Properties of Biochar from Anaerobically Digested Food Waste and Its Potential Use in Phosphorus Recovery and Soil Amendment
Abstract
1. Introduction
2. Materials and Methods
2.1. Biochar Production
2.2. Fourier Transform Infrared (FTIR) Analysis
2.3. Pyrolysis Yield and Volatile Matter
2.4. pH, Electrical Conductivity (EC) Values, and Elemental Composition
2.5. Surface Area (SBET)
2.6. Phosphorus Adsorption
2.7. Adsorption Kinetic Analysis
2.8. Pot Experiment
3. Results and Discussion
3.1. Pyrolysis Yield
3.2. pH Values and Electrical Conductivity (EC)
3.3. Surface Properties
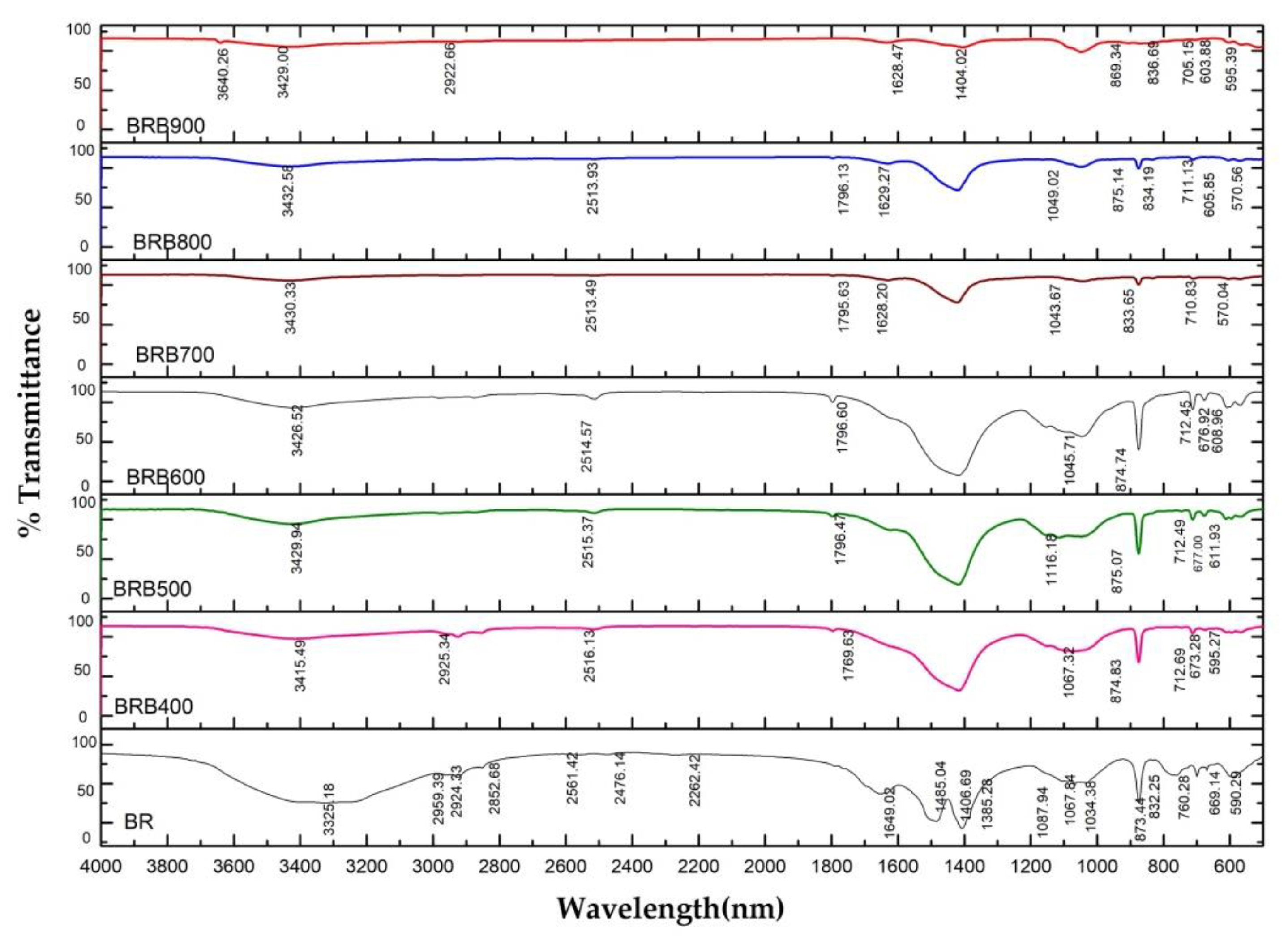
3.4. Changes in Chemical Bond Structure with Pyrolysis Temperature
3.5. Elemental Composition and Molecular Ratios
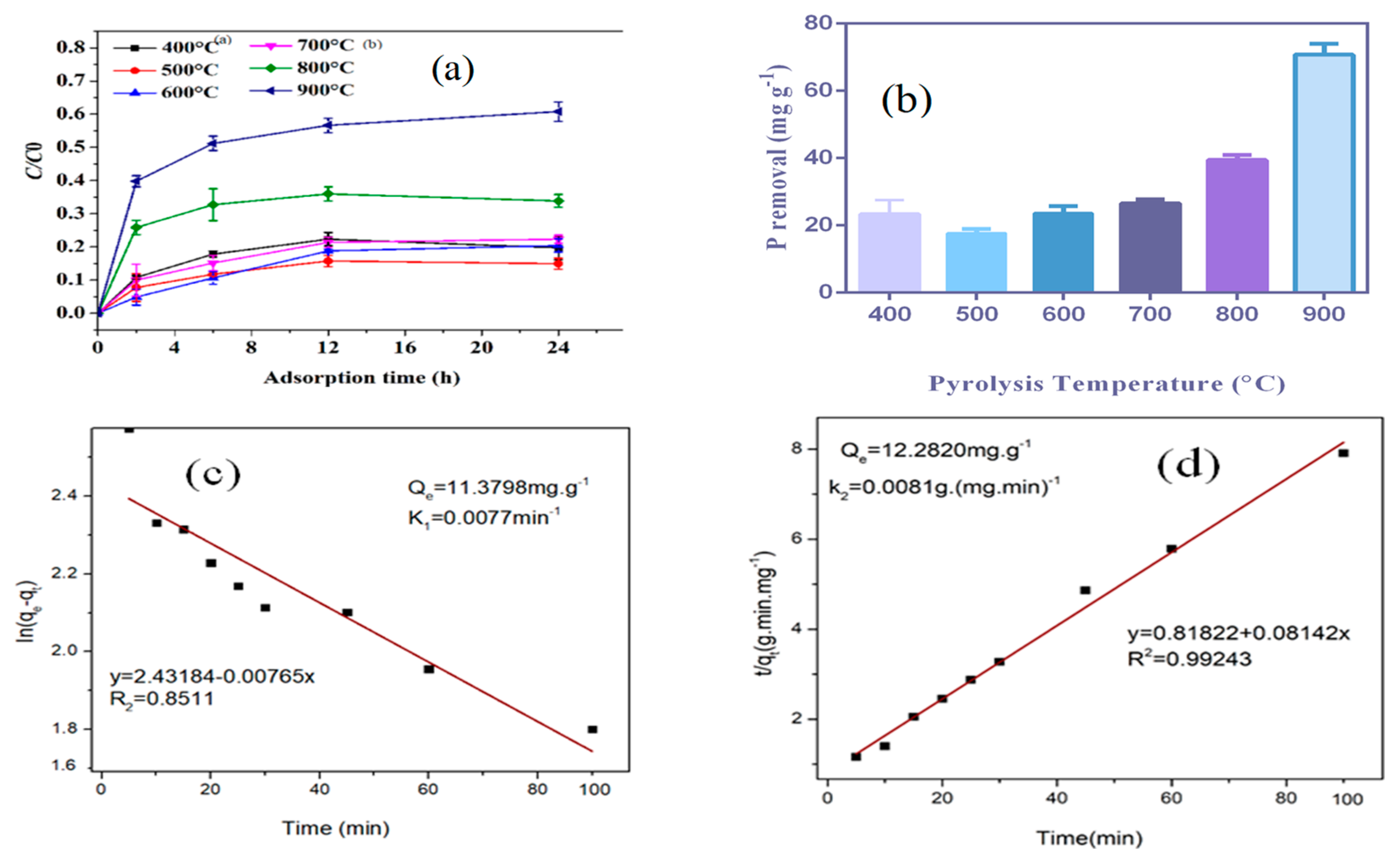
3.6. Phosphorus Adsorption and Soil Amendment
3.6.1. Adsorption Kinetics
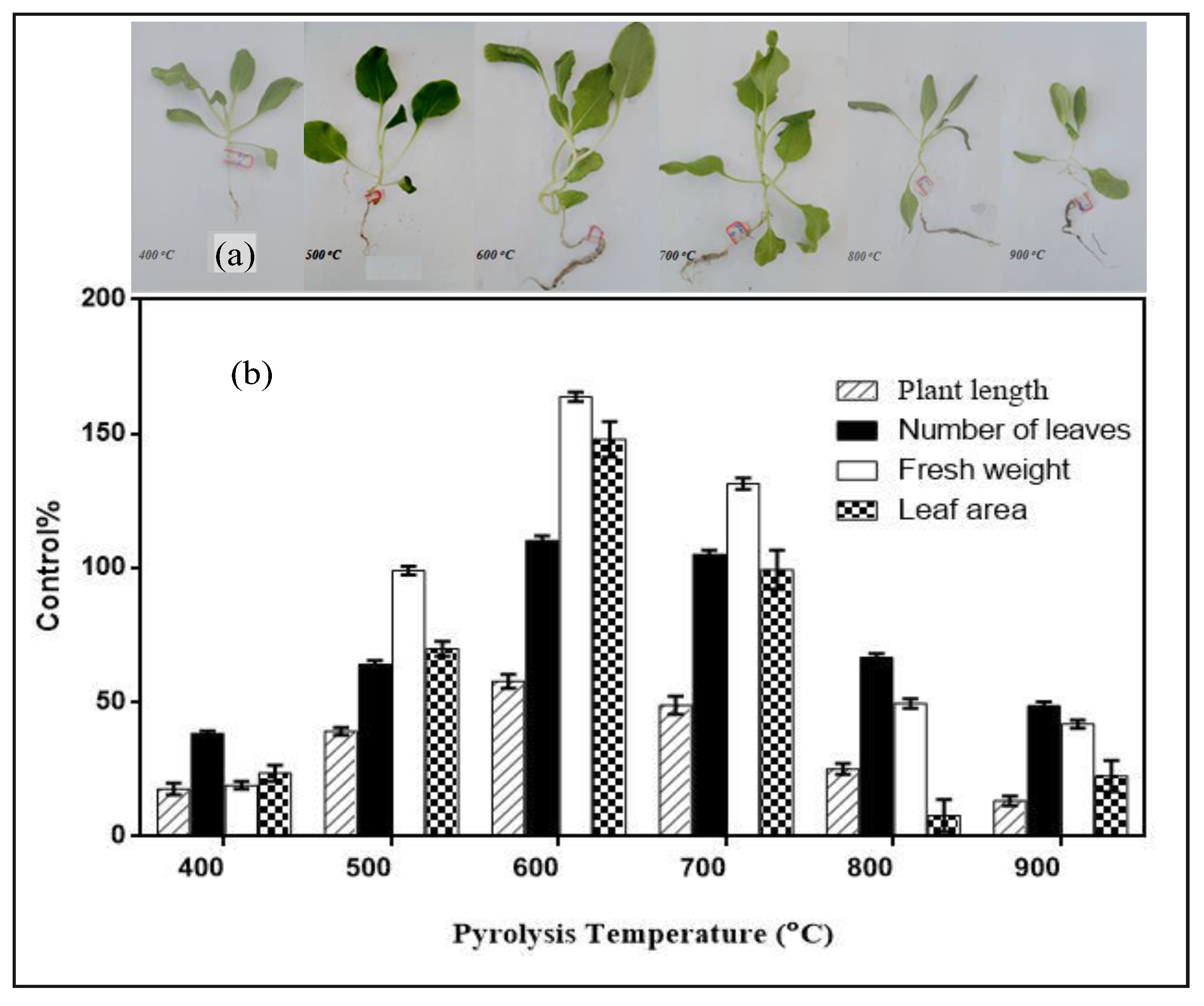
3.6.2. Soil Amendment
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, C.; Su, H.; Baeyens, J.; Tan, T. Reviewing the anaerobic digestion of food waste for biogas production. Renew. Sustain. Energy Rev. 2014, 38, 383–392. [Google Scholar] [CrossRef]
- Marcello, P. Does food sharing lead to food waste reduction? An experimental analysis to assess challenges and opportunities of a new consumption model. J. Clean. Prod. 2018, 185, 749–760. [Google Scholar]
- Aschemann-Witzel, J.; de Hooge, I.; Amani, P.; Bech-Larsen, T.; Oostindjer, M. Consumer-related food waste: Causes and potential for action. Sustainability 2015, 7, 6457–6477. [Google Scholar] [CrossRef]
- Michelini, L.; Principato, L.; Iasevoli, G. Understanding food sharing models to tackle sustainability challenges. Ecol. Econ. 2018, 145, 205–217. [Google Scholar] [CrossRef]
- Morone, P.; Falcone, P.M.; Lopolito, A. How to promote a new and sustainable food consumption model: A fuzzy cognitive map study. J. Clean. Prod. 2019, 208, 563–574. [Google Scholar] [CrossRef]
