Efficacy of N-Methyl-N-Nitrosourea Mutation on Physicochemical Properties, Phytochemicals, and Momilactones A and B in Rice
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Plant Materials
2.2. Measurement of Physicochemical Properties
2.3. Standards and Reagents
2.4. Extraction and Samples Preparation
2.5. Antioxidant Assays
2.5.1. DPPH Scavenging Assay
2.5.2. ABTS Cation Discoloration Assay
2.6. Determination of Total Phenolic Contents (TPC)
2.7. Determination of Total Flavonoid Contents (TFC)
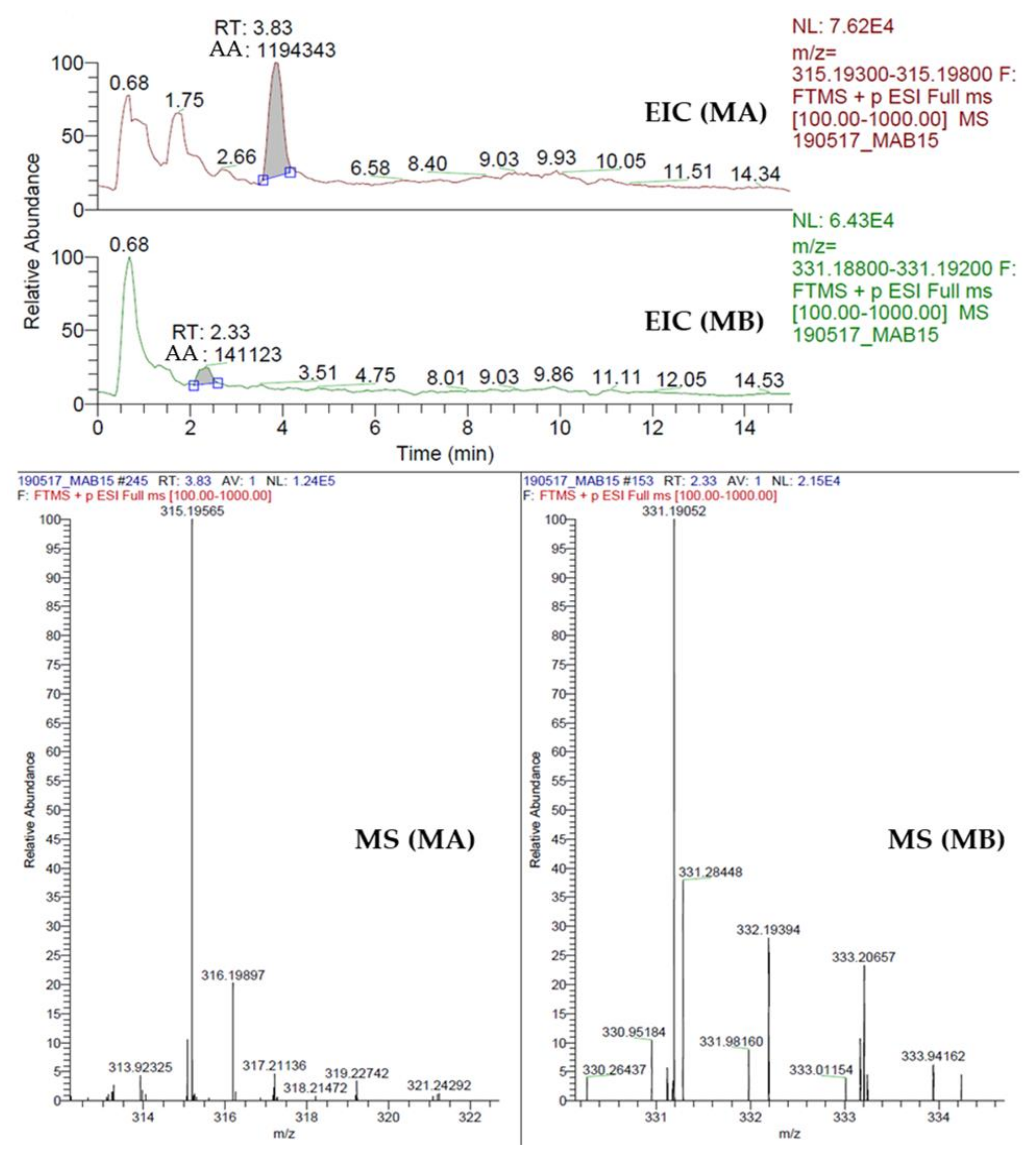
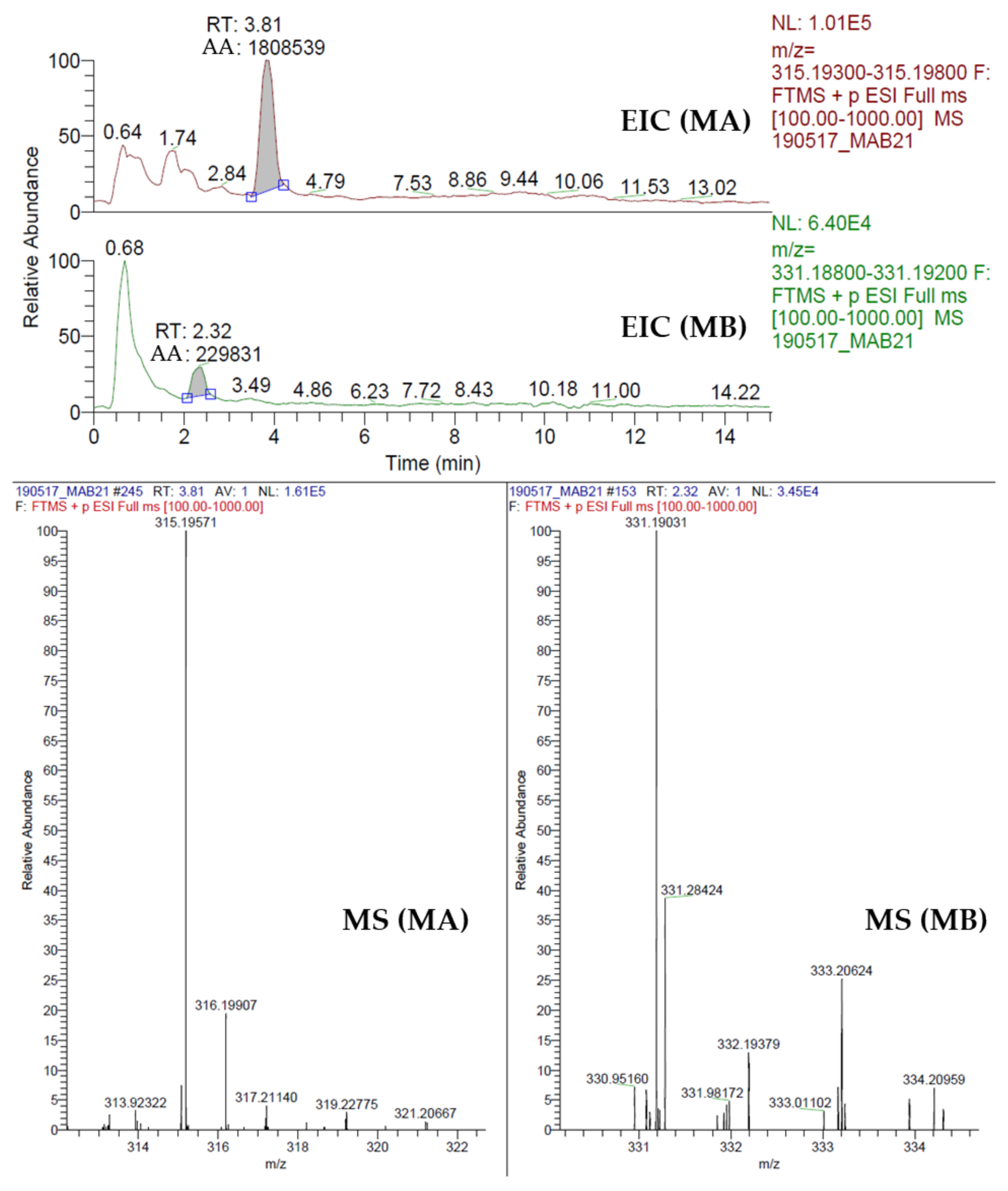
2.8. Quantification of Momilactone A (MA) and B (MB)
2.9. Statistical Analysis
3. Results
3.1. Antioxidant Activities
3.2. Total Phenolic and Flavonoid Contents
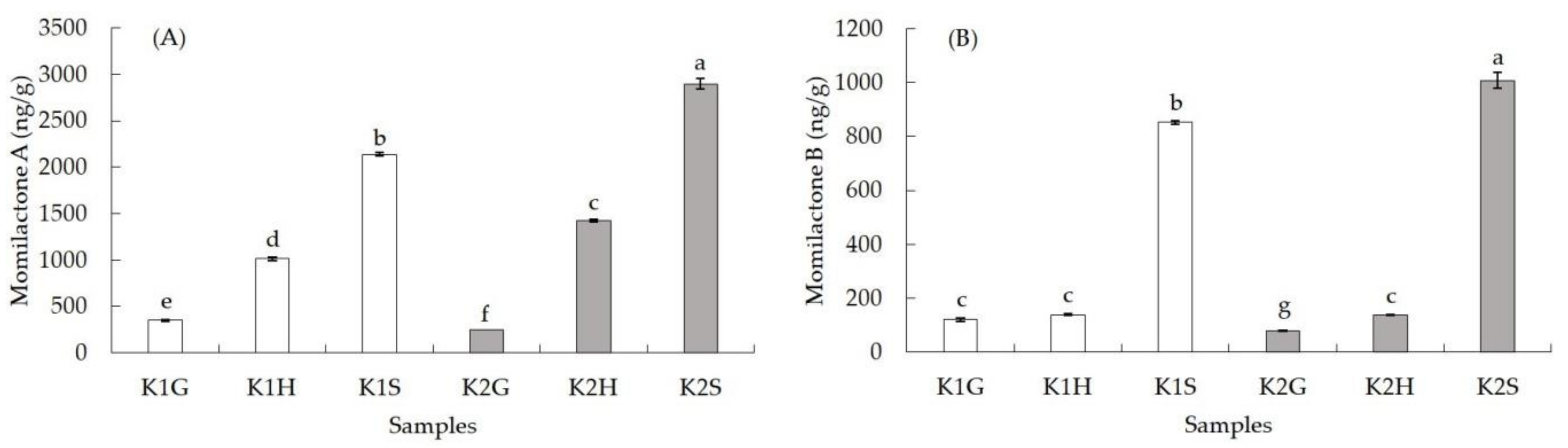
3.3. Momilactones A and B
3.4. Correlation of Biological Contents of Rice Grain
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rohman, A.; Helmiyati, S.; Hapsari, M.; Setyaningrum, D.L. Rice in health and nutrition. Int. Food Res. J. 2014, 21, 13. [Google Scholar]
- Kakar, K.; Xuan, T.D.; Haqani, M.I.; Rayee, R.; Wafa, I.K.; Abdiani, S.; Tran, H.D. Current situation and sustainable development for rice cultivation and production in Afghanistan. Agriculture 2019, 9, 49. [Google Scholar] [CrossRef]
- Ryan, E.P. Bioactive food components and health properties of rice bran. J. Am. Vet. Med. Assoc. 2011, 238, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, P.R.; Tamrakar, N.; Singh, L.; Tandon, A.; Sharma, D. Rice nutritional and medicinal properties: A. J. Pharmacogn. Phytochem. 2018, 7, 150–156. [Google Scholar]
- Bhat, F.M.; Riar, C.S. Health benefits of traditional rice varieties of temperate regions. Med. Aromat. Plants 2015, 4, 198. [Google Scholar]
- Asaduzzaman, M.; Haque, M.E.; Rahman, J.; Hasan, S.K.; Ali, M.A.; Akter, M.S.; Ahmed, M. Comparisons of physiochemical, total phenol, flavanoid content and functional properties in six cultivars of aromatic rice in Bangladesh. Afr. J. Food Sci. 2013, 7, 198–203. [Google Scholar] [CrossRef]
