Abstract
The growth of urban centers, along with the fragmentation of natural habitats, can interfere with the distribution pattern of species and their abundance, thus compromising the conservation of urban biodiversity. Principal players in this biodiversity are arthropods that help to decompose litterfall, favoring the recycling of nutrients and, hence, are an important part in sustaining the forest fragments that remain in urban areas. Therefore, it is important to study arthropod biodiversity in green urban areas, especially those areas where litterfall management is an important part of maintaining biodiversity. Accordingly, this study evaluated arthropod diversity associated with litterfall in three urban forests with different size and litterfall management practices, including Água Branca Park (ABP) and Tietê Ecological Park (TEP)—Núcleo Engenheiro Goulart in São Paulo City and Chico Mendes Park (CMP) in Osasco City, all belonging to the Metropolitan Region of São Paulo (MRSP), Brazil. Four litterfall harvests were carried out in each park between April and August of 2015 with twelve samples collected with a wood mold (30-cmx30-cm) at randomly points on the forest floor. The collected material was then screened in the laboratory and the arthropods were visually separated and preserved in alcohol 70%. Arthropods were classified by the order to which they belonged. Litterfall was dried in a forced air oven at 65 °C for seven days. Dried litterfall was then separated into leaves, branches, reproductive parts and miscellaneous fragments and weighed. Arthropod diversity was measured by Shannon, Margalef and Pielou indexes and non-metric multidimensional scaling analysis (NMDS) using the Manhattan distance index. Litterfall weight was compared by one-way ANOVA. The orders Hymenoptera, Collembola and Isopoda were dominant in CMP, ABP and TEP, respectively but multiple rare orders had a presence in all parks, albeit at different abundance. NMDS showed abundance similarity among the parks; however, TEP showed greater richness, Shannon diversity and evenness. The fractions of leaf litterfall and reproductive parts were different between the CMP and TEP. Overall, our results confirm that neither litterfall management, nor park size, is a final determinant of arthropod distribution, even though the abundance of dominant species was shown to differ in each park.
1. Introduction
Urban forests are small forest fragments inside urban environments. They form green patches in an immense gray matrix and play an important role in maintaining the biodiversity of species that occupy and/or transit into these areas [1,2]. These vegetation fragments in São Paulo City are managed locally and designated as areas for leisure and sports, as well as biodiversity conservation. Like larger forest fragments, these areas are subjected to such stresses as fire, pollution, urban growth and even vandalism [3,4].
Such stresses promote a particular dynamic of edaphic biodiversity in these forests and it can interfere with nutrient recycling of litterfall, an important part of organic matter that supports energy flow in ecosystems [5,6,7,8]. The largest fraction of litterfall in tropical regions consists of leaves, accounting for approximately 70% of the organic matter returned to the forest soils [9].
In urban areas, such as parks and forest fragments, litterfall can be managed according to the requirements of the area. In some parks, litterfall is swept and taken away from the forest, while in other parks, litterfall remains a part of forest soil. These different litterfall management practices can affect the dynamics of arthropods that feed, hide and hunt inside these fractions of deciduous material [10].
Several authors have reported on the benefits of ecosystem services associated with urban forests, such as groundwater recharge, surface runoff of rainwater, retention of particulate matters emitted by motors, as well as thermal comfort and carbon uptake [11]. In addition to maintaining the trophic structure of resistant communities in forest fragments, the insect community can alter the soil structure, thereby playing its own key role in providing such ecosystem services. Arthropods live in soil litterfall, feed on leaves, flowers and fruits that fall to the forest floor and then disperse this biological material to be decomposed by fungi and bacteria, all essential events for forest sustainability [12,13]. Since the arthropod community is essential to sustaining these urban forest fragments, different types of litterfall management practices that affect the dynamics of arthropod communities can certainly affect various ecosystem services [14].
The arthropods community might vary across the biome or ecosystems. Some mesofauna arthropods are predators in forest fragments [15], while organisms belonging to macrofauna have great mobility in the soil, excavating holes and galleries, fragmenting and incorporating organic residues into the soil, as well as fragmenting organic matter for the benefit of other microorganisms. These functions may, however, interfere with the niche of other species and thus affect the decomposition of litterfall, which, in turn, will modify soil structure [16,17,18].
Different edaphic organisms are organized in small patches across the forest floor, resulting in complex population dynamics. Thus, to better understand the structure of these communities, most studies rely on the identification of arthropods by their hierarchically higher taxonomic groups, such as class or order [16]. This type of identification provides an overview of the ecological complexity of soil community and points to functional groups [19].
