1. Introduction
Pollutants emitted from certain solids or liquids originate from anthropogenic activity or from the biogenic emissions of certain reactive hydrocarbon derivatives formed as chemical (by-)products of natural transformations [
1]. The odor-causing mixtures of sulfur-containing derivatives and their metabolites constitute a main class of irritant and toxic contaminants with increasingly detrimental impact on human health in the long-term exposure, being also classified as a potential mutagenic and carcinogenic risk factors [
2,
3]. A diverse ‘suite’ of environmental contaminants, potentially resistant to degradation, were designed and engineered for industrially-specific purposes becoming problematic, when released into the Nature since
recalcitrance correlates to persistence [
4]. On the other hand, natural gases also contain ‘sour’ impurities such as hydrogen sulfide (H
2S) or carbon dioxide (CO
2), the removal of which is technologically and/or economically relevant to ‘soften’ the natural/industrial (off)gases [
5,
6]. Consequently, the eradication of sulfur-containing compounds (e.g., H
2S, COS, mercaptans, organic sulfides) from unprocessed natural gases can be performed in the liquid-liquid extraction process, where industrial solvents are used in a ‘closed’ loop, recovered and regenerated
on-site, that theoretically prevent extractor disposal [
7,
8]. In this context questions about the ‘green’ solvents and manufacturing procedures used in the liquid-liquid extraction are looked upon favourably.
In fact, a range of solvents can be employed in the extraction process used in oil refining including (di/tri/tetra) ethylene glycol (D/T/TT/EG), diglycol amine (DGA),
n-methyl pyrolidone (NMP), dimethylsulphoxide (DMSO), dimethylformamide (DMF), morpholine and carbonate derivatives. An attractive alternative to commonly used industrial extractive liquids is sulfolane, an anthropogenic organosulfur medium regarded as
a versatile dipolar aprotic solvent [
9,
10]. Sulfolane (C
4H
8SO
2) is the generic name for hydrogenated sulfones of butadiene [
11]. In the present review we mainly focus on the sulfolane synthesis, application of sulfolane as an extractive solvent due to its ‘unique’ physicochemical properties as well as on the potential of sulfolane to cause equipment corrosion and subsequent spills. The possible risk for groundwater contamination, danger for human health and ways of sulfolane biodegradation are reviewed briefly as well. Interestingly, the analysis performed on data stored in the Reaxys database revealed an alternating tendency of waxing and waning interest in sulfolane during the space of the last fifty years. Roughly speaking, the revealed ‘wavy’ trends in sulfolane interests provide an illustration of the partial correlations with oil prices. Moreover, the primary goal of the presented case study was to verify applicability of industrial, multi-electrochemical technique for reliable detection of corrosion in low conductive process (sulfolane-based) fluids. Several aspects of corrosion measurement of general and localized corrosion modes were investigated briefly, since corrosion control and monitoring in sulfolane units does not find prominent presence in published literature. Incipient attempts to quantify impact of process parameters (temperature) and impurities (oxygen and chlorides) on stainless steel (UNS S304033) corrosion in pure sulfolane provided reasonably meaningful data and are detailed elsewhere in this paper. For the purpose of the proposed investigations a dedicated testing vessel was designed and constructed. It should be emphasized that there is a clear absence of detailed and quantitative evaluation of individual sulfolane impurities (like water, chlorides, etc.), their limits and interactions on corrosion rate of carbon and alloyed steels; therefore more detailed control of experiments, a wider range of solution conditions, a wider range of temperatures, and with this the corresponding surface analysis of the electrodes must be performed in the future.
2. Beware of Sulfolane
Formally, sulfolane is a cyclic sulfone, containing a four-membered carbon ring and sulfonyl functional group (R
2SO
2) with a sulfur atom double-bonded to two oxygen atoms with a quite large dipole moment (μ = 4.7 debye), dielectric constant (ε
υ = 43.4) and low vapor pressure [
12,
13]. The structure of sulfolane with its zwitterionic and dritterionic resonance structures is illustrated in
Figure 1.
Due to its combination of physical and chemical properties (inertness and stability), sulfolane is a selective solvent in liquid-liquid and liquid-vapour extraction processed, used for the removal of close-boiling alkanes from cycloalkanes (C
5–C
10 derived from petroleum fractions) or for the separation of compounds with different degree of saturation and polarity in the extractive rectification of arenes (BTX—benzene, toluene, xylene) from non-aromatic saturated hydrocarbon mixtures) [
14,
15]. Sulfolane is the preferred solvent for these processes due to its satisfactory selectivity for the aromatics of interest, low boiling temperature and its capacity for dissolving large quantities of aromatics in relatively low quantities of solvent—the sulfolane extraction process is less expensive to operate than similar processes using other solvents.