- Kakar, K.; Nitta, Y.; Asagi, N.; Komatsuzaki, M.; Shiotau, F.; Kokubo, T.; Xuan, T.D. Morphological analysis on comparison of organic and chemical fertilizers on grain quality of rice at different planting densities. Plant Prod. Sci. 2019, 22, 1–9. [Google Scholar] [CrossRef]
- Noori, Z.; Mujadidi, M.W.; Amin, M.W. Physicochemical properties and morphological observations of selected local rice varieties in northern Afghanistan. Int. J. Agric. Environ. Food Sci. 2018, 2, 99–103. [Google Scholar] [CrossRef][Green Version]
- Kakar, K.; Xuan, T.D.; Abdiani, S.; Wafa, I.K.; Noori, Z.; Attai, S.; Khanh, T.D.; Tran, H.D. Morphological observation and correlation of growth and yield characteristics with grain quality and antioxidant activities in exotic rice varieties of Afghanistan. Agriculture 2019, 9, 167. [Google Scholar] [CrossRef]
- Abacar, J.D.; Lin, Z.M.; Cheng, Z.X.; Qiang, D.C.; She, T.; Hui, L.Z.; Hua, W.S.; Feng, D.Y. Variation in yield and physicochemical quality traits among mutants of Japonica rice cultivar Wuyujing 3. Rice Sci. 2016, 23, 33–41. [Google Scholar] [CrossRef]
- Quan, N.V.; Xuan, T.D.; Tran, H.D.; Thuy, N.T.D.; Trang, L.T.; Huong, C.T.; Yusuf, A.; Tuyen, P.T. Antioxidant, α-amylase and α-glucosidase inhibitory activities and potential constituents of Canarium tramdenum Bark. Molecules 2019, 24, 605. [Google Scholar] [CrossRef] [PubMed]
- Tuyen, P.T.; Xuan, T.D.; Khang, D.T.; Ahmad, A.; Quan, N.T.; Anh, L.H.; Minh, T.N. Phenolic compositions and antioxidant properties in bark, flower, inner skin, kernel and leaf extracts of Castanea crenata Sieb. et Zucc. Antioxidants 2017, 6, 31. [Google Scholar] [CrossRef] [PubMed]
- Tuyen, P.T.; Khang, D.T.; Ha, P.T.T.; Hai, T.H.; Elzaawely, A.A.; Xuan, T.D. Antioxidant capacity and phenolic contents of three Quercus species. Int. Lett. Nat. Sci. 2016, 54, 85–99. [Google Scholar] [CrossRef]
- Liu, J.; Wang, C.; Wang, Z.; Zhang, C.; Lu, S.; Liu, J. The antioxidant and free-radical scavenging activities of extract and fractions from corn silk (Zea mays L.) and related flavone glycosides. Food Chem. 2011, 126, 261–269. [Google Scholar] [CrossRef]
