Sustainability of Pork Production with Immunocastration in Europe
Abstract
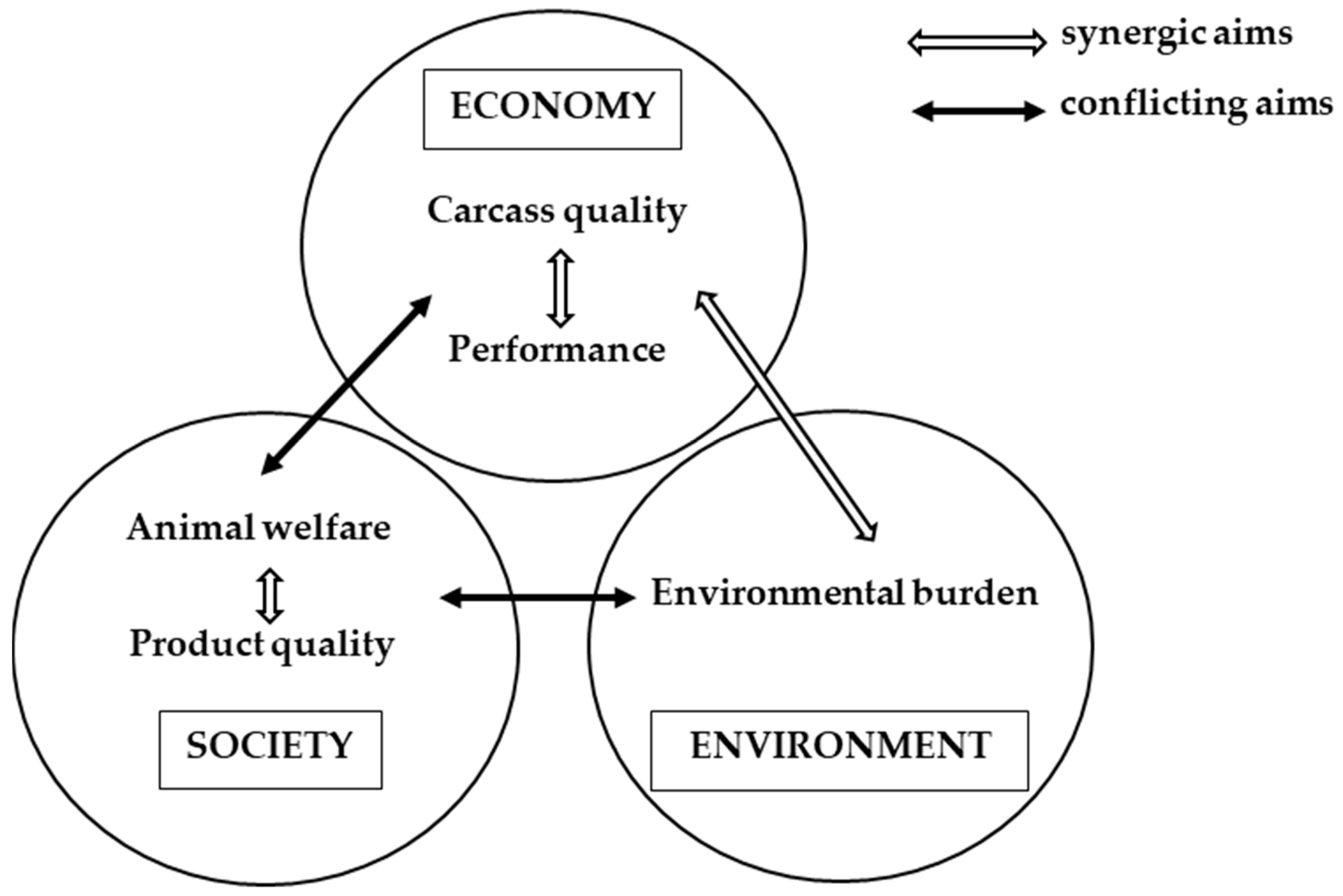
1. Introduction: The Castration Dilemma in Pork Production
2. The Principle of Immunocastration
3. Potential of Immunocastration for Sustainable Pork Production
3.1. On-Farm Application of Immunocastration
3.2. Reliability of Immunocastration
3.3. Consequences of Immunocastration for Animal Welfare, Behavior, and Health in Pork Production
3.4. Consequences of Immunocastration for Growth Performance, Carcass, and Meat Quality
3.5. Suitability of Immunocastration for Alternative and High Quality Production Systems
3.6. Consequences of Immunocastration for Feeding Requirements
3.7. Consequences of Immunocastration for the Environmental Burden
3.8. Consequences of Immunocastration for Economy
3.9. Societal Concerns and Immunocastration
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Krystallis, A.; de Barcellos, M.D.; Kügler, J.O.; Verbeke, W.; Grunert, K.G. Attitudes of European citizens towards pig production systems. Livest. Sci. 2009, 126, 46–56. [Google Scholar] [CrossRef]
- Van Loo, E.J.; Caputo, V.; Nayga, R.M.; Verbeke, W. Consumers’ valuation of sustainability labels on meat. Food Policy 2014, 49, 137–150. [Google Scholar] [CrossRef]
- Vanhonacker, F.; Verbeke, W. Public and Consumer Policies for Higher Welfare Food Products: Challenges and Opportunities. J. Agric. Environ. Eth. 2014, 27, 153–171. [Google Scholar] [CrossRef]
- Ingenbleek, P.T.M.; Immink, V.M.; Spoolder, H.A.M.; Bokma, M.H.; Keeling, L.J. EU animal welfare policy: Developing a comprehensive policy framework. Food Policy 2012, 37, 690–699. [Google Scholar] [CrossRef]
- EFSA. Opinion of the Scientific Panel on Animal Health and Welfare on a Request From the Commission Related to Welfare Aspects of the Castration of Piglets. Available online: https://www.efsa.europa.eu/en/efsajournal/pub/91 (accessed on 15 June 2019).