Apart from the traditional applications in the extraction of aromatics and in ‘sour gas sweetening’, (e.g., removal of acid gases from a natural gas stream) sulfolane can be used in a wide range of engineering and biomedical applications, e.g., fractionation of wood tars, curing agent for epoxy resins, production of electronics and polymers [
12]. Moreover, sulfolane and its analogues demonstrate the properties of inflammation inhibitors (sulfolane-induced hypothermia) and tumor cell growth inhibitors [
16,
17,
18]; however these effects are probably species-specific and
the extrapolation to human health is speculative, as noticed one of Reviewers.
Sulfolane with a high worldwide production volume (approximately 18 000–36 000 tons per year) is a readily available commodity chemical that can be purchased as anhydrous or aqueous chemical substance (3–5% water content prevents the transition to solid state) [
19]. According to ECHA data (European Chemical Agency), sulfolane is manufactured and/or imported to the European Economic Area in quantity of 1 000–10 000 tons annually [
20]. In theory, during the large-scale industrial usage of sulfolane disposal/leakage of solvent is not encountered due to its
on-site recovery and regeneration; however a typical loss rate is approximated to 5–10 ppm (parts per million) of the feed rate [
21]. Practically, some unpredicted/accidental spills as well as leaks from extraction units in refineries or gas plants were reported worldwide, resulting in inadvertent contamination of the vadose zone, groundwater and wetland ecosystems [
22]. Moreover, leachates from disposal areas, leakage during processes and seepage from producing wells and unlined storage ponds have led to sulfolane contaminated soil and groundwater around gas processing plants [
4]. High aqueous miscibility of sulfolane, combined with the low octanol-water partition (K
ow), carbon-water partition coefficient (K
oc) and pK
a value have resulted in substantial off-site migration from contaminated sites, therefore the environmental fate of the compound is being extensively studied [
23].
Reduction of leaks and spills of sulfolane highlight an important question about its corrosivity and impact on integrity of industrial systems (pipes, vessels, pumps etc.). It was noticed that impellers of sulfolane pumps are the components particularly sensitive to corrosion, due to the velocity of sulfolane against the metal. It seems that sulfolane-induced corrosion is purely industrial problem with no apparent crossover into the public realm, because research did not indicate that corrosion ever became severe enough to cause leakage/spills of sulfolane outside of the plant [
24]. In fact, pure sulfolane under standard operating conditions is considered to be a stable compound and non-aggressive to steel, but sulfolane systems, if contaminated by traces of oxygen and at typical process conditions (170–180 °C), may lead to sulfolane decomposition and formation of corrosive (by-)products—the corrosion of steel can be quite rapid, causing severe damage to industrial installations. Decomposition of sulfolane may be further enhanced by presence of water, used commonly as a sulfolane diluent, as well as by presence of oxygen and/or chlorides, respectively. Some general correlations between oxygen and chloride concentration leading to sulfolane corrosivity are known and described, e.g., oxygen above 0.5% and water above 3% accelerate sulfolane degradation and production of acidic corrodents [
25]. However, sulfolane-corrosion knowledge is still lacking detailed and quantitative evaluation of individual impurities, their limits and interactions on corrosion rate of carbon and alloyed steels.
Is it important to monitor the fate of sulfolane in the environment and the primary potential routes of human exposure to this solvent? The answer to above question is positive, because sulfolane poses a potential risk for contamination of surface and groundwater, domestic wells around sour gas plants and off-site migration—the transfer of contaminant mass between the liquid and solid phases was noticed [
22]. Sulfolane has been detected in groundwater in several regions nearby industrial areas, therefore some assessment of the potential hazard for human beings were undertaken with toxicity benchmarks specification including several genotoxicity studies, acute toxicity studies in multiple species via multiple routes of exposure or a chronic oral toxicity analysis as well [
19]. In fact, animal data suggest that reproductive and developmental toxicity is only likely to occur at concentrations much higher than those, that elicit other milder adverse effects. Sulfolane is known to be acutely toxic at doses over 200 mg/kg in lab rats and guinea pigs [
12]. Luckily,
sulfolane is not likely to be mutagenic, clastogenic or carcinogenic, or pose reproductive or developmental health risks except perhaps at very high exposure concentrations [
19].
