Applying Soil Health Indicators to Encourage Sustainable Soil Use: The Transition from Scientific Study to Practical Application
Abstract
1. Introduction
2. Ecosystem Services and the Soil Food Web
3. Measuring and Monitoring Soil Condition to Preserve Ecosystem Services
4. Measuring an Indicator Rather than the Actual Ecosystem Service
5. Considerations for the practical Use of Soil Health Indicators.
- “African soil crisis threatens food security” (2014) [58]
- “Careless farming adding to floods” (2014) [59];
- “EU pesticide bans could hit UK crops” (2014) [60];
- “UK faces significant shortage of farmland” (2014) [61];
- “Members of Parliament sound alarm over neglected soils” (2016) [62];
- “Nature loss linked to farming intensity” (2016) [63];
- “Prince of Wales joins soil boosting project” (2016) [64];
- “Scottish research finds soil crucial to climate change fight” (2016) [65];
- “Farm subsidies must be earned” (2017) [66];
- “Bread’s environmental costs are counted” (2017) [67];
6. Biological Indicator of Soil Quality (BISQ) in The Netherlands
- Soil biological indicators: earthworms, enchytraeids, nematodes, microarthropods, fungi, bacteria, N mineralisation, C mineralisation, and root mass (grassland only).
- Abiotic soil indicators: soil type and texture, penetration resistance, bulk density, organic matter parameters including labile fractions, pH, nutrients.
- System indicators: land use, vegetation, agricultural management (crop, rotation, tillage, fertilization, crop protection (pesticides), traffic) and groundwater level.
- If costs are a major aspect for the soil monitoring, these can only be reduced by reducing the number of indicators.
7. A Soil Health Test as a Practical Tool for Scottish Growers
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Synthesis; Island Press: Washington, DC, USA, 2005. [Google Scholar]
- Barnes, A.D.; Jochum, M.; Lefcheck, J.S.; Eisenhauer, N.; Scherber, C.; O’Connor, M.I.; de Ruiter, P.; Brose, U. Energy Flux: The Link between Multitrophic Biodiversity and Ecosystem Functioning. Trends Ecol. Evolut. 2018, 33, 186–197. [Google Scholar] [CrossRef] [PubMed]
- De Vries, F.T.; Wallenstein, M.D. Below-ground connections underlying above-ground food production: A framework for optimising ecological connections in the rhizosphere. J. Ecol. 2017, 105, 913–920. [Google Scholar] [CrossRef]
- Young, I.M.; Bengough, A.G. The search for the meaning of life in soil: An opinion. Eur. J. Soil Sci. 2018, 69, 31–38. [Google Scholar] [CrossRef]
- Bünemann, E.K.; Bongiorno, G.; Bai, Z.G.; Creamer, R.E.; De Deyn, G.; de Goede, R.; Fleskens, L.; Geissen, V.; Kuyper, T.W.; Mader, P.; et al. Soil quality—A critical review. Soil Biol. Biochem. 2018, 120, 105–125. [Google Scholar] [CrossRef]
- Bloem, J.; Lebbink, G.; Zwart, K.B.; Bouwman, L.A.; Burgers, S.L.G.E.; de Vos, J.A.; de Ruiter, P.C. Dynamics of microorganisms, microbivores and nitrogen mineralisation in winter wheat fields under conventional and integrated management. Agric. Ecosyst. Environ. 1994, 51, 129–143. [Google Scholar] [CrossRef]
- De Ruiter, P.C.; Moore, J.C.; Zwart, K.B.; Bouwman, L.A.; Hassink, J.; Bloem, J.; de Vos, J.A.; Marinissen, J.C.Y.; Didden, W.A.M.; Lebrink, G.; et al. Simulation of nitrogen minerlaization in the below-ground food webs of two winter wheat fields. J. Appl. Ecol. 1993, 30, 95–106. [Google Scholar] [CrossRef]
- Rashid, M.I.; de Goede, R.G.M.; Brussaard, L.; Bloem, J.; Lantinga, E.A. Production-ecological modelling explains the difference between potential soil N mineralisation and actual herbage N uptake. Appl. Soil Ecol. 2014, 84, 83–92. [Google Scholar] [CrossRef]
- Cosentino, D.; Chenu, C.; Le Bissonnais, Y. Aggregate stability and microbial community dynamics under drying-wetting cycles in a silt loam soil. Soil Biol. Biochem. 2006, 38, 2053–2062. [Google Scholar] [CrossRef]
- Spurgeon, D.J.; Keith, A.M.; Schmidt, O.; Lammertsma, D.R.; Faber, J.H. Land-use and land-management change: Relationships with earthworm and fungi communities and soil structural properties. BMC Ecol. 2013, 13, 46. [Google Scholar] [CrossRef] [PubMed]
