1. Introduction
Humans forget easily. Their origins, their destiny, their history, and their life often look confused to them, much like a veil of cold senselessness has fallen before their eyes. This is a weakness known since the dawn of time and clearly stated in Psalm 49:12 [
1], which reads: “Nevertheless man being in honor abideth not: he is like the beasts that perish.” Foolish confidence cannot save.
In this era of growing concerns over environmental issues, such words resound more powerfully than ever. For centuries, humans have exploited natural resources to the detriment of the environment at an increasingly fast rate, running after the profit of a few and in complete disregard of the most elementary rules of health and environmental protection [
2]. Blinded by the stunning resilience of nature, humans have always taken the environment for granted, assuming that natural resources are limitless, and that activities potentially damaging the environment can be scaled up regardless of their effects.
Unfortunately, activities relatively harmless on the small scale have become impressively destructive when translated on the global scale, resulting in the disproportionate, and ultimately lethal, pressure on Earth that characterizes the last decades [
2,
3,
4].
Nevertheless, the formulation of concepts strictly related to modern sustainability issues can be traced back to early 18th century. It is in those years that Hans Carl von Carlowitz, in his pioneering work [
5], pointed out the need for planned reforestation projects based on a simple balance between wood to be cut and wood able to regrow.
Since then, sustainability has been related to the capacity of identifying the most suitable conditions for the harmonious coexistence of human activities and environment [
6]. Admittedly, such a concept cannot but involve social, technological, economic and environmental issues, deeply intertwined in what industrialized countries refer to as modern civilization [
6,
7]. Therefore, it is not surprising that sustainability challenges encompass most of the archetypical trouble bearers, ranging from environmental pollution to industrial production, demographic crisis, misuse of lands and waste disposal [
6,
7,
8]. In facing these challenges, our societies are, stricto sensu, desperately seeking the most convenient compromise between the needs of the present generation and those of future ones.
An indication of this sense of urgency can be found, not only in scientific reports echoing the calls for commitment to properly address the sustainability conundrum in developed and developing countries. Philosophers, writers and thinkers have also pointed the finger at the dangers that technological and industrial development can bring. Their stories and ideas have deeply influenced the present capacity to imagine solutions and can be regarded as a measure of how strong the feeling is that humans have to undertake important actions to suitably cope with the limitedness of natural resources.
Reducing the population of human beings has been even imagined as a strategy to contrast contemporary global crises related to the world population increase [
9]. Certainly, significant downsizing would mitigate, or remove, any anthropic effect on the environment. The smaller scale would make the globe much bigger.
While such a scenario is unavoidably destined to be relegated to imaginary science, the small scale can still give interesting ideas to face the future. Moreover, it is precisely at the small scale that humans find a global wet biomass that is almost twice the size of common livestock [
10]. Terrestrial arthropods, the vast majority of which consists of insects, form a wet biomass that amounts up to 1.3 gigatons [
10]. Such a huge biomass represents a great wealth that Earth offers to humans and that can be potentially used to combat hunger and environmental issues related to traditional intensive farming. To this aim, intensive farming must be extended to insects.
Insect farming has remained an almost unknown practice until recently. Usually, insects were collected by wild harvest in Africa and Asia [
11]. The use of land for agriculture, desertification and urbanization have progressively induced a decline of insect wild catch [
11]. At the same time, the farming of edible insects has begun to rise as a valid, more sustainable alternative to wild harvesting [
12,
13]. At present, insect rearing is attracting increasing interest also in countries not traditionally involved in collecting insects, such as Europe and the United States of America, and insect-based products are increasing their market share. Insect farming, indeed, combines economic benefits with the production of food and feed ingredients that can beneficially affect the diet of population in both developed and developing countries.
Quite interestingly, insect rearing can significantly contribute to a sustainable development of modern societies. In particular, it fits well into the itinerary that leads modern economy to the transition from the old to the new paradigms in response to sustainability and environmental issues.
In this respect, it is worth noting that a possible response to some general questions raised by the concept of sustainability is rooted in the development of environmental movement in the early 1960s and, in particular, in the realization of the close relationship between environment deterioration and economic growth in both industrialized and underdeveloped countries [
14]. Shortly after, it was clearly recognized that an economic system has to properly fit to the surrounding environment, taking into due account the limitedness of its resources [
15]. This implies a closed economy, characterized by more or less severe constraints on the use of available resources with the aim of lengthening resources’ lifetimes in economic cycles.
Not yet a circular economy, this model knew a rapid evolution towards more sophisticated versions [
16,
17,
18,
19]. Closed loops have appeared in the economic thought, gradually displacing the so-called linear model based on a simple, light-hearted sequence of acts consisting of taking what is needed, making what is desired and eventually disposing what is exploited. Inspired by living systems, the economy has begun conjecturing about the benefits of regenerative terms in the equations of input and output of natural resources in the wide range of human activities from individual lifestyle to industrial processes [
20].
At present, circular economy is a multifaceted reality well established in the modern concept of sustainable development [
21,
22,
23]. Professed in speeches and reports of committees on various levels, taught in academic courses, invoked by many and still pursued by a few, it shows great promise. Aimed at keeping the value of resources produced and consumed as high as possible, circular economy proposes tangible strategies to preserve job positions and create new ones, thus stimulating economic growth while simultaneously protecting the environment. Combined with the raised awareness of environmental issues, it enhances the capability of more and less industrialized societies to suitably deal with the crucial issues arising on the way to further development [
24,
25].
Meeting the global food demand is one of the formidable challenges that mankind has to take up in the next future [
26,
27]. A huge increase in demand for primary foodstuffs is expected within a few decades, requiring a corresponding increase in food production. Drought and desertification, misuses of land and water, and the gradient between rich and poor countries have already given rise to an expanding gap in this regard, which can translate in significant food shortage in the absence of definite actions.
Emerging strategies to contrast this tendency and find viable solutions are discussed worldwide. Among them, protein production by insect farming is attracting increasing interest [
28]. While this practice displays enormous potential to support the fight against undernutrition and hidden hunger in specific areas of the planet, it can really enhance food security worldwide [
28]. Even more, insect farming is invoked as part of the solution to the global demand for primary foodstuffs, providing, in addition, an interesting case study for circular economy applied to agri-food industry [
28].
This short review focuses exactly on insect farming and the promise it shows for enhancing circularity and sustainability in animal feeding. It provides a short overview of the most relevant available literature in the field. First, we summarize the current state of the art, pointing out origins and perspectives of the most influential factors for the further development of more and less industrialized countries and the preservation of environment. Then, we highlight the considerable potential of insect farming in mitigating the societal challenges related to the increase of world population and the consequent increase of food and feed needs all across the world. The potential of insects to increase food security and contribute to the dietary quality of world population through vital nutrients and micronutrients is pointed out. After a general survey of the outstanding nutritional values of insect species, we discuss the potential associated with the intensive rearing of Hermetia illucens, Musca domestica and Tenebrio molitor, with special emphasis for aquaculture, i.e., the industrial activity within agri-food industry that is most sensitive to insect rearing and responsive to feeding innovation, and related feed production. Eventually, we analyze the traditional and innovative insect rearing methods. Specifically, we emphasize the challenges that researchers, entrepreneurs and stakeholders in general have to take on to allow the necessary development of the field and the realization of modern insect rearing systems.