- Razali, I.; Nor Asimiah, A.S.; Norhaya, H. Determination of antioxidants in palm oil products by high performance liquid chromatography. J. Oil Palm Res. 1997, 9, 25–33. [Google Scholar]
- Soong, Y.Y.; Barlow, P.J. Antioxidant activity and phenolic content of selected fruit seeds. Food Chem. 2004, 88, 411–417. [Google Scholar] [CrossRef]
- Chandra, S.; Khan, S.; Avula, B.; Lata, H.; Yang, M.H.; ElSohly, M.A.; Khan, I.A. Assessment of total phenolic and flavonoid content, antioxidant properties, and yield of aeroponically and conventionally grown leafy vegetables and fruit crops: A comparative study. Evid. Based Complement. Altern. Med. 2014, 2014, 1–9. [Google Scholar] [CrossRef]
- Walter, M.; Marchesan, E. Phenolic compounds and antioxidant activity of rice. Braz. Arch. Biol. Technol. 2011, 54, 371–377. [Google Scholar] [CrossRef]
- Parr, A.J.; Bolwell, G.P. Phenols in the plant and in man. The potential for possible nutritional enhancement of the diet by modifying the phenols content or profile. J. Sci. Food Agric. 2000, 80, 985–1012. [Google Scholar] [CrossRef]
- Stalikas, C.D. Extraction, separation, and detection methods for phenolic acids and flavonoids. J. Sep. Sci. 2007, 30, 3268–3295. [Google Scholar] [CrossRef]
- Saeed, N.; Khan, M.R.; Shabbir, M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement. Altern. Med. 2012, 12, 221. [Google Scholar] [CrossRef] [PubMed]
- Hyun, T.K.; Kim, H.C.; Ko, Y.J.; Kim, J.S. Antioxidant, α-glucosidase inhibitory and anti-inflammatory effects of aerial parts extract from Korean crowberry (Empetrum nigrum var. japonicum). Saudi J. Biol. Sci. 2016, 23, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Yawadio, R.; Tanimori, S.; Morita, N. Identification of phenolic compounds isolated from pigmented rices and their aldose reductase inhibitory activities. Food Chem. 2007, 101, 1616–1625. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J.; Czemerys, R. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chem. 2007, 105, 940–949. [Google Scholar] [CrossRef]
