Soil Microbial Community Structure and Physicochemical Properties in Amomum tsaoko-based Agroforestry Systems in the Gaoligong Mountains, Southwest China
Abstract
1. Introduction
2. Materials and Methods
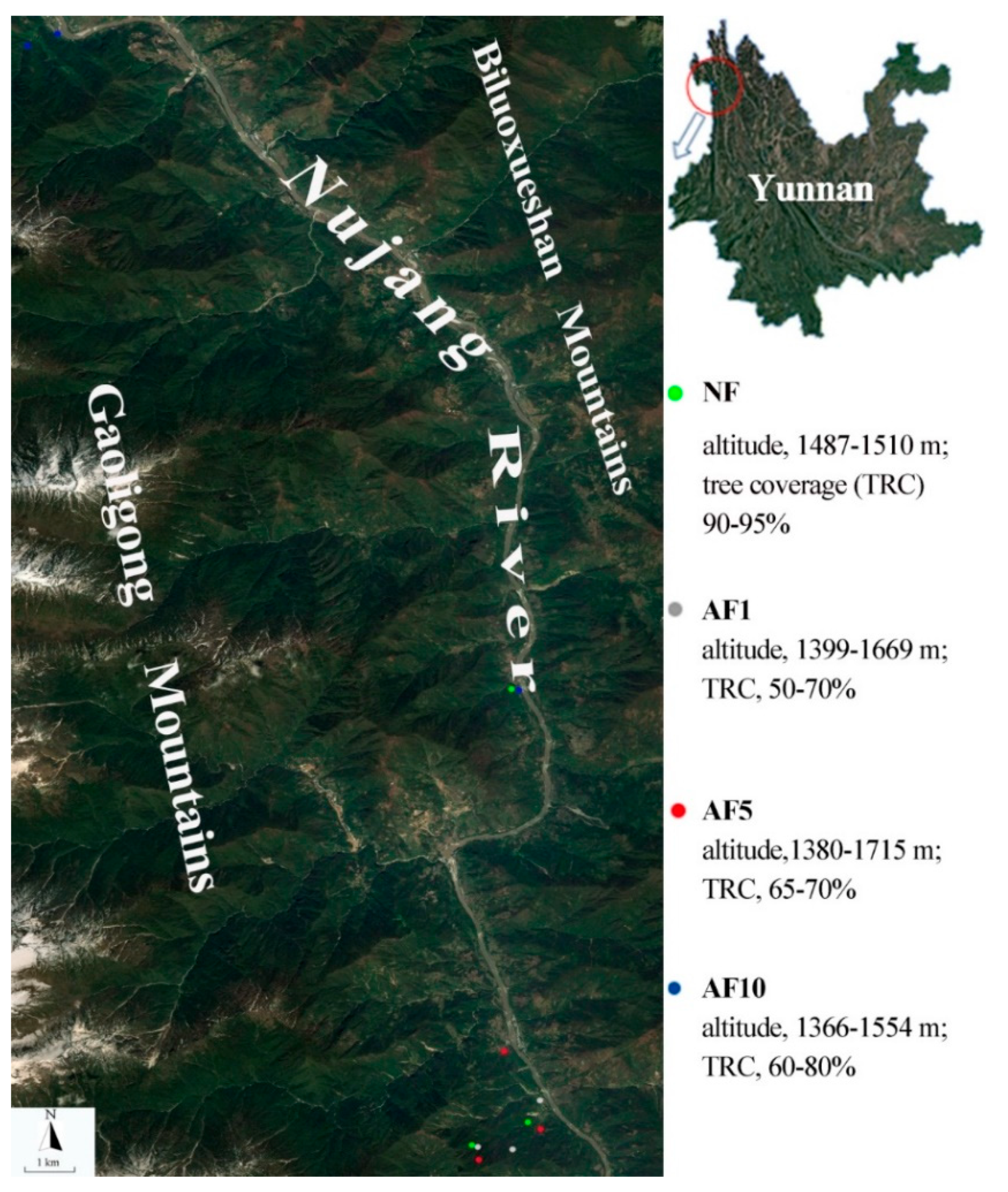
2.1. Study Area
2.2. Field Sampling and Soil Physicochemical Properties Determination
2.3. Analysis of Microbial Biomass and Structure
2.4. Data Processing and Analysis
3. Results
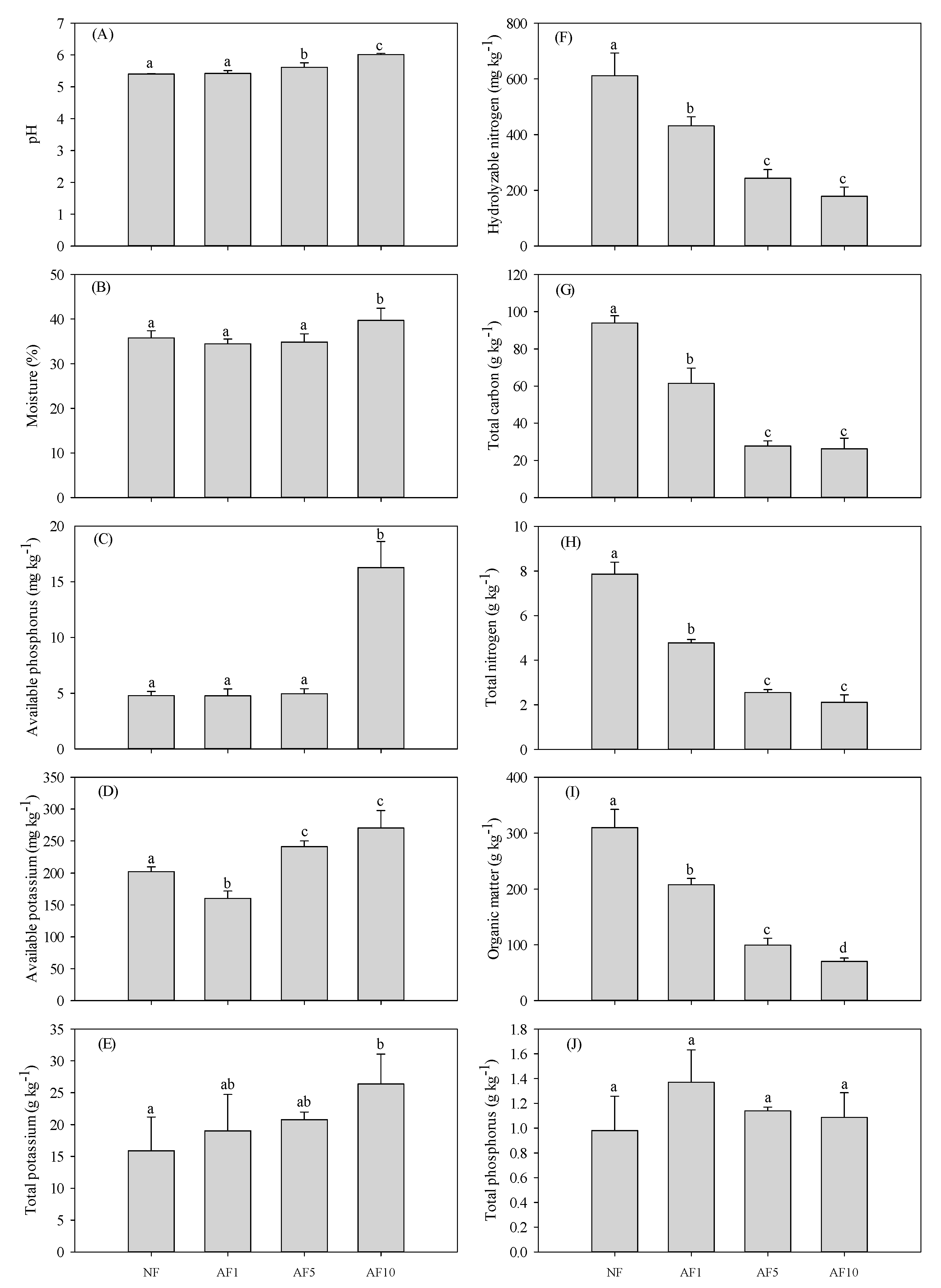
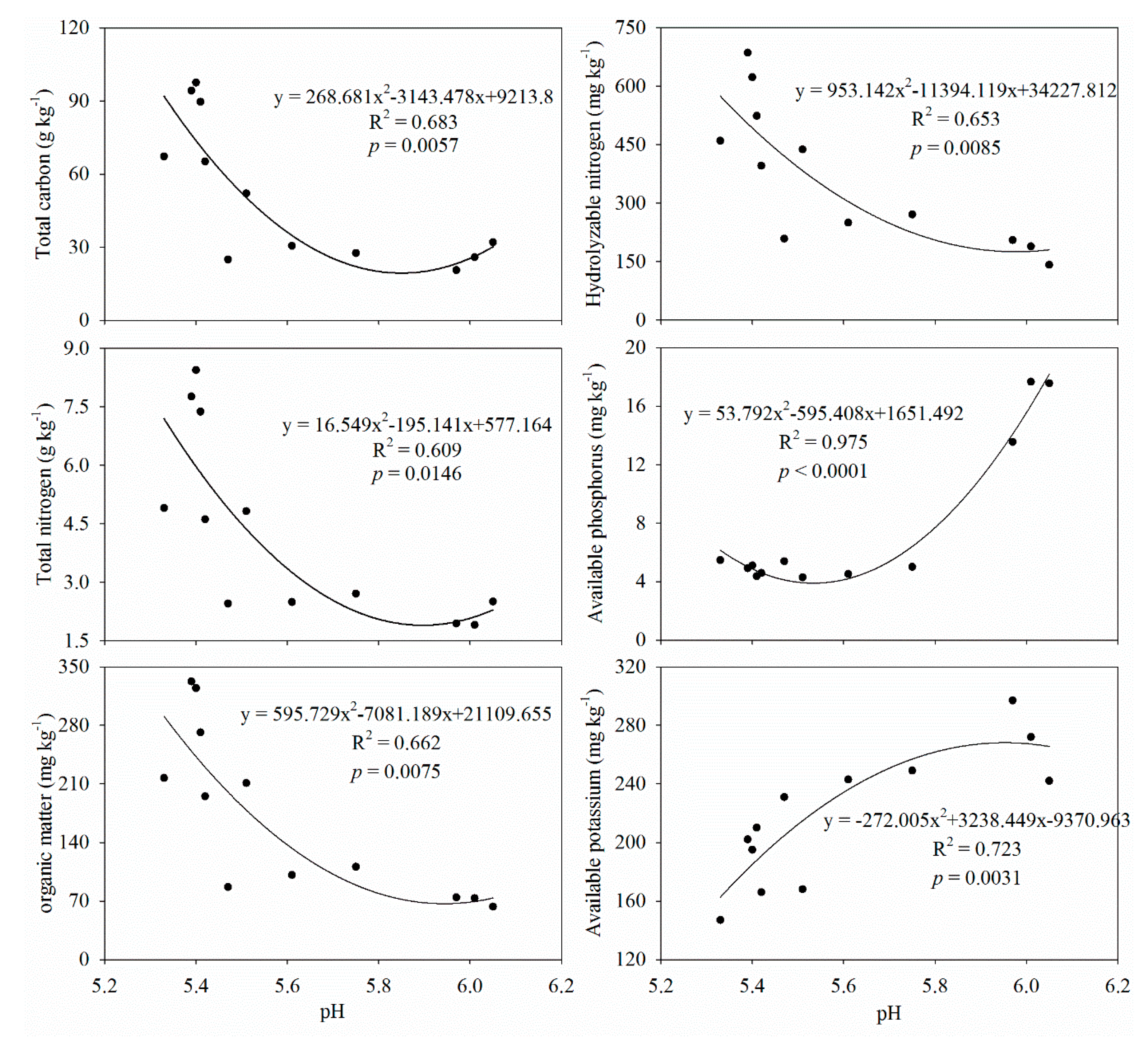
3.1. Soil Properties
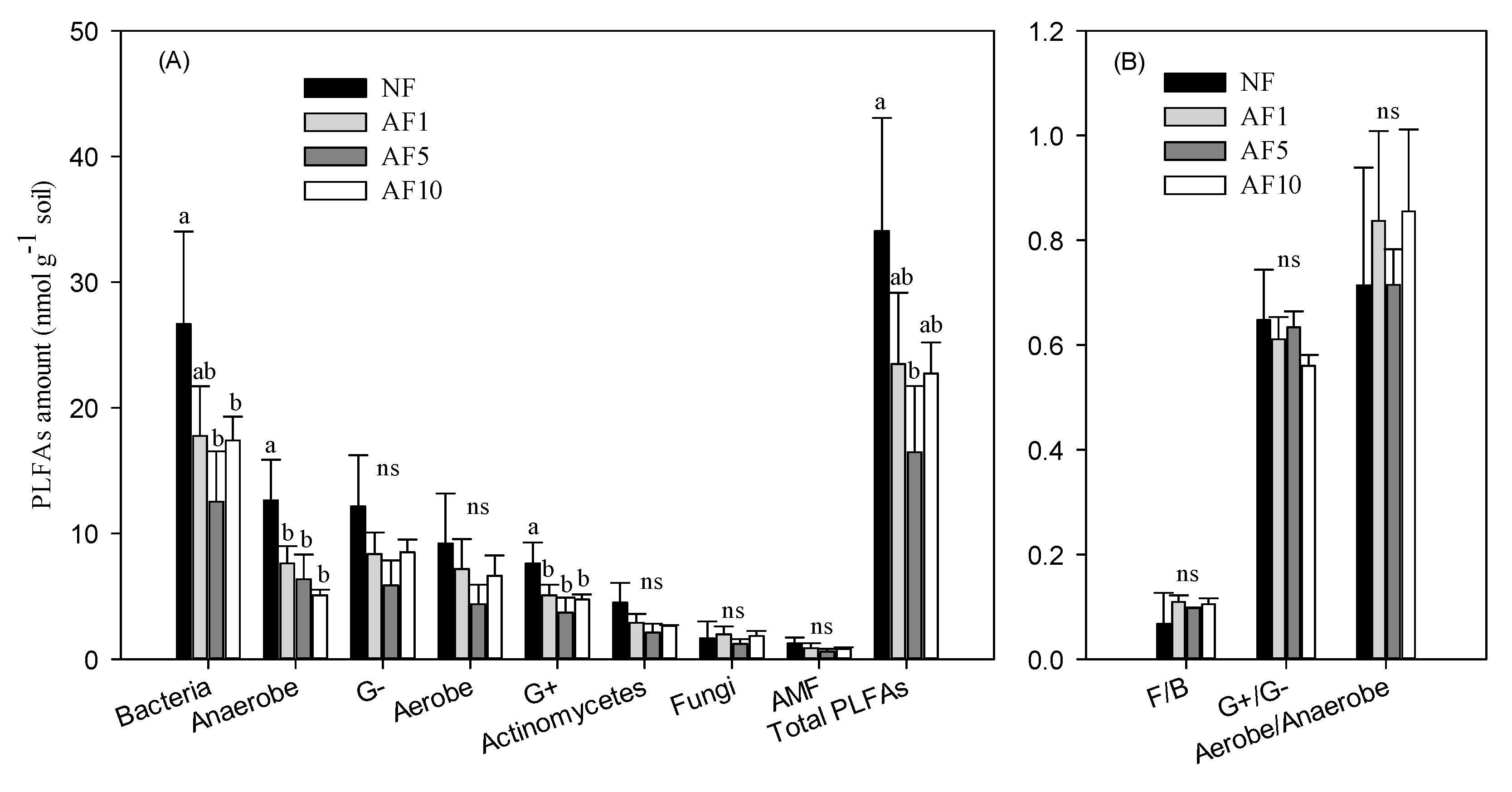
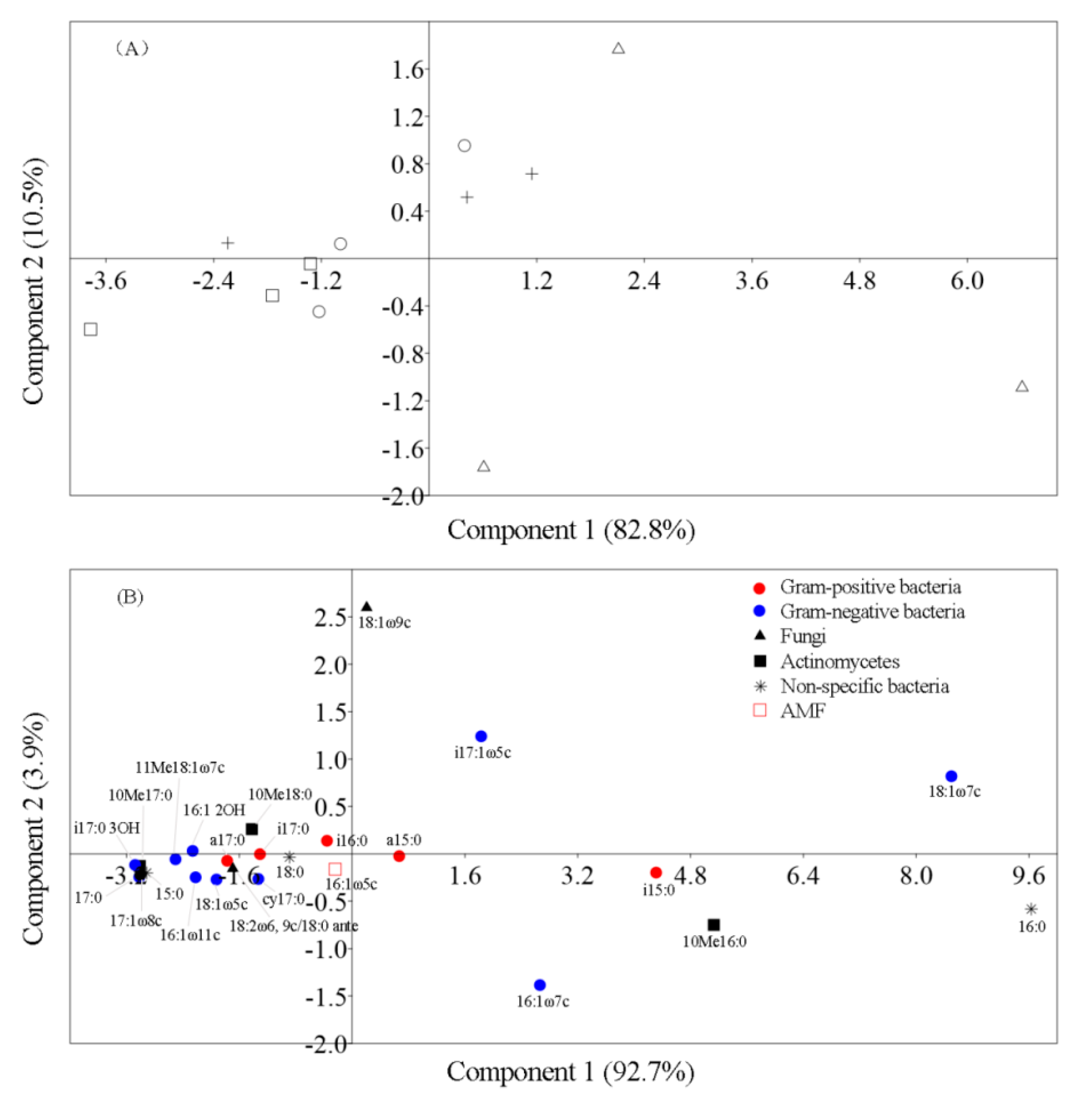
3.2. The Microbial Community Component
3.3. Correlations Between Microbial Community and Soil Characteristics
4. Discussion
4.1. AFs Effects on Soil Physicochemical Properties
4.2. AFs Effects on Soil Microbial Community Structure
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- MNRC. Bulletin of China National Land Resources. 2017. Available online: http://www.mnr.gov.cn/sj/tjgb/201807/P020180704391918680508.pdf (accessed on 16 January 2019).
- BNFGC. China Forestry Development Report; China Forestry Publishing House: Beijing, China, 2015. [Google Scholar]
- Yin, H.; Li, Y.; Ye, Z. Optimization principles and characteristics of agricultural industrial structure in Nujiang of Yunnan. Res. Agric. Mod. 2009, 30, 707–711. [Google Scholar]
- Banerjee, S.; Baah-Acheamfour, M.; Carlyle, C.N.; Bissett, A.; Richardson, A.E.; Siddique, T.; Bork, E.W.; Chang, S.X. Determinants of bacterial communities in Canadian agroforestry systems. Environ. Microbiol. 2016, 18, 1805–1816. [Google Scholar] [CrossRef] [PubMed]
- Moorhead, D.J.; Dickens, E.D. Agroforestry: A profitable land use. An overview of the 12th North American Agroforestry Conference. Agrofor. Syst. 2012, 86, 299–302. [Google Scholar] [CrossRef]
- Schwab, N.; Schickhoff, U.; Fischer, E. Transition to agroforestry significantly improves soil quality: A case study in the central mid-hills of Nepal. Agric. Ecosyst. Environ. 2015, 205, 57–69. [Google Scholar] [CrossRef]
- Silva, G.L.; Lima, H.V.; Campanha, M.M.; Gilkes, R.J.; Oliveira, T.S. Soil physical quality of Luvisols under agroforestry, natural vegetation and conventional crop management systems in the Brazilian semi-arid region. Geoderma 2011, 167–68, 61–70. [Google Scholar] [CrossRef]