- Andriuzzi, W.S.; Pulleman, M.M.; Schmidt, O.; Faber, J.F.; Brussaard, L. Anecic earthworms (Lumbricus terrestris) alleviate negative effects of extreme rainfall events on soil and plants in field mesocosms. Plant Soil 2015, 397, 103–113. [Google Scholar] [CrossRef]
- Paul, E.A.; Kravchenko, A.; Grandy, A.; Morris, S. Soil organic matter dynamics: Controls and management for sustainable ecosystem functioning. In The Ecology of Agricultural Landscapes: Long-Term Research on the Path to Sustainability; Hamilton, S.K., Doll, J.E., Robertson, G.P., Eds.; Oxford University Press: New York, NY, USA, 2015; pp. 104–134. [Google Scholar]
- Kallenbach, C.M.; Frey, S.D.; Grandy, A.S. Direct evidence for microbial-derived soil organic matter formation and its ecophysiological controls. Nat. Commun. 2016, 7, 13630. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, B.S.; Philippot, L. Insights into the resistance and resilience of the soil microbial community. FEMS Microbiol. Rev. 2013, 37, 112–129. [Google Scholar] [CrossRef] [PubMed]
- De Vries, F.T.; Liiri, M.E.; Bjørnlund, L.; Bowker, M.A.; Christensen, S.; Setälä, H.M.; Bardgett, R.D. Land use alters the resistance and resilience of soil food webs to drought. Nat. Clim. Chang. 2012, 2, 276–280. [Google Scholar] [CrossRef]
- De Vries, F.T.; Thébault, E.; Liiri, M.; Birkhofer, K.; Tsiafouli, M.A.; Bjørnlund, L.; Jørgensen, H.B.; Brady, M.V.; Christensen, S.; De Ruiter, P.C.; et al. Soil food web properties explain ecosystem services across European land use systems. Proc. Natl. Acad. Sci. USA 2013, 110, 14296–14301. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.F.; Paul, L.R.; Finlay, R.D. Microbial interactions in the mycorrhizosphere and their significance for sustainable agriculture. FEMS Microbiol. Ecol. 2004, 48, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nasim, G. The role of arbuscular mycorrhizae in inducing resistance to drought and salinity stress in crops. In Plant Adaptation and Phytoremediation; Ashraf, M., Ozturk, M., Ahmad, M.S.A., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 119–141. [Google Scholar]
- Santander, C.; Aroca, R.; Ruiz-Lozano, J.M.; Olave, J.; Cartes, P.; Borie, F.; Cornejo, P. Arbuscular mycorrhiza effects on plant performance under osmotic stress. Mycorrhiza 2017, 27, 639–657. [Google Scholar] [CrossRef] [PubMed]
- Postma, J.; Schilder, M.T.; Bloem, J.; van Leeuwen-Haagsma, W.K. Soil suppressiveness and functional diversity of the soil microflora in organic farming systems. Soil Biol. Biochem. 2008, 40, 2394–2406. [Google Scholar] [CrossRef]
- Kibblewhite, M.G.; Ritz, K.; Swift, M.J. Soil health in agricultural systems. Philos. Trans. R. Soc. B 2008, 363, 685–701. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.; Panagos, P.; Barcelo, S.; Bouraoui, F.; Bosco, C.; Dewitte, O.; Gardi, C.; Erhard, M.; Hervás, J.; Hiederer, R.; et al. The State of Soil in Europe. A Contribution of the JRC to the European Environment Agency’s Environment State and Outlook Report–SOER 2010; Publications Office of the European Union: Luxembourg, 2012. [Google Scholar]
- FAO. The State of the World’s Land and Water Resources for Food and Agriculture (SOLAW)–Managing Systems at Risk; Food and Agriculture Organization of the United Nations: Rome, Italy, 2011. [Google Scholar]
- Smith, P.; House, J.I.; Bustamante, M.; Sobocká, J.; Harper, R.; Pan, G.; West, P.C.; Clark, J.M.; Adhya, T.; Rumpel, C.; et al. Global pressures on soils from land use and management. Glob. Chang. Biol. 2015, 22, 1008–1028. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.S.; Ritz, K.; McGrath, S.P.; Quinton, J.N.; Goulding, K.W.T.; Jones, R.J.A.; Harris, J.A.; Bol, R.; Wallace, P.; Pilgrim, E.S.; et al. A review of the impacts of degradation threats on soil properties in the UK. Soil Use Manag. 2015, 31, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, J.P.; Saby, N.P.A.; Jones, A.; Louwagie, G.; Micheli, E.; Rutgers, M.; Schulte, R.P.O.; Spiegel, H.; Toth, G.; Creamer, R.E. Gap assessment in current soil monitoring networks across Europe for measuring soil functions. Environ. Res. Lett. 2017, 12, 124007. [Google Scholar] [CrossRef]