- Lee, C.W.; Yoneyama, K.; Takeuchi, Y.; Konnai, M.; Tamogami, S.; Kodama, O. Momilactones A and B in rice straw harvested at different growth stages. Biosci. Biotechnol. Biochem. 1999, 63, 1318–1320. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Xuan, T.D.; Minh, T.N.; Siddiqui, N.A.; Van Quan, N. Comparative extraction and simple isolation improvement techniques of active constituents’ momilactone A and B from rice husks of Oryza sativa by HPLC analysis and column chromatography. Saudi Pharm. J. 2019, 27, 17–24. [Google Scholar] [CrossRef]
- Quan, N.V.; Tran, H.D.; Xuan, T.D.; Ahmad, A.; Dat, T.D.; Khanh, T.D.; Teschke, R. Momilactones A and B are α-amylase and α-glucosidase inhibitors. Molecules 2019, 24, 482. [Google Scholar] [CrossRef]
- Kato-Noguchi, H. Convergent or parallel molecular evolution of momilactone A and B: Potent allelochemicals, momilactones have been found only in rice and the moss Hypnum plumaeforme. J. Plant Physiol. 2011, 168, 1511–1516. [Google Scholar] [CrossRef]
- Cartwright, D.W.; Langcake, P.; Pryce, R.J.; Leworthy, D.P.; Ride, J.P. Isolation and characterization of two phytoalexins from rice as momilactones A and B. Phytochemistry 1981, 20, 535–537. [Google Scholar] [CrossRef]
- Kato-Noguchi, H.; Hasegawa, M.; Ino, T.; Ota, K.; Kujime, H. Contribution of momilactone A and B to rice allelopathy. J. Plant Physiol. 2010, 167, 787–791. [Google Scholar] [CrossRef]
- Toyomasu, T.; Kagahara, T.; Okada, K.; Koga, J.; Hasegawa, M.; Mitsuhashi, W.; Sassa, T.; Yamane, H. Diterpene phytoalexins are biosynthesized in and exuded from the roots of rice seedlings. Biosci. Biotechnol. Biochem. 2008, 72, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Joung, Y.H.; Lim, E.J.; Kim, M.S.; Lim, S.D.; Yoon, S.Y.; Lim, Y.C.; Yoo, Y.B.; Ye, S.K.; Park, T.; Chung, I.M.; et al. Enhancement of hypoxia-induced apoptosis of human breast cancer cells via STAT5b by momilactone B. Int. J. Oncol. 2008, 33, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Minh, T.N.; Xuan, T.D.; Ahmad, A.; Elzaawely, A.; Teschke, R.; Van, T.M. Momilactones A and B: Optimization of yields from isolation and purification. Separations 2018, 5, 28. [Google Scholar] [CrossRef]