In the present paper, we focus on three urban forests in the São Paulo Metropolitan Region, São Paulo State, Brazil. Each one represents different litterfall management practices but all are dependent on arthropods for organic matter decomposition and nutrient recycling. Therefore, it is essential to understand how these organisms are organized in urban soils in order to understand how litterfall management practices might affect population dynamics on a micro level and forest biodiversity on a macro level. Therefore, we herein hypothesize that urban forests with different litterfall management practices necessarily present distinct distribution of arthropod abundances, which in turn, will affect litterfall decomposition and nutrient recycling.
2. Material and Methods
2.1. Study Areas
Three urban forests with different litterfall management practices in the Metropolitan Region of São Paulo (MRSP) were selected (Figure 1).
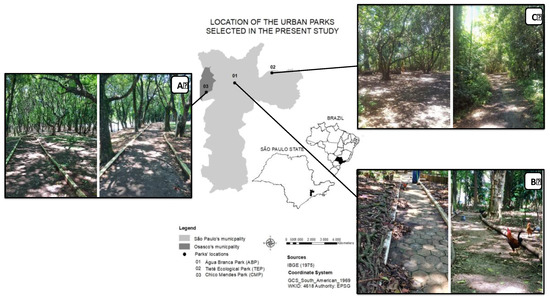
Figure 1.
Map of the study area (São Paulo and Osaco City); ABP—Água Branca Park; TEP—Tietê Ecological Park; CMP—Chico Mendes Park.
Chico Mendes Park, which is located in Osasco City (23°34′15.2″S and 46°47′02.2″W), northeast of the MRSP, covers approximately 11.4 ha of secondary forest and has a predominance of Atlantic Forest native vegetation. The local administrators do not manage the litterfall, and, as a result, it accumulates naturally (Figure 1A). Água Branca Park is a 13.7 ha space located in the Center-West zone of São Paulo City (23°31′48.1″S and 46°40′11.9″W). The park is manmade and is located in an area with intense foot traffic and contaminants from vehicles. The vegetation was planted in 1929 and the forest fragment has approximately 3000 adult trees belonging to 159 species [20]. The park was revitalized and registered in 1996 as a cultural, historical, architectural, touristic, technological and scenic asset by the Council for the Defense of Historic, Archaeological, Artistic and Tourist Heritage of the State of São Paulo (CONDEPHAAT). A particular feature of this park is the presence of wild chickens that control predator populations of edaphic fauna, such as spiders and scorpions, ensuring the safety of visitors while walking or sitting on the litterfall. In this urban forest, the litterfall is removed from the hiking trails and stored in vegetation islands inside the park (Figure 1B). Tietê Ecological Park—Núcleo Engenheiro Goulart is located on the east side of São Paulo City (23°29′41.7″S and 46°31′33.0″W) on the banks of the Tietê River. The park covers an area of 1,400 ha and was implemented in 1976 with the initial purpose of protecting the Tietê River floodplains and the local flora and fauna. This urban forest is located within an environmental preservation area named Várzeas do Tietê. Litterfall produced in the forest is not managed (Figure 1C).
2.2. Litterfall Sampling
Four litterfall harvests were carried out in each park between April and August of 2015, always in the morning, with twelve samples collected with a wood mold (30-cm × 30-cm) at randomly points on the forest floor. The collections were realized in different parts of the forests, including the edge and the core, occurring between 0–3 m far from the hiking trails (under the litter management area, when it occurred) and also in more distant areas, which means outside the litter management area (three or more meters far from the hiking trails). Only the upper layer of litterfall was sampled, without rotating any portion of the O horizon, i.e., the top, organic layer of soil, made up mostly of leaf litter and humus, or living roots. The sampled material was stored in separate plastic bags and properly identified with number, date and site and taken to the laboratory for analysis [9].
2.3. Screening of Litterfall Fractions
The litterfall fractions were dried in an oven with forced air circulation at 65 °C for four days, or until reaching constant weight. The dried material was separated into branches, leaves, reproductive and miscellaneous parts, according to the method of [21,22] and weighed.
2.4. Screening and Identification of Arthropods
Arthropods were visually screened, kept in flasks with 70% alcohol and labeled for identification. The identification was carried out with adult morphotypes that have already undergone stages of molting, at the order level. For taxonomic classification, the superorders Collembola and Acariformes, according to Reference [19], were considered.
2.5. Data Analysis
The Shannon index (indicator of heterogeneity) was used to evaluate the abundances of the taxonomic groups. The Margalef index was used as an indicator of species richness and the Pielou index measured species uniformity [23]. Non-metric multidimensional scaling analysis (NMDS) (NMDS) was performed with the data from the abundances of the taxonomic groups in the three urban forests. Even though the Bray-Curtis distance index is widely used in ecological studies, here the Manhattan coefficient was chosen, owing to the lower stress (<0.15). Dissimilarity between groups was tested with a multivariate analysis of Similarity (One-Way ANOSIM), using the Manhattan distance measure and considering Bonferroni-corrected p value ≤ 0.05 and R value < 1, as significant.
