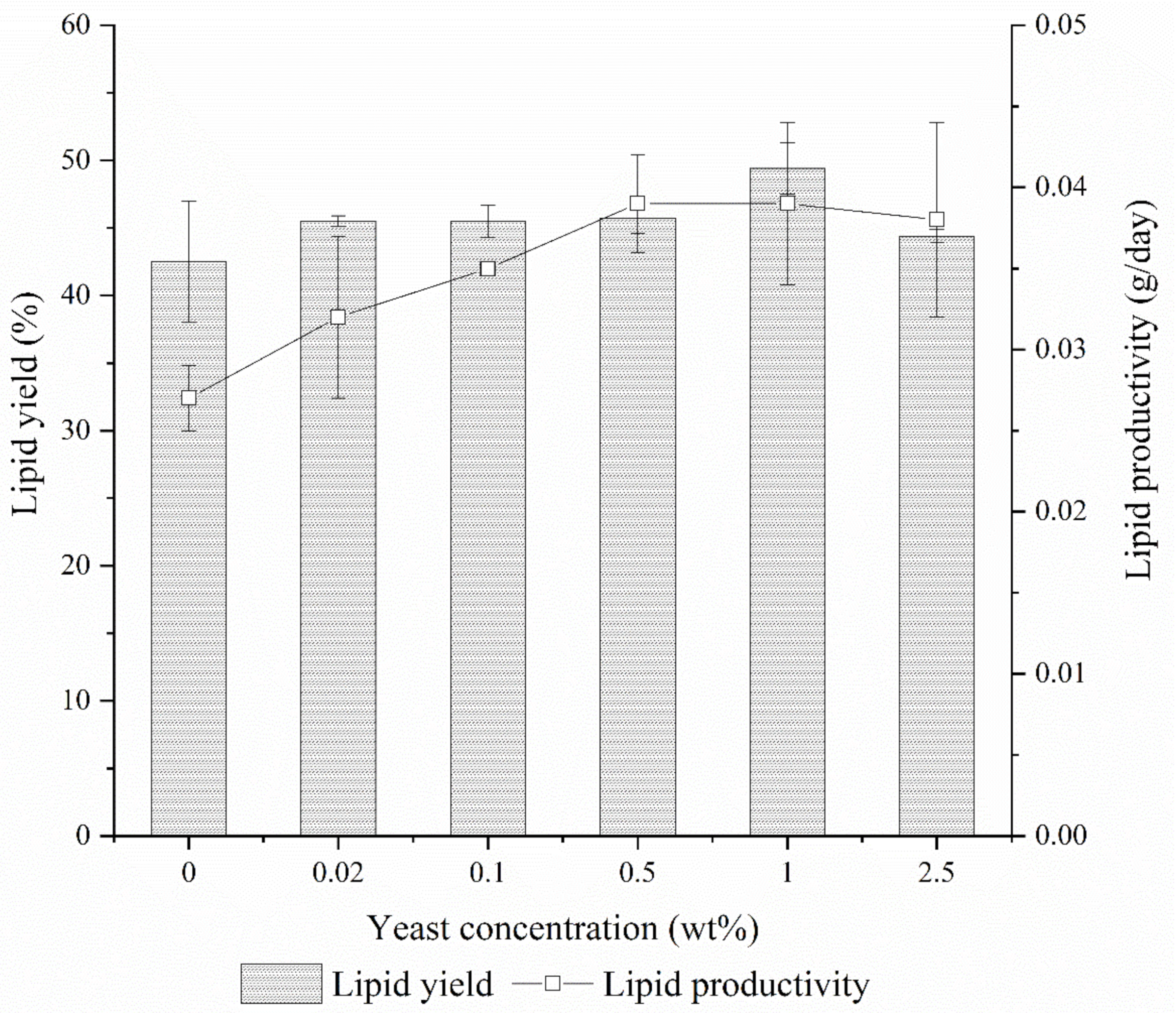
3.2. Lipid and Biodiesel from BSFL
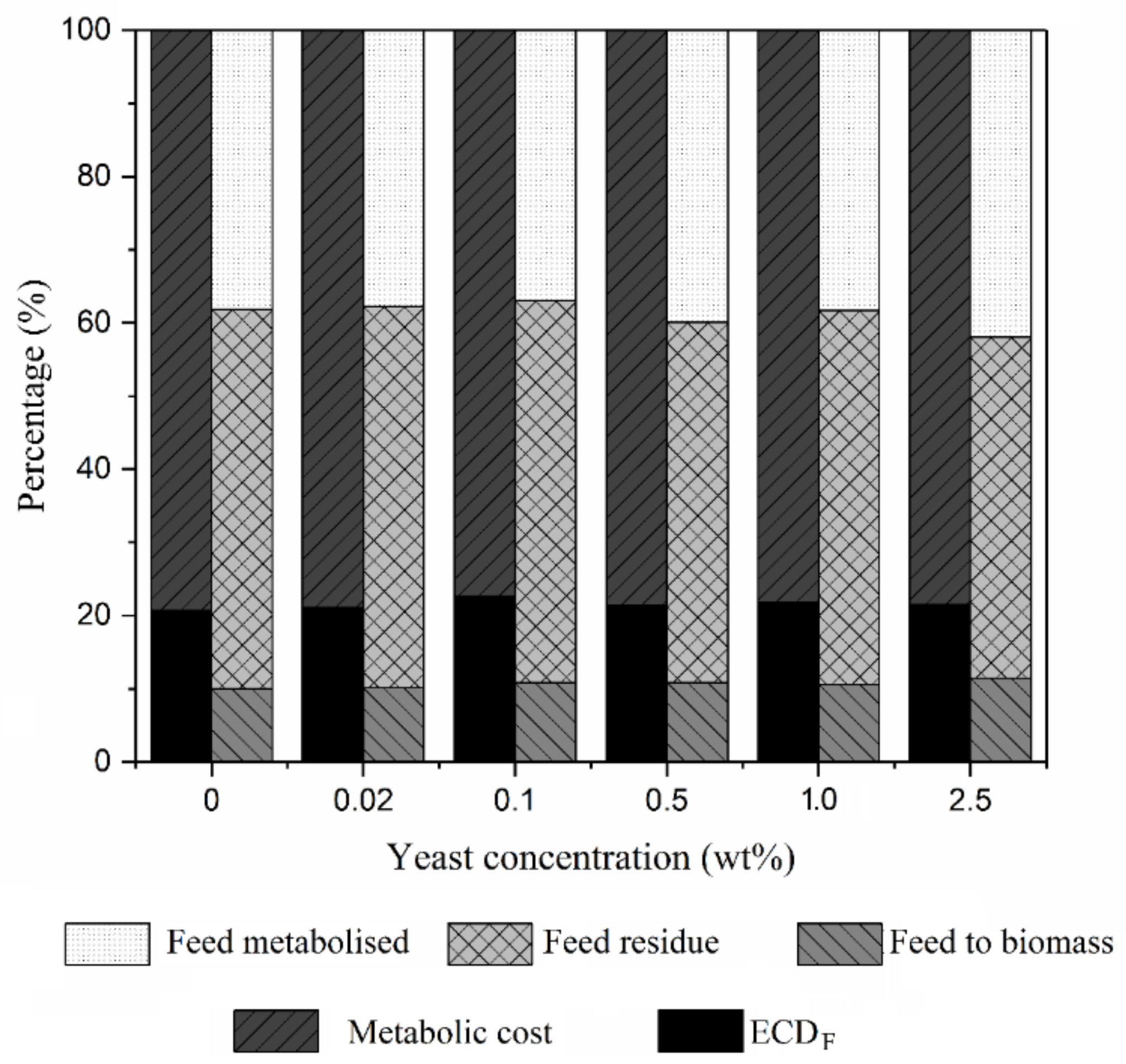
3.3. Waste Reduction of CEW by BSFL
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mathimani, T.; Pugazhendhi, A. Utilization of algae for biofuel, bio-products and bio-remediation. Biocatal. Agric. Biotechnol. 2019, 17, 326–330. [Google Scholar] [CrossRef]
- Raheem, A.; Prinsen, P.; Vuppaladadiyam, A.K.; Zhao, M.; Luque, R. A review on sustainable microalgae based biofuel and bioenergy production: Recent developments. J. Clean. Prod. 2018, 181, 42–59. [Google Scholar] [CrossRef]
- Adeniyi, O.M.; Azimov, U.; Burluka, A. Algae biofuel: Current status and future applications. Renew. Sustain. Energy Rev. 2018, 90, 316–335. [Google Scholar] [CrossRef]
- Gahukar, R. Edible insects farming: Efficiency and impact on family livelihood, food security, and environment compared with livestock and crops. In Insects as Sustainable Food Ingredients; Elsevier: Amsterdam, The Netherlands, 2016; pp. 85–111. [Google Scholar]
- Manzano-Agugliaroa, F.; Sanchez-Muros, M.J.; Barroso, F.G.; Martínez-Sánchez, A.; Rojo, S.; Pérez-Bãnón, C. Insects for biodiesel production. Renew. Sustain. Energy Rev. 2012, 16, 3744–3753. [Google Scholar] [CrossRef]
- Diclaro, J.W.; Kaufman, P.E. Black soldier fly hermetia illucens linnaeus (insecta: Diptera: Stratiomyidae). EENY 2009, 461, 1–3. [Google Scholar]
- Mohd-Noor, S.-N.; Wong, C.-Y.; Lim, J.-W.; Uemura, Y.; Lam, M.-K.; Ramli, A.; Bashir, M.J.; Tham, L. Optimization of self-fermented period of waste coconut endosperm destined to feed black soldier fly larvae in enhancing the lipid and protein yields. Renew. Energy 2017, 111, 646–654. [Google Scholar] [CrossRef]
- Surendra, K.C.; Oliver, R.; Tomberlin, J.K.; Jha, R.; Khanal, S.K. Bioconversion of organic wastes into biodiesel and animal feed via insect farming. Renew. Energy 2016, 98, 197–202. [Google Scholar] [CrossRef]
- Li, Q.; Zheng, L.; Cai, H.; Garza, E.; Yu, Z.; Zhou, S. From organic waste to biodiesel: Black soldier fly, hermetia illucens makes it feasible. Fuel 2011, 90, 1545–1548. [Google Scholar] [CrossRef]
