Woody Encroachment as a Social-Ecological Regime Shift
Abstract
1. Introduction
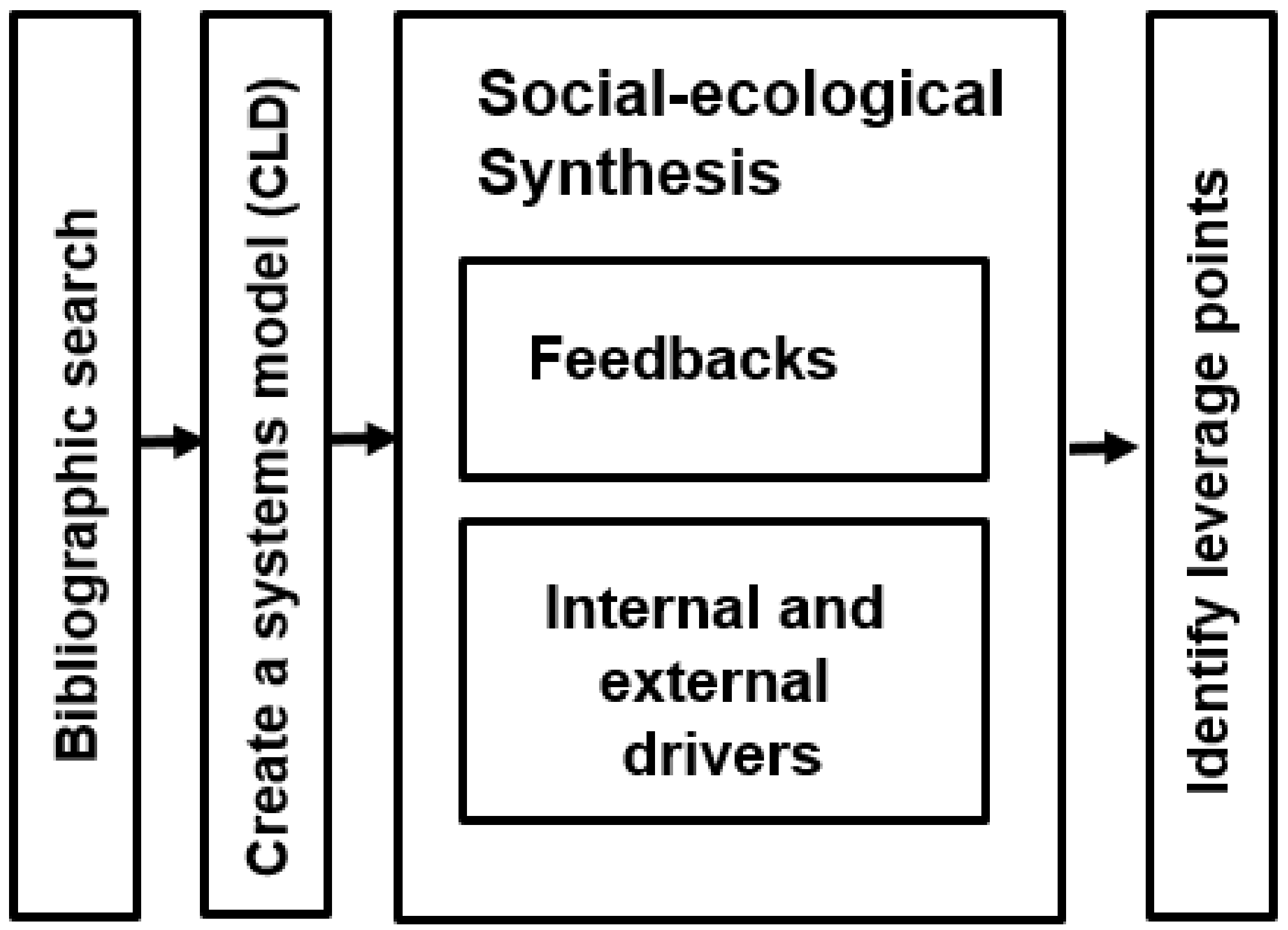
2. Methods
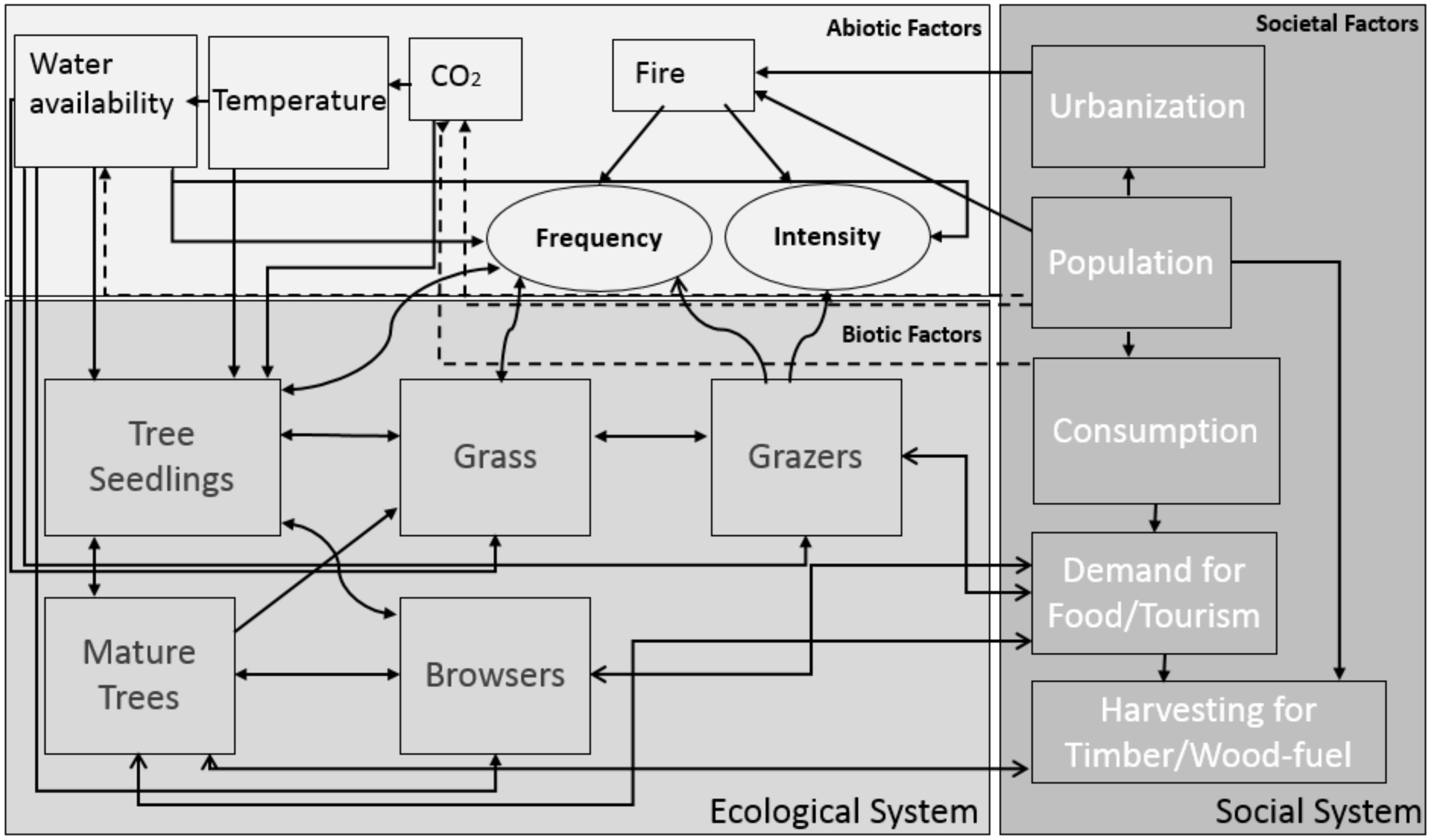
3. Regime Shift Synthesis
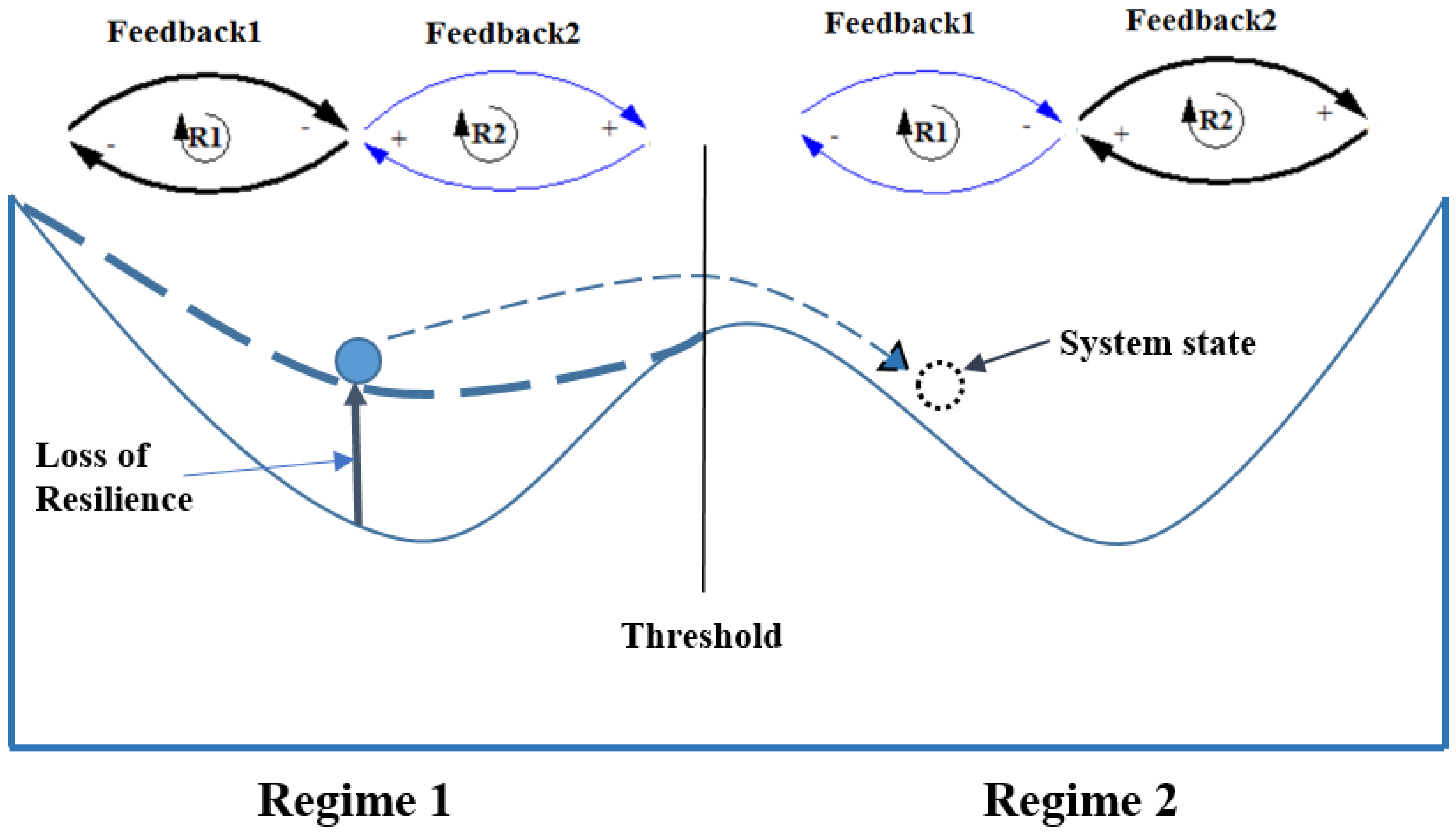
3.1. Alternative Regimes: Grassy and Woody
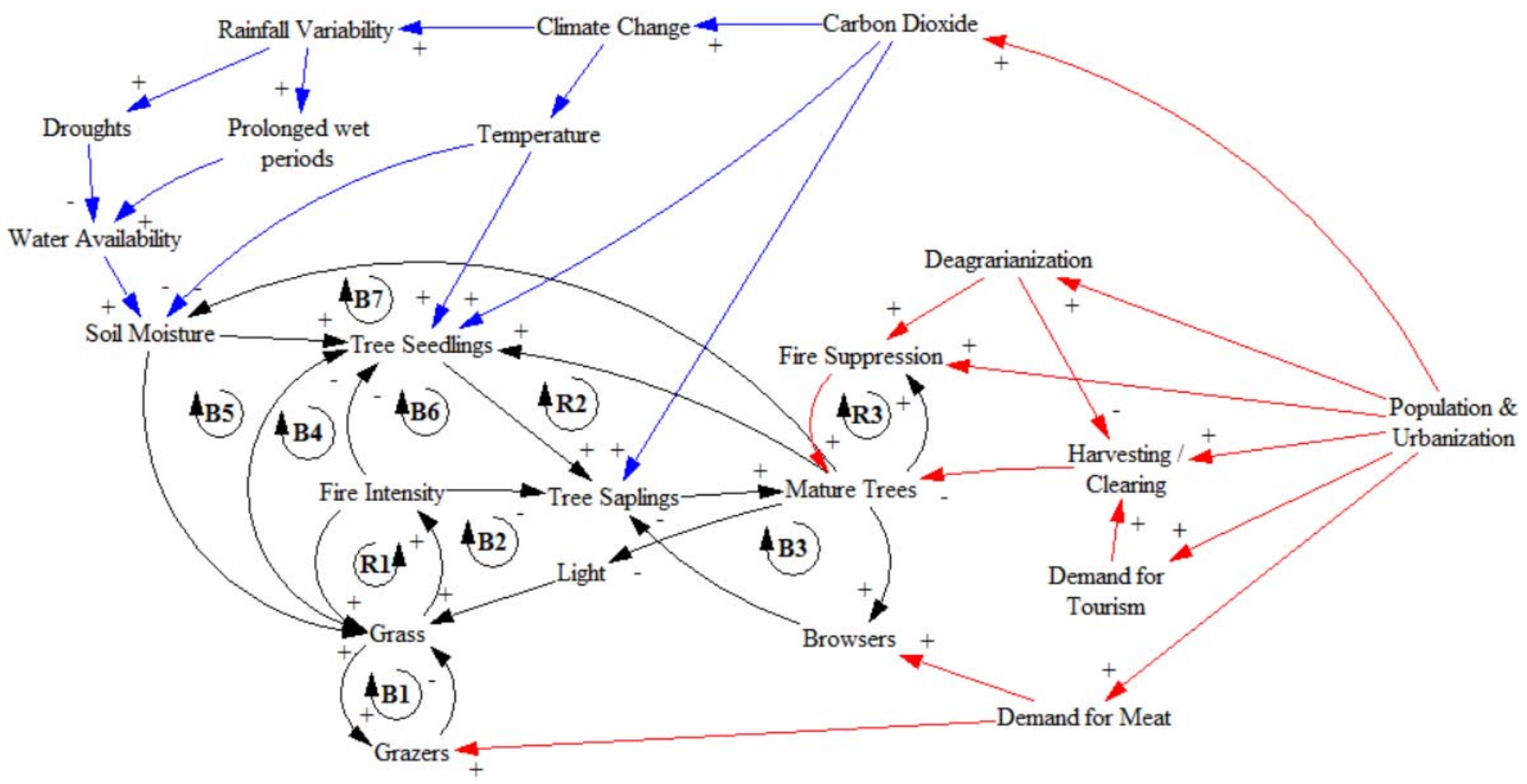
3.2. Feedback Mechanisms
3.2.1. Grassy Regime
3.2.2. Woody Regime
3.3. Drivers of Woody Encroachment
3.3.1. Internal System Changes
Tree Density
Grazers and Browsers
Soil Moisture
3.3.2. External Drivers
Population Growth and Urbanization
Land-Use, Institutional Arrangements and Worldviews
Carbon Dioxide
3.3.3. Shocks
3.4. Management Options and Leverage Points
4. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- O’Connor, T.G.; Puttick, J.R.; Hoffman, M.T. Bush Encroachment in Southern Africa: Changes and Causes. Afr. J. Range Forage Sci. 2014, 31, 67–88. [Google Scholar] [CrossRef]
- Anadón, J.D.; Sala, O.E.; Turner, B.L.; Bennett, E.M.; Anadon, J.D.; Sala, O.E.; Turner, B.L.; Bennett, E.M. Effect of Woody-Plant Encroachment on Livestock Production in North and South America. Proc. Natl. Acad. Sci. USA 2014, 111, 12948–12953. [Google Scholar] [CrossRef] [PubMed]
- Bond, W.J.; Midgley, G.F. Carbon Dioxide and the Uneasy Interactions of Trees and Savannah Grasses. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012, 367, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Twidwell, D.; Fuhlendorf, S.D.; Taylor, C.A.; Rogers, W.E. Refining Thresholds in Coupled Fire-Vegetation Models to Improve Management of Encroaching Woody Plants in Grasslands. J. Appl. Ecol. 2013, 50, 603–613. [Google Scholar] [CrossRef]
- Ratnam, J.; Bond, W.J.; Fensham, R.J.; Hoffmann, W.A.; Archibald, S.; Lehmann, C.E.R.; Anderson, M.T.; Higgins, S.I.; Sankaran, M. When Is a “forest” a Savanna, and Why Does It Matter? Glob. Ecol. Biogeogr. 2011, 20, 653–660. [Google Scholar] [CrossRef]
- Honda, E.A.; Durigan, G.; Bowman, D.; Walsh, A.; Milne, D.; Roques, K.; O’Connor, T.; Watkinson, A.; Kulmatiski, A.; Beard, K.; et al. Woody Encroachment and Its Consequences on Hydrological Processes in the Savannah. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2016, 371, 115–133. [Google Scholar] [CrossRef] [PubMed]
- Chidumayo, E.N.; Gumbo, D.J. The Dry Forests and Woodlands of Africa: Managing for Products and Services; Earthscan: London, UK, 2010. [Google Scholar]
- Wigley, B.J.; Bond, W.J.; Hoffman, M.T. Bush Encroachment under Three Contrasting Land-Use Practices in a Mesic South African Savanna. Afr. J. Ecol. 2009, 47, 62–70. [Google Scholar] [CrossRef]