- Fukuta, M.; Dang Xuan, T.; Deba, F.; Tawata, S.; Dang Khanh, T.; Min Chung, I. Comparative efficacies in vitro of antibacterial, fungicidal, antioxidant, and herbicidal activities of momilatones A and B. J. Plant Interact. 2007, 2, 245–251. [Google Scholar] [CrossRef]
- Kang, D.Y.; Darvin, P.; Joung, Y.H.; Byun, H.J.; Do, C.H.; Park, K.D.; Park, M.N.; Cho, K.H.; Yang, Y.M. Momilactone B inhibits ketosis in vitro by regulating the ANGPTL3-LPL pathway and inhibiting HMGCS2. Anim. Biotechnol. 2017, 28, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Kato-Noguchi, H.; Ino, T. Rice seedlings release momilactone B into the environment. Phytochemistry 2003, 63, 551–554. [Google Scholar] [CrossRef]
- Kato-Noguchi, H. Allelopathic substance in rice root exudates: Rediscovery of momilactone B as an allelochemical. J. Plant Physiol. 2004, 161, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Rayee, R.; Xuan, T.D.; Kakar, K.; Haqani, M.I. Antioxidant activity, quality parameters and grain characteristics of rice varieties of Afghanistan. Int. Lett. Nat. Sci. 2019, 73, 26. [Google Scholar] [CrossRef]
- Kakar, K.; Xuan, T.D.; Quan, N.V.; Wafa, I.K.; Tran, H.D.; Khanh, T.D.; Dat, T.D. Efficacy of MNU mutation on enhancing yield and quality of rice. Agriculture 2019, 9, 212. [Google Scholar] [CrossRef]
- Anh, T.T.T.; Khanh, T.D.; Dat, T.D.; Xuan, T.D. Identification of phenotypic variation and genetic diversity in rice (Oryza sativa L.) mutants. Agriculture 2018, 8, 30. [Google Scholar] [CrossRef]
- Quan, N.V.; Xuan, T.D.; Tran, H.D.; Thuy, N.T. Inhibitory activities of momilactones A, B, E, and 7-Ketostigmasterol isolated from rice husk on paddy and invasive weeds. Plants 2019, 8, 159. [Google Scholar] [CrossRef] [PubMed]
- Quan, N.V.; Khang, D.T.; Dep, L.T.; Minh, T.N.; Nakagoshi, N.; Xuan, T.D. The Potential use of a food-dyeing plant Peristrophe bivalvis (L.) Merr. in northern Vietnam. Int. J. Pharmacol. Phytochem. Ethnomed. 2016, 4, 14–26. [Google Scholar] [CrossRef]
- Quan, N.V.; Thien, D.D.; Khanh, T.D.; Tran, H.-D.; Xuan, T.D. Momilactones A, B, and tricin in rice grain and by-products are potential skin aging inhibitors. Foods 2019, 8, 602. [Google Scholar] [CrossRef] [PubMed]