This short review aims to represent a practical guide for specialized researchers, interested entrepreneurs and even common readers potentially attracted by the new business. It can be regarded as a brief introduction to the vast literature characterizing this broad field of study and its variegated and multidisciplinary areas of investigation. Currently available literature already includes comprehensive and detailed reviews and books on the most different aspects of insect rearing and insect utilization as sources of ingredients for food and feed. Most of such publications are mentioned in this short review.
2. The Reasons behind a Challenge
Perhaps, not everyone knows that sustainability is measured in planets. The unit of measure is the Earth itself, i.e., the resources that Earth is able to generate in a year. Every year, based on the data collected by the Global Footprint Network [
29], the day in which human beings finish consuming all the available resources on the planet for that current year is estimated. The day in which Earth goes beyond its limits is referred to as the Earth Overshoot Day and it is a simple intuitive measure of the way things are getting worse [
29]. In 2000, the Earth Overshoot Day fell in October. In 2017, it fell early in August, i.e., two months earlier. This means that humans used fewer natural resources in 2000 than in 2017. Specifically, humans used more water, exploited more land for farming, and caught more fish from seas, rivers and lakes in 2017 than in 2000. At the same time, humans consumed more fossil fuels and released more wastes in the environment.
A closer look at details reveals shocking results. Lifestyle has a planetary footprint [
29]. Numbers are crude. If the entire Earth’s population lived like Italians do, mankind would need 2.6 Earths every year. If the United States’ lifestyle was the standard, the number of Earths needed to satisfy the world’s population needs would increase up to 5.2. If all human beings on the planet lived like Australians, the number of Earths would reach 5.6.
The alarming scenario is determined by a combination of factors. Among them, climate changes and urbanization, short-sighted focus on the use of energy sources and overpopulation have outstanding importance.
It is, perhaps, the population growth that draws more attention [
30]. Its rate of increase is impressive. About 70 years ago, the Earth supported the life of about 2.6 billion people. Starting from the 1950s, population growth accelerated and about 5 billion people lived on the planet in 1987 and 1 billion more were registered in 1999. Twelve years later, global population was estimated to be around 7 billion. The current population of 7.7 billion people is expected to increase up to 9.7 billion in 2050 and up to 11 billion in 2100.
Despite the increase at a slower pace, such huge numbers bring far-reaching implications for the generations to come [
30]. First and foremost, food production has to be deeply reconsidered to secure their future [
30]. More than a quarter of mankind’s footprint is related to food, indeed. Therefore, important actions have to be undertaken worldwide to change societies and improve farming methods and food production processes.
Today, food supply and its uneven distribution represent crucial societal challenges. It is a terrible reality that world hunger has started to increase again in the recent years while overweight and obesity plague most opulent countries [
31]. It is not only a striking demonstration of inequity and senselessness, but it is also a clear indication of the inappropriateness of the current food production system.
From the 1980s to 2000, global demand for food underwent a constant increase, somehow mirroring the population growth [
31,
32]. In the meantime, food prices declined on the average, thus marking an important success in the battle against hunger. The trend reversed in 2004, when prices for grains started increasing due to the depletion of food stocks. Although food production was increased overall, production rate was unable to keep pace with the exponential growth in food demand [
31,
32]. In addition, disappointing harvests in countries with most important food productions determined further reduction in food availability in 2005. Other factors negatively affected food production costs in the subsequent years and caused other contractions. Finally, food security was challenged in many countries as a consequence of the global economic crisis that hit the planet in 2008 [
31,
32].
Actually, a significant fraction of the world’s population across the planet has, inexorably, run into food insecurity. The food for these people has become unaffordable or unavailable, and in some cases, potentially unsafe for human health. In addition, micronutrient deficiencies, also known as hidden hunger, have been estimated to afflict more than 2 billion persons worldwide [
33]. Characterized by devastating adverse effects involving poor health and mental impairment, and even death in the long term, it is particularly acute in children, compromising their existence [
33]. Often caused by local political and social crises, food insecurity is also deeply related to the inefficiency of the food production system [
31,
32].
In this regard, the terrible unbalance between pasture availability and food demand is one of the most evident examples [
34]. Overall, the reduction of pastures due to urbanization and desertification has to cope with the increased demand for food [
35]. Agriculture already uses approximately the 30% of available land on Earth [
31,
32]. However, up to 70% is exploited for industrial production of common livestock, i.e., the animals typically raised on a farm [
35]. This is one of the human activities most negatively impacting on planet resources [
35]. Capable of producing up to 58 million tons of animal proteins per year, livestock consumes more than 77 million tons of plant proteins [
35]. Furthermore, the obtained animal proteins are far from being evenly distributed across the countries, as most of the consumption is being located in industrialized countries [
35].
With a world population around 9.7 billion people by 2050, the present system for meat production will not be able to cope satisfactorily with the increase in meat demand [
35]. Economic models predict an increase in meat consumption per capita of about 9% in richest countries by 2030 [
35]. In the same period, crop price will experience an increase worldwide, determining, in turn, an increase of meat price around 20% [
36]. Restrictions on agri-food policies related to the reduction of carbon footprints are expected to require a deep modification of the animal feed, moving from soy meal to locally available feed [
37]. Moreover, it is no longer acceptable to extend the surface area of land devoted to agriculture, which means that the increasing population has to be fed using, more or less, the same surface area of land exploited at present.
Unfortunately, this is already quite challenging, in the light of the growing threat to available land represented by climate changes. The surface area of land involved in agricultural activities is progressively reducing, and, even worse, numerical simulations predict, for specific soil and climate conditions, that the yield per hectare in cereal and other important cultivations can decrease in the next future [
27,
32,
34].
Although the poorest countries will be the most severely affected by the effects of climate changes, suffering from an increase of malnutrition and poverty, the problem involves the Earth in its whole. The only way to contrast this gloomy scenario is a global policy addressing the improvement of social and economic conditions as well as of the access to food [
38]. In particular, it appears necessary to enhance the diversity and sustainability of food supply [
11]. However, what to do, and how to do it, is still a matter of debate. It is not clear what this global policy has to be and, moreover, governments and other international institutions are far from finding an agreement concerning the concrete actions that have to be undertaken [
28,
31,
32,
33,
34]. Sadly enough, questions regarding global policies represent societal challenges themselves.
At this difficult time, it is Nature itself that seems graciously inclined to show a possible solution. If man is looking for diversity and sustainability in food production, Nature recalls that he is literally surrounded by countless tiny living organisms that have been referred to, not by chance, as the real masters of the world [
39]. Often regarded as just a nuisance, insects include the largest number of species compared with other groups of organisms. About 900 thousand different kinds of insects have been recorded, which approximately represents 80% of the world’s species [
40]. The real number is still debated, with estimates ranging from 2 to 30 million. Overall, conservative estimates suggest that the number of individual living insects on Earth amounts to the inconceivable figure of over 10 quintillions [
40].
Far from being a nuisance, insects can really represent a great opportunity for mankind in its search of strategies for alternative food supply and environmental preservation and protection. In addition, although entomophagy is definitely far from the culture of the vast majority of industrialized countries, people have begun considering insects as a means to support the ever-increasing world’s population via the preparation of insect-based ingredients for food and feed.
