Reduction of Nitrates in Waste Water through the Valorization of Rice Straw: LIFE LIBERNITRATE Project
Abstract
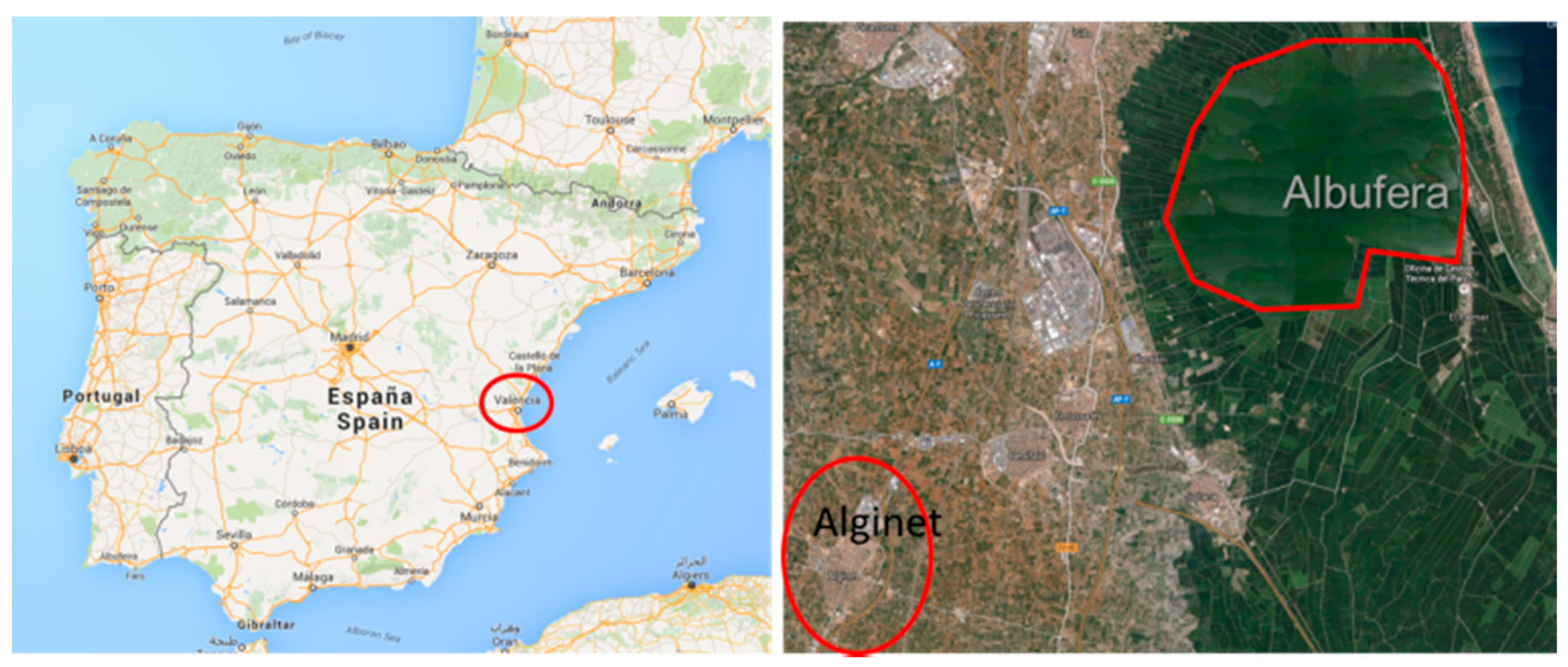
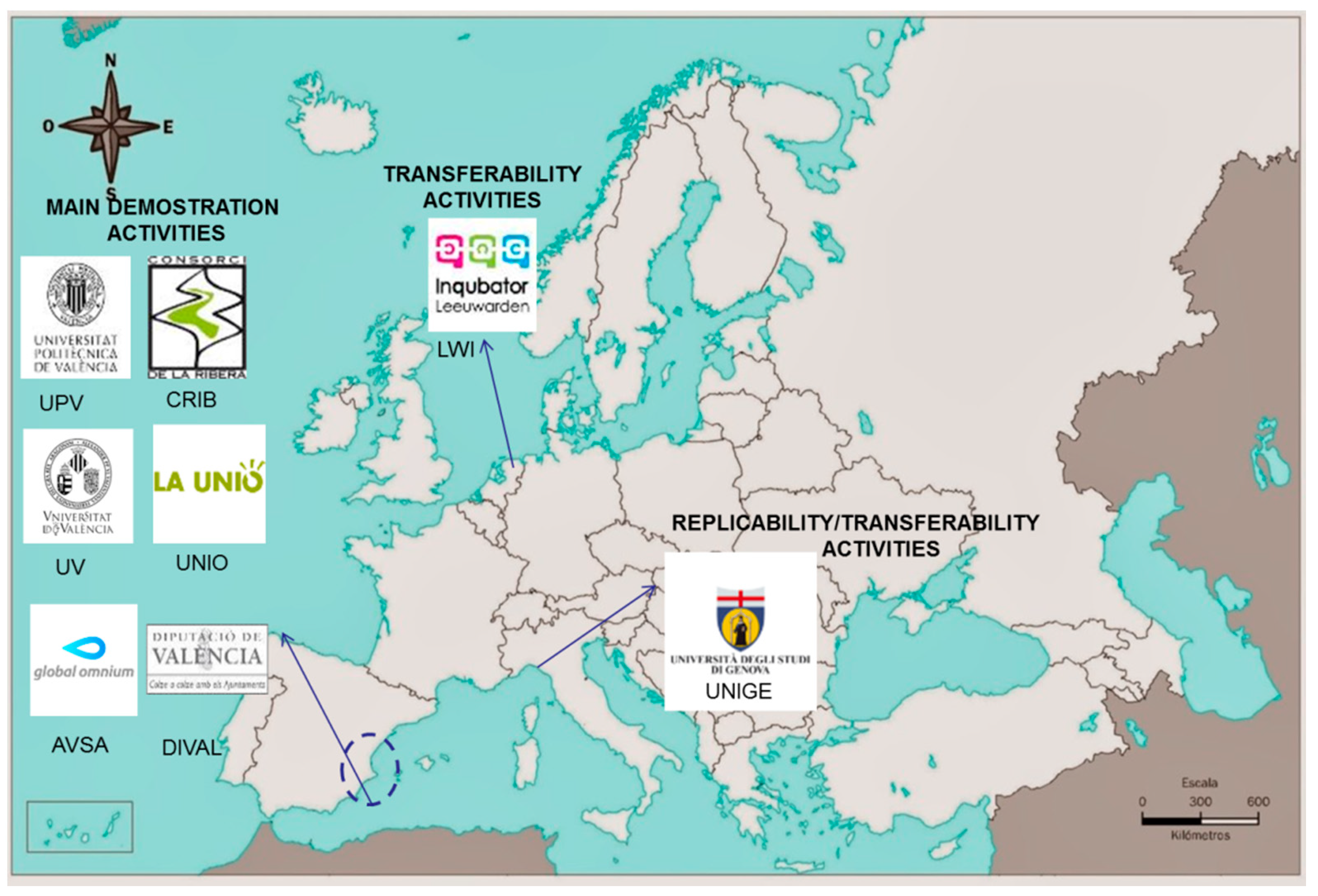
1. Framework of LIFE LIBERNITRATE Project
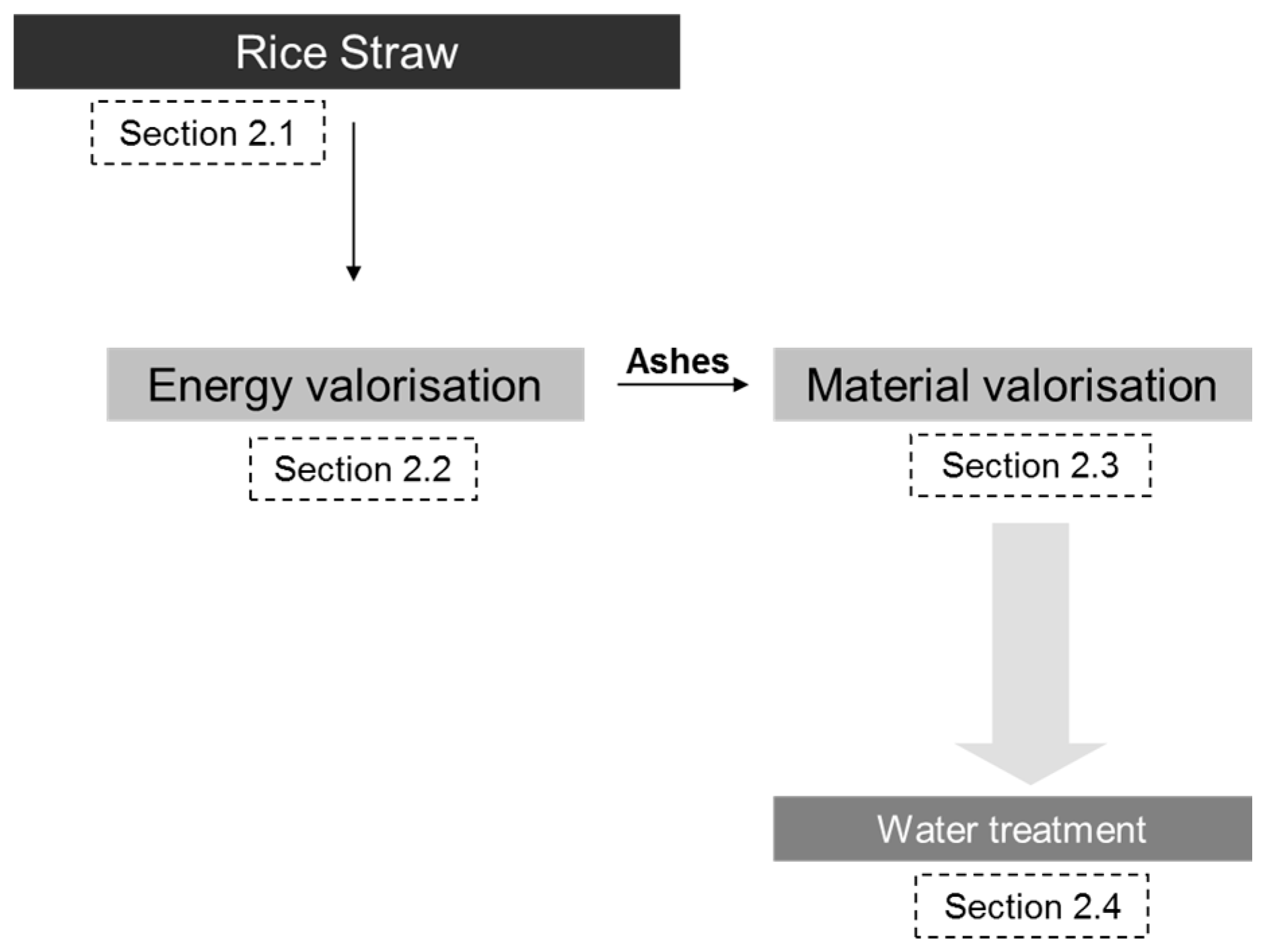
2. Context and Technological Background
2.1. Characterization and Pre-Treatment of Rice Straw
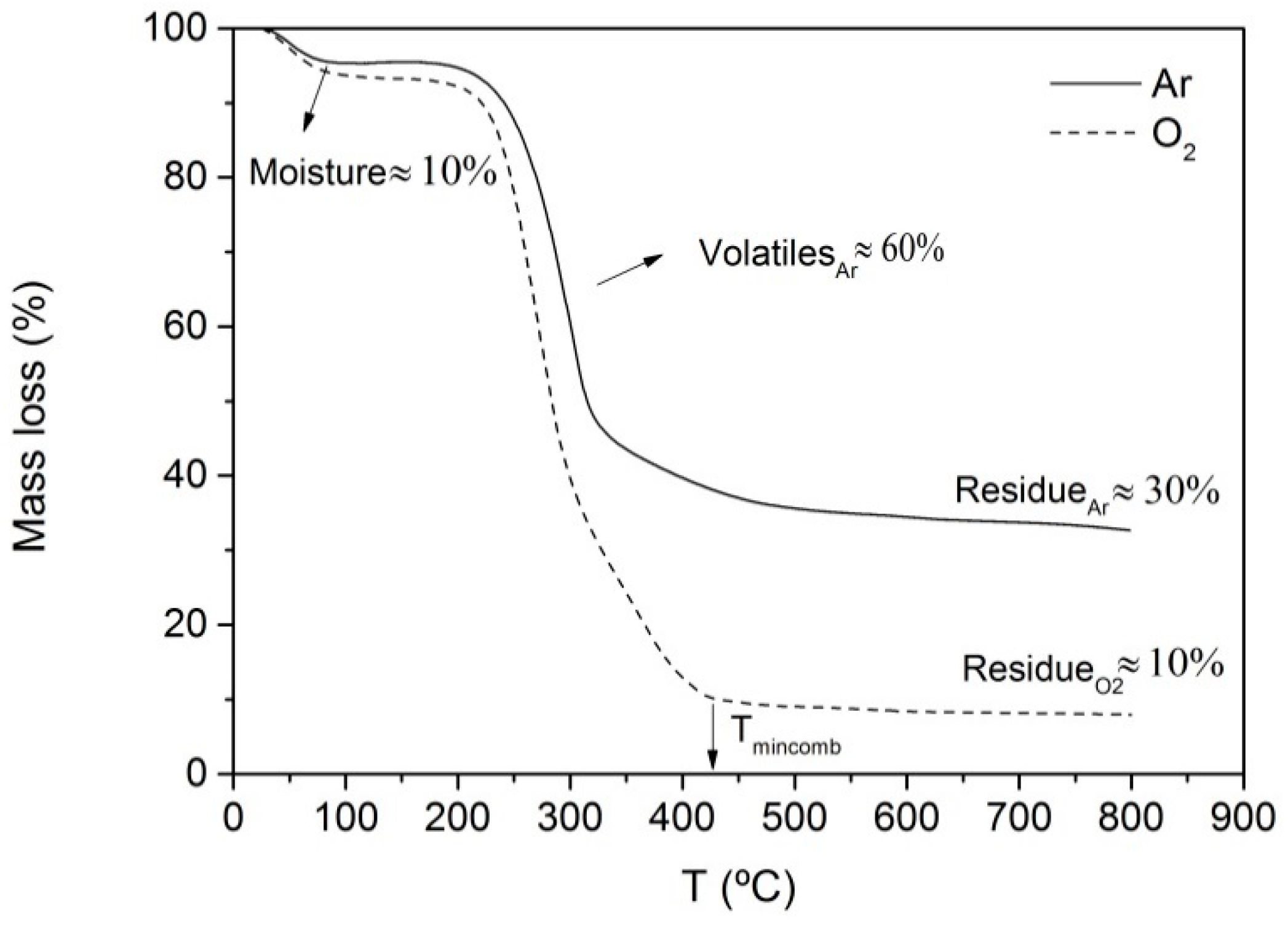
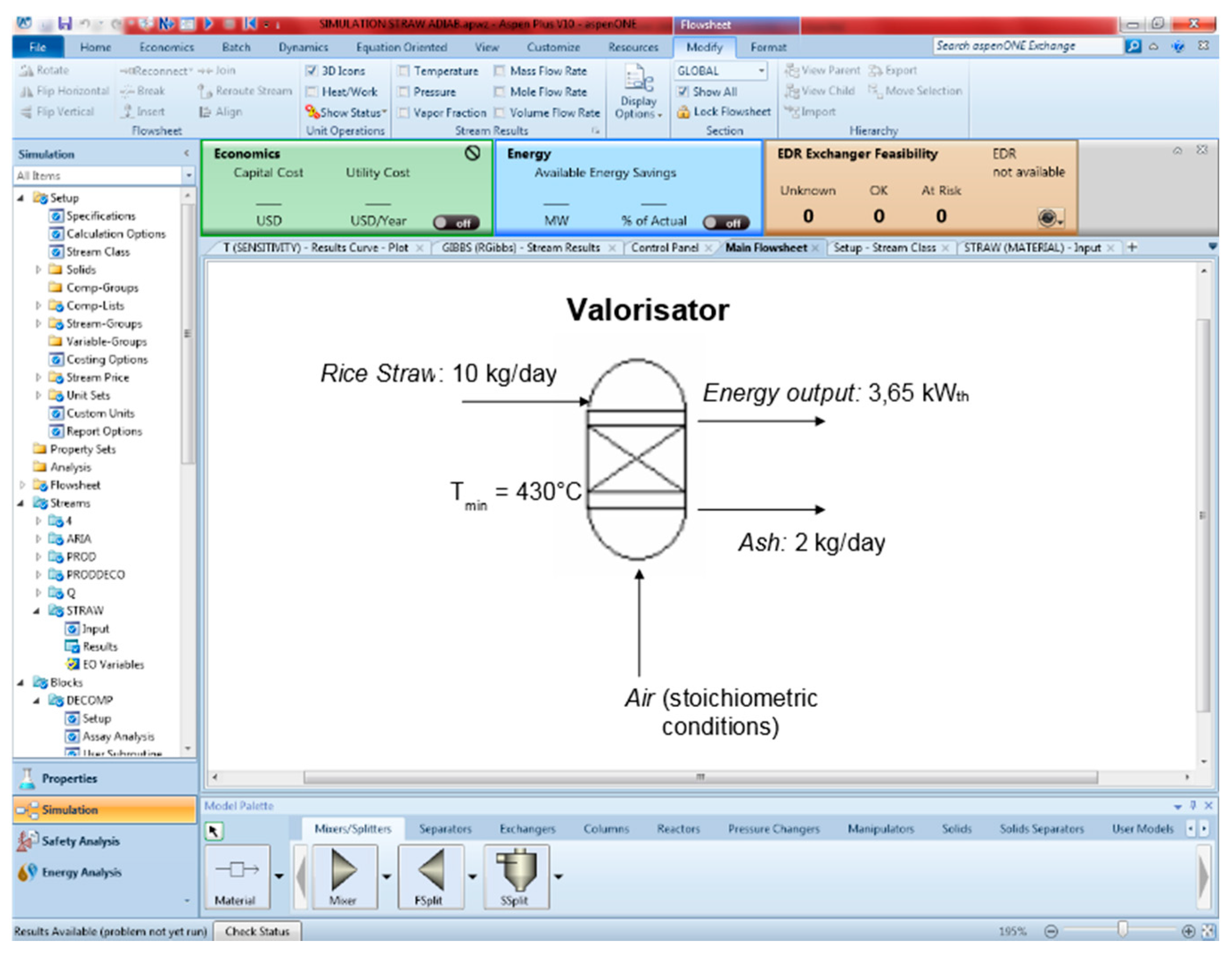
2.2. Energy Valorisation of Rice Straw
2.3. Material Valorisation of Rice Straw Ash
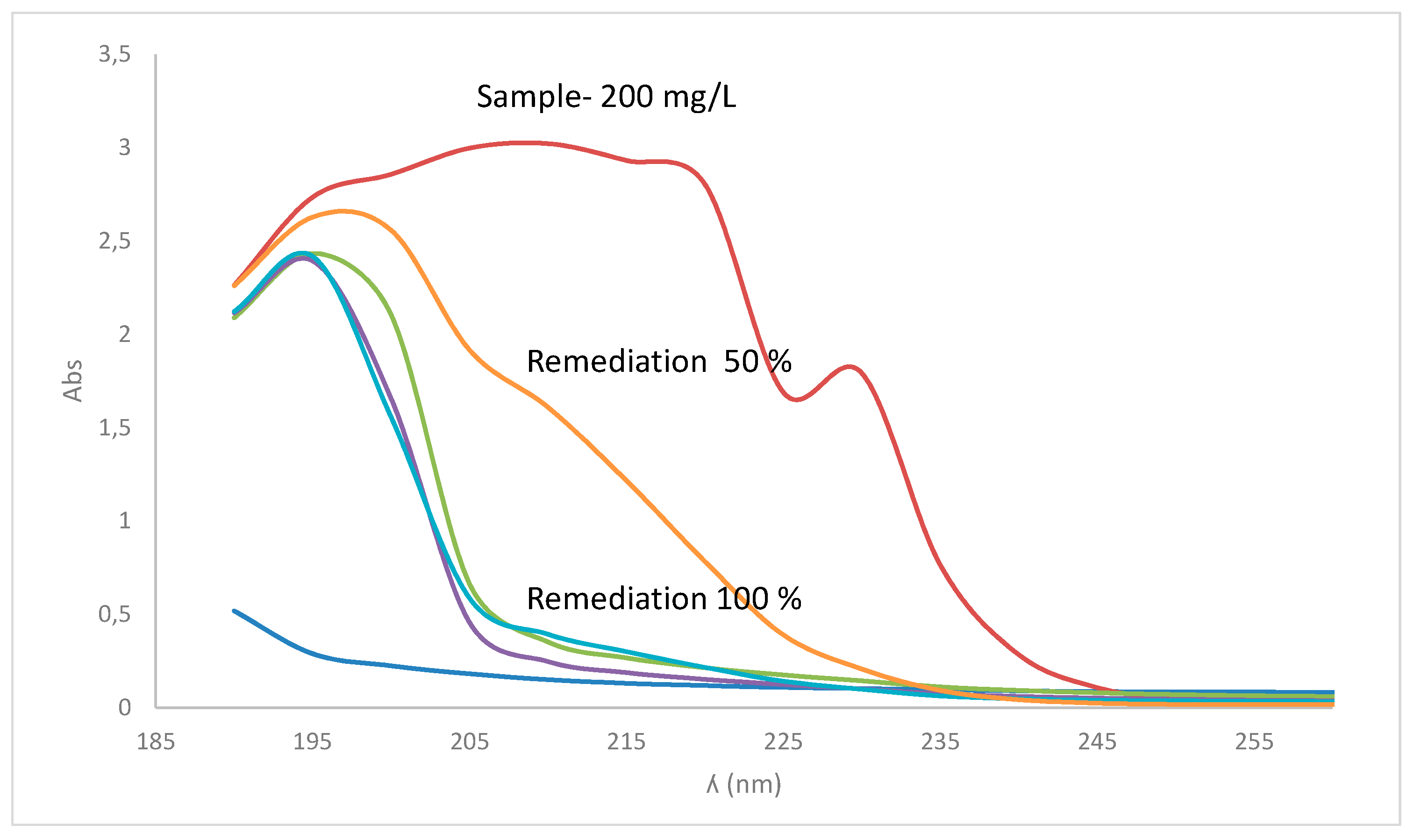
2.4. Product Assessment
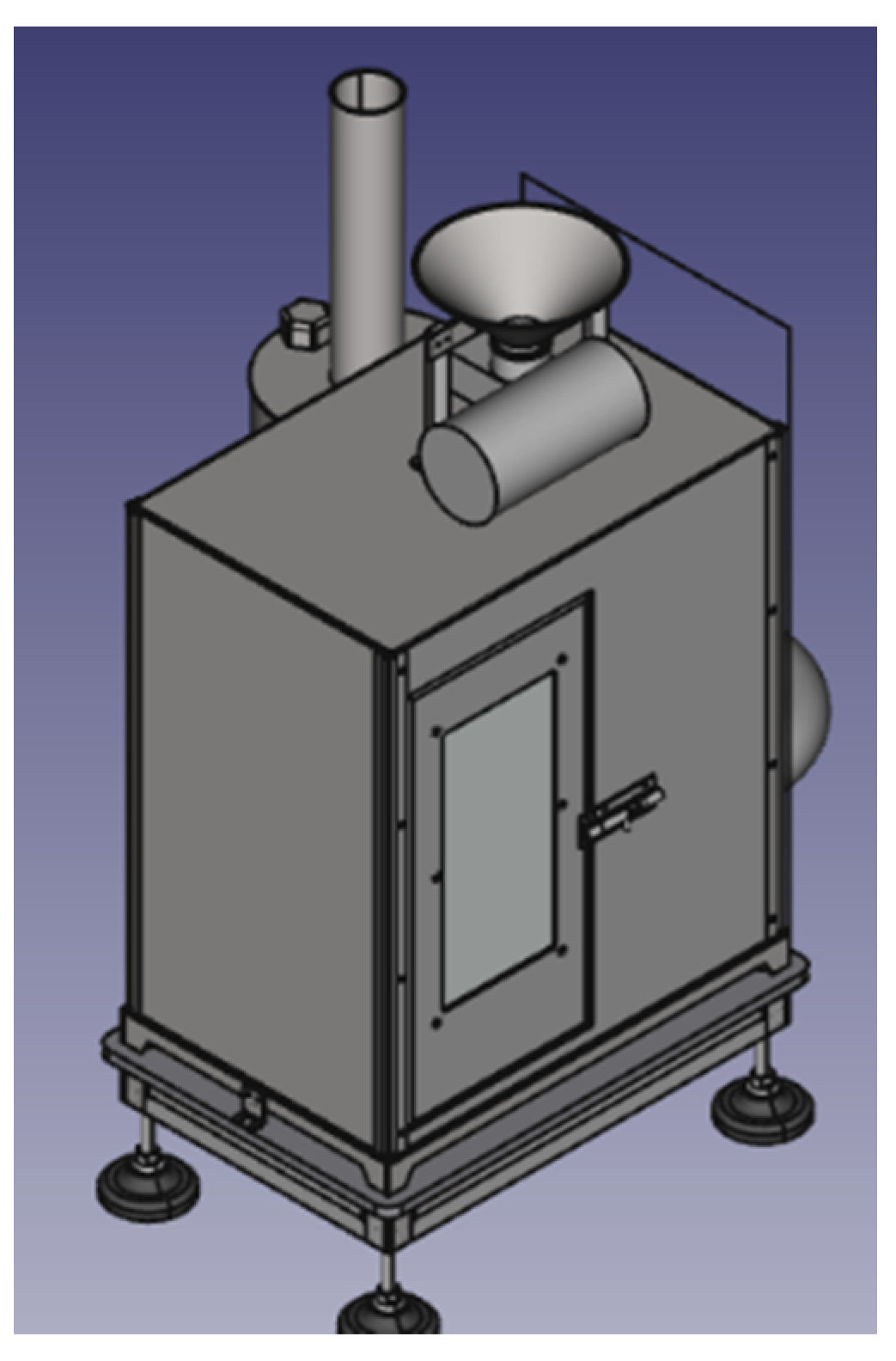
3. LIFE LIBERNITRATE: Project Overview
- -
- Direct purification of at least 26 m3/day of well water, reducing the nitrate concentration to at least 25 ppm. The prototype will be directly transferrable to municipalities of 200 inhabitants or fewer;
- -
