Mineral Carbonation as an Educational Investigation of Green Chemical Engineering Design
Abstract
1. Introduction
Aims and Objectives
2. Experimental Investigation Part A: Background of Mineral Carbonation
2.1. Reaction Rate and Kinetics
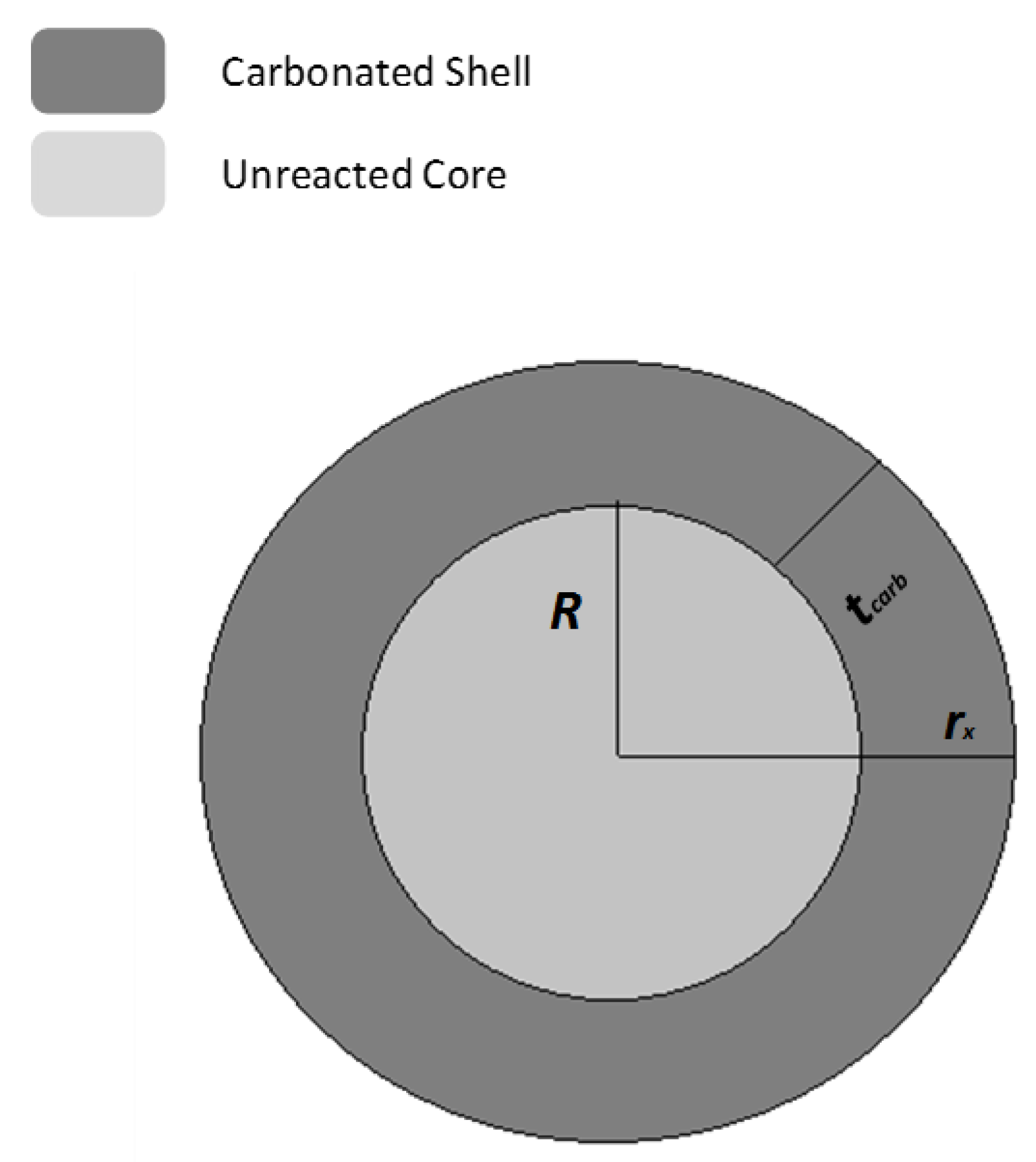
Reacted Particle Morphology
2.2. Effects of Process Parameters
2.2.1. Temperature and CO2 Pressure