- Gray, E.F.; Bond, W.J. Will Woody Plant Encroachment Impact the Visitor Experience and Economy of Conservation Areas? Koedoe 2013, 55, 1–9. [Google Scholar] [CrossRef]
- Reyers, B.; Biggs, R.; Cumming, G.S.; Elmqvist, T.; Hejnowicz, A.P.; Polasky, S. Getting the Measure of Ecosystem Services: A Social–ecological Approach. Front. Ecol. Environ. 2013, 11, 268–273. [Google Scholar] [CrossRef]
- Archibald, S. African Grazing Lawns: How Fire, Rainfall, and Grazer Numbers Interact to Affect Grass Community States. J. Wildl. Manag. 2013, 72, 492–501. [Google Scholar] [CrossRef]
- Higgins, S.I.; Bond, W.J.; February, E.C.; Bronn, A.; Euston-Brown, D.I.W.; Enslin, B.; Govender, N.; Rademan, L.; O’Regan, S.; Potgieter, A.L.F.; et al. Effects of Four Decades of Fire Manipulation on Woody Vegetation Structure in Savanna. Ecology 2007, 88, 1119–1125. [Google Scholar] [CrossRef] [PubMed]
- Sankaran, M.; Ratnam, J.; Hanan, N. Woody Cover in African Savannas: The Role of Resources, Fire and Herbivory. Glob. Ecol. Biogeogr. 2008, 17, 236–245. [Google Scholar] [CrossRef]
- Buitenwerf, R.; Bond, W.J.; Stevens, N.; Trollope, W.S.W. Increased Tree Densities in South African Savannas: >50 Years of Data Suggests CO2 as a Driver. Glob. Chang. Biol. 2012, 18, 675–684. [Google Scholar] [CrossRef]
- Scheffer, M.; Carpenter, S.; Foley, J.A.; Folke, C.; Walker, B. Catastrophic Shifts in Ecosystems. Nature 2001, 413, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Folke, C.; Carpenter, S.; Walker, B.; Scheffer, M.; Elmqvist, T.; Gunderson, L.; Holling, C.S. Regime Shifts, Resilience, and Biodiversity in Ecosystem Management. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 557–581. [Google Scholar] [CrossRef]
- Walker, B.; Holling, C.S.; Carpenter, S.R.; Kinzig, A. Resilience, Adaptability and Transformability in Social—Ecological Systems. Ecol. Soc. 2004, 9, 5. [Google Scholar] [CrossRef]
- Levin, S.; Xepapadeas, T.; Crépin, A.-S.; Norberg, J.; de Zeeuw, A.; Folke, C.; Hughes, T.; Arrow, K.; Barrett, S.; Daily, G.; et al. Social-Ecological Systems as Complex Adaptive Systems: Modeling and Policy Implications. Environ. Dev. Econ. 2012, 18, 111–132. [Google Scholar] [CrossRef]
- Levin, S.A. Ecosystems and the Biosphere as Complex Adaptive Systems. Ecosystems 1998, 1, 431–436. [Google Scholar] [CrossRef]
- Lade, S.J.; Niiranen, S.; Hentati-Sundberg, J.; Blenckner, T.; Boonstra, W.J.; Orach, K.; Quaas, M.F.; Österblom, H.; Schlüter, M. An Empirical Model of the Baltic Sea Reveals the Importance of Social Dynamics for Ecological Regime Shifts. Proc. Natl. Acad. Sci. USA 2015, 112, 11120–11125. [Google Scholar] [CrossRef] [PubMed]