3. An Advantageous Choice
The scientific community is nearly unanimous on the benefits deriving to society from the use of insects for nutrition purposes [
28]. Such opinion stems from a simple list of characteristic features that make insects advantageous from several perspectives. On the one hand, insects are everywhere. They can be found in abundance and can be grown literally all across the world. On the other hand, they are quite robust, overall, and, at least within a wide range of conditions, adapt easily to different environmental conditions. Furthermore, insects have short reproduction cycles, which translates into a quick biomass build-up. They also exhibit a remarkably high growth rate. This mirrors a very high efficiency in converting feed mass into body mass.
Admittedly, the feed conversion efficiency of insects is much higher than the one of mammalian livestock. Despite their intrinsic variability related to specific case studies, quantitative investigations indicate that, on the average, livestock have to be fed about 6 kg of plant proteins to produce 1 kg of high-quality animal proteins [
41]. The amount of feed needed to obtain 1 kg of meat, or, equivalently, a weight increase of 1 kg, depends significantly on the class of animals fed as well as on the farming system adopted. For instance, it has been estimated that 2.5 kg of feed are needed for chickens, 5 kg for pigs and up to 10 kg for cows [
42]. The difference with crickets is striking. Only 1.7 kg of feed are needed for crickets to produce 1 kg of weight increase [
43].
If edible weight is considered, ratios become even more advantageous for insects. Indeed, while the percentage of edibility is as high as 80% for crickets, it reduces to 55% for chickens and pork and drops to 40% for cattle [
44]. It follows that, compared with poultry, the feed conversion of house crickets (A. domestica) is twice, approximately four times higher than for pigs and more than twelve times higher than for cattle [
28].
It is also worth noting that the entire life cycle of insects is characterized by an extremely reduced environmental footprint compared with that of common livestock. Greenhouse gas emissions are significantly lower. While the 18% of total greenhouse gas emissions can be ascribed to the livestock sector, including direct and indirect emissions for livestock rearing, emissions per year related to insects are more than 100 times lower [
45]. The comparison of five insect species, pigs and beef cattle, in terms of CO
2 production per kilogram of mass gain, clearly shows that insect rearing can be extremely advantageous. While beef cattle produce, on the average, 2850 kg of CO
2 per kilogram of mass gain and pigs up to 1130 CO
2 per kilogram of mass gain,
Pachnoda marginata,
Blaptica dubia,
Locusta migratoria,
Tenebrio molitor and
Acheta domesticus produce, respectively, 122, 38, 18, 6 and 2 CO
2 per kilogram of mass gain [
45]. The difference is striking. Concerning organic wastes that can concur to environmental pollution, at least a tenfold difference between livestock and insects has been estimated [
45].
Insects appear to be particularly advantageous also in terms of water consumption. Simple calculations suggest that the production of 1 kg of animal proteins requires up to 20 times more water than the production of 1 kg of grain protein [
46]. However, the amount of water to be used to obtain 1 kg of meat can become as high as 100 times larger, if the water needed to grow forage is taken into account [
47]. Overall, it appears that about 2300 L of water are needed to produce 1 kg of proteins from chickens, but the figure increases up to 3500 L for 1 kg of protein from pigs and to 22,000 L to obtain 1 kg of proteins from cows [
46]. For the latter case, different estimates have been proposed that make the amount of water required equal to no less than 43,000 L [
48].
Studies also suggest that insects, and mealworms in particular, require much less land than other sources of animal proteins. It has been estimated that the amount of animal proteins produced from mealworms grown in 1 ha of land requires 2.5 ha of land if animal proteins are obtained from milk, up to 3.5 ha in the case of poultry and pigs, and no less than 10 ha in the case of cattle [
49].
The remarkably advantageous environmental footprint, emerging from information mentioned above, has to be combined with the nutritional value of insects. In this regard, insects happen to have a quality of nutrition definitely higher than traditional and conventional livestock [
50].
As a matter of fact, the nutritional profile is outstanding [
45]. The systematic evaluation of nutrients for 236 edible insects has shown that insects can often provide amounts of energy comparable with those provided by similar quantities of more conventional food [
51]. They contain proteins, lipids, monounsaturated and polyunsaturated fatty acids in particular, carbohydrates and vitamins [
51], and very low or undetectable levels of toxic elements [
52]. In addition, they exhibit high content of micronutrients such as copper, iron, magnesium, manganese, phosphorous, selenium and zinc [
51]. Riboflavin, pantothenic acid, biotin and, occasionally, folic acid have been also found [
51].
A considerable variability has been observed within the different species of insects and, depending on their development stage, the energy content of insects can be quite high. For instance, the Australian green (weaver) ant (
Oecophylla smaragdina) can provide 1272 kcal per 100 g of fresh weight [
53]. In contrast, the adult termite of Ivory Coast (
Macrotermes subhyalinus) exhibits an energy content of about 535 kcal per 100 g of fresh weight and the larvae of the yellow mealworm (
Tenebrio molitor) can reach an energy content of 138 kcal per 100 g of fresh weight [
53].
Similar considerations hold for the protein content. Variability ranges are wide within the same insect species and similar variability is observed moving from one species to the other. Investigations carried out on different species indicate that the protein content varies between the 13% and the 77% of the dry matter. Several insects compare remarkably well with chickens, pigs, cows and fishes [
28].
Rich in fatty acids and with a remarkably high content of dietary fiber, insects contain all the essential amino acids [
54]. Along the same line, it has been shown that mealworms and fish exhibit comparable relative contents of omega-3 and omega-6 fatty acids [
28]. Studies have also suggested that eating chitin can benefit health, as a consequence of both the enhancement of gut flora and the antibiotic properties [
54].
The systematic investigation of caloric contribution over almost a hundred insect species has demonstrated that insects have a caloric content, referred to dry matter, higher than maize in 90% of cases, higher than fish in 70%, higher than beef in 60% and higher than soybeans in 50% [
55].
Concerning micronutrients, some insect species contain substances that can be hardly found elsewhere [
28]. This is the case of riboflavin in termites and thiamine in silk moth larvae and palm weevils. Compared to chicken, these insects have twenty times more riboflavin and thiamine. Compared to beef, mealworms have a higher content of proteins including all the essential amino acids, calcium, vitamin C, thiamine, vitamin A and riboflavin [
56]. It is no surprise that the relative content undergoes variations depending on specific factors such as diet, gender, stage of life cycle and microclimate [
57].
Another important constituent of the insect nutritional profile is chitin. It is a polysaccharide consisting of long polymeric chains of 2-(acetylamino)-2-deoxy-D-glucose with remarkable mechanical strength because of the intramolecular hydrogen bonds contributing to structural stability [
58]. It is the primary constituent of the exoskeleton of insects, and arthropods in general, where it can be found in combination with other organic and inorganic compounds [
58]. Chitin mostly determines the carbohydrate content of insects, which typically ranges between 2.7 and 49.8 mg per kg of fresh matter [
50]. Although human gastric juices contain the chitinase enzyme, chitin is, substantially, an indigestible fiber. Therefore, its removal improves digestibility of insects. However, chitin is known to enhance the organism’s capability to defend against parasitic infections and allergic responses, playing the important role of microbiota modulation [
50].