The weight of each 30 × 30 cm2 litterfall sample was converted from grams to kilograms per hectare. One-way ANOVA was used to compare the mean dry mass values of litterfall fractions among the three study sites. The Tukey-Kramer post hoc test was used. All statistical analyses were performed with PAST, ver. 2.17 (Paleontological Statistics) [24].
3. Results
The three forest fragments presented different practices of managing litter dropped on the soil. During the study period, the fallen litter in Água Branca Park was removed from the walking trail and transferred to the forest floor in a vegetated area where visitors could walk and sit. At Chico Mendes Park, visitors were observed walking on the litterfall, a situation that was little observed at Tietê Ecological Park.
To demonstrate the biodiversity of arthropods associated with litterfall, a total of 1176 individuals were sampled, distributed into 19 orders, including 365 individuals belonging to 12 orders in CMP, 330 individuals in 16 orders in ABP and 481 individuals in 17 orders in TEP (Table 1). Three predominant orders were identified among the individuals sampled. The most abundant taxonomic group was Hymenoptera, representing 64% of the total sampled in CMP, 31% in ABP and 16% in TEP. The order Isopoda represented 50% of the individuals sampled in TEP, 12% in CMP and only 2% in ABP. Collembola represented 46% of the orders sampled in ABP; however, the order was less representative in CMP (6%) and TEP (5%). Other taxonomic groups with intermediate abundance were Araneae, Orthoptera, Acariformes and Coleoptera.

Table 1.
Arthropod order and abundance in each urban forest; use of litterfall; classification of the type of fauna based on the average size and functional aspect based on food niche.
The urban forest with the greatest abundance was TEP (n = 481), followed by CMP (n = 365) and ABP (n = 330). This value found in Tietê Ecological Park was significantly higher than in other areas (p > 0.05).
Some individuals sampled do not remain part of the fauna of the soil and litterfall when adults, especially Lepidoptera (Butterflies) in CMP and Diptera (Flies) in CMP and TEP. These individuals participate as edaphic fauna during the larval stage. According to the literature, other individuals identified in this study have not been reported as groups associated with deciduous plant material, such as Mallophaga, Ixodida and Siphonaptera, which are parasites of birds and mammals [25,26].
ABP had the highest proportion of mesofauna groups (55%), while TEP had the highest proportion of macrofauna groups and CMP showed the highest percentage of groups with individuals that can be classified as either macro or mesofauna (Figure 2).
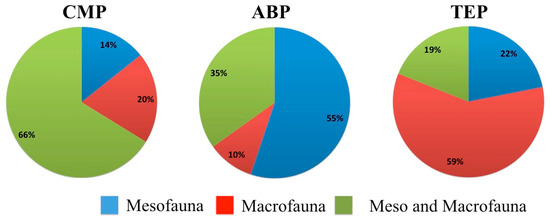
Figure 2.
Relative values of identified orders in the study with individuals of the mesofauna, macrofauna and belonging to both meso- and macrofauna.
The Shannon heterogeneity index showed similar arthropod diversity across the parks (F = 1.731, p > 0.05); with a subtle higher median value in TEP. The Margalef richness index and Pielou evenness index also trended in this direction (F = 0.752, p > 0.05 and F = 2.68, p > 0.05, respectively). The lack of the first quartile of boxplots regarding diversity, richness and uniformity may have resulted from the large number of samples with zero abundance of many taxonomic groups (Figure 3).
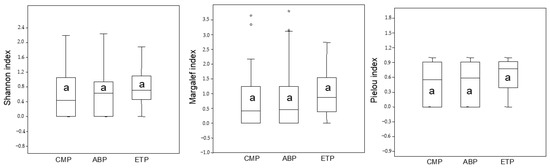
Figure 3.
Box plots showing the median values of diversity indexes analyzed in the study areas. Asterisks indicate extreme values for some samples. Lowercase letters indicate statistical differences between groups (α < 5%).
The Venn diagram (Figure 4) highlights the greatest taxonomic groups occurred in the parks. The order Neuroptera occurred only in ABP. The order Dermaptera occurred in ABP and TEP and the order Amphipoda occurred only in TEP. No exclusive or shared order was observed in CMP.

Figure 4.
Venn diagram highlighting shared abundances among parks. Font size and numbering refer to the abundance of the taxonomic group in fragments of urban forests.