- Meadows, D. Thinking in Systems: A Primer; Chelsea Green Publishing: London, UK, 2008. [Google Scholar]
- Hull, V.; Tuanmu, M.-N.; Liu, J. Synthesis of Human-Nature Feedbacks. Ecol. Soc. 2015, 20, 17. [Google Scholar] [CrossRef]
- Crépin, A.-S.; Biggs, R.; Polasky, S.; Troell, M.; de Zeeuw, A. Regime Shifts and Management. Ecol. Econ. 2012, 84, 15–22. [Google Scholar] [CrossRef]
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Synthesis; Island Press: Washington, DC, USA, 2005. [Google Scholar]
- Biggs, R.; Blenckner, T.; Folke, C.; Gordon, L.; Norström, A.; Nyström, M.; Peterson, G. Regime Shifts. In Encyclopedia of Theoretical Ecology; Hastings, A., Gross, L., Eds.; University of California Press: Ewing, NJ, USA, 2012; pp. 609–617. [Google Scholar]
- Biggs, R.; Schlüter, M.; Schoon, M.L. Principles for Building Resilience: Sustaining Ecosystem Services in Social-Ecological Systems; Biggs, R., Schlüter, M., Schoon, M.L., Eds.; Cambridge University Press: Cambridge, UK, 2015. [Google Scholar]
- Sankaran, M.; Hanan, N.P.; Scholes, R.J.; Ratnam, J.; Augustine, D.J.; Cade, B.S.; Gignoux, J.; Higgins, S.I.; Le Roux, X.; Ludwig, F.; et al. Determinants of Woody Cover in African Savannas. Nature 2005, 438, 846–849. [Google Scholar] [CrossRef] [PubMed]
- Staver, A.C.; Archibald, S.; Levin, S.A. The Global Extent and Determinants of Savanna and Forest as Alternative Biome States. Science 2011, 334, 230–232. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, C.E.R.; Archibald, S.A.; Hoffmann, W.A.; Bond, W.J. Deciphering the Distribution of the Savanna Biome. New Phytol. 2011, 191, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Scholes, R.J. Convex Relationships in Ecosystems Containing Mixtures of Trees and Grass. Environ. Resour. Econ. 2003, 26, 559–574. [Google Scholar] [CrossRef]
- Higgins, S.I.; Bond, W.J.; Trollope, W.S.W. Fire Resprouting and Variability a Recipe for Grass Tree Coexistence in Savanna. J. Ecol. 2000, 88, 213–229. [Google Scholar] [CrossRef]
- Rietkerk, M.; Dekker, S.C.; de Ruiter, P.C.; van de Koppel, J. Self-Organized Patchiness and Catastrophic Shifts in Ecosystems. Science 2004, 305, 1926–1929. [Google Scholar] [CrossRef] [PubMed]
- Bond, W.J.; Keeley, J.E. Fire as a Global ‘Herbivore’: The Ecology and Evolution of Flammable Ecosystems. Trends Ecol. Evol. 2005, 20, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Van Auken, O.W. Causes and Consequences of Woody Plant Encroachment into Western North American Grasslands. J. Environ. Manag. 2009, 90, 2931–2942. [Google Scholar] [CrossRef] [PubMed]