- Gupta, S.; Kua, H.W.; Koh, H.J. Application of biochar from food and wood waste as green admixture for cement mortar. Sci. Total Environ. 2018, 619, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.-Y.; Tsai, W.-T.; Chen, J.-W.; Lin, Y.-Q.; Chang, Y.-M. Characterization of biochar prepared from biogas digestate. Waste Manag. 2017, 66, 53–60. [Google Scholar] [CrossRef]
- Stefaniuk, M.; Oleszczuk, P. Characterization of biochars produced from residues from biogas production. J. Anal. Appl. Pyrolysis 2015, 115, 157–165. [Google Scholar] [CrossRef]
- Pituello, C.; Francioso, O.; Simonetti, G.; Pisi, A.; Torreggiani, A.; Berti, A.; Morari, F. Characterization of chemical–physical, structural and morphological properties of biochars from biowastes produced at different temperatures. J. Soils Sediments 2015, 15, 792–804. [Google Scholar] [CrossRef]
- Hossain, A.; Serrano, C.; Brammer, J.; Omran, A.; Ahmed, F.; Smith, D.; Davies, P. Combustion of fuel blends containing digestate pyrolysis oil in a multi-cylinder compression ignition engine. Fuel 2016, 171, 18–28. [Google Scholar] [CrossRef]
- Li, Q.; Xu, M.; Wang, G.; Chen, R.; Qiao, W.; Wang, X. Biochar assisted thermophilic co-digestion of food waste and waste activated sludge under high feedstock to seed sludge ratio in batch experiment. Bioresour. Technol. 2018, 249, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Liu, Z.; Niu, W.; Yang, L.; Zhou, T.; Qin, D.; Niu, Z.; Yuan, Q. Effects of pyrolysis temperature on the physicochemical properties of gas and biochar obtained from pyrolysis of crop residues. Energy 2018, 143, 746–756. [Google Scholar] [CrossRef]
- López-Cano, I.; Cayuela, M.; Mondini, C.; Takaya, C.; Ross, A.; Sánchez-Monedero, M. Suitability of different agricultural and urban organic wastes as feedstocks for the production of biochar—Part 1: Physicochemical characterisation. Sustainability 2018, 10, 2265. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Inyang, M.; Zimmerman, A.R.; Cao, X.; Pullammanappallil, P.; Yang, L. Biochar derived from anaerobically digested sugar beet tailings: Characterization and phosphate removal potential. Bioresour. Technol. 2011, 102, 6273–6278. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Chen, Q.; Sun, M.; Sun, P.; Shen, G. Pyrolysis temperature-induced changes in the catalytic characteristics of rice husk-derived biochar during 1, 3-dichloropropene degradation. Chem. Eng. J. 2017, 330, 804–812. [Google Scholar] [CrossRef]
- Inyang, M.; Gao, B.; Pullammanappallil, P.; Ding, W.; Zimmerman, A.R. Biochar from anaerobically digested sugarcane bagasse. Bioresour. Technol. 2010, 101, 8868–8872. [Google Scholar] [CrossRef]
- Troy, S.M.; Nolan, T.; Leahy, J.J.; Lawlor, P.G.; Healy, M.G.; Kwapinski, W. Effect of sawdust addition and composting of feedstock on renewable energy and biochar production from pyrolysis of anaerobically digested pig manure. Biomass Bioenergy 2013, 49, 1–9. [Google Scholar] [CrossRef]
- Chen, Q.; Qin, J.; Sun, P.; Cheng, Z.; Shen, G. Cow dung-derived engineered biochar for reclaiming phosphate from aqueous solution and its validation as slow-release fertilizer in soil-crop system. J. Clean. Prod. 2018, 172, 2009–2018. [Google Scholar] [CrossRef]
- Chen, B.; Chen, Z.; Lv, S. A novel magnetic biochar efficiently sorbs organic pollutants and phosphate. Bioresour. Technol. 2011, 102, 716–723. [Google Scholar] [CrossRef]
- Hammer, E.C.; Balogh-Brunstad, Z.; Jakobsen, I.; Olsson, P.A.; Stipp, S.L.; Rillig, M.C. A mycorrhizal fungus grows on biochar and captures phosphorus from its surfaces. Soil Biol. Biochem. 2014, 77, 252–260. [Google Scholar] [CrossRef]
- Chen, L.; Chen, Q.; Rao, P.; Yan, L.; Shakib, A.; Shen, G. Formulating and optimizing a novel biochar-based fertilizer for simultaneous slow-release of nitrogen and immobilization of cadmium. Sustainability 2018, 10, 2740. [Google Scholar] [CrossRef]