- Meneguz, M.; Schiavone, A.; Gai, F.; Dama, A.; Lussiana, C.; Renna, M.; Gasco, L. Effect of rearing substrate on growth performance, waste reduction efficiency and chemical composition of black soldier fly (hermetia illucens) larvae. J. Sci. Food Agric. 2018, 98, 5776–5784. [Google Scholar] [CrossRef]
- Nguyen, T.T.X.; Tomberlin, J.K.; Vanlaerhoven, S. Influence of resources on hermetia illucens (diptera: Stratiomyidae) larval deveopment. J. Med. Entomol. 2013, 50, 898–906. [Google Scholar] [CrossRef]
- Zheng, L.; Hou, Y.; Li, W.; Yang, S.; Li, Q.; Yu, Z. Biodiesel production from rice straw and restaurant waste employing black soldier fly assisted by microbes. Energy 2012, 47, 225–229. [Google Scholar] [CrossRef]
- Wong, C.-Y.; Rosli, S.-S.; Uemura, Y.; Ho, Y.C.; Leejeerajumnean, A.; Kiatkittipong, W.; Cheng, C.-K.; Lam, M.-K.; Lim, J.-W. Potential protein and biodiesel sources from black soldier fly larvae: Insights of larval harvesting instar and fermented feeding medium. Energies 2019, 12, 1570. [Google Scholar] [CrossRef]
- Chen, H. Modern Solid State Fermentation Theory and Practice; Springer Science: New York, NY, USA; London, UK, 2013. [Google Scholar]
- Garcí;a, C.C.G.; Mendoza, M.G.D.; González, M.S.; Cobos, P.M.; Ortega, C.M.E.; Ramirez, L.R. Effect of a yeast culture (saccharomyces cerevisiae) and monensin on ruminal fermentation and digestion in sheep. Anim. Feed Sci. Technol. 2000, 83, 165–170. [Google Scholar] [CrossRef]
- Kim, K.H.; Choi, I.S.; Kim, H.M.; Wi, S.G.; Bae, H.-J. Bioethanol production from the nutrient stress-induced microalga chlorella vulgaris by enzymatic hydrolysis and immobilized yeast fermentation. Bioresour. Technol. 2014, 153, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Harun, R.; Danquah, M.K.; Forde, G.M. Microalgal biomass as a fermentation feedstock for bioethanol production. J. Chem. Technol. Biotechnol. 2010, 85, 199–203. [Google Scholar] [CrossRef]
- Bradley, R.L. Moisture and total solids analysis. In Food Analysis; Springer: Boston, MA, USA, 2010; pp. 85–104. [Google Scholar]