- Staver, A.C.; Archibald, S.; Levin, S. Tree Cover in Sub-Saharan Africa: Rainfall and Fire Constrain Forest and Savanna as Alternative Stable States. Ecology 2011, 92, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Archibald, S.; Roy, D.P.; van Wilgen, B.W.; Scholes, R.J. What Limits Fire? An Examination of Drivers of Burnt Area in Southern Africa. Glob. Chang. Biol. 2009, 15, 613–630. [Google Scholar] [CrossRef]
- Archibald, S.; Staver, A.C.; Levin, S.A. Evolution of Human-Driven Fire Regimes in Africa. Proc. Natl. Acad. Sci. USA 2012, 109, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Hedenus, F.; Wirsenius, S.; Johansson, D.J.A. The Importance of Reduced Meat and Dairy Consumption for Meeting Stringent Climate Change Targets. Clim. Chang. 2014, 124, 79–91. [Google Scholar] [CrossRef]
- Bennett, J.E. Institutions and Governance of Communal Rangelands in South Africa. Afr. J. Range Forage Sci. 2013, 30, 77–83. [Google Scholar] [CrossRef]
- Fernandez, R.J.; Archer, E.R.M.; Ash, A.J.; Dowlatabadi, H.; Hiernaux, H.P.H.Y.; Reynolds, J.F.; Hvogel, C.; Walker, B.H.; Wiegand, T. Degradation and Recovery in Socio-Ecological Systems: A View from the Household/Farm Level. In Global Desertification: Do Humans Cause Deserts? Reynolds, J., Stafford Smith, D., Eds.; Dahlem University: Berlin, Germany, 2002; pp. 297–323. [Google Scholar]
- Kraaij, T.; Ward, D. Effects of Rain, Nitrogen, Fire and Grazing on Tree Recruitment and Early Survival in Bush-Encroached Savanna, South Africa. Plant Ecol. 2006, 186, 235–246. [Google Scholar] [CrossRef]
- Joubert, D.F.; Rothauge, A.; Smit, G.N. A Conceptual Model of Vegetation Dynamics in the Semiarid Highland Savanna of Namibia, with Particular Reference to Bush Thickening by Acacia Mellifera. J. Arid Environ. 2008, 72, 2201–2210. [Google Scholar] [CrossRef]
- Dohn, J.; Dembélé, F.; Karembé, M.; Moustakas, A.; Amévor, K.A.; Hanan, N.P. Tree Effects on Grass Growth in Savannas: Competition, Facilitation and the Stress-Gradient Hypothesis. J. Ecol. 2013, 101, 202–209. [Google Scholar] [CrossRef]
- Wiegand, K.; Saltz, D.; Ward, D. A Patch-Dynamics Approach to Savanna Dynamics and Woody Plant Encroachment—Insights from an Arid Savanna. Perspect. Plant Ecol. Evol. Syst. 2006, 7, 229–242. [Google Scholar] [CrossRef]
- Meyer, K.M.; Wiegand, K.; Ward, D.; Moustakas, A. SATCHMO: A Spatial Simulation Model of Growth, Competition, and Mortality in Cycling Savanna Patches. Ecol. Model. 2007, 209, 377–391. [Google Scholar] [CrossRef]
- Sage, R.F. The Evolution of C4 Photosynthesis. New Phytol. 2004, 161, 341–370. [Google Scholar] [CrossRef]