- Prunier, A.; Bonneau, M.; Von Borell, E.H.; Cinotti, S.; Gunn, M.; Fredriksen, B.; Giersing, M.; Morton, D.B.; Tuyttens, F.A.M.; Velarde, A. A review of the welfare consequences of surgical castration in piglets and the evaluation of non-surgical methods. Anim. Welf. 2006, 15, 277–289. [Google Scholar]
- Rault, J.-L.; Lay, D.C.; Marchant-Forde, J.N. Castration induced pain in pigs and other livestock. Appl. Anim. Behav. Sci. 2011, 135, 214–225. [Google Scholar] [CrossRef]
- Zamaratskaia, G.; Rasmussen, M.K. Immunocastration of Male Pigs–Situation Today. Procedia Food Sci. 2015, 5, 324–327. [Google Scholar] [CrossRef]
- Weiler, U.; Stefanski, V.; Von Borell, E. Die Kastration beim Schwein–Zielkonflikte und Lösungsansätze aus der Sicht des Tierschutzes. Züchtungskunde 2016, 88, 429–444. [Google Scholar]
- Čandek-Potokar, M.; Škrlep, M.; Zamaratskaia, G. Immunocastration as Alternative to Surgical Castration in Pigs. Theriogenology 2017, 6, 109–126. [Google Scholar] [CrossRef]
- von Borell, E.; Baumgartner, J.; Giersing, M.; Jäggin, N.; Prunier, A.; Tuyttens, F.A.M.; Edwards, S.A. Animal welfare implications of surgical castration and its alternatives in pigs. Animal 2009, 3, 1488–1496. [Google Scholar] [CrossRef]
- Bonneau, M. Use of entire males for pig meat in the European Union. Meat Sci. 1998, 49, 257–272. [Google Scholar] [CrossRef]
- Claus, R.; Weiler, U.; Herzog, A. Physiological aspects of androstenone and skatole formation in the boar—A review with experimental data. Meat Sci. 1994, 38, 289–305. [Google Scholar] [CrossRef]
- Wesoly, R.; Weiler, U. Nutritional Influences on Skatole Formation and Skatole Metabolism in the Pig. Animals 2012, 2, 221–242. [Google Scholar] [CrossRef]
- Kojima, M.; Degawa, M. Serum androgen level is determined by autosomal dominant inheritance and regulates sex-related CYP genes in pigs. Biochem. Biophys. Res. Commun. 2013, 430, 833–838. [Google Scholar] [CrossRef]
- Walstra, P.; Claudi-Magnussen, C.; Chevillon, P.; von Seth, G.; Diestre, A.; Matthews, K.R.; Homer, D.B.; Bonneau, M. An international study on the importance of androstenone and skatole for boar taint: Levels of androstenone and skatole by country and season. Livest. Prod. Sci. 1999, 62, 15–28. [Google Scholar] [CrossRef]
- Weiler, U.; Fischer, K.; Kemmer, H.; Dobrowolski, A.; Claus, R. Influence of androstenone sensitivity on consumer reactions to boar meat. In Boar Taint in Entire Male Pigs; Bonneau, M., Lundström, K., Malmfors, B., Eds.; EAAP Publication: Roma, Italy, 1998; Volume 92, pp. 147–151. [Google Scholar]
- Lunde, K.; Egelandsdal, B.; Skuterud, E.; Mainland, J.D.; Lea, T.; Hersleth, M.; Matsunami, H. Genetic Variation of an Odorant Receptor OR7D4 and Sensory Perception of Cooked Meat Containing Androstenone. PLoS ONE 2012, 7, e35259. [Google Scholar] [CrossRef]
- Weiler, U.; Font i Furnols, M.; Fischer, K.; Kemmer, H.; Oliver, M.A.; Gispert, M.; Dobrowolski, A.; Claus, R. Influence of differences in sensitivity of Spanish and German consumers to perceive androstenone on the acceptance of boar meat differing in skatole and androstenone concentrations. Meat Sci. 2000, 54, 297–304. [Google Scholar] [CrossRef]
- Font i Furnols, M.; Gispert, M.; Diestre, A.; Oliver, M.A. Acceptability of boar meat by consumers depending on their age, gender, culinary habits, and sensitivity and appreciation of androstenone odour. Meat Sci. 2003, 64, 433–440. [Google Scholar] [CrossRef]
- Weiler, U.; Isernhagen, M.; Stefanski, V.; Ritzmann, M.; Kress, K.; Hein, C.; Zöls, S. Penile Injuries in Wild and Domestic Pigs. Animals 2016, 6, 25. [Google Scholar] [CrossRef]
- Claus, R.; Weiler, U. Endocrine regulation of growth and metabolism in the pig: A review. Livest. Prod. Sci. 1994, 37, 245–260. [Google Scholar] [CrossRef]
- Batorek-Lukač, N.; Čandek-Potokar, M.; Bonneau, M.; Van Milgen, J. Meta-analysis of the effect of immunocastration on production performance, reproductive organs and boar taint compounds in pigs. Animal 2012, 6, 1330–1338. [Google Scholar] [CrossRef]
- Babol, J.; Squires, E.J. Quality of meat from entire male pigs. Food Res. Int. 1995, 28, 201–212. [Google Scholar] [CrossRef]
- Lundström, K.; Matthews, K.R.; Haugen, J.-E. Pig meat quality from entire males. Animal 2009, 3, 1497–1507. [Google Scholar] [CrossRef]
- Reiter, S.; Zöls, S.; Ritzmann, M.; Stefanski, V.; Weiler, U. Penile Injuries in Immunocastrated and Entire Male Pigs of One Fattening Farm. Animals 2017, 7, 71. [Google Scholar] [CrossRef]
- Rydhmer, L.; Zamaratskaia, G.; Andersson, H.K.; Algers, B.; Guillemet, R.; Lundström, K. Aggressive and sexual behaviour of growing and finishing pigs reared in groups, without castration. Acta Agric. Scand. Sect. A Anim. Sci. 2006, 56, 109–119. [Google Scholar] [CrossRef]
- Council Directive 2008/120/EC. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32008L0120 (accessed on 24 April 2018).