The NMDS diagram (Figure 5) presented stress of 0.14 and highlighted, as demonstrated by the Venn diagram, that most orders reported in the parks had similar abundance distribution, consequently promoting an overlap of the three study sites, since these orders recurred with similar abundances in the parks. This result is supported by statistical inference values of p > 0.05 and R = 0.04346, as determined by ANOSIM. The subtle distancing of sample units among the parks, as highlighted by the abundance of different taxonomic group, was strongly influenced by dominant orders at each site, emphasizing Isopoda in TEP, Hymenoptera in CMP and Collembola in ABP. According to the dominant orders in each park, it was observed that the distribution of abundances in ABP and TEP leaned toward similarity between the two study sites when compared to CMP, which presented greater sample unit distance, mainly influenced by the abundance of Hymenoptera.
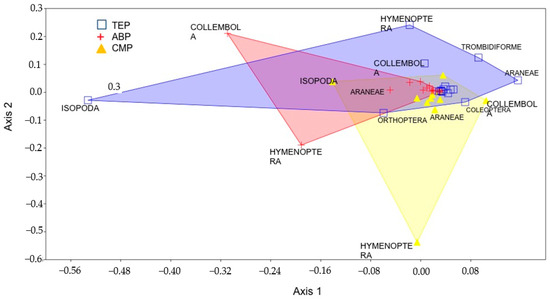
Figure 5.
Diagram of non-metric multidimensional scaling analysis (NMDS) using arthropod abundance and Manhattan distance among the three urban forests.
In order to evaluate the variation in the stocks of resources available for the arthropod niches realization (i.e., hiding, feeding or hunting) the biomass of litterfall fractions was obtained in the three study areas.
CMP had the highest leaf litterfall biomass among the three urban forests, an amount of branches and miscellaneous parts similar to the other two study sites but a greater number of reproductive parts than either ABP or TEP (Figure 6).
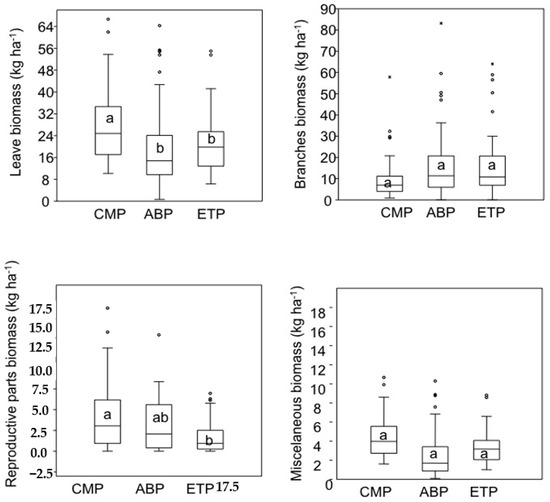
Figure 6.
Box plots showing the median values of litterfall fractions biomass analyzed in the study areas. Asterisks indicate extreme values for some samples. Lowercase letters indicate statistical differences between groups (α < 5%).
4. Discussion
It is common to have a few dominant species in nature, as well as many rare species [27]. Accordingly, each park presented a small group of dominant morphospecies with different functional aspects. The abundance of these taxons reflects the quantity of resources available and the influence of competitors and predators at the site. An example of this relationship is the presence of chickens in ABP, which can control Aranae and Scorpionidae populations, both important predators of the edaphic community [28].
The order with the largest number of individuals in the litterfall of all urban forests was Hymenoptera. This order is mainly represented by the Formicidae family and it has many cosmopolitan species [29]. Representatives of this order are frequently considered engineers of the ecosystem [30]. Environments under greater anthropic disturbance favor the colonization of ant species [31], which may be used as bioindicators of anthropization [32]. Ant individuals use deciduous leaves and branches for feeding and nesting [33] but this niche also plays an important role in the nesting process of other species. Thus, the niche amplitude of Formicidae (Hymenoptera) individuals favors the permanence of more stable populations in urban forest environments since herbivorous, saprophagous and predatory individuals are found in this group (Table 1), increasing the options to obtain food resources.
Studies in tropical forests with litterfall management have shown that arthropod abundance decreases with litterfall removal and increases with its addition when compared to litterfall deposited naturally on forest floor [34,35]. Since management practices at ABP include the removal of litterfall from the walking trails and pavements, moving it to the forest floor, this practice tends to increase the abundance of some taxonomic groups, especially Collembola, which, in this study, showed abundance almost six times higher than CMP and TEP. This order is important to the edaphic trophic web since individuals feed on microorganisms and are predated by other arthropods (Figure 7) [36].