- Weerasekara, C.; Udawatta, R.P.; Jose, S.; Kremer, R.J.; Weerasekara, C. Soil quality differences in a row-crop watershed with agroforestry and grass buffers. Agrofor. Syst. 2016, 90, 829–838. [Google Scholar] [CrossRef]
- Sharma, S.K.; Ramesh, A.; Sharma, M.P.; Joshi, O.P.; Govaerts, B.; Steenwerth, K.L.; Karlen, D.L. Microbial Community Structure and Diversity as Indicators for Evaluating Soil Quality; Springer: Berlin, Germany, 2010. [Google Scholar]
- Wall, D.H.; Nielsen, U.N.; Six, J. Soil biodiversity and human health. Nature 2015, 528, 69–76. [Google Scholar] [CrossRef]
- Vallejo, V.E.; Roldan, F.; Dick, R.P. Soil enzymatic activities and microbial biomass in an integrated agroforestry chronosequence compared to monoculture and a native forest of Colombia. Biol. Fertil. Soils 2010, 46, 577–587. [Google Scholar] [CrossRef]
- Gardi, C.; Montanarella, L.; Arrouays, D.; Bispo, A.; Lemanceau, P.; Jolivet, C.; Mulder, C.; Ranjard, L.; Rombke, J.; Rutgers, M.; et al. Soil biodiversity monitoring in Europe: Ongoing activities and challenges. Eur. J. Soil Sci. 2009, 60, 807–819. [Google Scholar] [CrossRef]
- Nannipieri, P.; Ascher, J.; Ceccherini, M.; Landi, L.; Pietramellara, G.; Renella, G. Mirobial diversity and soil functions. Eur. J. Soil Sci. 2003, 54, 655–670. [Google Scholar] [CrossRef]
- Berg, G. Plant-microbe interactions promoting plant growth and health: Perspectives for controlled use of microorganisms in agriculture. Appl. Microbiol. Biotechnol. 2009, 84, 11–18. [Google Scholar] [CrossRef]
- Balestrini, R.; Lumini, E.; Borriello, R.; Bianciotto, V. Soil Microbiology, Ecology and Biochemistry (Fourth Edition): Plant-Soil Biota Interactions; Elsevier: New York, NY, USA, 2015. [Google Scholar]
- Lavelle, P.; Decaens, T.; Aubert, M.; Barot, S.; Blouin, M.; Bureau, F.; Margerie, P.; Mora, P.; Rossi, J.P. Soil invertebrates and ecosystem services. Eur. J. Soil Biol. 2006, 42, S3–S15. [Google Scholar] [CrossRef]
- Colombo, F.; Macdonald, C.A.; Jeffries, T.C.; Powell, J.R.; Singh, B.K. Impact of forest management practices on soil bacterial diversity and consequences for soil processes. Soil Biol. Biochem. 2016, 94, 200–210. [Google Scholar] [CrossRef]
- Girmay, G.; Singh, B.R.; Mitiku, H.; Borresen, T.; Lal, R. Carbon stocks in Ethiopian soils in relation to land use and soil management. Land Degrad. Dev. 2008, 19, 351–367. [Google Scholar] [CrossRef]