- De Briyne, N.; Berg, C.; Blaha, T.; Temple, D. Pig castration: Will the EU manage to ban pig castration by 2018? Porc. Health Manag. 2016, 2, 29. [Google Scholar] [CrossRef]
- Backus, G.; Higuera, M.; Juul, N.; Nalon, E.; de Briyne, N. Second Progress Report 2015–2017 on the European Declaration on Alternatives to Surgical Castration of Pigs. Available online: https://www.boarsontheway.com/wp-content/uploads/2018/08/Second-progress-report-2015-2017-final-1.pdf (accessed on 26 April 2019).
- Fredriksen, B.; Font i Furnols, M.; Lundström, K.; Migdal, W.; Prunier, A.; Tuyttens, F.A.M.; Bonneau, M. Practice on castration of piglets in Europe. Animal 2009, 3, 1480–1487. [Google Scholar] [CrossRef]
- European Declaration on Alternatives to Surgical Castration of Pigs. Available online: https://ec.europa.eu/food/sites/food/files/animals/docs/aw_prac_farm_pigs_cast-alt_declaration_en.pdf (accessed on 31 March 2019).
- Thompson, D.L. Immunization against GnRH in male species (comparative aspects). Anim. Reprod. Sci. 2000, 60–61, 459–469. [Google Scholar] [CrossRef]
- Mancini, M.C.; Menozzi, D.; Arfini, F. Immunocastration: Economic implications for the pork supply chain and consumer perception. An assessment of existing research. Livest. Sci. 2017, 203, 10–20. [Google Scholar] [CrossRef]
- Kress, K.; Weiler, U.; Stefanski, V. Influence of housing conditions on antibody formation and testosterone after Improvac vaccinations. Adv. Anim. Biosci. 2018, 9, s19. [Google Scholar] [CrossRef]
- Claus, R.; Lacorn, M.; Danowski, K.; Pearce, M.C.; Bauer, A. Short-term endocrine and metabolic reactions before and after second immunization against GnRH in boars. Vaccine 2007, 25, 4689–4696. [Google Scholar] [CrossRef]
- Kubale, V.; Batorek-Lukač, N.; Škrlep, M.; Prunier, A.; Bonneau, M.; Fazarinc, G.; Čandek-Potokar, M. Steroid hormones, boar taint compounds, and reproductive organs in pigs according to the delay between immunocastration and slaughter. Theriogenology 2013, 79, 69–80. [Google Scholar] [CrossRef]
- Han, X.; Zhou, Y.; Zeng, Y.; Sui, F.; Liu, Y.; Tan, Y.; Cao, X.; Du, X.; Meng, F.; Zeng, X. Effects of active immunization against GnRH versus surgical castration on hypothalamic-pituitary function in boars. Theriogenology 2017, 97, 89–97. [Google Scholar] [CrossRef]
- European Medicines Agency. EPAR Summary for the Public. Available online: https://www.ema.europa.eu/en/documents/overview/improvac-epar-summary-public_en.pdf (accessed on 24 April 2019).
- Zamaratskaia, G.; Babol, J.; Madej, A.; Squires, E.J.; Lundström, K. Age-related Variation of Plasma Concentrations of Skatole, Androstenone, Testosterone, Oestradiol-17β, Oestrone Sulphate, Dehydroepiandrosterone Sulphate, Triiodothyronine and IGF-1 in Six Entire Male Pigs. Reprod. Domest. Anim. 2004, 39, 168–172. [Google Scholar] [CrossRef]
- Ayalew, G. A Review on the Effect of Immunocastration Against Gonadal Physiology and Boar Taint. Biomed. Nurs. 2019, 5, 26–40. [Google Scholar] [CrossRef]
- Chang, C.; Varamini, P.; Giddam, A.K.; Mansfeld, F.M.; D’Occhio, M.J.; Toth, I. Investigation of Structure–Activity Relationships of Synthetic Anti-Gonadotropin Releasing Hormone Vaccine Candidates. Chemmedchem 2015, 10, 901–910. [Google Scholar] [CrossRef]
- Claus, R.; Rottner, S.; Rueckert, C. Individual return to Leydig cell function after GnRH-immunization of boars. Vaccine 2008, 26, 4571–4578. [Google Scholar] [CrossRef]
- Einarsson, S.; Andersson, K.; Wallgren, M.; Lundström, K.; Rodriguez-Martinez, H. Short- and long-term effects of immunization against gonadotropin-releasing hormone, using ImprovacTM, on sexual maturity, reproductive organs and sperm morphology in male pigs. Theriogenology 2009, 71, 302–310. [Google Scholar] [CrossRef]
- Pinna, A.; Schivazappa, C.; Virgili, R.; Parolari, G. Effect of vaccination against gonadotropin-releasing hormone (GnRH) in heavy male pigs for Italian typical dry-cured ham production. Meat Sci. 2015, 110, 153–159. [Google Scholar] [CrossRef]
- Nautrup, B.P.; Vlaenderen, I.V.; Aldaz, A.; Mah, C.K. The effect of immunization against gonadotropin-releasing factor on growth performance, carcass characteristics and boar taint relevant to pig producers and the pork packing industry: A meta-analysis-ScienceDirect. Res. Vet. Sci. 2018, 119, 182–195. [Google Scholar] [CrossRef]
- Andersson, K.; Brunius, C.; Zamaratskaia, G.; Lundström, K. Early vaccination with Improvac®: Effects on performance and behaviour of male pigs. Animal 2012, 6, 87–95. [Google Scholar] [CrossRef]
- Sattler, T.; Sauer, F.; Schmoll, F. Effect of time of second GnRH vaccination on feed intake, carcass quality and fatty acid composition of male fatteners compared to entire boars and barrows. Berliner und Münchener Tierärztliche Wochenschrift 2014, 127, 290–296. [Google Scholar]
- Aluwé, M.; Degezelle, I.; Depuydt, L.; Fremaut, D.; Van den Broeke, A.; Millet, S. Immunocastrated male pigs: Effect of 4 v. 6 weeks time post second injection on performance, carcass quality and meat quality. Animal 2016, 10, 1466–1473. [Google Scholar] [CrossRef]
- Reiter, S.; Weiler, U.; Stefanski, V.; Ritzmann, M.; Zöls, S. Penile injuries in immunocastrated and entire male pigs of one fattening farm. Adv. Anim. Biosci. 2018, 9, s30. [Google Scholar]
- Škrlep, M.; Batorek-Lukač, N.; Prevolnik-Povše, M.; Čandek-Potokar, M. Teoretical and practical aspects of immunocastration. Stočarstvo Časopis za unapređenje stočarstva 2014, 68, 39–49. [Google Scholar]
- Miller, L.A.; Fagerstone, K.A.; Eckery, D.C. Twenty years of immunocontraceptive research: Lessons learned. J. Zoo Wildl. Med. 2013, 44, 84–96. [Google Scholar] [CrossRef]
- Sødring, S.; Naadland, T.H. High Androstenone in Norwegian Immunocastrates and the Effect on Vaccination Rate and Farmer Attitudes. Available online: http://www.ca-ipema.eu/oeiras-presentations (accessed on 26 April 2019).