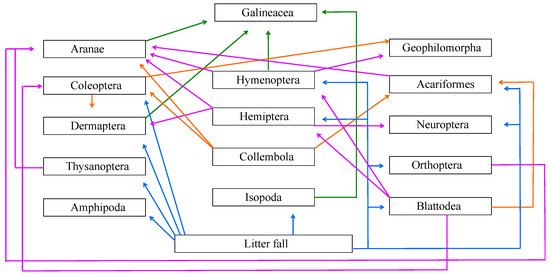
Figure 7.
Diagram of hypothetical arthropod trophic web in urban forests of São Paulo City based on the biodiversity of the three forests of this study.
Studies of trophic cascade involving spiders in the top-down control showed changes in litterfall decomposition when predators were removed, increasing the population of Colembolla [28,37]. Such relationship supports the abundance values found in this study. However, the increase of litterfall might promote an upward control of the community, essentially because the greater availability of resources can neutralize the top-down effect [38].
The most abundant order in TEP was Isopoda, a crustacean with saprophagous habits. Experiments on litterfall consumption by isopods showed that it stimulated microbial activity in the soil, increasing biomass and microbial respiration, resulting in a great availability of macronutrients on the surface of the edaphic system. In general, Isopoda are more sensitive to climatic variations, litterfall quality and air pollutants [39], thus explaining its low abundance at ABP. TEP has a larger area and is surrounded by residential neighborhoods with low buildings, favoring the dispersion of wind and air pollution from vehicles. This forest fragment has a lower canopy in relation to the other areas, creating milder microclimatic conditions, with lower thermal amplitude and higher humidity, favoring the ambient conditions required for the Isopoda development [39].
Five individuals of the Amphipoda order were found in TEP. The only terrestrial family in the order is Talitridae, which is found in environments with a humid microclimate. In Brazil, terrestrial individuals of this family are considered exotic and was probably introduced with exotic plant species used for forestry during last century [40]. Individuals of this family have occurrences registered in São Paulo, Rio de Janeiro and Espírito Santo states [40,41]. The Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Service (IPBES) and the Convention on Biological Biodiversity have posted warnings about the dangers of introducing exotic species based on their frequent competition for resources and lack of natural predators.
The abundance distribution of each morphospecies indicated in the Venn diagram and on NMDS showed that most taxons occurred across all study sites. According to [42], decreasing size of forest fragment correlates with increasing border effect and the influence of external factors. This may explain why medians of diversity, richness and uniformity were found to be higher in TEP, which has a larger preserved area than either CMP or ABP (Figure 3). The extent of border effects may vary from a few meters to the entire area of the fragment, depending on the shape and size of the fragment; however, urban forest fragments do not have typical border effect since their borders are usually managed [43].
ABP and CMP are directly affected by urbanization due to their small area. These areas also have greater exposure to wind, high temperature and low air humidity, factors that may directly or indirectly influence species abundance and distribution [44]. The richness and abundance of some taxons may not be directly related to the extent or complexity of the fragment but they are influenced by other factors, such as habitat heterogeneity, food availability and the microclimate inside of the forest fragment [45]. Thus, different litterfall conditions and management can directly interfere with the distribution of species abundance, which may, in turn, interfere with the patterns of nutrient recycling [46], given the particular ecological niches of arthropod species.
The litterfall composition evaluated showed little biomass difference among the study sites and the biomass of branches and miscellaneous parts were similar. This fact may have favored the realized niche of the found arthropods, thus contributing to the greater composition similarity of these animals. This may be an intrinsic characteristic of the floristic composition of each study site with a predominance of deciduous species. In the alternative, the topography or environment near each forest fragment may cause changes in wind or rainfall intensities, which would in turn, affect floristic composition. As previously reported, the intensity of the border effect is directly related to the area of the fragment [47], which could explain the smaller quantity of leaves and reproductive material stored in TEP and ABP, even considering the small difference in size between CMP and ABP. In addition, litterfall represents one of the nutrient outputs of forest systems, having associations with other ecosystem services, such as local climate regulation, surface runoff of rainwater and maintenance of ecosystem processes, such as litterfall decomposition [48,49].
The variation in litterfall fractions along the parks may be related to obtaining resources and, consequently, the distribution of abundances of some arthropod taxonomic groups. This means that increased nutritional availability, intentional litterfall management, or even trampling of deciduous material, may alter the occurrence of some individuals as well as the community structure in the soil, favoring or damaging the dynamics of arthropod populations.
5. Conclusions
The results of this study reflect the general pattern of recurrent biodiversity in nature, i.e., a predominance of many rare species and the presence of a few dominant groups of arthropods. Most of the taxa found, as well as the distribution of their abundances, were similar to those found in studies of other geographical locations, which used a similar methodology.