- Imaz, M.J.; Virto, I.; Bescansa, P.; Enrique, A.; Fernandez-Ugalde, O.; Karlen, D.L. Soil quality indicator response to tillage and residue management on semi-arid Mediterranean cropland. Soil Tillage Res. 2010, 107, 17–25. [Google Scholar] [CrossRef]
- Allison, V.J.; Miller, R.M.; Jastrow, J.D.; Matamala, R.; Zak, D.R. Changes in soil microbial community structure in a tallgrass prairie chronosequence. Soil Sci. Soc. Am. J. 2005, 69, 1412–1421. [Google Scholar] [CrossRef]
- Huang, Y.M.; Michel, K.; An, S.S.; Zechmeister-Boltenstern, S. Changes in microbial-community structure with depth and time in a chronosequence of restored grassland soils on the Loess Plateau in northwest China. J. Plant Nutr. Soil Sci. 2011, 174, 765–774. [Google Scholar] [CrossRef]
- Li, D.J.; Wen, L.; Jiang, S.; Song, T.Q.; Wang, K.L. Responses of soil nutrients and microbial communities to three restoration strategies in a karst area, southwest China. J. Environ. Manag. 2018, 207, 456–464. [Google Scholar] [CrossRef]
- Wu, Z.; Haack, S.E.; Lin, W.; Li, B.; Wu, L.; Fang, C.; Zhang, Z. Soil microbial community structure and metabolic activity of Pinus elliottii plantations across different stand ages in a subtropical area. PLoS ONE 2015, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Zhang, D.G.; Jiang, Z.H.; Sun, P.; Xiao, H.L.; Wu, Y.X.; Chen, J.G. Changes in the soil microbial communities of alpine steppe at Qinghai-Tibetan Plateau under different degradation levels. Sci. Total Environ. 2019, 651, 2281–2291. [Google Scholar] [CrossRef] [PubMed]
- Simpson, A.C.; Zabowski, D.; Rochefort, R.M.; Edmonds, R.L. Increased microbial uptake and plant nitrogen availability in response to simulated nitrogen deposition in alpine meadows. Geoderma 2019, 336, 68–80. [Google Scholar] [CrossRef]
- Wen, L.; Lei, P.; Xiang, W.; Yan, W.; Liu, S. Soil microbial biomass carbon and nitrogen in pure and mixed stands of Pinus massoniana and Cinnamomum camphora differing in stand age. For. Ecol. Manag. 2014, 328, 150–158. [Google Scholar] [CrossRef]
- Chen, F.; Zheng, H.; Zhang, K.; Ouyang, Z.; Lan, J.; Li, H.; Shi, Q. Changes in soil microbial community structure and metabolic activity following conversion from native Pinus massoniana plantations to exotic Eucalyptus plantations. For. Ecol. Manag. 2013, 291, 65–72. [Google Scholar] [CrossRef]