2.2.2. Agitation
2.3. Determination of the Reaction Extent
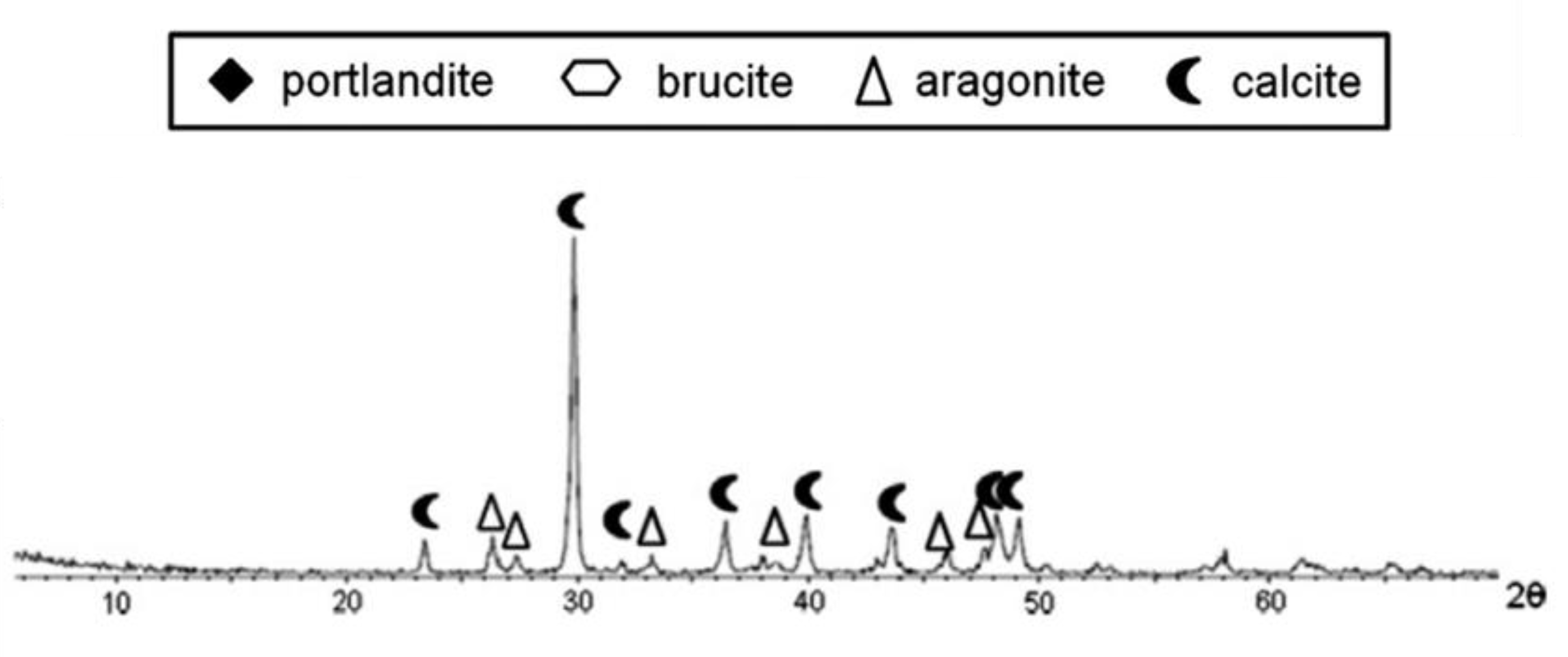
2.3.1. X-ray Diffraction (XRD)
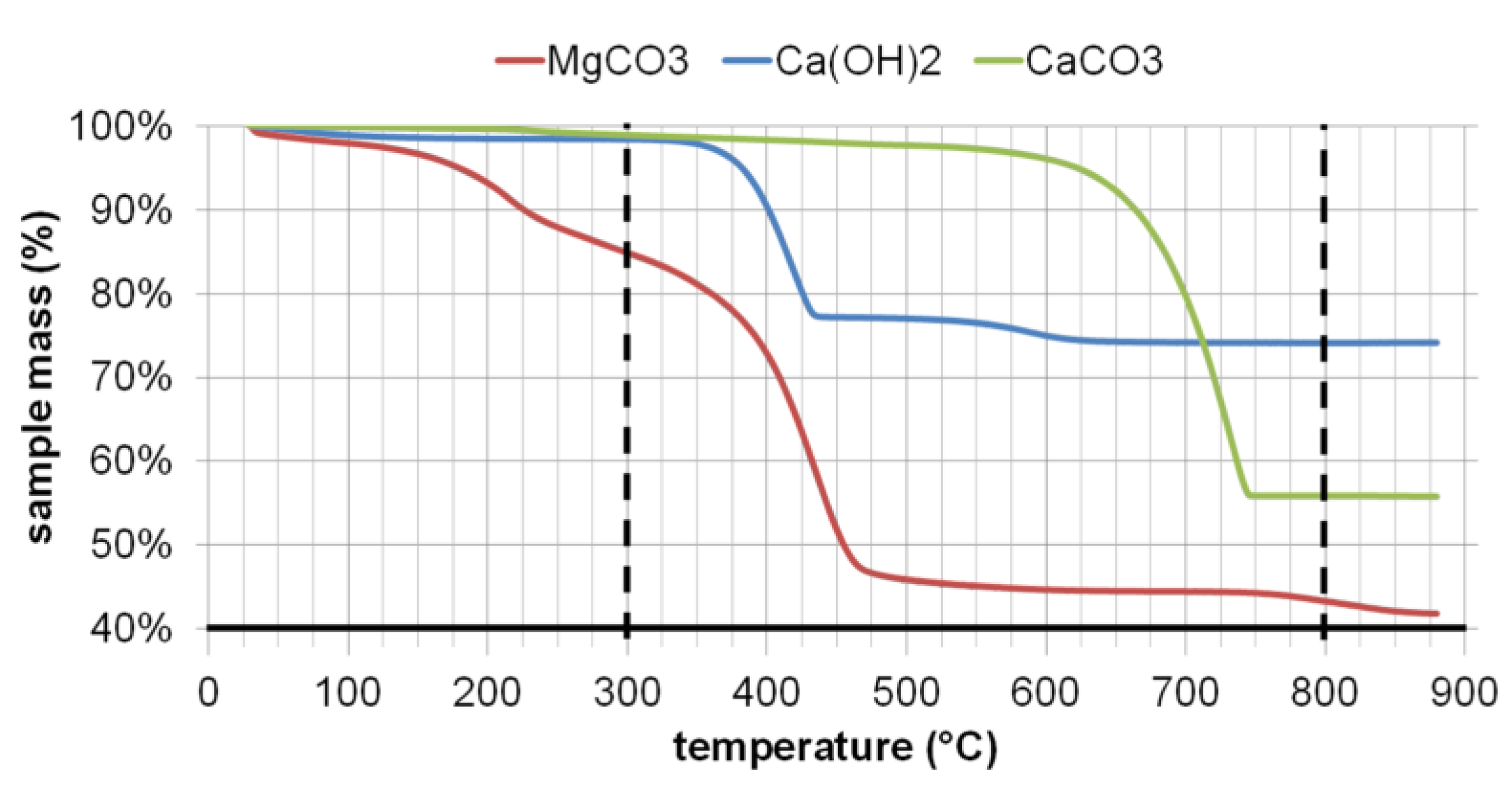
2.3.2. Thermogravimetric Analysis (TGA)
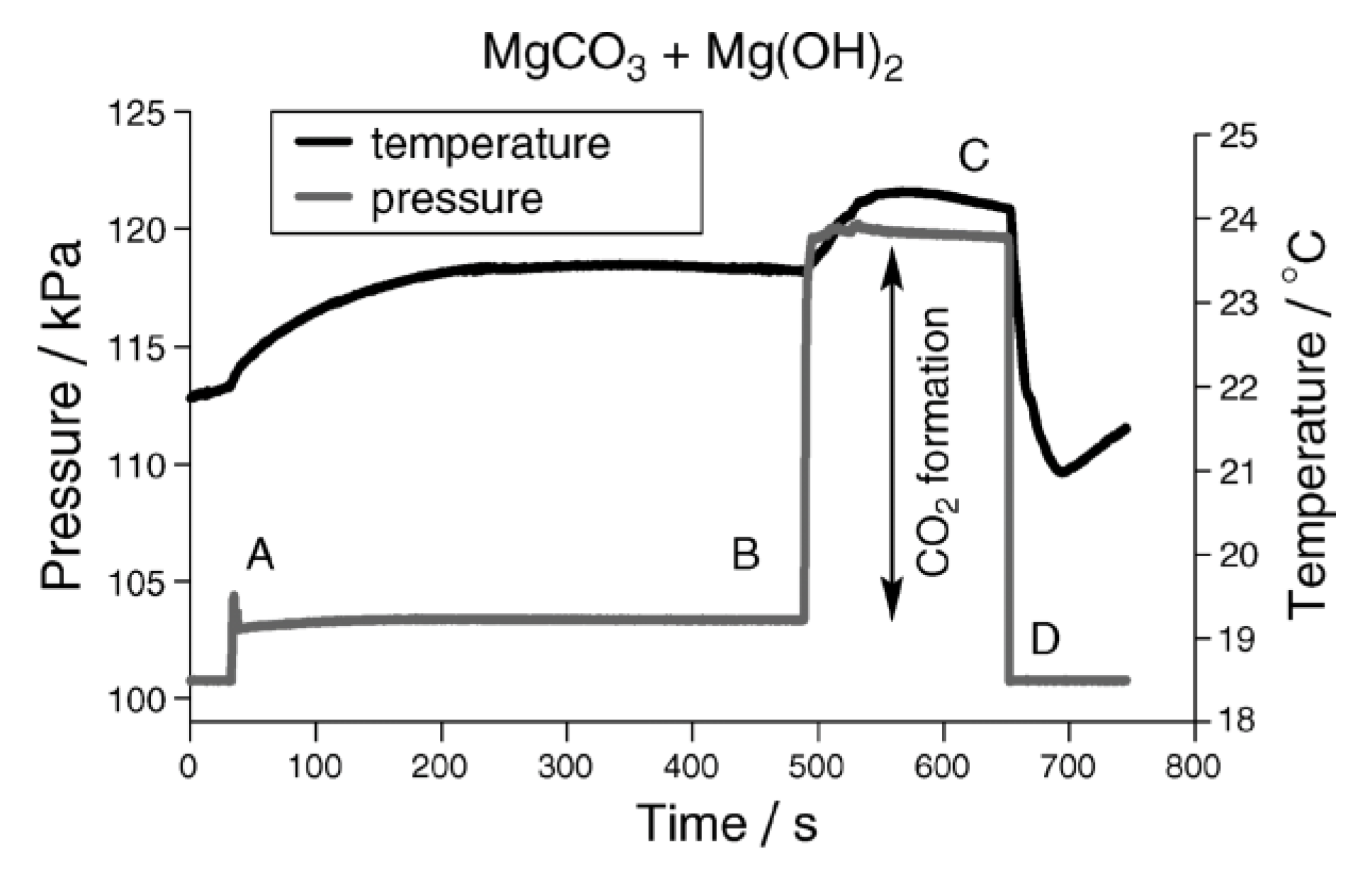
2.3.3. Calcimetry
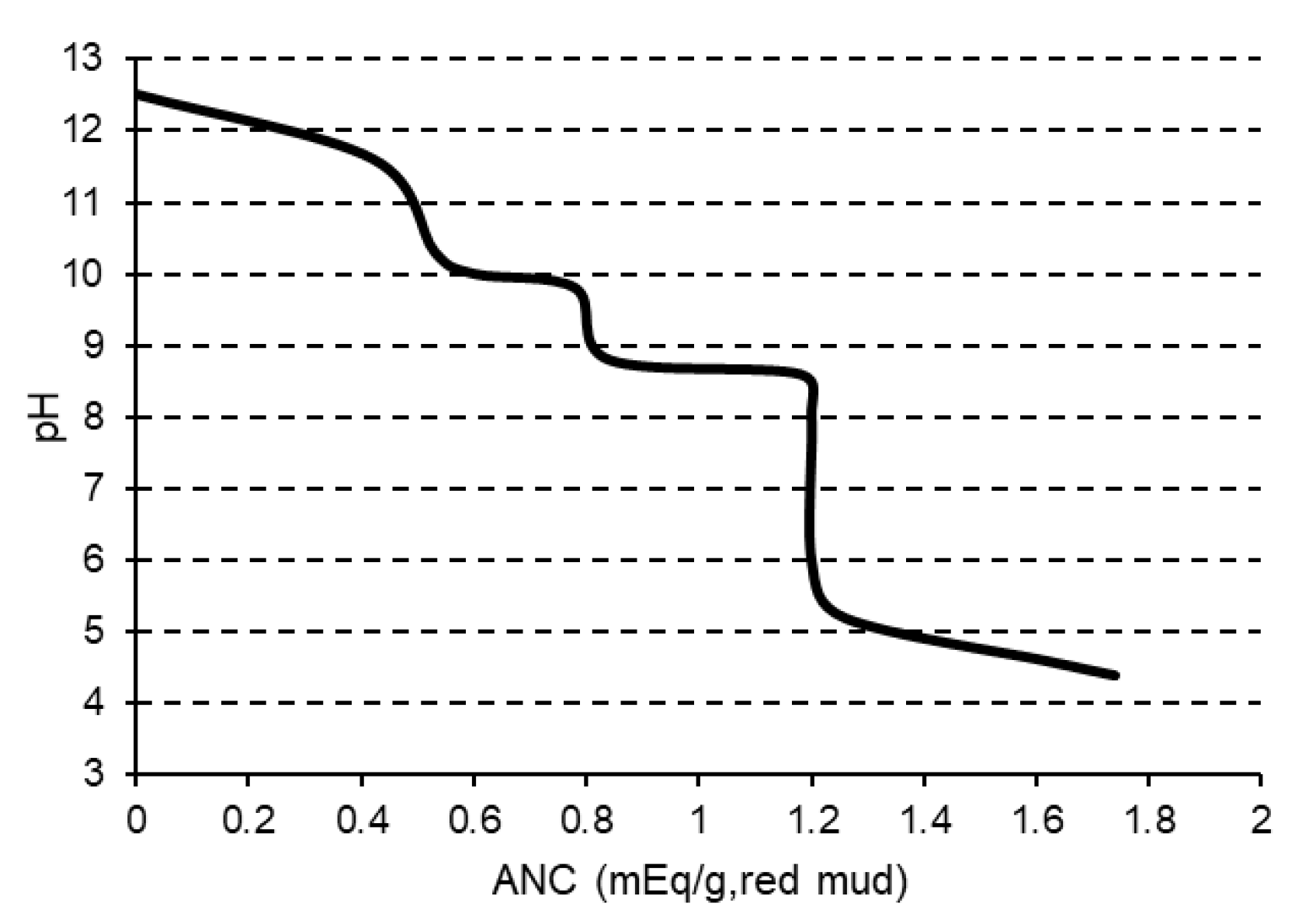
2.3.4. Acid-Base Titration
3. Experimental Investigation Part B: Materials and Methods
3.1. Mineral
3.2. Pressurized Reactor
3.3. Process Parameters
- The volume of water in the reactor: the stirrer blades were fixed to a certain height, so changing the amount of water would imply that mixing would become difficult if the water level became too low. Thus, a value that is well suited for the 1 L reactor was used: 800 mL H2O.