- Mathur, P.K.; ten Napel, J.; Bloemhof, S.; Heres, L.; Knol, E.F.; Mulder, H.A. A human nose scoring system for boar taint and its relationship with androstenone and skatole. Meat Sci. 2012, 91, 414–422. [Google Scholar] [CrossRef]
- Morales, J.; Dereu, A.; Manso, A.; de Frutos, L.; Piñeiro, C.; Manzanilla, E.G.; Wuyts, N. Surgical castration with pain relief affects the health and productive performance of pigs in the suckling period. Porc. Health Manag. 2017, 3, 18. [Google Scholar] [CrossRef]
- Needham, T.; Lambrechts, H.; Hoffman, L.C. Castration of male livestock and the potential of immunocastration to improve animal welfare and production traits: Invited Review. S. Afr. J. Anim. Sci. 2017, 47, 731–742. [Google Scholar] [CrossRef]
- McGlone, J.; Guay, K.; Garcia, A. Comparison of Intramuscular or Subcutaneous Injections vs. Castration in Pigs—Impacts on Behavior and Welfare. Animals 2016, 6, 52. [Google Scholar] [CrossRef]
- Einarsson, S. Vaccination against GnRH: Pros and cons. Acta Vet. Scand. 2006, 48, S10. [Google Scholar] [CrossRef]
- McNeil, M.M.; Weintraub, E.S.; Duffy, J.; Sukumaran, L.; Jacobsen, S.J.; Klein, N.P.; Hambidge, S.J.; Lee, G.M.; Jackson, L.A.; Irving, S.A.; et al. Risk of anaphylaxis after vaccination in children and adults. J. Allergy Clin. Immunol. 2016, 137, 868–878. [Google Scholar] [CrossRef]
- Gutzwiller, A.; Ampuero Kragten, S. Suppression of boar taint in cryptorchid pigs using a vaccine against the gonadotropin-releasing hormone. Schweizer Archiv für Tierheilkunde 2013, 155, 677–680. [Google Scholar] [CrossRef]
- Cronin, G.M.; Dunshea, F.R.; Butler, K.L.; McCauley, I.; Barnett, J.L.; Hemsworth, P.H. The effects of immuno- and surgical-castration on the behaviour and consequently growth of group-housed, male finisher pigs. Appl. Anim. Behav. Sci. 2003, 81, 111–126. [Google Scholar] [CrossRef]
- Baumgartner, J.; Laister, S.; Koller, M.; Pfützner, A.; Grodzycki, M.; Andrews, S.; Schmoll, F. The behaviour of male fattening pigs following either surgical castration or vaccination with a GnRF vaccine. Appl. Anim. Behav. Sci. 2010, 124, 28–34. [Google Scholar] [CrossRef]
- Puls, C.L.; Rojo, A.; Matzat, P.D.; Schroeder, A.L.; Ellis, M. Behavior of immunologically castrated barrows in comparison to gilts, physically castrated barrows, and intact male pigs. J. Anim. Sci. 2017, 95, 2345–2353. [Google Scholar] [CrossRef]
- dos Santos, R.d.K.S.; Caldara, F.R.; Moi, M.; dos Santos, L.S.; Nääs, I.A.; Foppa, L.; Garcia, R.G.; Borquis, R.R.A. Behavior of immunocastrated pigs. Revista Brasileira de Zootecnia 2016, 45, 540–545. [Google Scholar] [CrossRef][Green Version]
- Zamaratskaia, G.; Rydhmer, L.; Andersson, H.K.; Chen, G.; Lowagie, S.; Andersson, K.; Lundström, K. Long-term effect of vaccination against gonadotropin-releasing hormone, using Improvac, on hormonal profile and behaviour of male pigs. Anim. Reprod. Sci. 2008, 108, 37–48. [Google Scholar] [CrossRef]
- Rydhmer, L.; Lundström, K.; Andersson, K. Immunocastration reduces aggressive and sexual behaviour in male pigs. Animal 2010, 4, 965–972. [Google Scholar] [CrossRef]
- Guay, K.; Salgado, G.; Thompson, G.; Backus, B.; Sapkota, A.; Chaya, W.; McGlone, J.J. Behavior and handling of physically and immunologically castrated market pigs on farm and going to market. J. Anim. Sci. 2013, 91, 5410–5417. [Google Scholar] [CrossRef]
- Karaconji, B.; Lloyd, B.; Campbell, N.; Meaney, D.; Ahern, T. Effect of an anti-gonadotropin-releasing factor vaccine on sexual and aggressive behaviour in male pigs during the finishing period under Australian field conditions. Aust. Vet. J. 2015, 93, 121–123. [Google Scholar] [CrossRef]
- Schmidt, T.; Calabrese, J.M.; Grodzycki, M.; Paulick, M.; Pearce, M.C.; Rau, F.; von Borell, E. Impact of single-sex and mixed-sex group housing of boars vaccinated against GnRF or physically castrated on body lesions, feeding behaviour and weight gain. Appl. Anim. Behav. Sci. 2011, 130, 42–52. [Google Scholar] [CrossRef]