- Kim, W.-T.; Bae, S.-W.; Park, H.-C.; Park, K.-H.; Lee, S.-B.; Choi, Y.-C.; Han, S.-M.; Koh, Y.-H. The larval age and mouth morphology of the black soldier fly, hermetia illucens (diptera: Stratiomyidae). Int. J. Indust. Entomol. 2010, 21, 185–187. [Google Scholar]
- Li, Q.; Zheng, L.; Qiu, N.; Cai, H.; Tomberlin, J.K.; Yu, Z. Bioconversion of dairy manure by black soldier fly (diptera: Stratiomyidae) for biodiesel and sugar production. Waste Manag. 2011, 31, 1316–1320. [Google Scholar] [CrossRef]
- Lalander, C.; Diener, S.; Zurbrügg, C.; Vinnerås, B. Effects of feedstock on larval development and process efficiency in waste treatment with black soldier fly (hermetia illucens). J. Clean. Prod. 2019, 208, 211–219. [Google Scholar] [CrossRef]
- Parra, J.R.; Panizzi, A.R.; Haddad, M.L. Nutritional indices for measuring insect food intake and utilization. In Insect Bioecology and Nutrition for Integrated Pest Management; Panizzi, A.R., Parra, J.R., Eds.; CRC Press: London, UK; New York, NY, USA, 2012; p. 13. [Google Scholar]
- Pedrosa, R.; Tecelão, C.; Gil, M.M. Lipids in meat and seafood. In Methods in Food Analysis; Cruz, R.M., Khmelinskii, I., Vieira, M.C., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 142–200. [Google Scholar]
- Zheng, L.; Li, Q.; Zhang, J.; Yu, Z. Double the biodiesel yield: Rearing black soldier fly larvae, hermetia illucens, on solid residual fraction of restaurant waste after grease extraction for biodiesel production. Renew. Energy 2012, 41, 75–79. [Google Scholar] [CrossRef]
- Mohd-Sahib, A.-A.; Lim, J.-W.; Lam, M.-K.; Uemura, Y.; Isa, M.H.; Ho, C.-D.; Kutty, S.R.M.; Wong, C.-Y.; Rosli, S.-S. Lipid for biodiesel production from attached growth chlorella vulgaris biomass cultivating in fluidized bed bioreactor packed with polyurethane foam material. Bioresour. Technol. 2017, 239, 127–136. [Google Scholar] [CrossRef]
- Wiedmeier, R.D.; Arambel, M.J.; Walters, J.L. Effect of yeast culture and aspergillus oryzae fermentation extract on ruminal characteristics and nutrient digestibility1. J. Dairy Sci. 1987, 70, 2063–2068. [Google Scholar] [CrossRef]