- Mass of solids in the reactor: decreasing the liquid-to-solid (L/S) mass ratio could help promote particle-particle interaction, which might aid in carbonation conversion by eroding the silica layer, but, at too low values, mixing becomes problematic. To ensure good mixing, a relatively high L/S ratio was used: 20 (i.e., 40 g wollastonite material).
- Residence time: the higher the residence time, the higher the carbonation conversion [45]. However, to study the effect of process parameters on reaction extent, differences were magnified at intermediate conversion values, so the residence time should be limited. The reaction residence time was fixed to 30 min.
- Temperature: wollastonite reportedly carbonates best when the temperature is sufficiently high to aid in calcium leaching but not so high that it reduces the solubility of CO2 too greatly [45]. The temperature choice also impacts the duration of start-up, since heating from room temperature to the reaction temperature takes time, and of shut-down, since the reactor must be sufficiently cooled before it can be depressurized (<100 °C) and before its contents can be drained safely (<80 °C). Hence the temperature to be used was 120 °C (at this temperature, the steam pressure was 0.974 bar, gauge).
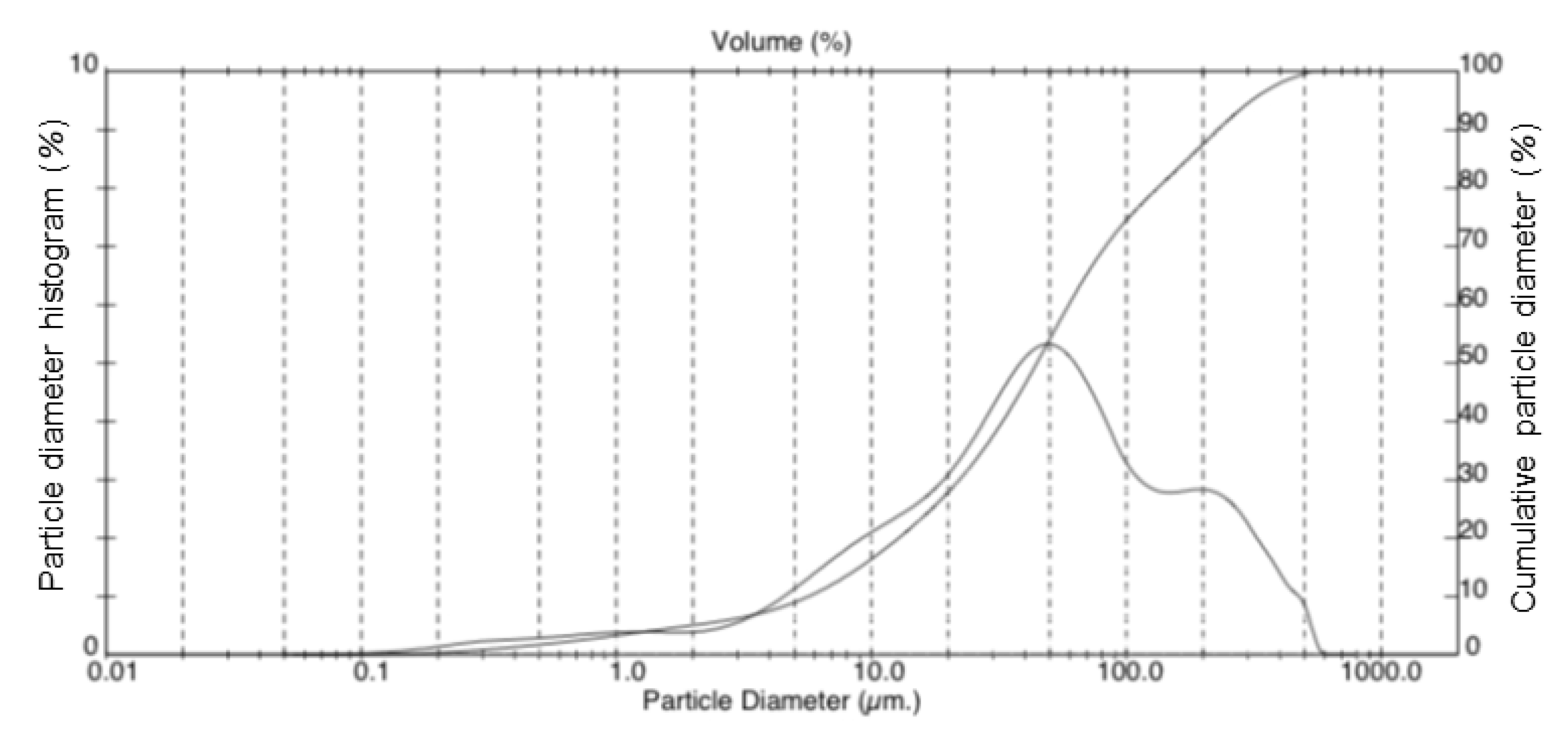
- Particle size: particle size is one of the most important parameters for mineral carbonation, as an increase in surface area (i.e., particle size reduction) contributes to the rate of carbonation (Equation (8)). However, reducing the particle size of mined minerals is costly, and the more milling is done, the lower the net CO2 sequestration for the process (the process can even become a net CO2 emitter). The material used in this project was milled by NYCO Minerals to the maximum extent that the company still deems economical for commercialization of the mineral; particle size data is presented in the Appendix. According to the results of Huijgen et al. [45], an average particle size below 100 µm is needed for satisfactory kinetics, which is the case for the present material. Hence, the material was used as is, and the initial particle size was constant for all experiments.
- CO2 gauge pressure (barg): 2, 5, 10, 40.
- Stirring rate (rpm): 100, 200, 400, 600.
3.4. Experimental Procedure
4. Experimental Investigation Part C: Calculations and Questions
4.1. Moisture Content in Collected Filter Cake
4.2. CO2 Content in Fresh and Carbonated Wollastonite
4.3. Laboratory Report Questions
- Present all experimental data in well-formatted lists and/or tables (in addition to the photocopy of your lab notebook notes).
- Present answers to the calculation questions and show calculations.
- Comment on your confidence in the acidification results for the original and carbonated solids, and make any additional remark about using this method for CO2 uptake determination.
- Comment on the carbonation extent achieved. Is it as expected based on the theory on process conditions and mechanism?
- How can the reaction progress be followed in real-time (on-line)? Research and summarize at least two instrumental methods that can be coupled to a pressurized high-temperature reactor.