- Bilskis, R.; Sutkeviciene, N.; Riskeviciene, V.; Januskauskas, A.; Zilinskas, H. Effect of active immunization against GnRH on testosterone concentration, libido and sperm quality in mature AI boars. Acta Vet. Scand. 2012, 54, 33. [Google Scholar] [CrossRef]
- Weiler, U.; Götz, M.; Schmidt, A.; Otto, M.; Müller, S. Influence of sex and immunocastration on feed intake behavior, skatole and indole concentrations in adipose tissue of pigs. Animal 2013, 7, 300–308. [Google Scholar] [CrossRef]
- Van den Broeke, A.; Leen, F.; Aluwé, M.; Ampe, B.; Van Meensel, J.; Millet, S. The effect of GnRH vaccination on performance, carcass, and meat quality and hormonal regulation in boars, barrows, and gilts. J. Anim. Sci. 2016, 94, 2811–2820. [Google Scholar] [CrossRef]
- Batorek-Lukač, N.; Škrlep, M.; Prunier, A.; Louveau, I.; Noblet, J.; Bonneau, M.; Čandek-Potokar, M. Effect of feed restriction on hormones, performance, carcass traits, and meat quality in immunocastrated pigs. J. Anim. Sci. 2012, 90, 4593–4603. [Google Scholar] [CrossRef]
- Turkstra, J.A.; Zeng, X.Y.; van Diepen, J.T.M.; Jongbloed, A.W.; Oonk, H.B.; van de Wiel, D.F.M.; Meloen, R.H. Performance of male pigs immunized against GnRH is related to the time of onset of biological response. J. Anim. Sci. 2002, 80, 2953–2959. [Google Scholar] [CrossRef]
- Millet, S.; Gielkens, K.; Brabander, D.D.; Janssens, G.P.J. Considerations on the performance of immunocastrated male pigs. Animal 2011, 5, 1119–1123. [Google Scholar] [CrossRef]
- Serrano, M.P.; Valencia, D.G.; Fuentetaja, A.; Lázaro, R.; Mateos, G.G. Effect of gender and castration of females and slaughter weight on performance and carcass and meat quality of Iberian pigs reared under intensive management systems. Meat Sci. 2008, 80, 1122–1128. [Google Scholar] [CrossRef]
- Martinez-Macipe, M.; Rodríguez, P.; Izquierdo, M.; Gispert, M.; Manteca, X.; Mainau, E.; Hernández, F.I.; Claret, A.; Guerrero, L.; Dalmau, A. Comparison of meat quality parameters in surgical castrated versus vaccinated against gonadotrophin-releasing factor male and female Iberian pigs reared in free-ranging conditions. Meat Sci. 2016, 111, 116–121. [Google Scholar] [CrossRef]
- Dalmau, A.; Velarde, A.; Rodríguez, P.; Pedernera, C.; Llonch, P.; Fàbrega, E.; Casal, N.; Mainau, E.; Gispert, M.; King, V.; et al. Use of an anti-GnRF vaccine to suppress estrus in crossbred Iberian female pigs. Theriogenology 2015, 84, 342–347. [Google Scholar] [CrossRef]
- Dalmau, A.; Temple, D.; Velarde, A. Relación entre el bienestar animal y el cerdo Ibérico en montanera. Suis 2011, 83, 22–29. [Google Scholar]
- Font i Furnols, M.; Gispert, M.; Soler, J.; Diaz, M.; Garcia-Regueiro, J.A.; Diaz, I.; Pearce, M.C. Effect of vaccination against gonadotrophin-releasing factor on growth performance, carcass, meat and fat quality of male Duroc pigs for dry-cured ham production. Meat Sci. 2012, 91, 148–154. [Google Scholar] [CrossRef]
- Daza, A.; Latorre, M.A.; Olivares, A.; López Bote, C.J. The effects of male and female immunocastration on growth performances and carcass and meat quality of pigs intended for dry-cured ham production: A preliminary study. Livest. Sci. 2016, 190, 20–26. [Google Scholar] [CrossRef]
- Xue, Y.; Zheng, W.; Zhang, F.; Rao, S.; Peng, Z.; Yao, W. Effect of immunocastration on growth performance, gonadal development and carcass and meat quality of SuHuai female pigs. Anim. Prod. Sci. 2019, 59, 794–800. [Google Scholar] [CrossRef]
- Grela, E.R.; Kowalczuk-Vasilev, E.; Klebaniuk, R. Performance, pork quality and fatty acid composition of entire males, surgically castrated or immunocastrated males, and female pigs reared under organic system. Pol. J. Vet. Sci. 2013, 16, 107–114. [Google Scholar] [CrossRef]
- Council Regulation EC (No) 834/2007. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:32007R0834&from=DE (accessed on 24 August 2017).
- European Commission Union. Register of Veterinary Medicinal Products. Available online: http://ec.europa.eu/health/documents/community-register/html/v095.htm (accessed on 24 April 2019).