- Quan, N.V.; Xuan, T.D.; Tran, H.D.; Ahmad, A.; Khanh, T.D.; Dat, T.D. Contribution of momilactones A and B to diabetes inhibitory potential of rice bran: Evidence from in vitro assays. Saudi Pharm. J. 2019, 27, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Robards, K.; Helliwell, S.; Blanchard, C. Composition and functional properties of rice. Int. J. Food Sci. Technol. 2002, 37, 849–868. [Google Scholar] [CrossRef]
- Chiang, A.N.; Wu, H.L.; Yeh, H.I.; Chu, C.S.; Lin, H.C.; Lee, W.C. Antioxidant effects of black rice extract through the induction of superoxide dismutase and catalase activities. Lipids 2006, 41, 797–803. [Google Scholar] [CrossRef]
- Shao, Y.; Xu, F.; Sun, X.; Bao, J.; Beta, T. Identification and quantification of phenolic acids and anthocyanins as antioxidants in bran, embryo and endosperm of white, red and black rice kernels (Oryza sativa L.). J. Cereal Sci. 2014, 59, 211–218. [Google Scholar] [CrossRef]
- Chung, M.J.; Walker, P.A.; Hogstrand, C. Dietary phenolic antioxidants, caffeic acid and Trolox, protect rainbow trout gill cells from nitric oxide-induced apoptosis. Aquat. Toxicol. 2006, 80, 321–328. [Google Scholar] [CrossRef]
- Ravishankar, D.; Rajora, A.K.; Greco, F.; Osborn, H.M. Flavonoids as prospective compounds for anti-cancer therapy. Int. J. Biochem. Cell Biol. 2013, 45, 2821–2831. [Google Scholar] [CrossRef]
- Goffman, F.D.; Bergman, C.J. Rice kernel phenolic content and its relationship with antiradical efficiency. J. Sci. Food Agric. 2004, 84, 1235–1240. [Google Scholar] [CrossRef]
- Zhang, M.W.; Guo, B.J.; Zhang, R.F.; Chi, J.W.; Wei, Z.C.; Xu, Z.H.; Zhang, Y.; Tang, X.J. Separation, purification and identification of antioxidant compositions in black rice. Agric. Sci. Chin. 2006, 5, 431–440. [Google Scholar] [CrossRef]
- Xuan, T.D.; Minh, T.N.; Khanh, T.D. Allelopathic momilactones A and B are implied in rice drought and salinity tolerance, not weed resistance. Agron. Sustain. Dev. 2016, 36, 52. [Google Scholar] [CrossRef]