- An expected reduction of 90% in the concentration of nitrates in 130 L/day of water effluent in drinking water to facilitate compliance with the Nitrates Directive, and in the reject water of a reverse osmosis plant;
4. Conclusions
Author Contributions
Funding

Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations. Coping with Water Scarcity: Challenge of the Twenty-First Century; Food and Agriculture Organization of the United Nations: Rome, Italy, 2007. [Google Scholar]
- Sousa, J.C.G.; Ribeiro, A.R.; Barbosa, M.O.; Pereira, M.F.R.; Silva, A.M.T. A review on environmental monitoring of water organic pollutants identified by EU guidelines. J. Hazard. Mater. 2018, 344, 146–162. [Google Scholar] [CrossRef] [PubMed]
- European Parliament, Council of the European Union. Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off. J. Eur. Parliam. 2000, L327, 1–82. [Google Scholar] [CrossRef]
- Escribano Francés, G.; Quevauviller, P.; San Martín González, E.; Vargas Amelin, E. Climate change policy and water resources in the EU and Spain. A closer look into the Water Framework Directive. Environ. Sci. Policy 2017, 69, 1–12. [Google Scholar] [CrossRef]
- Stavenhagen, M.; Buurman, J.; Tortajada, C. Saving water in cities: Assessing policies for residential water demand management in four cities in Europe. Cities 2018, 79, 187–195. [Google Scholar] [CrossRef]
- Bigalke, M.; Schwab, L.; Rehmus, A.; Tondo, P.; Flisch, M. Uranium in agricultural soils and drinking water wells on the Swiss Plateau. Environ. Pollut. 2018, 233, 943–951. [Google Scholar] [CrossRef] [PubMed]
- St-Hilaire, A.; Duchesne, S.; Rousseau, A.N. Floods and water quality in Canada: A review of the interactions with urbanization, agriculture and forestry. Can. Water Resour. J. Rev. Can. Ressour. Hydr. 2016, 41, 273–287. [Google Scholar] [CrossRef]
- Matysik, M.; Absalon, D.; Ruman, M. Surface Water Quality in Relation to Land Cover in Agricultural Catchments (Liswarta River Basin Case Study). Pol. J. Environ. Stud. 2015, 24, 175–184. [Google Scholar] [CrossRef]