- Collins, L.W.; Chalk, S.J.; Kingston, H.S. Atmospheric pressure microwave sample preparation procedure for the combined analysis of total phosphorus and kjeldahl nitrogen. Anal. Chem. 1996, 68, 2610–2614. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Wu, B.; Su, L.; Gao, X.; Chai, X.; Dai, X. Development of nano-CaO2-coated clinoptilolite for enhanced phosphorus adsorption and simultaneous removal of cod and nitrogen from sewage. Chem. Eng. J. 2017, 328, 35–43. [Google Scholar] [CrossRef]
- Yan, J.; Jiang, T.; Yao, Y.; Wang, J.; Cai, Y.; Green, N.W.; Wei, S. Underestimation of phosphorus fraction change in the supernatant after phosphorus adsorption onto iron oxides and iron oxide-natural organic matter complexes. J. Environ. Sci. 2017, 55, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Takaya, C.A.; Fletcher, L.A.; Singh, S.; Anyikude, K.U.; Ross, A.B. Phosphate and ammonium sorption capacity of biochar and hydrochar from different wastes. Chemosphere 2016, 145, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Belevi, H.; Moench, H. Factors determining the element behavior in municipal solid waste incinerators. 1. Field studies. Environ. Sci. Technol. 2000, 34, 2501–2506. [Google Scholar] [CrossRef]
- Thangalazhy-Gopakumar, S.; Adhikari, S.; Ravindran, H.; Gupta, R.B.; Fasina, O.; Tu, M.; Fernando, S.D. Physiochemical properties of bio-oil produced at various temperatures from pine wood using an auger reactor. Bioresour. Technol. 2010, 101, 8389–8395. [Google Scholar] [CrossRef]
- DeSisto, W.J.; Hill, N.; Beis, S.H.; Mukkamala, S.; Joseph, J.; Baker, C.; Ong, T.-H.; Stemmler, E.A.; Wheeler, M.C.; Frederick, B.G. Fast pyrolysis of pine sawdust in a fluidized-bed reactor. Energy Fuels 2010, 24, 2642–2651. [Google Scholar] [CrossRef]
- Cao, X.; Harris, W. Properties of dairy-manure-derived biochar pertinent to its potential use in remediation. Bioresour. Technol. 2010, 101, 5222–5228. [Google Scholar] [CrossRef]