- Sankaran, M.; Ratnam, J.; Hanan, N.P. Tree-Grass Coexistence in Savannas Revisited—Insights from an Examination of Assumptions and Mechanisms Invoked in Existing Models. Ecol. Lett. 2004, 7, 480–490. [Google Scholar] [CrossRef]
- Ward, D.; Wiegand, K.; Getzin, S. Walter’s Two-Layer Hypothesis Revisited: Back to the Roots! Oecologia 2013, 172, 617–630. [Google Scholar] [CrossRef] [PubMed]
- Scholes, R.J.; Archer, S.R. Tree Grass Interactions in Savannas. Annu. Rev. Ecol. Syst. 1997, 28, 517–544. [Google Scholar] [CrossRef]
- Van Langevelde, F.; van de Vijver, C.A.D. M.; Kumar, L.; van de Koppel, J.; de Ridder, N.; van Andel, J.; Skidmore, A.K.; Hearne, J.W.; Stroosnijder, L.; Bond, W.J.; et al. Effects of fire and herbivory on the stability of savanna ecosystems. Ecology 2003, 84, 337–350. [Google Scholar] [CrossRef]
- D’Antonio, C.M.; Bowman, D.M.J.S.; Doyle, J.C.; Balch, J.K.; Carlson, J.M.; Cochrane, M.A.; Artaxo, P.; DeFries, R.S.; Harrison, S.P.; Bond, W.J. Fire in the Earth System. Science 2009, 324, 481–484. [Google Scholar]
- Skarpe, C. Shrub Layer Dynamics under Different Herbivore Densities in an Arid Savanna, Botswana. J. Appl. Ecol. 1990, 27, 873–885. [Google Scholar] [CrossRef]
- Roques, K.G.G.; O’Connor, T.G.G.; Watkinson, A.R.R. Dynamics of Shrub Encroachment in an African Savanna: Relative Influences of Fire, Herbivory, Rainfall and Density Dependence. J. Appl. Ecol. 2001, 38, 268–280. [Google Scholar] [CrossRef]
- Ward, D.; Esler, K.J. What Are the Effects of Substrate and Grass Removal on Recruitment of Acacia Mellifera Seedlings in a Semi-Arid Environment? Plant Ecol. 2011, 212, 245–250. [Google Scholar] [CrossRef]
- February, E.; Higgins, S.I. Influence of Competition and Rainfall Manipulation on the Growth Responses of Savanna Trees and Grasses Responses of Savanna Trees and Grasses. Ecology 2013, 94, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Tedder, M.; Kirkman, K.; Morris, C.; Fynn, R. Tree-Grass Competition along a Catenal Gradient in a Mesic Grassland, South Africa. Grassl. Sci. 2014, 60, 1–8. [Google Scholar] [CrossRef]
- Bond, W.J. What Limits Trees in C 4 Grasslands and Savannas? Annu. Rev. Ecol. Evol. Syst. 2008, 39, 641–659. [Google Scholar] [CrossRef]
- O’Connor, T.G.; Goodman, P.S.; Clegg, B.; O’Connor, T.G.; Goodman, P.S.; Clegg, B.; O’Connor, T.G.; Goodman, P.S.; Clegg, B. A Functional Hypothesis of the Threat of Local Extirpation of Woody Plant Species by Elephant in Africa. Biol. Conserv. 2007, 136, 329–345. [Google Scholar] [CrossRef]