- Zhu, H.; Xu, Z.; Li, B.; Wang, H. A discussion on the loss of biodiversity of tropical rainforest by Amomum villosum planting underneath in south Yunnan. Guihaia 2002, 22, 55–60. [Google Scholar]
- Lin, J.; Zheng, Y.; Yu, Y.; Xia, P.; Wu, Z.; Chen, F.; Song, L. Analysis of essential oil from Amomum tsaoko by extraction of supercritical CO2 fluid. J. Chin. Med. Mater. 2000, 23, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Wei, R.; Huang, R. A preliminary study on the genetic system of Amomum tsaoko. J. Yunnan Univ. 1995, 17, 290–297. [Google Scholar]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Porta, J.; Lopez-Acevedo, M.; Roquero, C. Edafologia; Editions Mundi-Prensa: Madrid, Spain, 1994. [Google Scholar]
- Forestry Industry Standards of the People’s Republic of China. Forest Soil Analysis Method (Forestry Industry Standard, People’s Republic of China); Standards Press of China: Beijing, China, 2000. [Google Scholar]
- Bossio, D.; Scow, K.M. Impacts of carbon and flooding on soil microbial communities: Phospholipid fatty acid profiles and substrate utilization patterns. Microb. Ecol. 1998, 35, 265–278. [Google Scholar] [CrossRef]
- Cao, Y.; Fu, S.; Zou, X.; Cao, H.; Shao, Y.; Zhou, L. Soil microbial community composition under Eucalyptus plantations of different age in subtropical China. Eur. J. Soil Biol. 2010, 46, 128–135. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, T.; Gilliam, F.S.; Gundersen, P.; Zhang, W.; Chen, H.; Mo, J.M. Interactive Effects of Nitrogen and Phosphorus on Soil Microbial Communities in a Tropical Forest. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Zelles, L. Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: A review. Biol. Fertil. Soils 1999, 29, 111–129. [Google Scholar] [CrossRef]
- Li, C.; Shi, L.-L.; Ostermann, A.; Xu, J.; Li, Y.; Mortimer, P.E. Indigenous trees restore soil microbial biomass at faster rates than exotic species. Plant Soil 2015, 396, 151–161. [Google Scholar] [CrossRef]
- Bhandari, K.B.; West, C.P.; Acosta-Martinez, V.; Cotton, J.; Cano, A. Soil health indicators as affected by diverse forage species and mixtures in semi-arid pastures. Appl. Soil Ecol. 2018, 132, 179–186. [Google Scholar] [CrossRef]
- Federle, T.W. Microbial Distribution in Soil-New Techniques. In Perspectives in Microbial Ecology; Slovene Society for Microbiology: Ljubljana, Slovenia, 1986; pp. 493–498. [Google Scholar]