- O’Sullivan, L.; Bampa, F.; Knights, K.; Creamer, R.E. Soil protection for a sustainable future: Options for a soil monitoring network in Ireland. Soil Use Manag. 2017, 33, 346–363. [Google Scholar] [CrossRef]
- Cluzeau, D.; Guernion, M.; Chaussod, R.; Martin-Laurent, F.; Villenave, C.; Cortet, J.; Ruiz-Camacho, N.; Pernin, C.; Mateille, T.; Philippot, L.; et al. Integration of biodiversity in soil quality monitoring: Baselines for microbial and soil fauna parameters for different land-use types. Eur. J. Soil Biol. 2012, 49, 63–72. [Google Scholar] [CrossRef]
- European Union (EU). Communication from the Commission to the Council, the European Parliament, the Economic and Social Committee and the Committee of the Regions; Thematic Strategy for Soil Protection Plus Summary of the Impact Assessment; COM 231 (2006) Final; European Union: Brussels, Belgium, 2006. [Google Scholar]
- European Union (EU). Proposal for a Directive of the European Parliament and of the Council Establishing a Framework for the Protection of Soil and Amending Directive 2004/35/EC; COM 232 (2006) Final; European Union: Brussels, Belgium, 2006; 30p. [Google Scholar]
- Griffiths, B.S.; Römbke, J.; Schmelz, R.M.; Scheffczyk, A.; Faber, J.; Bloem, J.; Pérès, G.; Cluzeau, D.; Chabbi, A.; Suhadolc, M.; et al. Selecting cost effective and policy-relevant biological indicators for European monitoring of soil biodiversity and ecosystem function. Ecol. Ind. 2016, 69, 213–223. [Google Scholar] [CrossRef]
- Pulleman, M.; Creamer, R.; Hamer, U.; Helder, J.; Pelosi, C.; Pérès, G.; Rutgers, M. Soil biodiversity, biological indicators and soil ecosystem services—An overview of European approaches. Curr. Opin. Environ. Sustain. 2012, 4, 529–538. [Google Scholar] [CrossRef]
- Ritz, K.; Black, H.I.J.; Campbell, C.D.; Harris, J.A.; Wood, C. Selecting biological indicators for monitoring soils: A framework for balancing scientific and technical opinion to assist policy development. Ecol. Ind. 2009, 9, 1212–1221. [Google Scholar] [CrossRef]
- Stockdale, E.A.; Watson, C.A. Managing Soil Biota to Deliver Ecosystem Services; Natural England Commissioned Report NECR100; Natural England: York, UK, 2012.
- Francaviglia, R. Agricultural Soil Erosion and Soil Biodiversity: Developing Indicators for Policy Analyses; OECD: Paris, France, 2008. [Google Scholar]
- Havileck, E. Soil biodiversity and bioindication: From complex thinking to simple acting. Eur. J. Soil Biol. 2012, 49, 80–84. [Google Scholar]
- Faber, J.H.; Creamer, R.E.; Mulder, C.; Römbke, J.; Rutgers, M.; Sousa, J.P.; Stone, D.; Griffiths, B.S. The practicalities and pitfalls of establishing a policy-relevant and cost-effective soil biological monitoring scheme. Integr. Environ. Assess. Manag. 2013, 9, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Feld, C.K.; Martins da Silva, P.; Sousa, J.P.; De Bello, F.; Bugter, R.; Grandin, U.; Hering, D.; Lavorel, S.; Mountford, O.; Pardo, I.; et al. Indicators of biodiversity and ecosystem services: A synthesis across ecosystems and spatial scales. Oikos 2009, 118, 1862–1871. [Google Scholar] [CrossRef]
- Lemanceau, P.; Maron, P.-A.; Mazurier, S.; Mougel, C.; Pivato, B.; Plassart, P.; Ranjard, L.; Revellin, C.; Tardy, V.; Wipf, D. Understanding and managing soil biodiversity: A major challenge in agroecology. Agron. Sustain. Dev. 2015, 35, 67–81. [Google Scholar] [CrossRef]
- Gardi, C.; Montanarella, L.; Arrouays, D.; Bispo, A.; Lemanceau, P.; Jolivet, C.; Mulder, C.; Ranjard, L.; Römbke, J.; Rutgers, M.; et al. Soil biodiversity monitoring in Europe: Ongoing activities and challenges. Eur. J. Soil Sci. 2009, 60, 807–819. [Google Scholar] [CrossRef]
- Rutgers, M.; Schouten, A.J.; Bloem, J.; van Eekeren, N.; de Goede, R.G.M.; Jagers op Akkerhuis, G.A.J.M.; van der Wal, A.; Mulder, C.; Brussaard, L.; Breure, A.M. Biological measurements in a nationwide soil monitoring network. Eur. J. Soil Sci. 2009, 60, 820–832. [Google Scholar] [CrossRef]