- Yoon, S.-H.; Mukerjea, R.; Robyt, J.F. Specificity of yeast (saccharomyces cerevisiae) in removing carbohydrates by fermentation. Carbohydr. Res. 2003, 338, 1127–1132. [Google Scholar] [CrossRef]
- Li, W.; Li, M.; Zheng, L.; Liu, Y.; Zhang, Y.; Yu, Z.; Ma, Z.; Li, Q. Simultaneous utilization of glucose and xylose for lipid accumulation in black soldier fly. Biotechnol. Biofuels 2015, 8, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Boulton, C.; Quain, D. Brewing Yeast and Fermentation; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Nguyen, H.C.; Liang, S.-H.; Li, S.-Y.; Su, C.-H.; Chien, C.-C.; Chen, Y.-J.; Huong, D.T.M. Direct transesterification of black soldier fly larvae (hermetia illucens) for biodiesel production. J. Taiwan Inst. Chem. Eng. 2018, 85, 165–169. [Google Scholar] [CrossRef]
- Deshmukh, S.; Kumar, R.; Bala, K. Microalgae biodiesel: A review on oil extraction, fatty acid composition, properties and effect on engine performance and emissions. Fuel Process. Technol. 2019, 191, 232–247. [Google Scholar] [CrossRef]
- Arcos-García, J.L.; Castrejón, F.A.; Mendoza, G.D.; Pérez-Gavilán, E.P. Effect of two commercial yeast cultures with saccharomyces cerevisiae on ruminal fermentation and digestion in sheep fed sugar cane tops. Livest. Prod. Sci. 2000, 63, 153–157. [Google Scholar] [CrossRef]
| Fatty Acid Methyl Ester | This Study | BSFL Reared with Food Waste [8] | Chlorella vulgaris [31] | Jatropha [31] | Palm [31] |
|---|---|---|---|---|---|
| C10:0 | 1.3 ± 0.06 | n/a | n/a | n/a | n/a |
| C12:0 | 66.0 ± 1.44 | 44.9 | n/a | n/a | n/a |
| C14:0 | 14.4 ± 0.12 | 8.3 | 2.3 | 0.2 | 1.22 |
| C14:1 | 1.4 ± 0.01 | n/a | n/a | n/a | n/a |
| C15:0 | n/a | n/a | n/a | n/a | n/a |
| C16:0 | 6.6 ± 0.57 | 13.5 | 6 | 13.4 | 47.9 |
| C16:1 | 2.2 ± 0.15 | 2.4 | 16.4 | 0.8 | 0.04 |
| C17:0 | n/a | n/a | n/a | n/a | n/a |
| C18:0 | 0.4 ± 0.09 | 2.1 | 10.3 | 6.4 | 4.23 |
| C18:1 | 5.6 ± 0.45 | 12.0 | 26.5 | 36.5 | 37 |
| C18:2 | 2.3 ± 0.22 | 9.9 | n/a | 42.1 | 9.07 |