Although the urban forest fragments have different sizes and litterfall management, our results showed a certain degree of biodiversity similarity among the study sites, which highlights that neither litterfall management nor size of study site was sufficient to alter the variability or abundance of the identified arthropod orders. However, we did verify that the abundances of some dominant groups have unique characteristics, since each urban fragment presented a different predominant order.
On the other hand, variations in leaf and reproductive stock of litterfall may have interfered with the abundances of some taxonomic groups since the animals use these plant fractions to hide or feed. In addition, the present study shows the importance of gaining a better understanding of the ecological attributes of urban forests, either in relation to biodiversity or litterfall, in order to optimize the ecosystem services originated by this important component of the urban landscape.
Acknowledgments
The research was funded with own resources and the publication costs were divided between the authors of the paper.
Author Contributions
M.L.F. and L.C.d.S. conceived, designed and performed the experiment; M.L.F., A.R.T., K.G.d.S., L.C. and P.B.d.C. identified the animals and analyzed the data; M.L.F., L.C.d.S., D.d.M.C., C.C.Q. and C.T.K. wrote the paper; D.d.M.C., A.R.T. and M.L.F. translated and revised the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Seto, K.C.; Gueneralp, B.; Hutyra, L.R. Global forecasts of urban expansion to 2030 and direct impacts on biodiversity and carbon pools. Proc. Natl. Acad. Sci. USA 2012, 109, 16083–16088. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kim, B.H.S. The effect of urban green infrastructure on disaster mitigation in Korea. Sustainability 2017, 9, 26. [Google Scholar] [CrossRef]
- Nowak, D.J.; Noble, M.H.; Sisinni, S.M.; Dwyer, J.F. People and Trees: Assessing the US Urban Forest Resource. J. For. 2001, 99, 37–42. [Google Scholar]
- Li, H.; Li, Y.; Gao, Y.; Zou, C.; Yan, S.; Gao, J. Human impact on vegetation dynamics around Lhasa, southern Tibetan Plateau, China. Sustainability 2016, 8, 1146. [Google Scholar] [CrossRef]
- Vitousek, P.M. Litterfall, nutrient cycling, and nutrient limitation in tropical forests. Ecology 1984, 79, 10–22. [Google Scholar] [CrossRef]
- Brown, K.S.; Freitas, A.V. Butterfly communities of urban forest fragments in Campinas, Sao Paulo, Brazil: Structure, instability, environmental correlates, and conservation. J. Insect Conserv. 2002, 6, 217–231. [Google Scholar] [CrossRef]
- Bai, S.H.; Xu, Z.; Blumfield, T.J.; Reverchon, F. Human footprints in urban forests: Implication of nitrogen deposition for nitrogen and carbon storage. J. Soils Sediments 2015, 15, 1927–1936. [Google Scholar]
- Bednova, O.V.; Kuznetsov, V.A.; Tarasova, N.P. Transformation of urban forest ecosystems: Indication and integral assessment. Dokl. Akad. Nauk 2015, 463, 713–718. [Google Scholar] [CrossRef]
- Ferreira, M.L.; Uchiyama, E.A. Litterfall assessement in a fragment of secondary tropical forest, Ibiúna, SP, southeastern Brazil. Rev. Árvore 2015, 39, 791–799. [Google Scholar] [CrossRef]
- Ashford, O.S.; Foster, W.A.; Turner, B.L.; Sayer, E.J.; Sutcliffe, L.; Tanner, E.V.J. Litter manipulation and the soil arthropod community in a lowland tropical rainforest. Soil Biol. Biochem. 2013, 62, 5–12. [Google Scholar] [CrossRef]
- Panno, A.; Carrus, G.; Lafortezza, R.; Mariani, L.; Sanesi, G. Nature-based solutions to promote human resilience and wellbeing in cities during increasingly hot summers. Environ. Res. 2017, 159, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Kreutzweiser, D.P.; Good, K.P.; Capell, S.S.; Holmes, S.B. Leaf-litter decomposition and macroinvertebrate communities in boreal forest streams linked to upland logging disturbance. J. N. Am. Benthol. Soc. 2008, 27, 1–15. [Google Scholar] [CrossRef]