Finally, it is worth mentioning that several insect species exhibit the capability of converting efficiently organic wastes, side-streams and by-products of agricultural processes to animal proteins. The process is known as the bio-conversion process and it is attracting increasing interest as a valid alternative to more traditional and conventional methods to treat organic wastes [
59,
60]. It is precisely intensive insect rearing that offers the unique opportunity to break down significant amounts of organic wastes, side-streams and by-products while increasing the total insect biomass and producing a range of products from biofuels and fertilizers to fine chemicals for the pharmaceutical industry [
59,
60]. The method is particularly advantageous in terms of sustainability because it relies upon the use of organic wastes as the substrate for the intensive production of insect biomass [
59,
60]. In this way, insects become active agents in the waste management process, turning large amounts of organic wastes destined to disposal into valuable products [
59,
60]. For this reason, bio-conversion is receiving increasing attention from both researchers and entrepreneurs. At present, however, only a few studies are available, mostly focusing on a handful of species belonging to Diptera and Coleoptera orders [
59,
60]. It follows that great opportunities exist to investigate the subject and identify the best combinations of insects and organic wastes that allow maximizing both bio-conversion and insect farming processes.
4. Feed from Insects: Three Specific Case Studies
With their outstanding nutritional value, insects can contribute to a viable and environmentally friendly solution to the challenges involving the sustainable production of food and feed. While cultural aspects are detrimental for the rapid acceptance of entomophagy in more industrialized countries, the utilization of insects for the preparation of ingredients for animal feed is an option attracting considerable interest and already under testing.
Feed production involves a global market of primary importance for numerous countries around the world. The total feed produced worldwide in 2011 has been estimated to amount approximately to 870 million tons [
28]. Correspondingly, the total revenues generated from commercial feed manufacturing have been evaluated to about 350 billion US
$ [
28]. Although these numbers can seem huge, feed production has to increase by 70%, at least, to meet the expected global feed demand in 2050. Such rise in feed demand also stems from the need of increasing, by 60%, meat production from livestock because of the expected increase of the world’s population [
57].
At present, animal and fish feed typically include fishmeal, meat meal, fish oil, soybeans and several other grains as major ingredients. This brings with it significant limitations to future scenarios. Indeed, fishmeal and meat meals represent up to 70% of production costs, which is going to make the costs for feed production substantially prohibitive [
28].
Concerning fishmeal, 2010 and 2011 have seen a marked increase in the worldwide demand. The first effect has been an immediate rise of the prices, which have remained high until today. While this means less accessible fishmeal to end users, it also represents an opportunity for aquaculture [
28]. Being a vital industry for the production of animal feed, it can be reasonably expected to benefit from the increasing demand for fish, although obstacles exist.
Nowadays, fishmeal production involves approximately the 10% of global fish production [
53]. Fishmeal production is leaded by South America, which significantly relies upon the catch of anchoveta. For this activity, predictions are pessimistic [
53]. In 1994, up to 12.5 million tons of anchoveta were caught, but the catch yield reduced to 4.2 million tons in 2010. Further drops have been recorded recently [
53]. Additionally, the sector suffers from the tighter fishing quotas for the different countries, often a useful measure to maintain sustainable fish stocks and diversity, the control enhancement on irregular fishing, and the use of more convenient alternatives to fishmeal [
53]. As a consequence, the use of fishmeal for feed production is going through hard times.
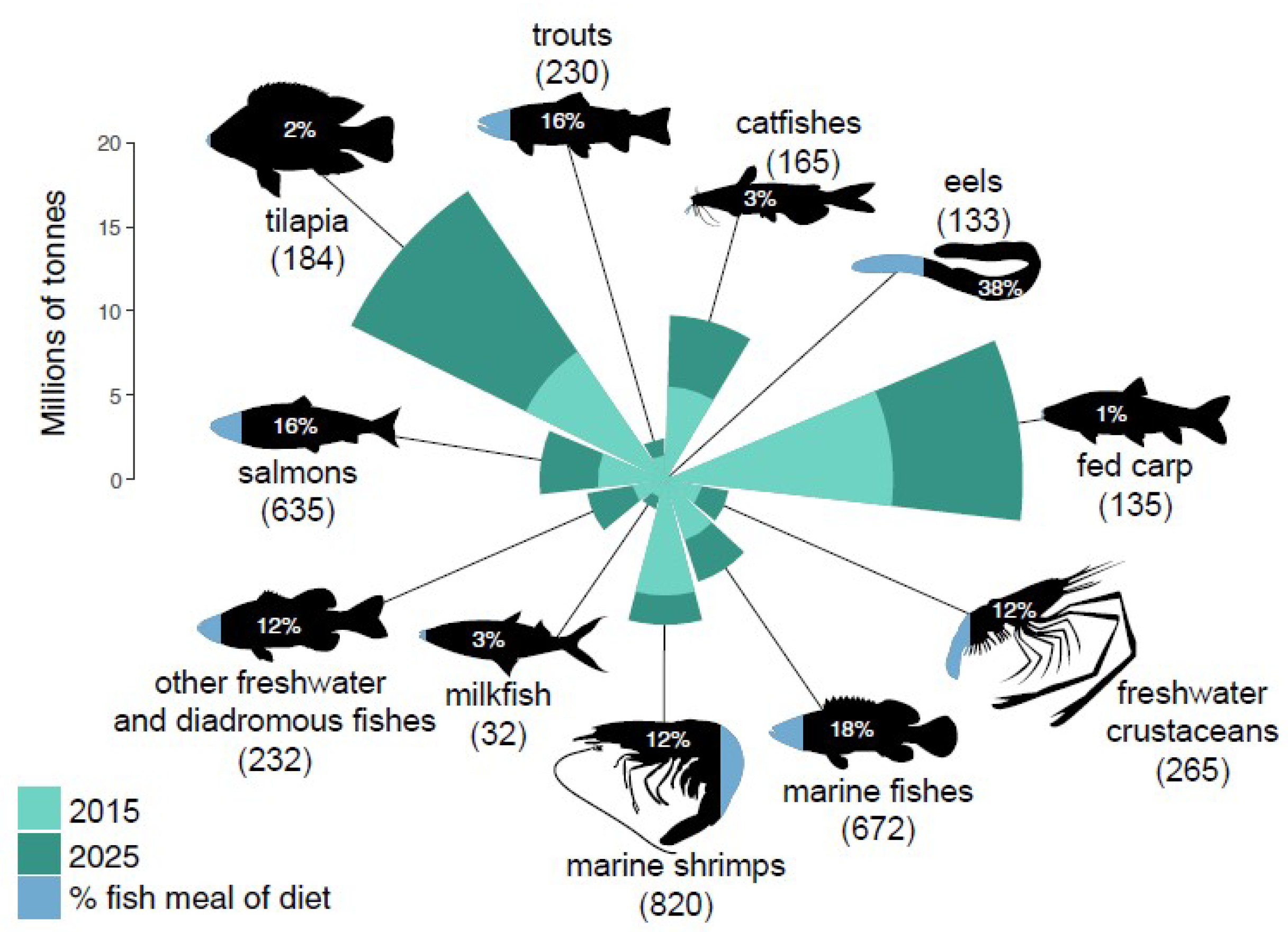
This represents a serious concern even for aquaculture itself. Fishmeal represents, indeed, an important ingredient to feed higher value species, as schematically shown in
Figure 1; particularly in the light of the predictions regarding the total production of feeds for aquaculture species, which is predicted to increase by 75% from 49.7 million tons in 2015 to 87.1 million tons in 2025 [
61].