- Zheng, X.; Su, Y.; Liu, B.; Lan, J.; Yang, S.; Lin, Y. Microbial community diversity in tea root zone soil at different elevations. Chin. J. Eco-Agric. 2010, 18, 866–871. [Google Scholar] [CrossRef]
- Olatunji, O.A.; Luo, H.; Pan, K.; Tariq, A.; Sun, X.; Chen, W.; Wu, X.; Zhang, L.; Xiong, Q.; Li, Z.; et al. Influence of phosphorus application and water deficit on the soil microbiota of N2-fixing and non-N-fixing tree. Ecosphere 2018, 9. [Google Scholar] [CrossRef]
- Lipski, A.; Spieck, E.; Makolla, A.; Altendorf, K. Fatty acid profiles of nitrite-oxidizing bacteria reflect their phylogenetic heterogeneity. Syst. Appl. Microbiol. 2001, 24, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Schwab, V.F.; Herrmann, M.; Roth, V.-N.; Gleixner, G.; Lehmann, R.; Pohnert, G.; Trumbore, S.; Kuesel, K.; Totsche, K.U. Functional diversity of microbial communities in pristine aquifers inferred by PLFA- and sequencing-based approaches. Biogeosciences 2017, 14, 2697–2714. [Google Scholar] [CrossRef]
- Luo, D.; Cheng, R.M.; Shi, Z.M.; Wang, W.X. Decomposition of Leaves and Fine Roots in Three Subtropical Plantations in China Affected by Litter Substrate Quality and Soil Microbial Community. Forests 2017, 8, 412. [Google Scholar] [CrossRef]
- Frostegard, A.; Baath, E.; Tunlid, A. Shifts in the structure of soil microbial communities in limed forests as revealed by phospholipid fatty-acid analysis. Soil Biol. Biochem. 1993, 25, 723–730. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Q.; Yao, H. Soil carbon, nitrogen and microbiological characteristics during bamboo high-speed growth. Acta Ecol. Sin. 2012, 32, 1412–1418. [Google Scholar] [CrossRef]
- Luo, S.; Huang, J.; Yuan, L. Nutrients and microorganisms in soils with wild Artemisia annua L. Acta Pedol. Sin. 2014, 51, 868–879. [Google Scholar]
- Wang, F.; Yuan, T.; Gu, S.; Wang, Z. Effects of organic and inorganic slow-release compound fertilizer on different soils microbial community structure. Environ. Sci. 2015, 36, 1461–1467. [Google Scholar]
- Olsson, P.A. Signature fatty acids provide tools for determination of the distribution and interactions of mycorrhizal fungi in soil. FEMS Microbiol. Ecol. 1999, 29, 303–310. [Google Scholar] [CrossRef]