- Oliviero, C.; Ollila, A.; Andersson, M.; Heinonen, M.; Voutila, L.; Serenius, T.; Peltoniemi, O. Strategic use of anti-GnRH vaccine allowing selection of breeding boars without adverse effects on reproductive or production performances. Theriogenology 2016, 85, 476–482. [Google Scholar] [CrossRef]
- Bauer, A.; Lacorn, M.; Claus, R. Effects of two levels of feed allocation on IGF-I concentrations and metabolic parameters in GnRH-immunized boars. J. Anim. Physiol. Anim. Nutr. 2009, 93, 744–753. [Google Scholar] [CrossRef]
- Huber, L.; Squires, E.J.; de Lange, C.F.M. Dynamics of nitrogen retention in entire male pigs immunized against gonadotropin-releasing hormone. J. Anim. Sci. 2013, 91, 4817–4825. [Google Scholar] [CrossRef]
- Dunshea, F.R.; Allison, J.R.D.; Bertram, M.; Boler, D.D.; Brossard, L.; Campbell, R.; Crane, J.P.; Hennessy, D.P.; Huber, L.; de Lange, C.; et al. The effect of immunization against GnRF on nutrient requirements of male pigs: A review. Animal 2013, 7, 1769–1778. [Google Scholar] [CrossRef]
- Batorek-Lukač, N.; Dubois, S.; Noblet, J.; Čandek-Potokar, M.; Labussière, E. Effect of high dietary fat content on heat production and lipid and protein deposition in growing immunocastrated male pigs. Animal 2016, 10, 1941–1948. [Google Scholar] [CrossRef]
- Quiniou, N.; Monziols, M.; Colin, F.; Goues, T.; Courboulay, V. Effect of feed restriction on the performance and behaviour of pigs immunologically castrated with Improvac®. Animal 2012, 6, 1420–1426. [Google Scholar] [CrossRef]
- Labussière, E.; Batorek-Lukač, N.; Besnard, J.-C.; Čandek-Potokar, M.; Noblet, J. Effet de la teneur en énergie nette du régime sur la consommation volontaire et les performances de croissance des porcs mâles immunocastrés. In Proceedings of the 46èmes Journées de la Recherche Porcine, Paris, France, 1 January 2014. [Google Scholar]
- Moore, K.L.; Mullan, B.P.; Kim, J.C.; Dunshea, F.R. Standardized ileal digestible lysine requirements of male pigs immunized against gonadotrophin releasing factor. J. Anim. Sci. 2016, 94, 1982–1992. [Google Scholar] [CrossRef]
- Labussière, E.; Dubois, S.; van Milgen, J.; Noblet, J. Partitioning of heat production in growing pigs as a tool to improve the determination of efficiency of energy utilization. Front. Physiol. 2013, 4. [Google Scholar] [CrossRef]
- van Milgen, J.; Noblet, J.; Dubois, S. Energetic efficiency of starch, protein and lipid utilization in growing pigs. J. Nutr. 2001, 131, 1309–1318. [Google Scholar] [CrossRef]
- Le Floc’h-Burban, N.; Prunier, A.; Louveau, I. Effect of chirurgical or immune castration on postprandial nutrient profiles in male pigs. In Proceedings of the Annual Meeting of the European Association for Animal Production, Nantes, France, 26–30 August 2013; Wageningen Academic Publishers: Wageningen, The Netherlands, 2013; p. 559. [Google Scholar]
- Quemeneur, K.; Labussiere, E.; Gall, M.L.; Lechevestrier, Y.; Montagne, L. Feeding behaviour and pre-prandial status affect post-prandial plasma energy metabolites and insulin kinetics in growing pigs fed diets differing in fibre concentration. Br. J. Nutr. 2019, 121, 625–636. [Google Scholar] [CrossRef]
- Fry, J.; Kingston, C. Life Cycle Assessment of Pork Report. Available online: https://pork.ahdb.org.uk/media/2344/lifecycelassmntofporklaunchversion.pdf (accessed on 19 April 2019).
- Stern, S.; Sonesson, U.; Gunnarsson, S.; Oborn, I.; Kumm, K.-I.; Nybrant, T. Sustainable development of food production: A case study on scenarios for pig production. Ambio 2005, 34, 402–407. [Google Scholar] [CrossRef]
- Kool, A.; Blonk, H.; Ponsioen, T.; Sukkel, W.; Vermeer, H.M.; de Vries, J.W.; Hoste, R. Carbon Footprints of Conventional and Organic Pork. Available online: http://library.wur.nl/WebQuery/wurpubs/fulltext/50314 (accessed on 16 April 2019).
- Reckmann, K.; Krieter, J. Environmental impacts of the pork supply chain with regard to farm performance. J. Agric. Sci. 2015, 153, 411–421. [Google Scholar] [CrossRef]
- Bandekar, P.A.; Leh, M.; Bautista, R.; Matlock, M.D.; Thoma, G.; Ulrich, R. Life cycle assessment of alternative swine management practices. J. Anim. Sci. 2019, 97, 472–484. [Google Scholar] [CrossRef]
- Aluwé, M.; Tuyttens, F.A.M.; Millet, S. Field experience with surgical castration with anaesthesia, analgesia, immunocastration and production of entire male pigs: Performance, carcass traits and boar taint prevalence. Animal 2015, 9, 500–508. [Google Scholar] [CrossRef]
- De Cuyper, C.; Van den Broeke, A.; Van linden, V.; Leen, F.; Aluwé, M.; Van Meensel, J.; Millet, S. L’impact du poids d’abattage et du sexe sur l’empreinte carbone de l’ingestion alimentaire des porcs. 2019, pp. 195–196. Available online: http://www.journees-recherche-porcine.com/texte/2019-gb.php (accessed on 15 June 2019).