- Would it make any difference to the reaction if impure CO2 was used (e.g. direct flue gas, or CO2 of lower purity than used)? Think about chemical and physical aspects.
- Explain the function of a rupture disk and how it is selected for a reactor. In what cases is a rupture disk used versus a relief valve?
- Research literature to find out what the pH and CO2 solubility (mol/L) at the operating temperature and pressure you used would be if the reaction medium was pure water (i.e. no mineral).
- Following up on question 8, do you think the pH and CO2 solubility with the reacting wollastonite present (as in the experiment) would be higher or lower than the values you found for pure water?
5. Discussion
- Q3)
- Students commented on the increase in the CO2 content of wollastonite after reaction versus the original mineral. Comments were also made regarding possible vapor losses from the acidification tube due to heating, and water condensation into the tube due to an ice bath, as potential sources of error in the quantitative determination.
- Q5)
- Two approaches to follow reaction progress in real-time were commonly mentioned. One is to use instrumentation that analyzes the reaction medium in-situ, such as in-situ FTIR. The other is by collecting samples while the reactor operates at elevated temperatures and pressures, using appropriate sampling techniques.
- Q6)
- Students recalled the concepts of partial and total pressures to answer this question. They realized that it is the CO2 partial pressure that drives the carbonation reaction, thus impure CO2, in the form of flue gases, would require greater total pressure to achieve similar reactivity as pure CO2. From a physical perspective, the implication of this includes the need for reactors that can withstand higher pressures and compressors that can pressure flue gases to very high pressures, both of which affect process costing.
- Q7)
- This question about rupture disks and relief valves can easily be answered by searching the internet, but it was selected for this lab for a special purpose. Sheridan students perform another lab experiment using a climbing film evaporator, which uses a boiler that has a pressure relief valve. Thus answering this question enabled the students to recall that experiential learning event, which is a pedagogical approach to reinforce learned concepts, thus making the experience of researching the answer to this question more engaging.
- Q8)
- The answer to this question comes from published literature and can be most readily be found in journal articles. This question is thus designed to expose students to journal literature, and an example of a paper that students found to answer this question is that of Duan and Sun [63].
- Q9)
- Students analyzed the effect of wollastonite on CO2 solubility, and consequently pH, by looking at chemical equilibrium equations. They verified that wollastonite consumes acidity, thus shifts the carbonic acid equilibria towards the greater dissolution of CO2 and increases the pH.
5.1. Energetic Analysis and Design Project
- Energetic analysis: (a) explain how you adapted the proposed procedure to reach the energy consumption and net CO2 sequestration results (what assumptions had to be made, what changes you made, if any, to the proposed procedure, etc.); (b) show values (and sources) of all parameters used in the equations; (c) show the energy consumption value of each individual process/equipment; (d) show the calculation procedure of converting energy consumption to CO2 emissions (footprint) and compare the CO2 emissions rate to the CO2 sequestration rate to determine if net CO2 sequestration is achieved; (e) discuss what process changes you may make, if more experimental or modeling information, to improve the efficiency of the process; (f) write conclusions and recommendations on whether accelerated mineral carbonation can become an industrially feasible process for CO2 sequestration.
- Reaction rate law: (a) explain the procedure used to develop the rate law(s), including data and graphs used; (b) explain assumptions taken and any limitation of the applicability of the rate law(s); (c) comment on what you learned about the mineral carbonation reaction based on the rate law(s) obtained; (d) comment on how the rate law(s) can aid in improving the process design and energy consumption presented earlier.
- Mass transfer analysis: (a) explain the procedure used in the analysis, including data and graphs used; (b) explain assumptions taken and any limitation on validity of the analysis results; (c) comment on what you learned about the mineral carbonation reaction based on the results obtained; (d) comment on how the analysis can aid in improving the process design and energy consumption presented earlier.