In contrast, the same scenario is favorable to insects. They are already utilized in aquaculture as well as in the production of animal feed. Therefore, the further development of insect proteins for aquaculture and livestock is highly desirable and, overall, insects are currently regarded as a very attractive option for novel feed production. It is also worthwhile to say that insects are a well-documented part of fish diets in nature for both carnivorous and omnivorous species of continental fish [
62,
63] as well as marine fish and fish that tolerate a wide range of salinity that can be found in brackish water [
64]. In this regard, nutritional aspects related to insect meals in fish nutrition have been recently reviewed [
65].
Among the others, three different insect species emerge for their ease of breeding, their global diffusion and, additionally, their remarkable efficiency in converting organic wastes. Harmless for human beings and for the environment, the black soldier fly (Hermetia illucens), the common housefly (Musca domestica) and the yellow mealworm (Tenebrio molitor) show significant promise for future production scenarios embedded in the circular economy paradigm. These insects not only are seemingly suitable to mass production, but they also provide a meal that allows partial or total replacement of fishmeal.
In the following, we briefly describe the three insect species and their use in the production of feed for fish. For the sake of clarity, black soldier fly, common housefly, and yellow mealworm are analyzed separately.
4.1. Black Soldier Fly
Hermetia illucens (HI), best known as the black soldier fly, is a native species of the American continent that can be found widely distributed worldwide in temperate and tropical areas [
66]. Not being a vector of diseases for humans, animals or plants, its capacity of transforming low-value residual organic streams into high-value protein products made HI, at present, the most used insect species to produce feed for animals [
67,
68]. Until 2018, around 80% of EU insect-producing companies based their business on HI [
69].
HI lifecycle includes eggs, larvae, pre-pupa, pupae and adult stages, taking around 40–45 days to complete [
66]. Representative pictures are shown in
Figure 2a,b. Larvae are white, while pre-pupae and pupae are black, which allows easy identification [
70,
71].
HI pre-pupae show appealing nutritional values. The protein content of dry matter can be as large as 55%, while about 35% of dry matter is fat. As shown in
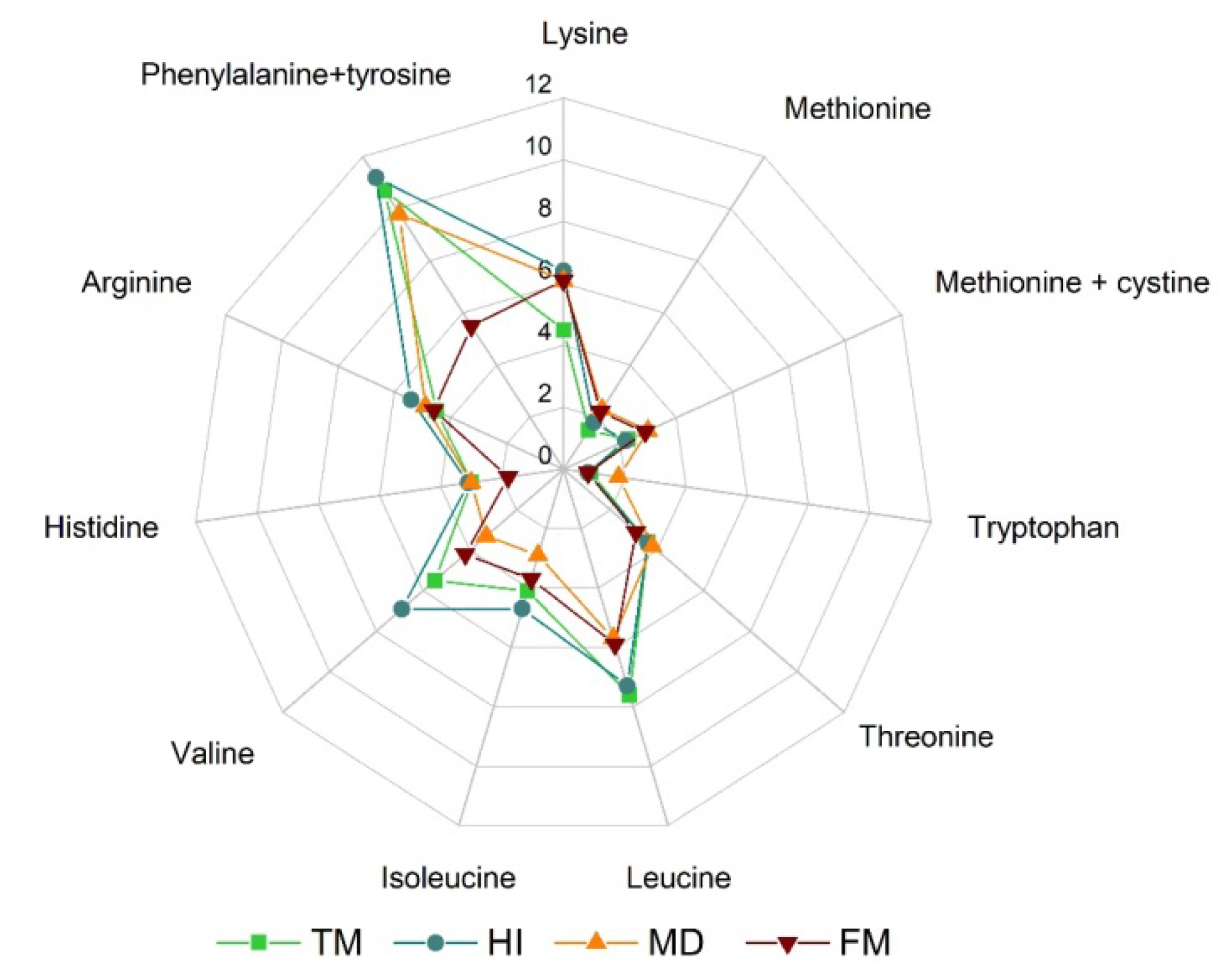
Figure 3, HI exhibits a well-balanced profile of essential amino acids, comparable with those of
Musca domestica,
Tenebrio molitor and, in several cases, definitely better than that of fishmeal. However, lipids can be reduced by up to 5% by suitable defatting processes [
77]. Nutritional values strongly depend on the substrate used in the rearing process [
78]. Large variations in body composition also occur throughout the course of larval development. For example, crude protein content decreases with increasing age. The highest percentage was reported for larvae 5 days old (61%), while it was less in larvae 15 (44%) and 20 (42%) days old [
79]. HI larvae contain high concentrations of minerals such as manganese (Mn), iron (Fe), zinc (Zn), copper (Cu), phosphorus (P) and calcium (Ca) [
80,
81].
Several studies have pointed out that HI pre-pupae can partially or completely substitute fishmeal in the diet of different fish species [
79,
80]. However, only a relatively low inclusion is suggested to obtain results similar to those of traditional feedstuff [
79,
80]. Best substitution percentages change with larvae meal quality, fish species and fish diet formulation [
79,
80].
Although additional investigations are needed to understand how different factors can affect HI meal performances, literature already provides hints in this respect. For instance, a pre-pupae HI meal with no fat removal has been used to partially substitute fishmeal and fish oil in the diet of rainbow trout [
81]. Specifically, 15% and 30% of HI meal were used to substitute, respectively, the 25% and 50% of fishmeal and the 36% and 72% of fish oil. No significant differences on growth performances were reported for HI meal at the lowest replacement percentages. Above such level, chitin contained in pre-pupae may reduce digestibility, i.e., nutrient availability for fish. Moreover, the large lipid content, in particular, saturated fats, can play an important role. Partially defatted HI meal resulted, indeed, in improved fish growth even when used to substitute the 40% of fishmeal [
83].