- Zak, D.R.; Ringelberg, D.B.; Pregitzer, K.S.; Randlett, D.L.; White, D.C.; Curtis, P.S. Soil microbial communities beneath Populus grandidentata crown under elevated atmospheric CO2. Ecol. Appl. 1996, 6, 257–262. [Google Scholar] [CrossRef]
- Moche, M.; Gutknecht, J.; Schulz, E.; Langer, U.; Rinklebe, J. Monthly dynamics of microbial community structure and their controlling factors in three floodplain soils. Soil Biol. Biochem. 2015, 90, 169–178. [Google Scholar] [CrossRef]
- Si, G.H.; Peng, C.L.; Yuan, J.F.; Xu, X.Y.; Zhao, S.J.; Xu, D.B.; Wu, J.S. Changes in soil microbial community composition and organic carbon fractions in an integrated rice-crayfish farming system in subtropical China. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Bossio, D.A.; Fleck, J.A.; Scow, K.M.; Fujii, R. Alteration of soil microbial communities and water quality in restored wetlands. Soil Biol. Biochem. 2006, 38, 1223–1233. [Google Scholar] [CrossRef]
- Chen, H.; Ning, D.; Li, Y.; Liao, Y.; Xiao, L. High-yield cultivation technique of Amomum tsaoko. China For. Sci. Technol. 2012, 26, 105–107. [Google Scholar]
- Li, J. Study on the cultivation techniques of Amomum tsaoko under forest. J. Green Sci. Technol. 2017, 29–30. [Google Scholar]
- Li, X.H. Causes of wilthering death and its prevent and control measures of Amomum tsaoko. South China Agric. 2017, 11, 33–34. [Google Scholar]
- Yang, Z.; Yu, Y. A summary on cultivation and diseases and pests control of Amomum tsaoko in Jiakedi, Yunnan. Agric. Dev. Equip. 2015, 12, 153. [Google Scholar]
- Thomazini, A.; Mendonca, E.S.; Cardoso, I.M.; Garbin, M.L. SOC dynamics and soil quality index of agroforestry systems in Atlantic rainforest of Brazil. Geoderma Reg. 2015, 5, 15–24. [Google Scholar] [CrossRef]
- Jin, M.; Cai, C.; Liu, G.; Ma, C. Response of Amomum tsaoko productions and benefits to different environmental factors and cultural measures in the canyon region of Nujiang. Chin. J. Trop. Crops 2016, 37, 446–455. [Google Scholar]
- Zhang, Y. High-yield cultivation of Amomum Tsaoko in Longling. For. Inventory Plann. 2011, 36, 113–116. [Google Scholar]
- Wang, X.; Cao, Y.; Zhang, F. Nitrogen phosphorus and potassium in soil nature of resources and integrated management. Acta Agric. Univ. Pekin. 1995, 21, 89–93. [Google Scholar]
- Jensen, T.L. Soil pH and the Availability of Plant Nutrients. IPNI Plant Nutrition Today No. 2. , 2010. Available online: www.ipni.net/pnt (accessed on 16 January 2019).