- Trulli, E.; Torretta, V.; Rada, E.C.; Istrate, I.A.; Papa, E.A. Modeling for use of water in agriculture. INMATEH Agric. Eng. 2014, 44, 121–128. [Google Scholar]
- Food and Agriculture Organization of the United Nations. World Fertilizer Trends and Outlook to 2018 Table; Food and Agriculture Organization of the United Nations: Rome, Italy, 2015; ISBN 9789251086926. [Google Scholar]
- Huang, J.; Xu, C.; Ridoutt, B.G.; Wang, X.; Ren, P. Nitrogen and phosphorus losses and eutrophication potential associated with fertilizer application to cropland in China. J. Clean. Prod. 2017, 159, 171–179. [Google Scholar] [CrossRef]
- Han, J.; Shi, J.; Zeng, L.; Xu, J.; Wu, L. Effects of nitrogen fertilization on the acidity and salinity of greenhouse soils. Environ. Sci. Pollut. Res. 2015, 22, 2976–2986. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, K.M.; King, A.M.; Harter, T. Identifying sources of groundwater nitrate contamination in a large alluvial groundwater basin with highly diversified intensive agricultural production. J. Contam. Hydrol. 2013, 151, 140–154. [Google Scholar] [CrossRef] [PubMed]
- Wick, K.; Heumesser, C.; Schmid, E. Groundwater nitrate contamination: Factors and indicators. J. Environ. Manag. 2012, 111, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, M.F.; Barzetti, S.; Hastie, D.R. The production of atmospheric NOx and N2O from a fertilized agricultural soil. Atmos. Environ. Part A Gen. Top. 1991, 25, 1961–1969. [Google Scholar] [CrossRef]
- Costagliola, A.; Roperto, F.; Benedetto, D.; Anastasio, A.; Marrone, R.; Perillo, A.; Russo, V.; Papparella, S.; Paciello, O. Outbreak of fatal nitrate toxicosis associated with consumption of fennels (Foeniculum vulgare) in cattle farmed in Campania region (Southern Italy). Environ. Sci. Pollut. Res. 2014, 21, 6252–6257. [Google Scholar] [CrossRef] [PubMed]
- Grossman, M.R. Nitrates from Agriculture in Europe: The EC Nitrates Directive and its Implementation in England. B.C. Environ. Aff. Law Rev. 2000, 27, 567–629. [Google Scholar]