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
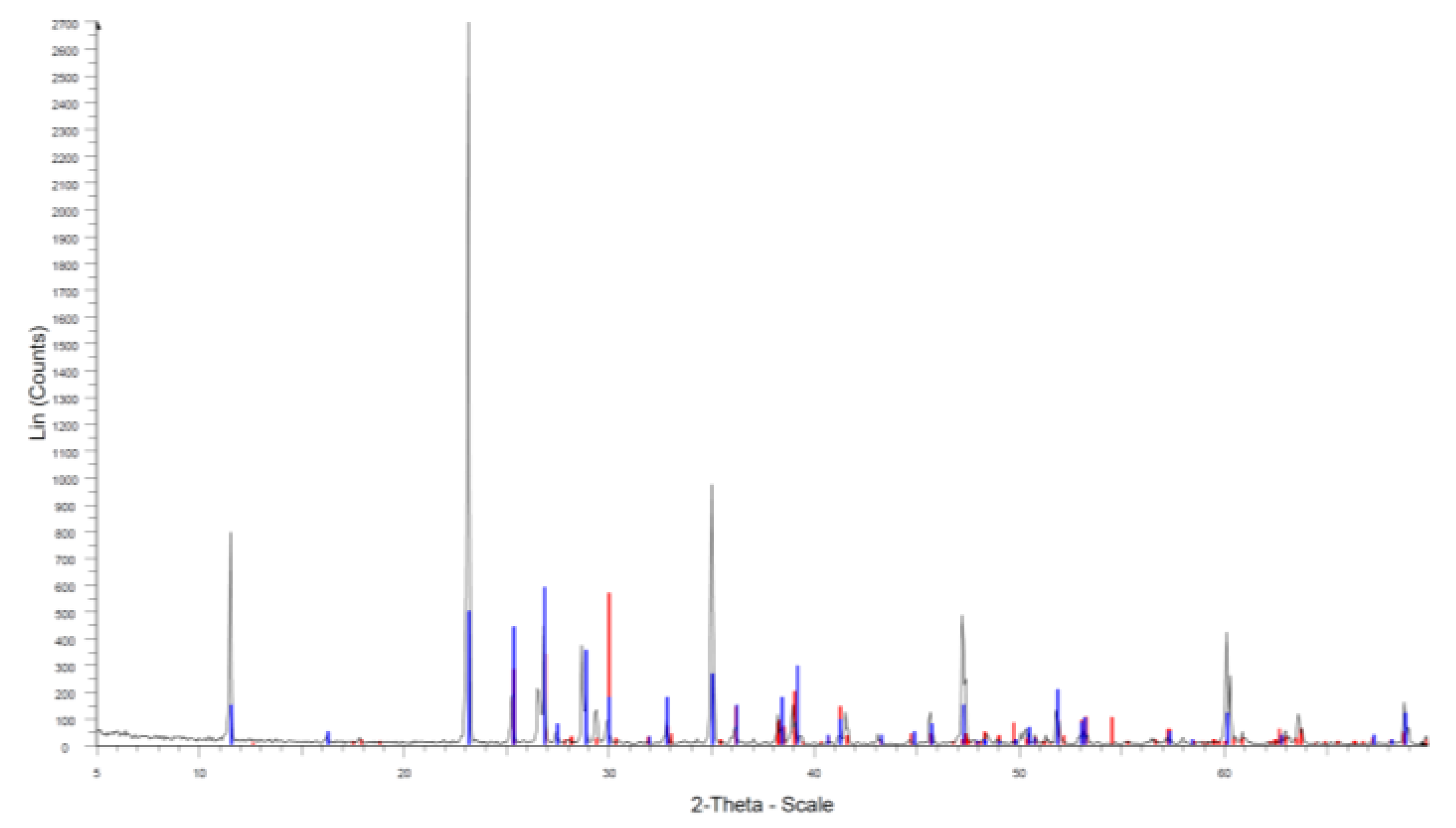
References
- UNESCO. United nations educational, scientific and cultural organization: Educating for a sustainable future: A transdisciplinary vision for concerted action. In Proceedings of the International Conference on Environment and Society: Education and Public Awareness for Sustainability, Thessalonika, Greece, 8–12 December 1997. [Google Scholar]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice, 12 Principles of Green Chemistry; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Bastin, L.D.; Dicks, A.P. Advances in green chemistry education. Green Chem. Lett. Rev. 2019, 12, 101. [Google Scholar] [CrossRef]
- Summerton, L.; Hurst, G.A.; Clark, J.H. Facilitating active learning within green chemistry. Curr. Opin. Green Sustain. Chem. 2018, 13, 56–60. [Google Scholar] [CrossRef]
- Dicks, A.P. Teaching reaction efficiency through the lens of green chemistry: Should students focus on the yield, or the process? Curr. Opin. Green Sustain. Chem. 2018, 13, 27–31. [Google Scholar] [CrossRef]
- Dym, C.L.; Agogino, A.M.; Eris, O.; Frey, D.D.; Leifer, L.J. Engineering Design Thinking, Teaching, and Learning. J. Eng. Educ. 2005, 94, 103–120. [Google Scholar] [CrossRef]
- Eilks, I.; Byers, B. (Eds.) Innovative Methods of Teaching and Learning Chemistry in Higher Education; RSC Publishing: Cambridge, UK, 2009. [Google Scholar]
- Feisel, L.D.; Rosa, A.J. The Role of the Laboratory in Undergraduate Engineering Education. J. Eng. Educ. 2005, 94, 121–130. [Google Scholar] [CrossRef]
- Martínez, K.P.; Campos, J.L.B. Developing scientific thinking skills through teaching chemical reaction with inquiry based teaching. Educ. Quím. 2019, 30, 93. [Google Scholar] [CrossRef]
- McKim, A.S. Overcoming sustainability barriers within the chemical industry. Curr. Opin. Green Sustain. Chem. 2018, 14, 10–13. [Google Scholar] [CrossRef]
- Parmentier, M.; Gabriel, C.M.; Guo, P.; Isley, N.A.; Zhou, J.; Gallou, F. Switching from organic solvents to water at an industrial scale. Curr. Opin. Green Sustain. Chem. 2017, 7, 13–17. [Google Scholar] [CrossRef]
- Metz, B.; Davidson, O.; de Coninck, H.; Loos, M.; Meyer, L. (Eds.) IPCC Special Report on Carbon Dioxide Capture and Storage; Cambridge University Press: New York, NY, USA, 2005. [Google Scholar]
- Oelkers, E.H.; Gislason, S.R.; Matter, J. Mineral Carbonation of CO2. Elements 2008, 4, 333–337. [Google Scholar] [CrossRef]
- Saran, R.K.; Arora, V.; Yadav, S. CO2 sequestration by mineral carbonation: A review. Glob. Nest J. 2018, 20, 497–503. [Google Scholar] [CrossRef]
- Baena-Moreno, F.M.; Rodríguez-Galán, M.; Vega, F.; Alonso-Fariñas, B.; Vilches Arenas, L.F.; Navarrete, B. Carbon capture and utilization technologies: A literature review and recent advances. Energy Sources, Part A Recover. Util. Environ. Eff. 2019, 41, 1403–1433. [Google Scholar] [CrossRef]
- Kelektsoglou, K. Carbon capture and storage: A review of mineral storage of CO2 in Greece. Sustainability 2018, 10, 4400. [Google Scholar] [CrossRef]
- National Oceanic and Atmospheric Administration (NOAA). Available online: https://www.noaa.gov (accessed on 18 May 2019).
- The Intergovernmental Panel on Climate Change (IPCC). Summary for Policymakers. In Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Edenhofer, O., Pichs-Madruga, R., Sokona, Y., Farahani, E., Kadner, S., Seyboth, K., Adler, A., Baum, I., Brunner, S., Eickemeier, P., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014. [Google Scholar]
- Georgakopoulos, E.; Santos, R.M.; Chiang, Y.W.; Manovic, V. Two-way Valorization of Blast Furnace Slag: Synthesis of Precipitated Calcium Carbonate and Zeolitic Heavy Metal Adsorbent. J. Vis. Exp. 2017, 120, e55062. [Google Scholar] [CrossRef] [PubMed]
- Azadi, M.; Edraki, M.; Farhang, F.; Ahn, J. Opportunities for mineral carbonation in Australia’s mining industry. Sustainability 2019, 11, 1250. [Google Scholar] [CrossRef]
- Ibrahim, M.; El-Naas, M.; Benamor, A.; Al-Sobhi, S.; Zhang, Z. Carbon Mineralization by Reaction with Steel-Making Waste: A Review. Processes 2019, 7, 115. [Google Scholar] [CrossRef]
- Dindi, A.; Quang, D.V.; Vega, L.F.; Nashef, E.; Abu-Zahra, M.R.M. Applications of fly ash for CO2 capture, utilization, and storage. J. CO2 Util. 2019, 29, 82–102. [Google Scholar] [CrossRef]
- Carbon cure Technologies. Available online: https://www.carboncure.com/ (accessed on 28 June 2019).
- Carbon8 Systems. Available online: http://c8s.co.uk/ (accessed on 28 June 2019).
- Mineral Carbonation International. Available online: https://www.mineralcarbonation.com/ (accessed on 28 June 2019).
- Solidia Technologies. Available online: https://solidiatech.com/ (accessed on 28 June 2019).