- del Martínez-Ballesta, M.C.; Silva, C.; Lopez-Berenguer, C.; Cabañero, F.; Carvajal, M. Plant aquaporins: New perspectives on water and nutrient uptake in saline environment. Plant Biol. 2006, 8, 535–546. [Google Scholar] [CrossRef]
- Al-Wabel, M.I.; Al-Omran, A.; El-Naggar, A.H.; Nadeem, M.; Usman, A.R. Pyrolysis temperature induced changes in characteristics and chemical composition of biochar produced from conocarpus wastes. Bioresour. Technol. 2013, 131, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Keiluweit, M.; Nico, P.S.; Johnson, M.G.; Kleber, M. Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef]
- Mukome, F.N.; Parikh, S.J. Chemical, physical, and surface characterization of biochar. In Biochar: Production, Characterization, and Applications; Ok, Y.S., Uchimiya, S.M., Chang, S.X., Bolan, N., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 68–96. [Google Scholar]
- Onorevoli, B.; da Silva Maciel, G.P.; Machado, M.E.; Corbelini, V.; Caramão, E.B.; Jacques, R.A. Characterization of feedstock and biochar from energetic tobacco seed waste pyrolysis and potential application of biochar as an adsorbent. J. Environ. Chem. Eng. 2018, 6, 1279–1287. [Google Scholar] [CrossRef]
- El Ouaqoudi, F.Z.; El Fels, L.; Lemée, L.; Amblès, A.; Hafidi, M. Evaluation of lignocelullose compost stability and maturity using spectroscopic (ftir) and thermal (tga/tda) analysis. Ecol. Eng. 2015, 75, 217–222. [Google Scholar] [CrossRef]
- Özsin, G.; Pütün, A.E. Kinetics and evolved gas analysis for pyrolysis of food processing wastes using tga/ms/ft-ir. Waste Manag. 2017, 64, 315–326. [Google Scholar] [CrossRef]
- Zbytniewski, R.; Buszewski, B. Characterization of natural organic matter (nom) derived from sewage sludge compost. Part 1: Chemical and spectroscopic properties. Bioresour. Technol. 2005, 96, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Tandy, S.; Healey, J.R.; Nason, M.A.; Williamson, J.C.; Jones, D.L.; Thain, S.C. Ft-ir as an alternative method for measuring chemical properties during composting. Bioresour. Technol. 2010, 101, 5431–5436. [Google Scholar] [CrossRef]