- Winton, E.F.; Tardiff, R.G.; McCabe, L.J. Nitrate in drinking water. J. Am. Water Works Assoc. 1971, 63, 95–98. [Google Scholar] [CrossRef]
- Europe’s Water: An Indicator-Based Assessment. Available online: http://www.mcs-international.org/downloads/029_europes_water.pdf (accessed on 23 August 2018).
- Commission to the Council and the European Parliament. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A52013DC0683 (accessed on 23 August 2018).
- Moliner, C.; Bosio, B.; Arato, E.; Ribes, A. Thermal and thermo-oxidative characterisation of rice straw for its use in energy valorisation processes. Fuel 2016, 180, 71–79. [Google Scholar] [CrossRef]
- Sulyman, M.; Namiesnik, J.; Gierak, A. Low-cost Adsorbents Derived from Agricultural By-products/Wastes for Enhancing Contaminant Uptakes from Wastewater: A Review. Pol. J. Environ. Stud. 2017, 26, 479–510. [Google Scholar] [CrossRef]
- Ali, I.; Asim, M.; Khan, T.A. Low cost adsorbents for the removal of organic pollutants from wastewater. J. Environ. Manag. 2012, 113, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Khalid, N.; Ahmad, S.; Kiani, S.N.; Jamil, A. Removal of mercury from aqueous solutions by adsorption to rice husks. Sep. Sci. Technol. 1999, 34, 3139–3153. [Google Scholar] [CrossRef]
- Kumar, U.; Bandyopadhyay, M. Sorption of cadmium from aqueous solution using pretreated rice husk. Bioresour. Technol. 2006, 97, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Orlando, U.S.; Baes, A.U.; Nishijima, W.; Okada, M. Preparation of agricultural residue anion exchangers and its nitrate maximum adsorption capacity. Chemosphere 2002, 48, 1041–1046. [Google Scholar] [CrossRef]
- Diagboya, P.N.E.; Dikio, E.D. Silica-based mesoporous materials; emerging designer adsorbents for aqueous pollutants removal and water treatment. Microporous Mesoporous Mater. 2018, 266, 252–267. [Google Scholar] [CrossRef]
- LIFE LIBERNITRATE. Available online: http://www.lifelibernitrate.eu/ (accessed on 23 August 2018).