- Bispo, A.; Cluzeau, D.; Creamer, R.; Dombos, M.; Graefe, U.; Krogh, P.H.; Sousa, J.P.; Pérès, G.; Rutgers, M.; Winding, A.; et al. Indicators for monitoring soil biodiversity. Integr. Environ. Assess. Manag. 2009, 5, 717–719. [Google Scholar] [CrossRef] [PubMed]
- Black, H.I.J.; Parekh, N.R.; Chaplow, J.S.; Monson, F.; Watkins, J.; Creamer, R.; Potter, E.D.; Poskitt, J.M.; Rowland, P.; Ainsworth, G.; et al. Assessing soil biodiversity across Great Britain: National trends in the occurrence of heterotrophic bacteria and invertebrates in soil. J. Environ. Manag. 2003, 67, 255–266. [Google Scholar] [CrossRef]
- Ditzler, C.A.; Tugel, A.J. Soil quality field tools: Experiences of USDA-NRCS soil quality institute. Agron. J. 2002, 94, 33–38. [Google Scholar] [CrossRef]
- Fusaro, S.; Squartini, A.; Paoletti, M.G. Functional biodiversity, environmental sustainability and crop nutritional properties: A case study of horticultural crops in north-eastern Italy. Appl. Soil Ecol. 2018, 123, 699–708. [Google Scholar] [CrossRef]
- Turbé, A.; De Toni, A.; Benito, P.; Lavelle, P.; Lavelle, P.; Ruiz, N.; Van der Putten, W.H.; Labouze, E.; Mudgal, S. Soil Biodiversity: Functions, Threats and Tools for Policy Makers. Bio Intelligence Service, IRD, and NIOO; Report for European Commission (DG Environment); European Communities: Paris, France, 2010. [Google Scholar]
- Aalders, I.; Hough, R.L.; Towers, W.; Black, H.I.J.; Ball, B.C.; Griffiths, B.S.; Hopkins, D.W.; Lilly, A.; McKenzie, B.M.; Rees, R.M.; et al. Considerations for Scottish soil monitoring in the European context. Eur. J. Soil Sci. 2009, 60, 833–843. [Google Scholar] [CrossRef]
- Paz-Ferreiro, J.; Fu, S. Biological indices for soil quality evaluation: Perspectives and limitations. Land Degrad. Dev. 2016, 27, 14–25. [Google Scholar] [CrossRef]
- Doran, J.W.; Zeiss, M.R. Soil health and sustainability: Managing the biotic component of soil quality. Appl. Soil Ecol. 2000, 15, 3–11. [Google Scholar] [CrossRef]
- Reynolds, W.D. Saturated Hydraulic Properties: Ring Infiltrometer. In Soil Sampling and Methods of Analysis, 2nd ed.; Carter, M.R., Gregorich, E.G., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 1043–1056. [Google Scholar]
- Ehlers, W. Observations on earthworm channels and infiltration on tilled and untilled loess soil. Soil Sci. 1975, 119, 242–249. [Google Scholar] [CrossRef]
- Bouché, M.B. Stratégies lombriciennes. In Soil Organisms as Components of Ecosystems; Lohm, U., Persson, T., Eds.; Ecology Bulletin; NFR: Stockholm, Sweden, 1977; pp. 122–132. [Google Scholar]
- Bouché, M.B.; Al-Addan, F. Earthworms, water infiltration and soil stability: Some new assessments. Soil Biol. Biochem. 1997, 29, 441–452. [Google Scholar] [CrossRef]
- Six, J.; Bossuyt, H.; Degryze, S.; Denef, K. A history of research on the link between (micro)aggregates, soil biota, and soil organic matter dynamics. Soil Till. Res. 2004, 79, 7–31. [Google Scholar] [CrossRef]
- Wardle, D.A. Impacts of disturbance on detritus food webs in agro-ecosystems of contrasting tillage and weed management practices. In Advances in Ecological Research; Begon, M., Fitter, A.H., Eds.; Academic Press: Cambridge, MA, USA, 1995; Volume 26, pp. 105–185. [Google Scholar]
- Guide to British earthworms. Available online: https://www.opalexplorenature.org/?q=Earthwormguide (accessed on 23 August 2018).
- Paoletti, M.G.; Sommaggio, D.; Fusaro, S. An earthworm soil quality index proposal (QBS-e) applied to agroecosystems. Biol. Ambient. 2013, 27, 25–43. [Google Scholar]
- African Soil Crisis Threatens Food Security. Available online: https://www.bbc.co.uk/news/science-environment-30277514 (accessed on 23 August 2018).
- Careless Farming Adding to Floods. Available online: http://www.bbc.co.uk/news/science-environment-26466653 (accessed on 23 August 2018).
- EU Pesticide BANS Could Hit UK Crops. Available online: http://www.bbc.co.uk/news/uk-29699449 (accessed on 23 August 2018).