- Van den Broeke, A.; Leen, F.; Aluwé, M.; Van Meensel, J.; Millet, S. Effect of slaughter weight and sex on carcass composition and N-and P-efficiency of pigs. In Book of Abstracts of the 68th Annual Meeting of the European Association for Animal Production; Wageningen Academic Publishers: Wageningen, The Netherlands, 2017. [Google Scholar]
- McGlone, J.J. The Future of Pork Production in the World: Towards Sustainable, Welfare-Positive Systems. Animals 2013, 3, 401–415. [Google Scholar] [CrossRef]
- De Moraes, P.J.U.; Allison, J.; Robinson, J.A.; Baldo, G.L.; Boeri, F.; Borla, P. Life cycle assessment (lca) and environmental product declaration (epd) of an immunological product for boar taint control in male pigs. J. Environ. Assess. Policy Manag. 2013, 15, 1350001. [Google Scholar] [CrossRef]
- Sherman, J.; Le, C.; Lamers, V.; Eckelman, M. Life cycle greenhouse gas emissions of anesthetic drugs. Anesth. Analg. 2012, 114, 1086–1090. [Google Scholar] [CrossRef]
- D’Souza, D.N.; Hewitt, R.J.E.; van Barneveld, R.J. Pork production with entire males and immunocastrates in Australia. Adv. Anim. Biosci. 2018, 9, s58. [Google Scholar] [CrossRef]
- Zoetis Workshop zu Tierwohl und zur Impfung gegen Ebergeruch. Available online: https://www.zoetis.de/news-and-media/workshop-zu-tierwohl-und-zur-impfung-gegen-ebergeruch.aspx (accessed on 24 April 2019).
- USDA Livestock and Poultry: World Markets and Trade. Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/73666448x/ws859p59c/4x51hs663/livestock_poultry.pdf (accessed on 24 April 2019).
- European Commission. Establishing Best Practices on the Production, the Processing and the Marketing of Meat from Uncastrated Pigs or Pigs Vaccinated Against Boar Taint (Immunocastrated). 2019. Available online: https://ec.europa.eu/food/sites/food/files/animals/docs/aw_prac_farm_pigs_cast-alt_establishing-best-practices.pdf (accessed on 24 April 2019).
- de Roest, K.; Montanari, C.; Fowler, T.; Baltussen, W. Resource efficiency and economic implications of alternatives to surgical castration without anaesthesia. Animal 2009, 3, 1522–1531. [Google Scholar] [CrossRef]
- Verhaagh, M.; Deblitz, C. Wirtschaftlichkeit der Alternativen zur betäubungslosen Ferkelkastration–Aktualisierung und Erweiterung der betriebswirtschaftlichen Berechnungen. Thünen Work. Pap. 2019, 110. [Google Scholar] [CrossRef]
- Vondeling, G.T.; Cao, Q.; Postma, M.J.; Rozenbaum, M.H. The Impact of Patent Expiry on Drug Prices: A Systematic Literature Review. Appl. Health Econ. Health Policy 2018, 16, 653–660. [Google Scholar] [CrossRef]
- Niemi, J.K.; Voutila, L.; Valros, A.; Oliviero, C.; Heinonen, M.; Peltoniemi, O. Economic aspects of immunocastration in the pigs. In Proceedings of the 25th Nordic Association of Agricultural Congress, Riga, Latvia, 16–18 June 2015. [Google Scholar]
- Backus, G.; Støier, S.; Courat, M.; Bonneau, M.; Higuera, M. First Progress Report from the European Declaration on Alternatives to Surgical Castration of Pigs (16/12/2010). Available online: https://ec.europa.eu/food/sites/food/files/animals/docs/aw_prac_farm_pigs_cast-alt_declaration_progress-report_20141028.pdf (accessed on 26 April 2019).
- Tuyttens, F.A.M.; Vanhonacker, F.; Langendries, K.; Aluwé, M.; Millet, S.; Bekaert, K.; Verbeke, W. Effect of information provisioning on attitude toward surgical castration of male piglets and alternative strategies for avoiding boar taint. Res. Vet. Sci. 2011, 91, 327–332. [Google Scholar] [CrossRef]
- Schübeler, A.S.; Koch, L. The search for the best way. Animal welfare vs. boar taint-immunocastration in comparison to the alternatives from the producer and master views. Fleischwirtschaft 2018, 98, 21–23. [Google Scholar]
- Aluwé, M.; Vanhonacker, F.; Millet, S.; Tuyttens, A.M. Influence of hands-on experience on pig farmers’ attitude towards alternatives for surgical castration of male piglets. Res. Vet. Sci. 2015, 103, 80–86. [Google Scholar] [CrossRef]
- Di Pasquale, J.; Nannoni, E.; Sardi, L.; Rubini, G.; Salvatore, R.; Bartoli, L.; Adinolfi, F.; Martelli, G. Towards the Abandonment of Surgical Castration in Pigs: How is Immunocastration Perceived by Italian Consumers? Animals 2019, 9, 198. [Google Scholar] [CrossRef]
- Mancini, M.C.; Menozzi, D.; Arfini, F.; Veneziani, M. Chapter 13-How Do Firms Use Consumer Science to Target Consumer Communication? The Case of Animal Welfare. In Case Studies in the Traditional Food Sector; Cavicchi, A., Santini, C., Eds.; Woodhead Publishing: Cambridge, UK, 2018; pp. 337–357. [Google Scholar]
- Heid, A.; Hamm, U. Consumer Attitudes Towards Alternatives to Piglet Castration Without Pain Relief in Organic Farming: Qualitative Results from Germany. J. Agric. Environ. Eth. 2012, 25, 687–706. [Google Scholar] [CrossRef]
- Fredriksen, B.; Johnsen, A.M.S.; Skuterud, E. Consumer attitudes towards castration of piglets and alternatives to surgical castration. Res. Vet. Sci. 2011, 90, 352–357. [Google Scholar] [CrossRef]
- Mörlein, D.; Schübeler, A.S. This is how the dialogue with the customers succeeds: Vaccination against boar taint-How can the procedure be best communicated? Fleischwirtschaft 2017, 97, 36–40. [Google Scholar]
- Čandek-Potokar, M.; Prevolnik-Povše, M.; Škrlep, M.; Font i Furnols, M.; Batorek-Lukač, N.; Kress, K.; Stefanski, V. Acceptability of Dry-Cured Belly (Pancetta) from Entire Males, Immunocastrates or Surgical Castrates: Study with Slovenian Consumers. Foods 2019, 8, 122. [Google Scholar] [CrossRef]
- European Medicines Agency. EPAR-Scientific Discussion. Available online: https://www.ema.europa.eu/en/documents/scientific-discussion/improvac-epar-scientific-discussion_en.pdf (accessed on 22 April 2019).