- Quan, N.T.; Xuan, T.D. Foliar application of vanillic and p-hydroxybenzoic acids enhanced drought tolerance and formation of phytoalexin momilactones in rice. Arch. Agron. Soil Sci. 2018, 64, 1831–1846. [Google Scholar] [CrossRef]
- Minh, T.; Xuan, T.; Ahmad, A.; Elzaawely, A.; Teschke, R.; Van, T.T.M. Efficacy from different extractions for chemical profile and biological activities of rice husk. Sustainability 2018, 10, 1356. [Google Scholar] [CrossRef]
- Kim, S.J.; Park, H.R.; Park, E.; Lee, S.C. Cytotoxic and antitumor activity of momilactone B from rice hulls. J. Agric. Food Chem. 2007, 55, 1702–1706. [Google Scholar] [CrossRef]
- Kalapathy, U.; Proctor, A.; Shultz, J. Silicate thermal insulation material from rice hull ash. Ind. Eng. Chem. Res. 2003, 42, 46–49. [Google Scholar] [CrossRef]
- Tan, B.L.; Norhaizan, M.E. Scientific evidence of rice by-products for cancer prevention: Chemopreventive properties of waste products from rice milling on carcinogenesis in vitro and in vivo. BioMed Res. Int. 2017, 2017, 1–18. [Google Scholar] [CrossRef]
- Butsat, S.; Siriamornpun, S. Antioxidant capacities and phenolic compounds of the husk, bran and endosperm of Thai rice. Food Chem. 2010, 119, 606–613. [Google Scholar] [CrossRef]
- Fardet, A.; Rock, E.; Rémésy, C. Is the in vitro antioxidant potential of whole-grain cereals and cereal products well reflected in vivo? J. Cereal Sci. 2008, 48, 258–276. [Google Scholar] [CrossRef]
| Code | Origin | Descriptions | Status |
|---|---|---|---|
| K1 | DT84 | A sticky and traditional rice cultivar in the north of Vietnam with good quality | Cultivar |
| K2 | Mutated DT84 | F2 (self-pollination from the mutated DT84 F1) | Mutant line |
| Samples | IC50 of DPPH (µg/mL) | IC50 of ABTS (µg/mL) | TPC (µg/g) | TFC (µg/g) | |
|---|---|---|---|---|---|
| K1 | Grain | 2009.0 ± 181.2 b | 766.5 ± 20.6 a | 75.8 ± 6.3 d | 1.4 ± 0.1 e |
| Husk | 503.4 ± 18.3 d | 72.6 ± 6.9 e | 462.5 ± 27.0 c | 75.9 ± 1.8 c | |
| Straw | 2372.7 ± 153.3 a | 601.7 ± 27.5 b | 893.3 ± 39.5 b | 197.2 ± 5.3 b | |
| K2 | Grain | 1267.1 ± 68.8 c | 642.0 ± 10.7 b | 80.3 ± 9.9 d | 0.8 ± 0.1 e |
| Husk | 972.9 ± 69.7 c | 137.6 ± 3.5 d | 405.9 ± 3.0 c | 54.7 ± 2.1 d | |