- Brodie, E.; Edwards, S.; Clipson, N. Bacterial community dynamics across a floristic gradient in a temperate upland grassland ecosystem. Microb. Ecol. 2002, 44, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.C.; Ren, C.Q.; Cheng, H.T.; Zou, Y.K.; Bughio, M.A.; Li, Q.F. Conversion of rainforest into agroforestry andmonoculture plantation in China: Consequences for soil phosphorus forms and microbial community. Sci. Total Environ. 2017, 595, 769–778. [Google Scholar] [CrossRef]
- Du, M.; Xie, J. Study on the chemical constituents of the essential oil from the stems of Amomum tsaoko. Yunnan Chem. Technol. 1994, 3, 5,17,18. [Google Scholar]
- Min, Y.; Zhang, W.; Yao, L.; Liu, W.; Wang, H. Study on the essential oil chemical ingredients of Amomum tsaoko leaf from different regions of Yunnan province. J. Anhui Agric. Sci. 2011, 39, 3298–3300. [Google Scholar]
- Yang, Y.; Yan, R.W.; Cai, X.Q.; Zheng, Z.L.; Zou, G.L. Chemical composition and antimicrobial activity of the essential oil of Amomum tsaoko. J. Sci. Food Agric. 2008, 88, 2111–2116. [Google Scholar] [CrossRef]
- Liu, X. Studies on extraction, application and safety evaluation of antibacterial substances from Amomum tsaoko. Master’s Thesis, Hainan University, Haikou, China, 2011. [Google Scholar]
- Meng, D.; Li, W.; Wang, P.; Ren, H.; Lu, Z. Comparison of aromatic components and essential oil from Amomum tasoko. J. Harbin Univ. Commer. 2012, 28, 703–707. [Google Scholar]
- Leckie, S.E.; Prescott, C.E.; Grayston, S.J.; Neufeld, J.D.; Mohn, W.W. Characterization of humus microbial communities in adjacent forest types that differ in nitrogen availability. Microb. Ecol. 2004, 48, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Hannam, K.D.; Quideau, S.A.; Kishchuk, B.E. Forest floor microbial communities in relation to stand composition and timber harvesting in northern Alberta. Soil Biol. Biochem. 2006, 38, 2565–2575. [Google Scholar] [CrossRef]
- Grayston, S.J.; Campbell, C.D.; Bardgett, R.D.; Mawdsley, J.L.; Clegg, C.D.; Ritz, K.; Griffiths, B.S.; Rodwell, J.S.; Edwards, S.J.; Davies, W.J.; et al. Assessing shifts in microbial community structure across a range of grasslands of differing management intensity using CLPP, PLFA and community DNA techniques. Appl. Soil Ecol. 2004, 25, 63–84. [Google Scholar] [CrossRef]
- Rivest, D.; Lorente, M.; Olivier, A.; Messier, C. Soil biochemical properties and microbial resilience in agroforestry systems: Effects on wheat growth under controlled drought and flooding conditions. Sci. Total Environ. 2013, 463, 51–60. [Google Scholar] [CrossRef]
- Frostegard, A.; Tunlid, A.; Baath, E. Microbial biomass measured as total lipid phosphate in soils of different organic content. J. Microbiol. Methods 1991, 14, 151–163. [Google Scholar] [CrossRef]
- Sun, Y.; Wu, J.; Shao, Y.; Zhou, L.; Mai, B.; Lin, Y.; Fu, S. Responses of soil microbial communities to prescribed burning in two paired vegetation sites in southern China. Ecol. Res. 2011, 26, 669–677. [Google Scholar] [CrossRef]
- Green, C.T.; Scow, K.M. Analysis of phospholipid fatty acids (PLFA) to characterize microbial communities in aquifers. Hydrol. J. 2000, 8, 126–141. [Google Scholar] [CrossRef]
- Yu, H.Y.; Ma, Q.H.; Liu, X.D.; Xu, Z.Z.; Zhou, G.S.; Shi, Y.H. Short- and long-term warming alters soil microbial community and relates to soil traits. Appl. Soil Ecol. 2018, 131, 22–28. [Google Scholar] [CrossRef]
| Total PLFA | Fungi | Bacteria | G+ | G− | F/B | G+/G− | Actinomycetes | Aerobe | Anaerobe | AMF | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | −0.302 | 0.035 | −0.304 | −0.377 | −0.236 | 0.236 | −0.474 | −0.346 | −0.230 | −0.380 | −0.288 |
| Moisture | 0.111 | −0.004 | 0.127 | 0.064 | 0.194 | −0.037 | −0.467 | 0.042 | 0.147 | 0.052 | 0.089 |
| Ctotal | 0.681 * | 0.104 | 0.686 * | 0.759 ** | 0.615 * | −0.385 | 0.380 | 0.693 * | 0.533 | 0.750 ** | 0.558 |
| Ntotal | 0.714 ** | 0.142 | 0.714 ** | 0.783 ** | 0.643 * | −0.369 | 0.355 | 0.729 ** | 0.571 | 0.776 ** | 0.592 * |