- Staver, A.C.; Bond, W.J.; Stock, W.D.; van Rensburg, S.J.; Waldram, M.S. Browsing and Fire Interact to Suppress Tree Density in an African Savanna. Ecol. Appl. 2009, 19, 1909–1919. [Google Scholar] [CrossRef] [PubMed]
- Staver, A.C.; Bond, W.J. Is There a ‘Browse Trap’? Dynamics of Herbivore Impacts on Trees and Grasses in an African Savanna. J. Ecol. 2014, 102, 595–602. [Google Scholar] [CrossRef]
- Stevens, N.; Erasmus, B.F.N.; Archibald, S.; Bond, W.J. Woody Encroachment over 70 Years in South African Savannahs: Overgrazing, Global Change or Extinction Aftershock? Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20150437. [Google Scholar] [CrossRef] [PubMed]
- Daskin, J.H.; Stalmans, M.; Pringle, R.M. Ecological Legacies of Civil War: 35-Year Increase in Savanna Tree Cover Following Wholesale Large-Mammal Declines. J. Ecol. 2016, 104, 79–89. [Google Scholar] [CrossRef]
- Skowno, A.L.; Thompson, M.W.; Hiestermann, J.; Ripley, B.; West, A.G.; Bond, W.J. Woodland Expansion in South African Grassy Biomes Based on Satellite Observations (1990–2013): General Patterns and Potential Drivers. Glob. Chang. Biol. 2017, 23, 2358–2369. [Google Scholar] [CrossRef] [PubMed]
- Jolly, W.M.; Nemani, R.; Running, S.W. A Generalized, Bioclimatic Index to Predict Foliar Phenology in Response to Climate. Glob. Chang. Biol. 2005, 11, 619–632. [Google Scholar] [CrossRef]
- Ward, D. Do We Understand the Causes of Bush Encroachment in African Savannas? Afr. J. Range Forage Sci. 2005, 22, 101–105. [Google Scholar] [CrossRef]
- Vadigi, S.; Ward, D. Herbivory Effects on Saplings Are Influenced by Nutrients and Grass Competition in a Humid South African Savanna. Perspect. Plant Ecol. Evol. Syst. 2014, 16, 11–20. [Google Scholar] [CrossRef]
- Shackleton, C.; Shackleton, S.; Netshiluvhi, T.; Mathabela, F. The Contribution and Direct-Use Value of Livestock to Rural Livelihoods in the Sand River Catchment, South Africa. Afr. J. Range Forage Sci. 2005, 22, 127–140. [Google Scholar] [CrossRef]
- Castley, J.G.; Boshoff, A.F.; Kerley, G.I.H. Compromising South Africa’s Natural Biodiversity—Inappropriate Herbivore Introductions. S. Afr. J. Sci. 2001, 97, 344–348. [Google Scholar]
- Maciejewski, K.; Kerley, G.I.H. Understanding Tourists’ Preference for Mammal Species in Private Protected Areas: Is There a Case for Extralimital Species for Ecotourism? PLoS ONE 2014, 9, e88192. [Google Scholar] [CrossRef] [PubMed]
- Guldemond, R.; van Aarde, R. A Meta-Analysis of the Impact of African Elephants on Savanna Vegetation. J. Wildl. Manag. 2008, 72, 892–899. [Google Scholar]
- Banks, D.I.; Griffin, N.J.; Shackleton, C.M.; Shackleton, S.E.; Mavrandonis, J.M. Wood Supply and Demand around Two Rural Settlements in a Semi-Arid Savanna, South Africa. Biomass Bioenergy 1996, 11, 319–331. [Google Scholar] [CrossRef]