- Clarke, I.J.; Walker, J.S.; Hennessy, D.; Kreeger, J.; Nappier, J.M.; Crane, J.S. Inherent Food Safety of a Synthetic Gonadotropin-Releasing Factor (GnRF) Vaccine for the Control of Boar Taint in Entire Male Pigs. Int. J. Appl. Res. Vet. Med. 2008, 6, 7–14. [Google Scholar]
- Dorn, C.; Griesinger, G. GnRH-Analoga in der Reproduktionsmedizin. Gynäkologische Endokrinologie 2009, 7, 161–170. [Google Scholar] [CrossRef]
- D’Occhio, M.J. Immunological suppression of reproductive functions in male and female mammals. Anim. Reprod. Sci. 1993, 33, 345–372. [Google Scholar] [CrossRef]
- Simms, M.S.; Scholfield, D.P.; Jacobs, E.; Michaeli, D.; Broome, P.; Humphreys, J.E.; Bishop, M.C. Anti-GnRH antibodies can induce castrate levels of testosterone in patients with advanced prostate cancer. Br. J. Cancer 2000, 83, 443–446. [Google Scholar] [CrossRef][Green Version]
| Country | Boars (%) | Immunocastrates (%) | Barrows (%) | Pig Population (×1000) |
|---|---|---|---|---|
| Germany | 20 | <1 | 80 | 28,046 |
| Spain | 80 | 5 | 15 | 25,495 |
| Denmark | <2 | 0 | >97 | 12,402 |
| Netherlands | 65 | 0 | 35 | 12,013 |
| France | 22 | <0.1 | 78 | 11,835 |
| Italy | 2 | 5 | 93 | 8561 |
| Belgium | 8 | 15 | 80 | 6351 |
| Romania | 0 | 5 | 95 | 5180 |
| UK | 98 | <1 | 2 | 4383 |
| Hungary | 1 | 0 | 99 | 2935 |
| Austria | 5 | 0 | 95 | 2846 |
| Portugal | 85 | 2.5 | 12.5 | 2014 |
| Norway | <1 | 6 | 94 | 1644 |
| Switzerland | 5 | 2.5 | 92.5 | 1573 |
| Czech | 5 | 5 | 90 | 1548 |
| Ireland | 100 | 0 | 0 | 1468 |
| Sweden | 1 | 9 | 90 | 1354 |
| Finland | 4 | 0 | 96 | 1258 |
| Slovakia | 0 | 10 | 90 | 637 |
| Latvia | 0 | 0 | 100 | 368 |
| Estonia | 0 | 0 | 100 | 359 |
| Slovenia | 1 | 0 | 99 | 288 |
| Macedonia | 0 | 0 | 100 | 200 |
| Luxembourg | 1 | 0 | 99 | 90 |
| Iceland | 0 | 0 | 99 | 36 |
| Total | 34 | 2.8 | 63 | 132,884 |
| Country | Year | Alternatives Implemented |
|---|---|---|
| Norway | 2002 | Local anesthesia (lidocaine) with analgesia (meloxicam) |
| Netherlands | 2009 | Surgical castration under anesthesia (CO2) for export market |
| Germany | 2009 | End of surgical castration without anesthesia postponed until 2020. Meanwhile analgesia (meloxicam); anesthesia (esp. isoflurane) required only in some organic programs; immunocastration in some high-quality meat programs |
| Switzerland | 2010 | Anesthesia (isoflurane) |
| Denmark | 2011/2019 | Analgesia (meloxicam); from 2019 on, plus local anesthesia (procaine) |
| Belgium | 2011 | Surgical castration with analgesia (meloxicam) for export market; immunocastration domestic retail market |
| France | 2013 | Analgesia (meloxicam) |
| Sweden | 2016 | Local anesthesia (lidocaine) with analgesia (meloxicam); one smaller retailer prefers immunocastration |
| Austria | 2017 | Analgesia (meloxicam) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kress, K.; Millet, S.; Labussière, É.; Weiler, U.; Stefanski, V. Sustainability of Pork Production with Immunocastration in Europe. Sustainability 2019, 11, 3335. https://doi.org/10.3390/su11123335
Kress K, Millet S, Labussière É, Weiler U, Stefanski V. Sustainability of Pork Production with Immunocastration in Europe. Sustainability. 2019; 11(12):3335. https://doi.org/10.3390/su11123335
Chicago/Turabian StyleKress, Kevin, Sam Millet, Étienne Labussière, Ulrike Weiler, and Volker Stefanski. 2019. "Sustainability of Pork Production with Immunocastration in Europe" Sustainability 11, no. 12: 3335. https://doi.org/10.3390/su11123335
APA StyleKress, K., Millet, S., Labussière, É., Weiler, U., & Stefanski, V. (2019). Sustainability of Pork Production with Immunocastration in Europe. Sustainability, 11(12), 3335. https://doi.org/10.3390/su11123335