- Olajire, A.A. A review of mineral carbonation technology in sequestration of CO2. J. Pet. Sci. Eng. 2013, 109, 364–392. [Google Scholar] [CrossRef]
- Li, J.; Jacobs, A.D.; Hitch, M. Direct aqueous carbonation on olivine at a CO2 partial pressure of 6.5 MPa. Energy 2019, 173, 902–910. [Google Scholar] [CrossRef]
- Farhang, F.; Oliver, T.K.; Rayson, M.S.; Brent, G.F.; Molloy, T.S.; Stockenhuber, M.; Kennedy, E.M. Dissolution of heat activated serpentine for CO2 sequestration: The effect of silica precipitation at different temperature and pH values. J. CO2 Util. 2019, 30, 123–129. [Google Scholar] [CrossRef]
- Haque, F.; Santos, R.M.; Dutta, A.; Thimmanagari, M.; Chiang, Y.W. Co-Benefits of Wollastonite Weathering in Agriculture: CO2 Sequestration and Promoted Plant Growth. ACS Omega 2019, 4, 1425–1433. [Google Scholar] [CrossRef]
- Stopic, S.; Dertmann, C.; Modolo, G.; Kegler, P.; Neumeier, S.; Kremer, D.; Wotruba, H.; Etzold, S.; Telle, R.; Rosani, D.; et al. Synthesis of Magnesium Carbonate via Carbonation under High Pressure in an Autoclave. Metals 2018, 8, 993. [Google Scholar] [CrossRef]
- Nowamooz, A.; Dupuis, J.C.; Beaudoin, G.; Molson, J.; Lemieux, J.M.; Horswill, M.; Fortier, R.; Larachi, F.; Maldague, X.; Constantin, M.; et al. Atmospheric Carbon Mineralization in an Industrial-Scale Chrysotile Mining Waste Pile. Environ. Sci. Technol. 2018, 52, 8050–8057. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Lee, H.-K. Mineral Sequestration of Carbon Dioxide in Circulating Fluidized Bed Combustion Boiler Bottom Ash. Minerals 2017, 7, 237. [Google Scholar] [CrossRef]
- Haque, F.; Chiang, E.; Santos, R.M. Alkaline Mineral Soil Amendment: A Climate Change ‘Stabilization Wedge’? Energies 2019, 12, 2299. [Google Scholar] [CrossRef]
- Casiflux F Technical Data; Sibelco Specialty Minerals Europe: Antwerp, Belgium, 2011.
- Daval, D.; Martinez, I.; Corvisier, J.; Findling, N.; Goffé, B.; Guyot, F. Carbonation of Ca-bearing silicates, the case of wollastonite: Experimental investigations and kinetic modeling. Chem. Geol. 2009, 265, 63–78. [Google Scholar] [CrossRef]
- Liu, W.; Song, L.; Xu, C.; Rohani, S.; Chen, M.; Liang, B.; Li, C. Combined synthesis of Li4SiO4 sorbent with high CO2 uptake in the indirect carbonation of blast furnace slag process. Chem. Eng. J. 2019, 370, 71–80. [Google Scholar] [CrossRef]
- Rashid, M.I.; Benhelal, E.; Farhang, F.; Oliver, T.K.; Rayson, M.S.; Brent, G.F.; Stockenhuber, M.; Kennedy, E.M. Development of Concurrent grinding for application in aqueous mineral carbonation. J. Clean. Prod. 2019, 212, 151–161. [Google Scholar] [CrossRef]
- Rigopoulos, I.; Ioannou, I.; Delimitis, A.; Efstathiou, A.; Kyratsi, T. Ball Milling Effect on the CO2 Uptake of Mafic and Ultramafic Rocks: A Review. Geosciences 2018, 8, 406. [Google Scholar] [CrossRef]
- Wang, F.; Dreisinger, D.B.; Jarvis, M.; Hitchins, T. The technology of CO2 sequestration by mineral carbonation: Current status and future prospects. Can. Metall. Q. 2018, 57, 46–58. [Google Scholar] [CrossRef]
- Dudhaiya, A.; Santos, R.M. How Characterization of Particle Size Distribution Pre- and Post-Reaction Provides Mechanistic Insights into Mineral Carbonation. Geosciences 2018, 8, 260. [Google Scholar] [CrossRef]
- Ptáček, P.; Nosková, M.; Brandštetr, J.; Šoukal, F.; Opravil, T. Mechanism and kinetics of wollastonite fibre dissolution in the aqueous solution of acetic acid. Powder Technol. 2011, 206, 338–344. [Google Scholar] [CrossRef]
- Georgakopoulos, E.; Santos, R.M.; Chiang, Y.W.; Manovic, V. Influence of process parameters on carbonation rate and conversion of steelmaking slags—Introduction of the ‘carbonation weathering rate’. Greenhouse Gases Sci. Technol. 2016, 6, 470–491. [Google Scholar] [CrossRef]
- Daval, D.; Martinez, I.; Guigner, J.-M.; Hellmann, R.; Corvisier, J.; Findling, N.; Dominici, C.; Goffé, B.; Guyot, F. Mechanism of wollastonite carbonation deduced from micro- to nanometer length scale observations. Am. Mineral. 2009, 94, 1707–1726. [Google Scholar] [CrossRef]
- Huijgen, W.J.J.; Witkamp, G.J.; Comans, R.N.J. Mechanisms of aqueous wollastonite carbonation as a possible CO2 sequestration process. Chem. Eng. Sci. 2006, 64, 4242–4251. [Google Scholar] [CrossRef]
- Peuble, S.; Godard, M.; Gouze, P.; Leprovost, R.; Martinez, I.; Shilobreeva, S. Control of CO2 on flow and reaction paths in olivine-dominated basements: An experimental study. Geochim. Cosmochim. Acta 2019, 252, 16–38. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, C.J.; Jiang, M.F.; Saxén, H.; Zevenhoven, R. Preparation of magnesium hydroxide from serpentinite by sulfuric acid leaching for CO2 mineral carbonation. Miner. Eng. 2015, 79, 116–124. [Google Scholar] [CrossRef]
- Abu Fara, A.; Rayson, M.R.; Brent, G.F.; Oliver, T.K.; Stockenhuber, M.; Kennedy, E.M. Formation of magnesite and hydromagnesite from direct aqueous carbonation of thermally activated lizardite. Environ. Prog. Sustain. Energy 2019. [Google Scholar] [CrossRef]
- Helgeson, H.C. Kinetics of mass transfer among silicates and aqueous solutions. Geochim. Cosmochim. Acta 1971, 35, 421–469. [Google Scholar] [CrossRef]