- Madubansi, M.; Shackleton, C.M. Changes in Fuelwood Use and Selection Following Electrification in the Bushbuckridge Lowveld, South Africa. J. Environ. Manag. 2007, 83, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Christiaensen, L.; Todo, Y. Poverty Reduction during the Rural–Urban Transformation—The Role of the Missing Middle. World Dev. 2014, 63, 43–58. [Google Scholar] [CrossRef]
- Christiaensen, L.; de Weerdt, J.; Todo, Y. Urbanization and Poverty Reduction: The Role of Rural Diversification and Secondary Towns. Agric. Econ. 2013, 44, 435–447. [Google Scholar] [CrossRef]
- Shackleton, R.; Shackleton, C.; Shackleton, S.; Gambiza, J. Deagrarianisation and Forest Revegetation in a Biodiversity Hotspot on the Wild Coast, South Africa. PLoS ONE 2013, 8, e76939. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.T. Changing Patterns of Rural Land Use and Land Cover in South Africa and Their Implications for Land Reform. J. South. Afr. Stud. 2014, 40, 707–725. [Google Scholar] [CrossRef]
- Lynam, T.; Brown, K. Mental Models in Human-Environment Interactions: Theory, Policy Implications, and Methodological Explorations. Ecol. Soc. 2012, 17, 24. [Google Scholar] [CrossRef]
- Van Wilgen, B.W.; Forsyth, G.G.; de Klerk, H.; Das, S.; Khuluse, S.; Schmitz, P. Fire Management in Mediterranean-Climate Shrublands: A Case Study from the Cape Fynbos, South Africa. J. Appl. Ecol. 2010, 47, 631–638. [Google Scholar] [CrossRef]
- Polley, H.W. Implications of Rising Atmospheric Carbon Dioxide Concentration for Rangelands. J. Range Manag. 1997, 50, 562–577. [Google Scholar] [CrossRef]
- Stevens, N.; Seal, C.E.; Archibald, S.; Bond, W. Increasing Temperatures Can Improve Seedling Establishment in Arid-Adapted Savanna Trees. Oecologia 2014, 175, 1029–1040. [Google Scholar] [CrossRef] [PubMed]
- Buitenwerf, R.; Rose, L.; Higgins, S.I. Three Decades of Multi-Dimensional Change in Global Leaf Phenology. Nat. Clim. Chang. 2015, 5, 364–368. [Google Scholar] [CrossRef]
- Leakey, A.D.B.; Ainsworth, E.A.; Bernacchi, C.J.; Rogers, A.; Long, S.P.; Ort, D.R. Elevated CO2 Effects on Plant Carbon, Nitrogen, and Water Relations: Six Important Lessons from FACE. J. Exp. Bot. 2009, 60, 2859–2876. [Google Scholar] [CrossRef] [PubMed]
- Nowak, R.S.; Ellsworth, D.S.; Smith, S.D. Functional Responses of Plants to Elevated Atmospheric CO2- Do Photosynthetic and Productivity Data from FACE Experiments Support Early Predictions? New Phytol. 2004, 162, 253–280. [Google Scholar] [CrossRef]