The importance of insect meal production protocols becomes evident from the comparison of the effects on Atlantic salmon of two HI meals obtained through different nutrient isolation and processing methods [
84]. Experimental evidence showed that HI meal can be used to replace up to 100% of the traditional food if the proper processing method is adopted. No differences emerged from the histological examination of salmon. Additionally, trained panelists analyzed sensory attributes of fillets of fish from diets containing fishmeal (control) or 25% of HI meal (100% of fishmeal substitution) after 105 days of feeding. Odor, taste, flavor and texture analyses of the fish fed with HI meal were performed, revealing no significant difference with respect to the control case study.
While the nutritional value of HI larvae meal has been extensively studied, its impact on fish health, and gut health in particular, has been less investigated so far. However, according to a recent study, total replacement of fishmeal with HI larvae meal does not compromise the gut health of Atlantic salmon. Rather, dietary insect meal inclusion seems to reduce excessive lipid deposition within enterocytes (steatosis) in the proximal intestine [
85].
4.2. Common Housefly
The common housefly is the most widespread fly species in the world. Its binomial name is
Musca domestica (MD). MD larvae can grow on a variety of substrates, showing the ability of transforming organic wastes into a valuable biomass, rich in protein and fat useful as feed. For this reason, the production of MD larvae under controlled conditions to feed farm animals has been investigated since the late 1960s [
86], and it is nowadays particularly implemented in China [
71].
MD shares with HI the very short lifecycle and growth stages. The various stages of the lifecycle are shown in
Figure 2c. Eggs hatch after 8 to 12 h, while the larval and pupal stages last about 5 days. The 10-day cycle can be reduced to 6 days under controlled conditions [
87].
MD exhibits a nutritional profile quite similar to the HI one, thus showing large protein and lipid contents. Changes in composition have been reported for the different growth stages, also depending on the rearing substrate. For MD larvae, proteins and lipids range between 40% and 60% and between 9% and 26% of dry matter, respectively. Interestingly, the protein amount can increase up to about 70% in MD pupae. The amino acid profile is shown in
Figure 3. It compares remarkably well with those of HI and TM. Moreover, it is also quite close to the one of fishmeal. P, K, Na, Mg and Fe contents are similar to HI ones, while Ca and Mg levels are lower by 15 and 3 times, respectively [
87].
The use of MD larvae as feed in fish farms has been studied extensively since the late 2000s, with particular focus on pond species. The results obtained display high variability depending on several factors including the fish species and the feeding substrate for insects.
Overall, literature provides different case studies where MD meal emerged as a promising alternative to fishmeal. For instance, it has been found that a 50% replacement of fishmeal with MD larvae meal improves the growth of Nile tilapia [
88]. A recent, more comprehensive study on the same fish species reports on a 10-week feeding trial to assess the effect of the progressive inclusion of MD larvae meal [
89]. Diets with 25%, 50%, 75% and 100% of fishmeal substitution were used and several qualitative parameters were evaluated and compared with those of fish fed with a control diet. No significant difference in feed intake and apparent digestibility coefficient between the treatments was observed. Furthermore, the replacement of up to 75% of fishmeal does not affect the growth performance and ingredient utilization. Rather, the 25% fishmeal substitution positively influenced the flesh quality of Nile tilapia. Nevertheless, the complete replacement of fishmeal caused a significantly lower survival rate, weight gain, specific growth rate and higher feed conversion rate [
89].
4.3. Mealworm Beetle
Mealworm beetle is an indigenous species to Europe, but distributed nowadays all around the planet. Its binomial name is
Tenebrio molitor (TM). Compared with HI and MD, it has a lifecycle definitely much longer, with a variable length ranging from 280 to 630 days [
87].
The two fundamental stages of the TM lifecycle are shown in
Figure 2d,e. At room temperature, larvae typically hatch after 10 to 12 days and become mature after 8 to 20 growth stages that take around 3 to 4 months overall [
87]. Mature larvae can live up to 18 months and are characterized by a light yellow-brown color. Typically, they are 20 to 32 mm long and their weight ranges between 130 and 160 mg. The pupal stage normally lasts 7 to 9 days at room temperature, but it can take up to 20 days if temperature is lowered [
87]. The adult TM lives for 2 to 3 months [
87].
TM and its larvae, which are commonly known as mealworms, are pests of grain, flour and food stores. They are omnivorous and have the ability to recycle different kinds of organic food by-products into high-quality feed rich in calories, protein and fat in relatively short times [
90]. Mealworms contain large amounts of proteins (47%-60%), with essential amino acids in appealing proportion as shown in
Figure 3. They also have a relatively large fat content (31%-43%). The fatty acid composition is closer to that of MD larvae meal, except for lauric and linoleic acids, which are, respectively, much less and much more in TM. Mealworms also exhibit large amounts of minerals such as P, K, Na, Mg, Fe and Cu. However, they contain much less Ca and Mn than HI [
87].
The commercial production of mealworms started in the United States of America in the 1950s. The market was initially for fishing bait, but mealworms were soon used as feed for exotic pets and zoo animals [
80]. More recently, mealworms also showed their high potential for the partial replacement of fishmeal in the diets of different fishes [
85]. Positive feedbacks have been reported also for the use of relatively large TM meal contents. For instance, mealworms being highly palatable, they can replace up to 40% of the fishmeal component in practical diets for African catfish without any significant reduction in growth performance and feed efficiency ratio. Moreover, good growth performances were observed for diets with up to 80% replacement of fishmeal [
86]. TM meal was successfully used also in rainbow trout [
87], where it can replace up to 50% of dietary levels [
88]. Interesting results have been obtained also for marine carnivorous species such as seabream [
88] and European sea bass [
89], where fish growth was not significantly influenced at fishmeal replacements of 25%. Small European sea bass can be fed for 6 weeks with 25% of TM larval meal with a significant anti-inflammatory response and improved anti-parasite activity [
90]. It is also worth noting that the inclusion of a full-fat TM larvae meal does not result in negative effects on most quality traits of the fish flesh of rainbow trout, except for the fatty acid profile [
82].
5. Toward Large-Scale Biorefineries for Efficient Insect Rearing
Insect farming for commercial purposes has deep roots in tropical areas of Asia, Africa, and Latin America, where insects represent an important component of local diet. The practice has progressively expanded to Western countries following, first, the diffusion of exotic pets and, then, the increasing demand for novel and more sustainable feeds and foods [
82,
91,
92,
93,
94,
95,
96,
97,
98].
5.1. The Market
The European market has undergone a rapid growth during the last years, especially after the EU commission allowed the use of insects in feed for pets and aquaculture animals starting from July 2017 [
69]. Several start-ups, small/medium enterprises and a few large-scale companies have been founded across Europe with the aim of improving insect rearing methods, thus attracting the interest of important investors. The International Platform of Insects for Food and Feed (IPIFF), an EU non-profit organization, promotes the use of insects and insect-derived products as a source of nutrients for human consumption and animal feed, and was soon established. IPIFF has recently declared that its member companies raised 600 million €, managing to produce more than 6000 tons of insects in 2019. Such production could increase up to 5 million tons by 2030 [
99].