- Bodor, M.; Santos, R.M.; Kriskova, L.; Elsen, J.; Vlad, M.; Van Gerven, T. Susceptibility of mineral phases of steel slags towards carbonation: Mineralogical, morphological and chemical assessment. Eur. J. Mineral. 2013, 25, 533–549. [Google Scholar] [CrossRef]
- Santos, R.M.; Van Bouwel, J.; Vandevelde, E.; Mertens, G.; Elsen, J.; Van Gerven, T. Accelerated mineral carbonation of stainless steel slags for CO2 storage and waste valorization: Effect of process parameters on geochemical properties. Int. J. Greenhouse Gas Control 2013, 17, 32–45. [Google Scholar] [CrossRef]
- Santos, R.M.; Thijs, J.; Georgakopoulos, E.; Chiang, Y.W.; Creemers, A.; Van Gerven, T. Improving the Yield of Sonochemical Precipitated Aragonite Synthesis by Scaling up Intensified Conditions. Chem. Eng. Commun. 2016, 203, 1671–1680. [Google Scholar] [CrossRef][Green Version]
- Faust, G.T. Thermal analysis studies on carbonates I. aragonite and calcite. Am. Mineral. 1950, 35, 207–224. [Google Scholar]
- Santos, R.M.; Bodor, M.; Dragomir, P.N.; Vraciu, A.; Vlad, M.; Gerven, T.V. Magnesium chloride as a leaching and aragonite-promoting self-regenerative additive for the mineral carbonation of calcium-rich materials. Miner. Eng. 2014, 59, 71–81. [Google Scholar] [CrossRef]
- Santos, R.M.; Ling, D.; Sarvaramini, A.; Guo, M.; Elsen, J.; Larachi, F.; Beaudoin, G.; Blanpain, B.; Van Gerven, T. Stabilization of basic oxygen furnace slag by hot-stage carbonation treatment. Chem. Eng. J. 2012, 203, 239–250. [Google Scholar] [CrossRef]
- Apesteguia, M.; Plante, A.F.; Virto, I. Methods assessment for organic and inorganic carbon quantification in calcareous soils of the Mediterranean region. Geoderma Reg. 2018, 12, 39–48. [Google Scholar] [CrossRef]
- Fonnesbeck, B.B.; Boettinger, J.L.; Lawley, J.R. Improving a Simple Pressure-Calcimeter Method for Inorganic Carbon Analysis. Soil Sci. Soc. Am. J. 2013, 77, 1553–1562. [Google Scholar] [CrossRef]
- Rajesh, R.; Brindha, K.; Elango, L. Groundwater Quality and its Hydrochemical Characteristics in a Shallow Weathered Rock Aquifer of Southern India. Water Qual. Expo. Heal. 2015, 7, 515–524. [Google Scholar] [CrossRef]
- Harvey, D. Section 9.2: Acid-Base Titrations. Modern Analytical Chemistry 2.0. Available online: http://dpuadweb.depauw.edu/harvey_web/eTextProject/pdfFiles/AnalChem2.0.pdf (accessed on 25 July 2019).
- Fagerlund, J.; Son, M. Gasometric determination of CO2 released from carbonate materials. J. Chem. Educ. 2010, 87, 1372–1376. [Google Scholar] [CrossRef]
- Bajpai, P. Pulping Calculations. In Biermann’s Handbook of Pulp and Paper, 3rd ed; Elsevier: Dehradun, India, 2018; Volume 1, pp. 353–373. [Google Scholar]
- Khaitan, S.; Dzombak, D.A.; Lowry, G.V. Chemistry of the Acid Neutralization Capacity of Bauxite Residue. Environ. Eng. Sci. 2009, 26, 873–881. [Google Scholar] [CrossRef]
- Duan, Z.; Sun, R. An improved model calculating CO2 solubility in pure water and aqueous NaCl solutions from 273 to 533 K and from 0 to 2000 bar. Chem. Geol. 2003, 193, 257–271. [Google Scholar] [CrossRef]
- Santos, R.M.; Van Audenaerde, A.; Chiang, Y.W.; Iacobescu, R.I.; Knops, P.; Van Gerven, T. Nickel Extraction from Olivine: Effect of Carbonation Pre-Treatment. Metals 2015, 5, 1620–1644. [Google Scholar] [CrossRef]
- Moomaw, W.; Burgherr, P.; Heath, G.; Lenzen, M.; Nyboer, J.; Verbruggen, A. Annex II: Methodology. In IPCC Special Report on Renewable Energy Sources and Climate Change Mitigation; Edenhofer, O., Pichs-Madruga, R., Sokona, Y., Seyboth, K., Matschoss, P., Kadner, S., Zwickel, T., Eickemeier, P., Hansen, G., Schlömer, S., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2011. [Google Scholar]
- Santos, R.M.; Francois, D.; Mertens, G.; Elsen, J.; Van Gerven, T. Ultrasound-intensified mineral carbonation. Appl. Therm. Eng. 2013, 57, 154–163. [Google Scholar] [CrossRef]
- Costa, G.; Polettini, A.; Pomi, R.; Stramazzo, A.; Zingaretti, D. Energetic assessment of CO2 sequestration through slurry carbonation of steel slag: A factorial study. Greenhouse Gases Sci. Technol. 2016, 7, 530–541. [Google Scholar] [CrossRef]




| Oxide | Wt % |
|---|---|
| CaO | 51.2 ± 0.6 |
| SiO2 | 46.4 ± 0.3 |
| Al2O3 | 0.79 ± 0.04 |
| MgO | 0.70 ± 0.05 |
| Fe2O3 | 0.52 ± 0.06 |
| K2O | 0.14 ± 0.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fantucci, H.; Sidhu, J.S.; Santos, R.M. Mineral Carbonation as an Educational Investigation of Green Chemical Engineering Design. Sustainability 2019, 11, 4156. https://doi.org/10.3390/su11154156
Fantucci H, Sidhu JS, Santos RM. Mineral Carbonation as an Educational Investigation of Green Chemical Engineering Design. Sustainability. 2019; 11(15):4156. https://doi.org/10.3390/su11154156
Chicago/Turabian StyleFantucci, Hugo, Jaspreet S. Sidhu, and Rafael M. Santos. 2019. "Mineral Carbonation as an Educational Investigation of Green Chemical Engineering Design" Sustainability 11, no. 15: 4156. https://doi.org/10.3390/su11154156
APA StyleFantucci, H., Sidhu, J. S., & Santos, R. M. (2019). Mineral Carbonation as an Educational Investigation of Green Chemical Engineering Design. Sustainability, 11(15), 4156. https://doi.org/10.3390/su11154156