| C18:3 | n/a | 0.1 | 20.8 | 0.3 | 0.26 |
| Yeast Concentration (wt %) | Overall Degradation (%) | Waste Reduction Index (g/day) |
|---|---|---|
| 0 | 48.10 ± 0.39 | 0.31 ± 0.02 |
| 0.02 | 47.87 ± 0.95 | 0.33 ± 0.02 |
| 0.1 | 47.79 ± 1.27 | 0.34 ± 0.01 |
| 0.5 | 50.80 ± 0.73 | 0.38 ± 0.01 |
| 1.0 | 48.94 ± 0.07 | 0.36 ± 0.02 |
| 2.5 | 53.43 ± 1.62 | 0.40 ± 0.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wong, C.Y.; Mohd Aris, M.N.; Daud, H.; Lam, M.K.; Yong, C.S.; Abu Hasan, H.; Chong, S.; Show, P.L.; Hajoeningtijas, O.D.; Ho, Y.C.; et al. RETRACTED: In-Situ Yeast Fermentation to Enhance Bioconversion of Coconut Endosperm Waste into Larval Biomass of Hermetia illucens: Statistical Augmentation of Larval Lipid Content. Sustainability 2020, 12, 1558. https://doi.org/10.3390/su12041558
Wong CY, Mohd Aris MN, Daud H, Lam MK, Yong CS, Abu Hasan H, Chong S, Show PL, Hajoeningtijas OD, Ho YC, et al. RETRACTED: In-Situ Yeast Fermentation to Enhance Bioconversion of Coconut Endosperm Waste into Larval Biomass of Hermetia illucens: Statistical Augmentation of Larval Lipid Content. Sustainability. 2020; 12(4):1558. https://doi.org/10.3390/su12041558
Chicago/Turabian StyleWong, Chung Yiin, Muhammad Naeim Mohd Aris, Hanita Daud, Man Kee Lam, Ching Seong Yong, Hadura Abu Hasan, Siewhui Chong, Pau Loke Show, Oetami Dwi Hajoeningtijas, Yeek Chia Ho, and et al. 2020. "RETRACTED: In-Situ Yeast Fermentation to Enhance Bioconversion of Coconut Endosperm Waste into Larval Biomass of Hermetia illucens: Statistical Augmentation of Larval Lipid Content" Sustainability 12, no. 4: 1558. https://doi.org/10.3390/su12041558
APA StyleWong, C. Y., Mohd Aris, M. N., Daud, H., Lam, M. K., Yong, C. S., Abu Hasan, H., Chong, S., Show, P. L., Hajoeningtijas, O. D., Ho, Y. C., Goh, P. S., Kausarian, H., Pan, G.-T., & Lim, J. W. (2020). RETRACTED: In-Situ Yeast Fermentation to Enhance Bioconversion of Coconut Endosperm Waste into Larval Biomass of Hermetia illucens: Statistical Augmentation of Larval Lipid Content. Sustainability, 12(4), 1558. https://doi.org/10.3390/su12041558