- Bond, W.J.; Midgley, G.F. A Proposed CO2-Controlled Mechanism of Woody Plant Invasion in Grasslands and Savannas. Glob. Chang. Biol. 2000, 6, 865–869. [Google Scholar] [CrossRef]
- Crowlely, G.; Garnett, S.; Shepard, S. Impact of Storm-Burning on Melaleuca Viridiflora Invasion of Grasslands and Grassy Woodlands on Cape York Peninsula, Australia. Aust. Ecol. 2009, 34, 196–209. [Google Scholar] [CrossRef]
- Smit, I.P.J.; Asner, G.P.; Govender, N.; Vaughn, N.R.; van Wilgen, B.W. An Examination of the Potential Efficacy of High-Intensity Fires for Reversing Woody Encroachment in Savannas. J. Appl. Ecol. 2016, 53, 1623–1633. [Google Scholar] [CrossRef]
- Dublin, H.T. Decline of the Mara Woodlands: The Role of Fire and Elephants. Ph.D. Thesis, University of British Columbia, Vancouver, BC, Canada, 1986. [Google Scholar]
- Smit, G.N. An Approach to Tree Thinning to Structure Southern African Savannas for Long-Term Restoration from Bush Encroachment. J. Environ. Manag. 2004, 71, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Angassa, A.; Oba, G. Bush Encroachment Control Demonstrations in Southern Ethiopia: 1. Woody Species Survival Strategies with Implications for Herder Land Management. Afr. J. Ecol. 2009, 47, 63–76. [Google Scholar] [CrossRef]
- Wigley, B.J.; Bond, W.J.; Hoffman, M.T. Thicket Expansion in a South African Savanna under Divergent Land Use: Local vs. Global Drivers? Glob. Chang. Biol. 2010, 16, 964–976. [Google Scholar] [CrossRef]
- Moncrieff, G.R.; Scheiter, S.; Bond, W.J.; Higgins, S.I. Increasing Atmospheric CO2 Overrides the Historical Legacy of Multiple Stable Biome States in Africa. New Phytol. 2014, 201, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Bond, W.J.; Woodward, F.I.; Midgley, G.F. The Global Distribution of Ecosystems in a World without Fire. New Phytol. 2005, 165, 525–538. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luvuno, L.; Biggs, R.; Stevens, N.; Esler, K. Woody Encroachment as a Social-Ecological Regime Shift. Sustainability 2018, 10, 2221. https://doi.org/10.3390/su10072221
Luvuno L, Biggs R, Stevens N, Esler K. Woody Encroachment as a Social-Ecological Regime Shift. Sustainability. 2018; 10(7):2221. https://doi.org/10.3390/su10072221
Chicago/Turabian StyleLuvuno, Linda, Reinette Biggs, Nicola Stevens, and Karen Esler. 2018. "Woody Encroachment as a Social-Ecological Regime Shift" Sustainability 10, no. 7: 2221. https://doi.org/10.3390/su10072221
APA StyleLuvuno, L., Biggs, R., Stevens, N., & Esler, K. (2018). Woody Encroachment as a Social-Ecological Regime Shift. Sustainability, 10(7), 2221. https://doi.org/10.3390/su10072221