- Agrafioti, E.; Bouras, G.; Kalderis, D.; Diamadopoulos, E. Biochar production by sewage sludge pyrolysis. J. Anal. Appl. Pyrolysis 2013, 101, 72–78. [Google Scholar] [CrossRef]
- Chan, K.; Xu, Z. 2009biochar: Nutrient properties and their enhancement. In Biochar for Environmental Management: Science and Technology; Earthscan: London, UK, 2009; p. 6784. [Google Scholar]
- Krull, E.S.; Baldock, J.A.; Skjemstad, J.O.; Smernik, R.J. Characteristics of biochar: Organo-chemical properties. In Biochar for Environmental Management: Science and Technology; Earthscan: London, UK, 2009. [Google Scholar]
- Atkinson, C.J.; Fitzgerald, J.D.; Hipps, N.A. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant Soil 2010, 337, 1–18. [Google Scholar] [CrossRef]
- Kloss, S.; Sass, O.; Geitner, C.; Prietzel, J. Soil properties and charcoal dynamics of burnt soils in the tyrolean limestone alps. Catena 2012, 99, 75–82. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Chen, J.; Yang, L. Engineered biochar reclaiming phosphate from aqueous solutions: Mechanisms and potential application as a slow-release fertilizer. Environ. Sci. Technol. 2013, 47, 8700–8708. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Gao, B.; Zhang, M.; Inyang, M.; Zimmerman, A.R. Effect of biochar amendment on sorption and leaching of nitrate, ammonium, and phosphate in a sandy soil. Chemosphere 2012, 89, 1467–1471. [Google Scholar] [CrossRef] [PubMed]
| Biogas Residue | Biogas Residue–Derived Biochar | ||||||
|---|---|---|---|---|---|---|---|
| 400 °C | 500 °C | 600 °C | 700 °C | 800 °C | 900 °C | ||
| PY | 70.35 ± 0.08 | 68.25 ± 0.13 | 66.02 ± 0.01 | 64.38 ± 0.62 | 56.33 ± 0.46 | 43.65 ± 0.08 | |
| VM | 55.36 ± 0.07 | 40.67 ± 0.16 | 34.90 ± 0.09 | 33.31 ± 0.18 | 30.52 ± 0.04 | 20.68 ± 0.05 | 4.49 ± 0.05 |