- Chapin, F.S., III; Matson, P.A.; Vitousek, P.; Chapin, M.C. Principles of Terrestrial Ecosystem Ecology, 2nd ed.; Springer: New York, NY, USA, 2011; p. 529. [Google Scholar]
- Livesley, S.J.; McPherson, E.G.; Calfapietra, C. The urban forest and ecosystem services: Impacts on urban water, heat, and pollution cycles at the tree, street, and city scale. J. Environ. Qual. 2016, 45, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Crowther, T.W.; Boddy, L.; Jones, T.H. Outcomes of fungal interactions are determined by soil invertebrate grazers. Ecol. Lett. 2011, 14, 1134–1142. [Google Scholar] [CrossRef] [PubMed]
- Meyer, W.M.; Ostertag, R.; Cowie, R.H. Macro-invertebrates accelerate litter decomposition and nutrient release in a Hawaiian rainforest. Soil Biol. Biochem. 2011, 43, 206–211. [Google Scholar] [CrossRef]
- Siqueira, T.; Bini, L.M.; Roque, F.O.; Couceiro, S.R.M.; Trivinho-Strixino, S.; Cottenie, K. Common and rare species respond to similar niche processes in macroinvertebrate metacommunities. Ecography 2012, 35, 183–192. [Google Scholar] [CrossRef]
- Bottinelli, N.; Jouquet, P.; Capowiez, Y.; Podwojewski, P.; Grimaldi, M.; Peng, X. Why is the influence of soil macrofauna on soil structure only considered by soil ecologists? Soil Tillage Res. 2015, 146, 118–124. [Google Scholar] [CrossRef]
- Moço, M.K.S.; Rodrigues, E.F.G.; Rodrigues, A.C.G.; Correia, M.E.F. Caracterização da fauna edáfica em diferentes coberturas vegetais na região Norte Fluminense. Rev. Bras. Ciênc. Solo 2005, 29, 555–564. [Google Scholar] [CrossRef]
- Augusto, E.L. Morcegos do Parque Chico Mendes, Osasco, São Paulo, como dispersores de sementes. Rev. PIBIC 2004, 1, 15–19. [Google Scholar]
- Ferreira, M.L.; Silva, J.L.; Pereira, E.E.; Ferreira, A.P.N.L. Litter fall production and decomposition in a fragment of secondary Atlantic Forest of São Paulo, SP, Southeastern Brazil. Rev. Árvore 2014, 38, 591–600. [Google Scholar] [CrossRef]
- Jesus, P.R.; Filho, H.O.L.; Ferreira, M.L. Litterfall production in three different sites with different abundance of eucalyptus (Eucalyptus spp.) at the Piqueri Park, SP. Braz. J. Ecol. 2014, 1–2, 59–65. [Google Scholar]
- Magurran, A.E. Measuring Biological Diversity; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Ash, J.S. A study of the Mallophaga of birds with particular reference to their ecology. IBIS 1960, 102, 93–110. [Google Scholar] [CrossRef]
- Evans, D.E.; Martins, J.R.; Guglielmone, A.A. A review of the ticks (Acari, Ixodida) of Brazil, their hosts and geographic distribution—1. The State of Rio Grande do Sul, Southern Brazil. Mem. Inst. Oswaldo Cruz 2000, 95, 453–470. [Google Scholar] [CrossRef] [PubMed]
- Lyons, K.G.; Brigham, C.A.; Traut, B.H.; Schwartz, M.W. Rare species and ecosystem functioning. Conserv. Biol. 2005, 19, 1019–1023. [Google Scholar] [CrossRef]
- Lawrence, K.L.; Wise, D.H. Spider predation on forest-floor Collembola and evidence for indirect effects on decomposition. Pedobiologia 2000, 44, 33–39. [Google Scholar] [CrossRef]
- Wetterer, J.K. Geographic Distribution of Strumigenys lanuginosa (Hymenoptera: Formicidae). Trans. Am. Entomol. Soc. 2017, 143, 729–733. [Google Scholar] [CrossRef]
- Folgarait, P.J. Ant biodiversity and its relationship to ecosystem functioning: A review. Biodivers. Conserv. 1998, 7, 1221–1244. [Google Scholar] [CrossRef]
- Gippet, J.M.; Mondy, N.; Diallo-Dudek, J.; Bellec, A.; Dumet, A.; Mistler, L.; Kaufmann, B. I’m not like everybody else: Urbanization factors shaping spatial distribution of native and invasive ants are species-specific. Urban Ecosyst. 2017, 20, 157–169. [Google Scholar] [CrossRef]
- Brancher, D.; Roza-Gomes, M.F. Survey of edaphic fauna in forest fragment in the municipality of Anchieta (SC, Brazil). Biota Neotrop. 2012, 12, 94–98. [Google Scholar] [CrossRef]