| Ptotal | −0.284 | 0.500 | −0.334 | −0.358 | −0.271 | 0.684 * | −0.260 | −0.332 | −0.007 | −0.336 | −0.322 |
| Ktotal | −0.385 | −0.171 | −0.363 | −0.424 | −0.331 | 0.062 | −0.284 | −0.441 | −0.379 | −0.442 | −0.375 |
| Nhy | 0.753 ** | 0.119 | 0.753 ** | 0.808 ** | 0.697 * | −0.380 | 0.269 | 0.776 ** | 0.643 * | 0.810 ** | 0.666 * |
| Pavail | −0.086 | 0.168 | −0.091 | −0.168 | −0.026 | 0.216 | −0.551 | −0.173 | −0.017 | −0.182 | −0.070 |
| Kavail | −0.278 | −0.184 | −0.260 | −0.306 | −0.216 | 0.011 | −0.261 | −0.294 | −0.282 | −0.316 | −0.257 |
| OM | 0.734 ** | 0.142 | 0.733 ** | 0.793 ** | 0.672 * | −0.360 | 0.300 | 0.753 ** | 0.618 * | 0.793 ** | 0.633 * |
| PLFA Marks | Signature of | Soil Chemical Variables | ||||
|---|---|---|---|---|---|---|
| Ctotal | Ntotal | OM | Ptotal | Nhy | ||
| 15:0 | non-specific bacteria | 0.794 ** | 0.818 ** | 0.821 ** | −0.394 | 0.830 ** |
| 16:0 | non-specific bacteria | 0.705 * | 0.732 ** | 0.741 ** | −0.405 | 0.755 ** |
| 18:0 | non-specific bacteria | 0.706 * | 0.742 ** | 0.755 ** | −0.346 | 0.772 ** |
| 17:0 | General bacteria | 0.529 | 0.578 * | 0.585 * | −0.466 | 0.606 * |
| a15:0 | Gram-positive bacteria | 0.584 * | 0.623 * | 0.662 * | −0.307 | 0.695 * |
| i15:0 | Gram-positive bacteria | 0.739 ** | 0.756 ** | 0.762 ** | −0.405 | 0.775 ** |
| i16:0 | Gram-positive bacteria | 0.828 ** | 0.850 ** | 0.807 ** | −0.382 | 0.793 ** |
| a17:0 | Gram-positive bacteria | 0.702 * | 0.744 ** | 0.778 ** | −0.251 | 0.803 ** |
| i17:0 | Gram-positive bacteria | 0.785 ** | 0.783 ** | 0.811 ** | −0.172 | 0.824 ** |
| 16:1 2OH | Gram-negative bacteria | 0.758 ** | 0.708 ** | 0.727 ** | 0.122 | 0.716 ** |
| 16:1ω7c | Gram-negative bacteria | 0.510 * | 0.545 | 0.596 * | −0.352 | 0.636 * |
| 16:1ω11c | Gram-negative bacteria | 0.507 | 0.548 | 0.608 * | −0.266 | 0.651 * |
| 17:1ω8c | Gram-negative bacteria | 0.591 * | 0.623 * | 0.663 * | −0.369 | 0.695 * |
| i17:1ω5c | Gram-negative bacteria | −0.109 | −0.123 | −0.223 | −0.535 | −0.249 |
| i17:0 3OH | Gram-negative bacteria | 0.300 | 0.250 | 0.175 | −0.372 | 0.134 |
| cy17:0 | Gram-negative bacteria | 0.512 | 0.548 | 0.594 * | −0.336 | 0.631 * |
| 18:1ω5c | Gram-negative bacteria | 0.706 * | 0.740 ** | 0.763 ** | −0.359 | 0.785 ** |
| 18:1ω7c | Gram-negative bacteria | 0.618 * | 0.646 * | 0.686 * | −0.020 | 0.706 * |
| 11Me18:1ω7c | Gram-negative bacteria | 0.666 * | 0.679 ** | 0.676 * | −0.077 | 0.674 * |
| 18:1ω9c | Fungi | −0.133 | −0.108 | −0.092 | 0.660 * | −0.109 |
| 18:2ω6,9c/18:0ante | Fungi | 0.740 ** | 0.784 ** | 0.730 ** | −0.490 | 0.715 ** |
| 16:1ω5c | Arbuscular mycorrhizal fungi | 0.558 | 0.592 * | 0.633 * | −0.322 | 0.666 * |
| 10Me16:0 | Actinomycetes | 0.661 * | 0.699 * | 0.728 ** | −0.336 | 0.754 ** |
| 10Me17:0 | Actinomycetes | 0.784 ** | 0.794 ** | 0.754 ** | −0.338 | 0.740 ** |
| 10Me18:0 | Actinomycetes | 0.598 * | 0.621 * | 0.625 * | −0.180 | 0.636 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, G.; Jin, M.; Cai, C.; Ma, C.; Chen, Z.; Gao, L. Soil Microbial Community Structure and Physicochemical Properties in Amomum tsaoko-based Agroforestry Systems in the Gaoligong Mountains, Southwest China. Sustainability 2019, 11, 546. https://doi.org/10.3390/su11020546
Liu G, Jin M, Cai C, Ma C, Chen Z, Gao L. Soil Microbial Community Structure and Physicochemical Properties in Amomum tsaoko-based Agroforestry Systems in the Gaoligong Mountains, Southwest China. Sustainability. 2019; 11(2):546. https://doi.org/10.3390/su11020546
Chicago/Turabian StyleLiu, Guizhou, Man Jin, Chuantao Cai, Chaonan Ma, Zhongsuzhi Chen, and Lunlun Gao. 2019. "Soil Microbial Community Structure and Physicochemical Properties in Amomum tsaoko-based Agroforestry Systems in the Gaoligong Mountains, Southwest China" Sustainability 11, no. 2: 546. https://doi.org/10.3390/su11020546
APA StyleLiu, G., Jin, M., Cai, C., Ma, C., Chen, Z., & Gao, L. (2019). Soil Microbial Community Structure and Physicochemical Properties in Amomum tsaoko-based Agroforestry Systems in the Gaoligong Mountains, Southwest China. Sustainability, 11(2), 546. https://doi.org/10.3390/su11020546