5.2. Insect Production Methods
Although insect rearing looks set to a brilliant future, little information is available concerning insect production methods and technologies, especially on the large scale [
59,
100,
101]. One of the reasons lies in the understandable reluctance of private companies to share details involving the heart of their business. A few patents are available, but they do not disclose sensitive data and performances [
102,
103,
104,
105,
106,
107]. In contrast, much more is known about traditional rearing methods, which represent the background for any technological improvement. For instance, a comprehensive guide to medium-scale HI production in an Indonesian insect farm has been published [
108].
In general, insect farming involves at least two main separate units, namely, one for the maintenance of the breeding colonies in captivity and the other for growing larvae from eggs [
69]. If business focuses on mature insects, an additional rearing area is needed. This is the case of cricket farms [
71]. Advanced systems often include an area to process insects and refine derived products. Production wastes, such as substrate leftovers and frass, can be used to produce fertilizers in a dedicated facility, thus contributing to circularity and sustainability.
In fact, recycling mineral sources in the food system rather than using exogenous sources helps in reducing the impact that the excess of some substances, e.g., N and P, can have on the environment [
109]. In this regard, it is worth noting that the frass produced by insects fed with by-products is rich in minerals, such as N, P and K, that are already used in agriculture. Thus, it shows the potential of partially or completely replacing mineral fertilizers [
110]. Therefore, a proper valorization of the by-products of insect rearing can provide an additional economic benefit to insect farming activity while creating new opportunities in the context of sustainable food chains.
In this regard, it is also worth noting that another economically valuable by-product of insect farming is chitin. At present, the market demand for this substance is even more important than the whole supply chain, which mainly comes from the seafood waste. Moreover, chitin from insects contains a lower amount of calcium carbonate (<6%) compared to crustaceans (30–50%), and, as a consequence, it can be more easily extracted, resulting in a more environmentally friendly process [
111].
Insects show the considerable advantage to thrive in densely populated areas, which allows intensive production even in confined spaces. Usually, larvae and pupae are placed together with a feeding substrate in small trays that can be made of different materials such as wood, high-density polyethylene or fiberglass. Trays used for fattening TM larvae is a standard 65×50×15 cm
3 box, easy to handle and deep enough to prevent larvae or adults from escaping [
71]. A recent study based on an EU pilot mill designed to produce 17 tons of fresh TM larvae per year reports that mealworms can be reared with a density of 5 larvae cm
-2 [
101].
Rearing boxes can be held in multilevel shelves to minimize the space used per insect mass produced. In some cases, stackable boxes are used. In others, the boxes are set on wagons or pallets to allow free movement within the rearing area [
71]. A similar set-up can be appreciated in
Figure 4, where a rearing unit used for TM mealworms is showed [
71]. Multilevel shelves can occupy the whole rearing area surface, potentially enabling the production of thousands tons of larvae per year.
In this regard, the project DESIRABLE (DESIgning the Insect bioRefinery to contribute to a more sustainABLE agro-food industry), funded by the French National Research Agency (ANR), has provided a few interesting case studies on the practical organization of a medium-scale (400 tons per year), large-scale (2000 tons per year), and very large-scale (10000 tons per year) biorefinery of TM larvae [
69]. About 13,000 boxes can be placed in a 400 m
2 building, leading to a production of about 250 kg of larvae m
-2 per year. At the end of the fattening stage, larvae are harvested and processed to obtain the final insect meal.
So-called farrow-to-wean areas where adults breed and females deposit eggs, while larvae are continuously moved to the fattening area after they hatch, are needed. Part of the newly-born larvae is left in the farrow-to-wean-area to be reared up to the adult stage with the aim of restoring the breeding population. In the case of TM, one-tenth of the annual larvae production suffices for this purpose [
69].
In the breeding area, adults are kept in cages that are provided with feeding substrate and water. Except for insect species that can fly or jump vertically, boxes for larvae and adult breeding are quite similar. Dividers or other structural elements can be added to ensure more efficient space utilization, as for the case of crickets [
71]. Precautions such as higher banks and smooth finish can be adopted for species that can crawl vertically. Closed cages are required for flying insects such as HI or MD [
69]. In any case, oviposition sites should be reduced to specific locations inside the breeding cages to allow easier egg collection. For instance, small boxes sealed with moisture can be used where females can preferentially spawn. Decomposing grain saturated with water, peat moss, coconut husks and other organic matter can be also used depending on the insect species [
71].
5.3. Rearing Conditions
The efficiency of insect farms in terms of feed conversion strictly depends on rearing conditions. Insects do not have a metabolic regulation of their own body temperature. Therefore, suitable temperature and humidity must be ensured to optimize the insect growth depending on the species and growth stage. Typically, temperatures range between 20 and 35 °C and humidity levels from 55% to 75% [
69,
71]. Air must be circulated to avoid undesired air stratification, proliferation of fungi, bacteria or viruses, and the accumulation of CO
2 and other dangerous gases that can have detrimental effects on the health of the colony and workers.
Climate control is particularly important for at least two reasons. First, even temperature differences of 2 or 3 °C can lead to significant time shifts in the insect development, thus resulting in uncontrolled farming outcomes [
71]. Second, the climate control system contributes to the energy demand, especially for insect farms located in temperate countries with cold winters and warm summers, where heating during winter and cooling during summer are required. In this regard, an innovative system was recently developed to separately monitor a small cluster of cages, thus enabling more accurate and efficient climate control [
107].
5.4. Insect Processing
The full production from adults to insect meal involves several steps that, in turn, require employees. Concerning the farrow-to-wean area, efforts are mainly related to feeding adults, removing the dead and frass, monitoring oviposition, and releasing new adults into the colony. Eggs must be periodically collected and moved to the rearing area after they hatch. Here, the new larvae are located in trays with the proper amount of substrate and water. Concerning the fattening area, larvae must be periodically moved to clean trays with new substrate and water. Finally, the larvae that have reached the desired size must be removed and processed. During this stage, larvae that have undergone a slower growth can be separated and moved to the subsequent batch [
71].
Insect growth facilities must also include an area intended for feed processing and storage. Similar to other feed manufacturing activities, e.g., fishmeal production, reared insects, larvae or pupae must be processed to avoid their spoilage and properly stored until the final use. Lack of hygiene during processing can be detrimental to the quality of the insect meal and potentially dangerous for the health of fed animals. Biological hazards can arise by the uncontrolled proliferation of bacteria, parasites, viruses, or fungi that may come from the insect itself, from the rearing substrate or from external contamination during the manufacturing process [
98]. In this regard, both traditional and innovative technologies available for insect processing have been recently reviewed [
112].
A common pretreatment used in the commercialization of insects and related products is blanching. It consists in a short boiling step followed by a rapid cooling in flowing cold water to reduce microbial counts and to inactivate the enzymes responsible for spoilage and food poisoning. However, blanching is ineffective on mesophilic bacterial spores and a further drying process to reduce the water content is needed.
Several technologies such as sun drying, freeze drying, microwave drying, smoke drying and oven drying can be applied to insect manufacturing [
112]. Other methods are often used to ensure a better ratio of macronutrients in the final insect feed, e.g., to reduce lipid or chitin contents, or to obtain derived products such as oil and protein powders and pellets. This allows more effective risk management [
98].