- Food and Agriculture Organization of the United Nations. World Agriculture: Towards 2015–2030: Summary Report; Food and Agriculture Organization of the United Nations: Rome, Italy, 2002. [Google Scholar]
- Moliner, C.; Bosio, B.; Arato, E.; Ribes-Greus, A. Comparative study for the energy valorisation of rice straw. In Chemical Engineering Transactions; Italian Association of Chemical Engineering—AIDIC: Milano, Italy, 2014; Volume 37, pp. 241–246. [Google Scholar]
- Moliner, C.; Aguilar, K.; Bosio, B.; Arato, E.; Ribes, A. Thermo-oxidative characterisation of the residues from persimmon harvest for its use in energy recovery processes. Fuel Process. Technol. 2016, 152, 421–429. [Google Scholar] [CrossRef]
- Bove, D.; Moliner, C.; Curti, M.; Rovero, G.; Baratieri, M.; Bosio, B.; Arato, E.; Garbarino, G.; Marchelli, F. Experimental studies on the gasification of the residues from prune of apple trees with a spouted bed reactor. In Proceedings of the European Biomass Conference and Exhibition Proceedings, Amsterdam, The Netherlands, 6–9 June 2016; Volume 2016, pp. 858–862. Available online: http://www.etaflorence.it/proceedings/?detail=13062 (accessed on 23 August 2018).
- Moliner, C.; Badia, J.D.; Bosio, B.; Arato, E.; Kittikorn, T.; Strömberg, E.; Teruel-Juanes, R.; Ek, M.; Karlsson, S.; Ribes-Greus, A. Thermal and thermo-oxidative stability and kinetics of decomposition of PHBV/sisal composites. Chem. Eng. Commun. 2018, 205, 226–237. [Google Scholar] [CrossRef]
- Sudhagar, M.; Lope, T.; Sokhansanj, S. An overview of compaction of biomass grinds. Powder Handl. Process. 2003, 15, 160–168. [Google Scholar]
- Liu, X.; Liu, Z.; Fei, B.; Cai, Z.; Jiang, Z.; Liu, X. Comparative properties of bamboo and rice straw pellets. BioResources 2013, 8, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Said, N.; Abdel Daiem, M.M.; García-Maraver, A.; Zamorano, M. Influence of densification parameters on quality properties of rice straw pellets. Fuel Process. Technol. 2015, 138, 56–64. [Google Scholar] [CrossRef]
- Moliner, C.; Bove, D.; Bosio, B.; Ribes, A.; Arato, E. Feasibility studies on the energy valorisation of agricultural residues using Aspen Plus (c). In Proceedings of the European Biomass Conference and Exhibition, Viena, Austria, 1–4 June 2015; pp. 803–809. [Google Scholar]
- Aspen Technology, Inc. Available online: https://web.ist.utl.pt/ist11038/acad/Aspen/AspUserGuide10.pdf (accessed on 23 August 2018).
- Moliner, C.; Bosio, B.; Molins, C.; Campins, P.; Ribes, A.; Arato, E. From rice straw residues to energy and silica-based products. In Proceedings of the 6th International Symposium on Energy from Biomass and Waste, Venice, Italy, 14–17 November 2016. [Google Scholar]



| ρapparent (g/L) | ρbulk (g/L) | ε (−) | ρbulk-pellets (g/L) [36] |
|---|---|---|---|
| 239 | 56 | 0.76 | 600 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moliner, C.; Teruel-Juanes, R.; Primaz, C.T.; Badia, J.D.; Bosio, B.; Campíns-Falcó, P.; Molíns-Legua, C.; Hernandez, F.; Sanjuan-Navarro, L.; Madramany, P.; et al. Reduction of Nitrates in Waste Water through the Valorization of Rice Straw: LIFE LIBERNITRATE Project. Sustainability 2018, 10, 3007. https://doi.org/10.3390/su10093007
Moliner C, Teruel-Juanes R, Primaz CT, Badia JD, Bosio B, Campíns-Falcó P, Molíns-Legua C, Hernandez F, Sanjuan-Navarro L, Madramany P, et al. Reduction of Nitrates in Waste Water through the Valorization of Rice Straw: LIFE LIBERNITRATE Project. Sustainability. 2018; 10(9):3007. https://doi.org/10.3390/su10093007
Chicago/Turabian StyleMoliner, Cristina, Roberto Teruel-Juanes, Carmem T. Primaz, Jose David Badia, Barbara Bosio, Pilar Campíns-Falcó, Carmen Molíns-Legua, Francesc Hernandez, Lorenzo Sanjuan-Navarro, Plàcid Madramany, and et al. 2018. "Reduction of Nitrates in Waste Water through the Valorization of Rice Straw: LIFE LIBERNITRATE Project" Sustainability 10, no. 9: 3007. https://doi.org/10.3390/su10093007
APA StyleMoliner, C., Teruel-Juanes, R., Primaz, C. T., Badia, J. D., Bosio, B., Campíns-Falcó, P., Molíns-Legua, C., Hernandez, F., Sanjuan-Navarro, L., Madramany, P., Morán, J., Castro, J., Sanchis, F. J., Martínez, J. D., Hiddink, F., Ribes-Greus, A., & Arato, E. (2018). Reduction of Nitrates in Waste Water through the Valorization of Rice Straw: LIFE LIBERNITRATE Project. Sustainability, 10(9), 3007. https://doi.org/10.3390/su10093007