| Straw | 1247.9 ± 77.4 c | 371.8 ± 26.0 c | 1156.6 ± 94.7 a | 481.6 ± 14.8 a | |
| BHT | 28.95 ± 0.8 e | 30.63 ± 0.6 f | - | - | |
| AC | PC | LC | TS | GY | 1/DPPH | 1/ABTS | TPC | TFC | MA | |
|---|---|---|---|---|---|---|---|---|---|---|
| PC | 0.610 | |||||||||
| LC | 0.138 | 0.212 | ||||||||
| TS | −0.829 * | −0.772 | −0.549 | |||||||
| GY | −0.958 ** | −0.481 | −0.359 | 0.858 * | ||||||
| 1/DPPH | −0.992 *** | −0.549 | −0.122 | 0.798 * | 0.961 ** | |||||
| 1/ABTS | −0.970 *** | −0.560 | −0.302 | 0.830 * | 0.972 *** | 0.957 ** | ||||
| TPC | −0.204 | 0.306 | 0.151 | −0.139 | 0.238 | 0.308 | 0.209 | |||
| TFC | 0.913 ** | 0.634 | 0.466 | −0.915 ** | −0.949 ** | −0.919 ** | −0.929 ** | −0.259 | ||
| MA | 0.946 ** | 0.478 | 0.371 | −0.832 * | −0.989 *** | −0.958 ** | −0.972 *** | −0.344 | 0.967 ** | |
| MB | 0.921 ** | 0.414 | 0.407 | −0.814 * | −0.987 *** | −0.938 ** | −0.956 ** | −0.360 | 0.954 ** | 0.996 *** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kakar, K.; Xuan, T.D.; Quan, N.V.; Wafa, I.K.; Tran, H.-D.; Khanh, T.D.; Dat, T.D. Efficacy of N-Methyl-N-Nitrosourea Mutation on Physicochemical Properties, Phytochemicals, and Momilactones A and B in Rice. Sustainability 2019, 11, 6862. https://doi.org/10.3390/su11236862
Kakar K, Xuan TD, Quan NV, Wafa IK, Tran H-D, Khanh TD, Dat TD. Efficacy of N-Methyl-N-Nitrosourea Mutation on Physicochemical Properties, Phytochemicals, and Momilactones A and B in Rice. Sustainability. 2019; 11(23):6862. https://doi.org/10.3390/su11236862
Chicago/Turabian StyleKakar, Kifayatullah, Tran Dang Xuan, Nguyen Van Quan, Imran Khan Wafa, Hoang-Dung Tran, Tran Dang Khanh, and Tran Dang Dat. 2019. "Efficacy of N-Methyl-N-Nitrosourea Mutation on Physicochemical Properties, Phytochemicals, and Momilactones A and B in Rice" Sustainability 11, no. 23: 6862. https://doi.org/10.3390/su11236862
APA StyleKakar, K., Xuan, T. D., Quan, N. V., Wafa, I. K., Tran, H.-D., Khanh, T. D., & Dat, T. D. (2019). Efficacy of N-Methyl-N-Nitrosourea Mutation on Physicochemical Properties, Phytochemicals, and Momilactones A and B in Rice. Sustainability, 11(23), 6862. https://doi.org/10.3390/su11236862