It is worth noting that food processing may significantly affect the feed quality and must be opportunely selected. For instance, high temperatures may cause protein denaturation and lipid oxidation. Thus, suitable conditions must be chosen for specific insect species to improve antimicrobial activity while reducing quality loss. Processing can also reduce acceptability of insect meal due to changes in flavor, aroma, texture, color, and other properties [
69,
71]. This can negatively affect fish growth and, thus, fish farm productivity [
91].
5.5. Challenges
Despite the remarkable interest attracted in recent years and the impressive work of research and analysis carried out by a few scientists [
28], insect rearing for food and feed production is still in its infancy. It represents one of those rare fields of study in which origins and evolution make fundamental and applied research extremely close to engineering and industrial application.
The driving force underlying the rapid scientific and technological development and the interest increasingly attracted from entrepreneurs and industrialists can be readily identified in the growth of the world’s population. Demographic studies predict a dramatic scenario, with more than 9 billion people living on Earth by 2050. This number has been shown to require the doubling of the total food and feed presently produced worldwide. In turn, this involves suitably coping with the scarcity of land that can be additionally used for cultivation and farming, the impoverishment of seas in terms of fishing resources and the consequences of climate changes that already affect adversely the food and feed production in several developed and developing countries.
All of this represents challenges to be overcome. Humans have to re-evaluate and modify the current food and feed production systems. It is necessary to go beyond the enhancement of production efficiencies and the reduction of wastes. Specifically, it seems that modern societies have to identify new food and feed sources.
Insects can be part of a rational and profitable solution to meet the food and feed demands in the next future. However, the use of insects as food and feed sources brings with it several problems on different levels. Some of them are clearly discernible today, but it is likely that other issues will emerge as the field of study evolves further and insect farming becomes a more common practice.
The first main challenge that deserves mention is related to the acceptance of insects and insect-based products in modern societies. Although edible insects have been part of the human diet in various regions around the globe since ancient times, people living in modern societies, and particularly in Western countries, feel uncomfortable with the idea of consuming food with ingredients deriving from insects. The feeling of disgust is relieved, but still present, for feed containing ingredients obtained by insect biomass. Therefore, actions have to be undertaken to raise awareness of the opportunities offered by insects for food and feed production. Important initial steps have been taken to address this societal challenge, but much remains to be done. The public at large must be involved as well as political decision-makers, researchers and entrepreneurs, international agencies and food and feed industries.
The second main challenge concerns the accurate evaluation of the factors involved in insect rearing. The transition from the traditional wild catch of edible insects from their habitats to massive farming requires careful study. The subject is extremely broad and only a truly multidisciplinary approach can assure significant progress in a relatively short time. Specific expertise is needed to advance knowledge regarding the effects of rearing conditions and insect diet on the insect’s health, its growth rate in the different growth stages, its nutritional profile, and its capability of converting agricultural wastes and by-products into useful biomass.
Similar problems arise in connection with the optimal use of insect-derived nutrients and micronutrients such as amino acids, fats, chitin and metallic elements. The processing of wet and dry insect biomass to efficiently and effectively extract the desired nutrients and micronutrients has still to be properly developed. At present, there exists significant opportunities for innovation, which can beneficially impact the sector. Considerable room for improvement also exists for the by-products of the insect farming activity.
The third main challenge involves the intensive insect rearing itself. A fair balance between mechanization, automation, and manual labor must be found. Most of the insect farming methods utilized are labor-intensive. Only a few stages are automated, which makes a huge staff necessary when rearing scales up. Therefore, products available on the market are still overpriced compared with other feed sources such as soy meal.
Several stages are yet to be optimized. For instance, insect selection connected with the growth stages, reproduction cycle, breeding conditions and feeding. For these reasons, academic research and research institutes are collaborating with EU companies to foster innovation [
69]. In a few cases, the interaction between researchers and entrepreneurs has led to significant improvements and technology patenting. This is the case of a Dutch company that has recently developed a centralized system for dispersing olfactory triggers aimed at controlling oviposition in HI adult colonies [
104]. An automated device to transport newborn larvae from the adult cage to a system allowing their counting, dozing and analysis has also been designed [
105]. The same company has also patented a breeding cage equipped with pipes for receiving and discharging cleaning liquids for making any cleaning process more effective [
106].
6. Conclusions
Global food and feed demands are growing rapidly. Agriculture and livestock expansions definitely show detrimental long-term implications for the environment. Within this framework, insect farming appears as an increasingly attractive candidate for a strategic response to the need of sustainable food and feed production. Insects represent an abundant food source that can be reared using organic side-streams, thus enhancing circularity and sustainability, while alleviating food and feed insecurity.
Insects exhibit high nutritional values, give rise to low emissions of greenhouse gases, require much less land than traditional agricultural and farming practices and display a very high efficiency food-to-feed conversion. Therefore, insects can replace, at least in part, protein ingredients utilized in preparing compound feeds for common livestock and aquaculture.
At present, commercial insect rearing is growing an industrial sector across North America and Europe. Nevertheless, the full potential of insects in terms of food and feed security can be actually realized, only if a formidable amount of work will be done by a range of stakeholders in the next future. Contributions from academia and research institutes are fundamental to provide new insights on biological, chemical and technological aspects concerning the production and the use of insects as food and feed. Nevertheless, the direct and constant dialogue with the final users, such as consumers and farmers, must be improved to overcome both practical obstacles and skepticisms. Moreover, public institutions and policy makers should ensure a proper support to the industrial sector involved in insect farming, thus recognizing that it is an essential sector for developing more sustainable food chains.
The feedstock industry is going to require enormous amounts of insect biomass to replace the ingredients that are typically used at present such as fishmeal, fish oil and soy. Several major and minor factors have to be considered making insect rearing efficient and effective. Not only insect lifecycles must be well known and controlled, but actions related to automation and technological transfer must be properly implemented.
Improving automation, and technology in general, can allow insect rearing to enable a virtuous circular economy. It shows a remarkable capacity to convert organic matters into proteins and, then, to save natural resources from consumption. The whole agri-food system would improve its sustainability levels if insects are used to generate innovative business models. Indeed, sustainability might be enhanced both upstream, i.e., saving and recycling organic wastes, and downstream, e.g., by reducing the use of fishmeal in aquaculture.
A critical point concerns the economic features related to sustainability. For insect rearing to be competitive and robust, this new industry has to reach large-scale, cost-effective and reliable production. However, insect farming is far from economic convenience today, especially under small-scale rearing conditions. Main costs are related to raw materials used for feeding insects, which have a relatively high price [
113]. At the same time, the introduction of insect meals in animal diet, e.g., in fish farming, is often not advantageous in economic terms for farmers due to the fact that the price for insect meal is higher than for traditional meals [
114].
The scientific and economic literature on this issue is quite scarce and the corresponding lack of information does not allow generalizing the considerations mentioned above. In spite of this, costs of both insect rearing and insect meals can be reasonably expected to decrease in a virtuous circular economy scenario, particularly in the case of large-scale, technologically innovated insect farming. The inherent capacity of exploiting organic matter and the efficiency of the rearing process can, indeed, assure significant advantage.
Issues related to food and feed safety must be suitably tackled, and processing stages for obtaining proteins for food industry or protein meals for animal or fish feedstock have to be designed.
Overall, a formidable challenge has emerged, that can be taken on only through an intensive multidisciplinary approach based on a strong interaction between research institutions, enterprises and industry, and policy makers.