- Uk Faces Significant Shortage of Farmland. Available online: https://www.bbc.co.uk/news/science-environment-28003435 (accessed on 23 August 2018).
- Members of Parliament Sound Alarm over Neglected Soils. Available online: http://www.bbc.co.uk/news/science-environment-36428361 (accessed on 23 August 2018).
- Nature Loss Linked to Farming Intensity. Available online: http://www.bbc.co.uk/news/science-environment-37298485 (accessed on 23 August 2018).
- Prince of Wales Joins Soil Boosting Project. Available online: https://www.bbc.co.uk/news/science-environment-37766919 (accessed on 23 August 2018).
- Scottish Research Finds Soil Crucial to Climate Change Fight. Available online: https://www.scotsman.com/news/education/scottish-research-finds-soil-crucial-to-climate-change-fight-1-4093629#ixzz45DwfHpQY (accessed on 23 August 2018).
- Farm Subsidies Must Be Earned. Available online: http://www.bbc.co.uk/news/science-environment-40673559 (accessed on 23 August 2018).
- Bread’s Environmental Costs Are Counted. Available online: http://www.bbc.co.uk/news/science-environment-39106180 (accessed on 23 August 2018).
- Lancaster, J. The ridiculous notion of assessing ecological health and identifying the useful concepts underneath. Hum. Ecol. Risk Assess. 2000, 6, 213–222. [Google Scholar] [CrossRef]
- Bloem, J.; Schouten, A.J.; Sørensen, S.J.; Rutgers, M.; van der Werf, A.; Breure, A.M. Monitoring and evaluating soil quality. In Microbiological Methods for Assessing Soil Quality; Bloem, J., Hopkins, D.W., Benedetti, A., Eds.; CABI: Wallingford, UK, 2006; pp. 23–49. [Google Scholar]
- UNCED. United Nations Conference in Environment and Development; Agenda 21; UNCED: Rio de Janeiro, Brazil, 1992. [Google Scholar]
- Rutgers, M.; Schouten, T.; Bloem, J.; Buis, E.; Dimmers, W.; van Eekeren, N.; de Goede, R.G.M.; Jagers op Akkerhuis, G.A.J.M.; Keidel, H.; Korthals, G.; et al. Een Indicatorsysteem voor Ecosysteemdiensten van de Bodem: Life Support Functions Revisited; RIVM Rapport 2014-0145; RIVM: Bilthoven, The Netherlands, 2014; 129p, Available online: http://edepot.wur.nl/345145 (accessed on 23 August 2018).