- Boucher, P.; Hébert, C.; Francoeur, A.; Sirois, L. Postfire Succession of Ants (Hymenoptera: Formicidae) Nesting in Dead Wood of Northern Boreal Forest. Environ. Entomol. 2015, 44, 1316–1327. [Google Scholar] [CrossRef] [PubMed]
- Barajas-Gusmán, G.; Alvarez-Sánchez, J. The relationships between litter fauna and rates of litter decomposition in a tropical rain forest. Appl. Soil Ecol. 2003, 24, 91–100. [Google Scholar] [CrossRef]
- Sayer, E.J.; Tanner, E.V.J.; Lacey, A.L. Effects of litter manipulation on early-stage decomposition and meso-arthropod abundance in a tropical moist forest. For. Ecol. Manag. 2006, 229, 285–293. [Google Scholar] [CrossRef]
- Sánchez, D.P.; Alexandra, S.M.; Parada, M.R.P. Poblaciones de acaros, colembolos y otra mesofauna en un inceptisol bajo diferentes. Rev. Fac. Nac. Agron. Medellin 2015, 68, 7411–7422. [Google Scholar] [CrossRef]
- Coleman, D.C.; Crossley, D.A., Jr. Fundamentals of Soil Ecology, 2nd ed.; Academic Press: Amsterdam, The Netherlands, 2004; p. 408. [Google Scholar]
- Ferguson, S.H. Changes in trophic abundance of soil arthropods along a grass–shrub–forest gradient. Can. J. Zool. 2001, 79, 457–464. [Google Scholar] [CrossRef]
- Agodi, A.; Conti, G.O.; Barchitta, M.; Quattrocchi, A.; Lombardo, B.M.; Montesanto, G.; Messina, G.; Fiore, M.; Ferrante, M. Validation of Armadillo officinalis Dumèril, 1816 (Crustacea, Isopoda, Oniscidea) as a bioindicator: In vivo study of air benzene exposure. Ecotoxicol. Environ. Saf. 2015, 114, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Bregenski, M.A. Variação Espacial e Temporal de Talitroides Topitotum (BURT, 1934) (Crustacea, Amphipoda, Talitridae), em um Remanescente de Floresta Ombrófila Mista, no Parque Municipal do Iguaçu, Curitiba, PR. Master’s Thesis, Universidade Federal do Paraná, Curitiba, Brazil, 2008; p. 49. [Google Scholar]
- Eutropio, F.B.; Krohling, W. First record of Amphipoda Talitroides topitotum (Burt, 1934) (Gammaridea, Talitridae) in the State of Espírito Santo, Brazil. Acta Sci. Biol. Sci. 2013, 35, 37–39. [Google Scholar] [CrossRef][Green Version]
- Saunders, D.A.; Hobbs, R.J.; Margules, C.R. Biological consequences of ecosystem fragmentation: A review. Conserv. Biol. 1991, 5, 18–32. [Google Scholar] [CrossRef]
- Hamberg, L.; Lehvävirta, S.; Malmivaara-Lämsä, M.; Rita, H.; Kotze, D.J. The effects of habitat edges and trampling on understorey vegetation in urban forests in Helsinki, Finland. Appl. Veg. Sci. 2008, 11, 83–98. [Google Scholar] [CrossRef]
- Schwarz, K.; Herrmann, D.L.; Mchale, M.R. Abiotic Drivers of Ecological Structure and Function in Urban Systems. In Urban Wildlife Conservation; Springer: New York, NY, USA, 2014; pp. 55–74. [Google Scholar]
- Ossola, A.; Hahs, A.K.; Nash, M.A.; Livesley, S.J. Habitat complexity enhances comminution and decomposition processes in urban ecosystems. Ecosystems 2016, 19, 927–941. [Google Scholar] [CrossRef]
- Chen, Y.A.; Forschler, B.T. Elemental concentrations in the frass of saproxylic insects suggest a role in micronutrient cycling. Ecosphere 2016, 7, e01300. [Google Scholar] [CrossRef]
- Sizer, N.C.; Tanner, E.V.J.; Ferraz, I.D.K. Edge effects on litterfall mass and nutrient concentrations in forest fragments in central Amazonia. J. Trop. Ecol. 2000, 16, 853–863. [Google Scholar] [CrossRef]
- Zhang, J.H.; Hou, X.; Shu, Q.S. Research on the hydrological effects of forest litter-fall. In Energy Science and Applied Technology: Proceedings of the 2nd International Conference on Energy Science and Applied Technology (ESAT 2015); CRC Press: Boca Raton, FL, USA, 2015; p. 85. [Google Scholar]
- Ferreira, M.L.; Ribeiro, A.P.; Albuquerque, C.R.; Lamano-Ferreira, A.P.N.; Figueira, R.C.L.; Lafortezza, R. Air contaminants and litter fall decomposition in urban forest areas: The case of São Paulo, SP, Brazil. Environ. Res. 2017, 155, 314–320. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).