| pH | 7.95 ± 0.03 | 9.19 ± 0.08 | 9.28 ± 0.02 | 10.60 ± 0.25 | 11.56 ± 0.20 | 12.51 ± 0.05 | 12.52 ± 0.21 |
| EC | 3.88 ± 0.39 | 2.23 ± 0.10 | 3.05 ± 0.04 | 3.17 ± 0.04 | 3.20 ± 0.01 | 12.66 ± 0.05 | 11.50 ± 0.15 |
| SBET | 8.48 | 17.91 | 19.05 | 21.68 | 25.96 | 29.42 | 66.15 |
| Sample | C (%) | H (%) | N (%) | S (%) | O (%) | H/C | C/N |
|---|---|---|---|---|---|---|---|
| BR | 20.34 | 3.32 | 2.41 | 0.84 | 28.48 | 1.95 | 10.59 |
| BRB400 | 19.81 | 1.51 | 1.41 | 0.99 | 21.73 | 0.92 | 17.55 |
| BRB500 | 14.34 | 0.60 | 0.68 | 1.23 | 21.59 | 0.50 | 26.38 |
| BRB600 | 13.95 | 0.41 | 0.58 | 1.41 | 21.21 | 0.35 | 30.11 |
| BRB700 | 13.17 | 0.32 | 0.38 | 1.37 | 19.09 | 0.29 | 43.38 |
| BRB800 | 9.70 | 0.30 | 0.19 | 1.26 | 14.63 | 0.37 | 64.12 |
| BRB900 | 7.07 | 0.63 | 0.14 | 1.57 | 4.925 | 1.07 | 63.33 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alghashm, S.; Qian, S.; Hua, Y.; Wu, J.; Zhang, H.; Chen, W.; Shen, G. Properties of Biochar from Anaerobically Digested Food Waste and Its Potential Use in Phosphorus Recovery and Soil Amendment. Sustainability 2018, 10, 4692. https://doi.org/10.3390/su10124692
Alghashm S, Qian S, Hua Y, Wu J, Zhang H, Chen W, Shen G. Properties of Biochar from Anaerobically Digested Food Waste and Its Potential Use in Phosphorus Recovery and Soil Amendment. Sustainability. 2018; 10(12):4692. https://doi.org/10.3390/su10124692
Chicago/Turabian StyleAlghashm, Shakib, Shiying Qian, Yinfeng Hua, Jian Wu, Haitao Zhang, Weihua Chen, and Guoqing Shen. 2018. "Properties of Biochar from Anaerobically Digested Food Waste and Its Potential Use in Phosphorus Recovery and Soil Amendment" Sustainability 10, no. 12: 4692. https://doi.org/10.3390/su10124692
APA StyleAlghashm, S., Qian, S., Hua, Y., Wu, J., Zhang, H., Chen, W., & Shen, G. (2018). Properties of Biochar from Anaerobically Digested Food Waste and Its Potential Use in Phosphorus Recovery and Soil Amendment. Sustainability, 10(12), 4692. https://doi.org/10.3390/su10124692