- De Vries, F.T.; Bååth, E.; Kuyper, T.W.; Bloem, J. High turnover of fungal hyphae in incubation experiments. FEMS Microbiol. Ecol. 2009, 67, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Anderson, J.P.E.; Domsch, K.H. A physiological method for the quantitative measurement of microbial biomass in soils. Soil Biol. Biochem. 1978, 10, 215–221. [Google Scholar] [CrossRef]
- Schipper, L.A.; Sparling, G.P. Performance of soil condition indicators across taxonomic groups and land uses. Soil Sci. Soc. Am. J. 2000, 64, 300–311. [Google Scholar] [CrossRef]
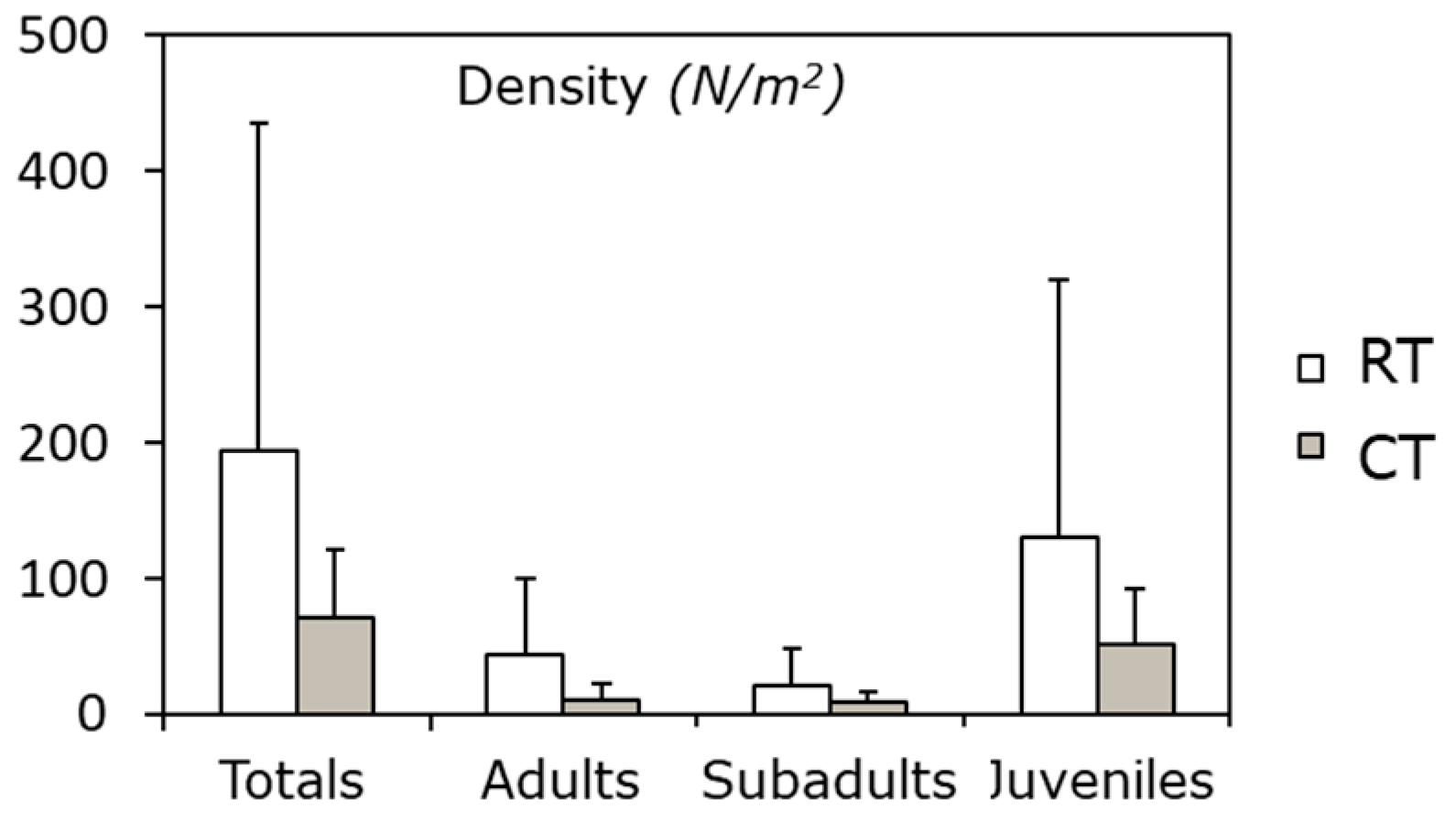
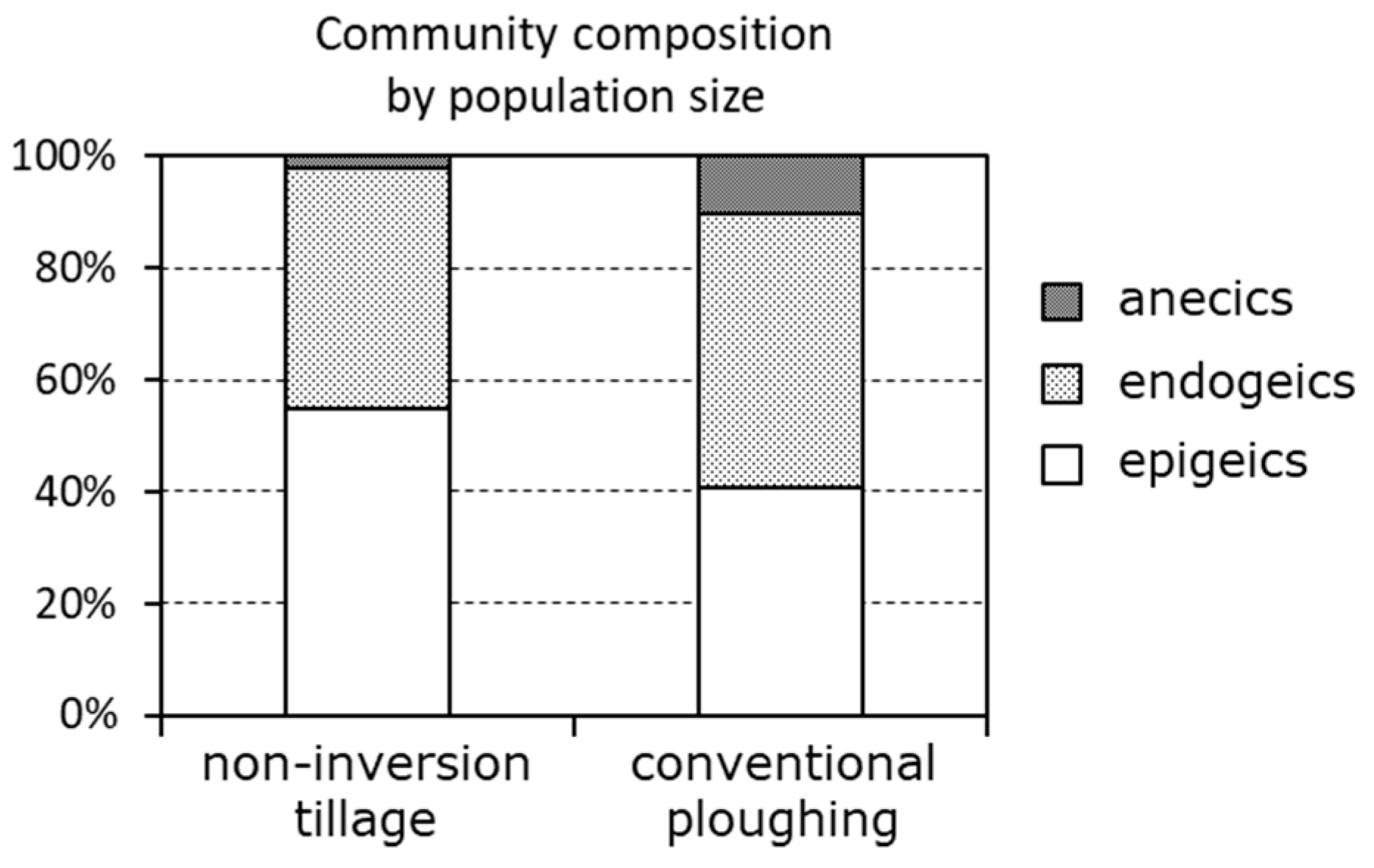
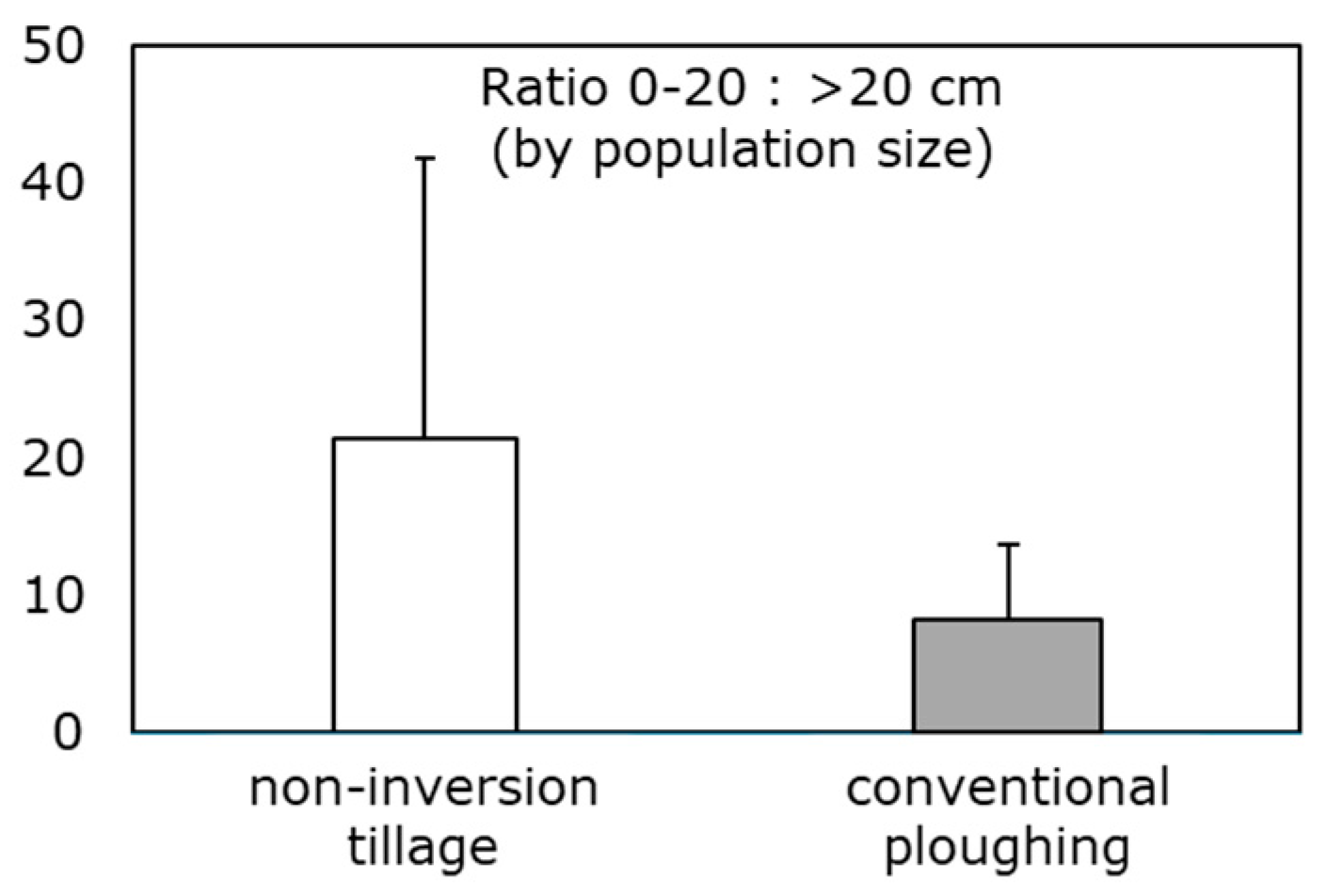
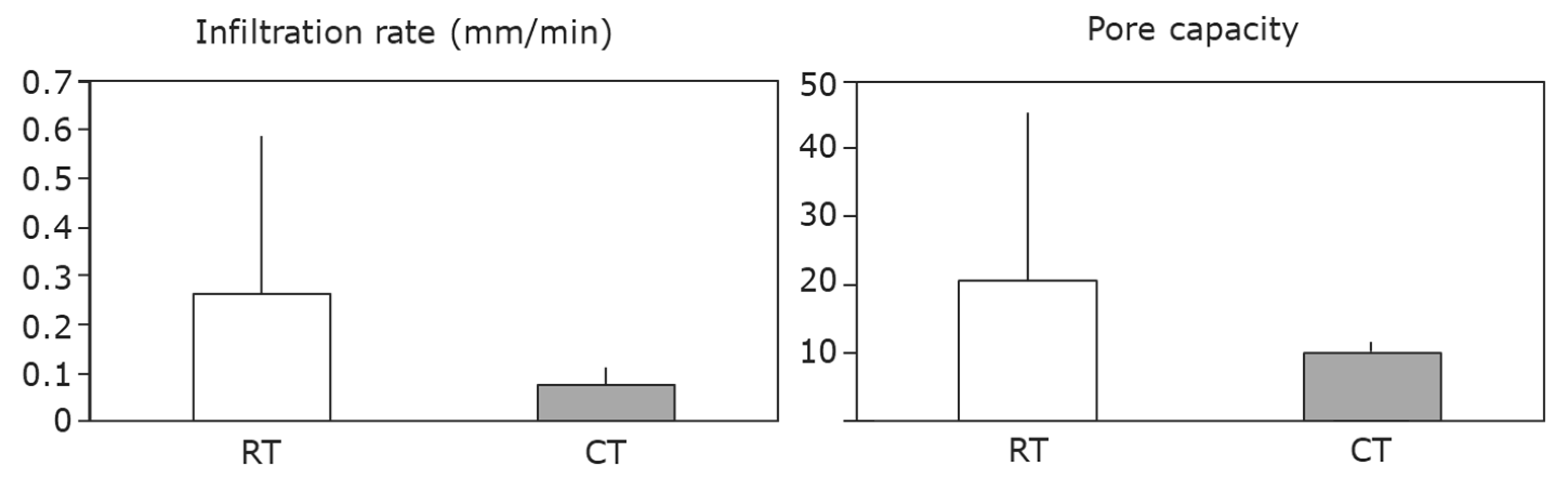
| Measure | Overview | Score | Target range |
|---|---|---|---|
| Potentially Mineralisable N | 28.7 mg kg−1 | >21 mg kg−1 | |
| Organic Matter (LOI) | 5.96 % | >9.5 % | |
| pH | 6.1 | 6.5–7.5 | |
| Extractable Phosphorus | 4.39 mg L−1 | 4.5–13.5 mg L−1 | |
| Extractable Potassium | 87.9 mg L−1 | >76 mg L−1 | |
| Extractable Magnesium | 154 mg L−1 | 61–1000 mg L−1 | |
| Extractable Calcium | 1500 mg L−1 | >3000 mg L−1 | |
| Extractable Sodium | 11.2 mg L−1 | >50 mg L−1 | |
| Visual Evaluation of Soil Structure | 2.75 | <2.4 | |
| Earthworm count | 6.25 per 20cm−2 | >8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Griffiths, B.S.; Faber, J.; Bloem, J. Applying Soil Health Indicators to Encourage Sustainable Soil Use: The Transition from Scientific Study to Practical Application. Sustainability 2018, 10, 3021. https://doi.org/10.3390/su10093021
Griffiths BS, Faber J, Bloem J. Applying Soil Health Indicators to Encourage Sustainable Soil Use: The Transition from Scientific Study to Practical Application. Sustainability. 2018; 10(9):3021. https://doi.org/10.3390/su10093021
Chicago/Turabian StyleGriffiths, Bryan S., Jack Faber, and Jaap Bloem. 2018. "Applying Soil Health Indicators to Encourage Sustainable Soil Use: The Transition from Scientific Study to Practical Application" Sustainability 10, no. 9: 3021. https://doi.org/10.3390/su10093021
APA StyleGriffiths, B. S., Faber, J., & Bloem, J. (2018). Applying Soil Health Indicators to Encourage Sustainable Soil Use: The Transition from Scientific Study to Practical Application. Sustainability, 10(9), 3021. https://doi.org/10.3390/su10093021