Generally, two procedures of removing sulfolane-containing contaminants from industrial/environmental areas can be distinguished based on the nature of applied transformations: physical (filtration) and chemical (oxidation). The first technique comprises transfer and/or concentration of pollutants in a liquid phase onto solid phase (activated carbon adsorption), whereas the second one is related to the destruction/decomposition of molecules by thermal/catalytic oxidation, flaring or degradation by microorganism community to harmless (by-)products [
26]. The complete oxidation of sulfolane can be described as follows:
Exploiting the metabolic capabilities of microorganisms naturally occurring in areas of contamination, using techniques such as monitored natural attenuation or biostimulation, may be a way to remediate sulfolane-contaminated aquifers according to ‘clean at the end of pipe’ paradigm [
4]. Biodegradation of sulfolane using microbes residing in an activated sludge system, in wastewater treatment or in laboratory microcosm using contaminated aquifer sediments was evaluated under aerobic and anaerobic conditions, respectively. It turned out that sulfolane-contaminated environmental samples are pretty easily biodegradable in nutrient-enriched aerobic microcosms [
27,
28].
2.1. Synthesis and Manufacturing
Sulfolane is a non-reactive, water-soluble dipolar aprotic industrial solvent that was first engineered and patented by Shell Oil Company in the early 1940s [
10]. The original process of sulfolane synthesis is based on the hydrogenation of 3-sulfolene (
3) obtained as a product of sulfur dioxide (
2) and butadiene (
1) reaction (
Scheme 1).
We illustrated some selected examples of sulfolane synthesis as well. In 1982 the synthesis of sulfolane was proposed with 1,4-dihalogenobutane as the starting reagent and sulfoxide as the intermediate [
29]., A few years later, the silica-supported titanium/tartaric acid catalysts were used in the sulfide→sulfoxide oxidation [
30]. In fact, several titanium derivatives supported on silica or molecular sieves suspended in a solution containing hydrogen peroxide (H
2O
2) led to high yields of the selective oxidation of sulfides to sulfoxides. Due to the significance of sulfoxides as intermediates in organic synthesis, the selective sulfide→sulfoxide oxidation has been a challenging issue; therefore a large number of supported reagents (hydrogen peroxide, chromic acid, nitric acid, ozone, manganese dioxide, peracids) was tested revealing some process drawbacks such as extended reaction time or formation of toxic and irritant intermediates [
31]. In 1999 the use of urea-hydrogen peroxide (UHP) adduct under solvent-free conditions was proposed in response to the need for mild and selective/efficient methods for conversion of sulfide to the corresponding sulfoxides or sulfone [
32]. Highly chemoselective and non-aqueous procedure of sulfides→sulfones oxidation with magnesium monoperoxyphthalate (MMPP) on hydrated silica gel support in methylene chloride solvent was demonstrated by Ali et al. [
33]. The employment of magnesium reagent resulted in a simple and environmentally friendly procedure with high yields. Similarly, the excellent yield (≥94%) and selectivity (>95%) of sulfides→sulfoxides oxidation was reached using tetra-(tetraalkylammonium)octamolybdate catalysts with 30% aqueous hydrogen peroxide as oxidant in high ratio of substrate to catalyst (10000:1) [
34]. Following the principles of ‘green chemisty’ a new environmentally friendly method for the sulfide oxidation at the ambient temperature by H
2O
2/H
2O (oxidant/solvent) mixture was documented with high yields and non-toxicity of the corresponding (by-)products [
35]. In 2017 an eco-friendly and catalyst-free method for oxidation of sulfides to sulfones, using Oxone (a stable 2:1:1 ternary composite of KHSO
5, K
2SO
4 and KHSO
4) was provided by Kupwade et al. [
36]. The previously available protocols in the chemoselective sulfides→sulfoxides oxidation by Oxone suffered from the drawbacks of thermal activation; therefore the energy-efficient combination of Oxone-KBr was proposed as an oxidant. Moreover, the chromatography-free and scalable procedure for the transformation of sulfides to sulfones with an unprecedented catalyst-oxidant combination (diethylamine-Oxone) was provided as well.
It should be emphasized that recyclable pollution-free catalysts reused in the mild reaction conditions with halogen-free solvents and ‘green’ oxidants provide a benign alternative to environmentally unfavorable (by-)products formed in the other reactions. The detailed description of all available methods for sulfolane synthesis is beyond the scope of this paper, but can be found elsewhere [
35].
Is sulfolane still an eye-catching compound for the scientific community? In fact, we found some highly interesting regularity while mining the commercially available Reaxys database [
37]. Interestingly, the analysis performed on data stored in the Reaxys database revealed an alternating tendency of waxing and waning interest in sulfolane during the space of the last fifty years as illustrated in
Figure 2. Extensive database screening was performed to identify 1644 hits, where word ‘sulfolane’ was queried in the title or abstract of the paper published during last five decades (from 1969 to 2018). Most of them describe sulfolane physicochemical properties (704), its chemical transformations (462), physicochemical analysis methods (450) or quantum chemical calculation procedures (33), respectively. More detailed analysis revealed, that many papers referred to solubilizers (320), ionic liquids (168), viscosity (140), solubility (106), organic solvents (100), solvent extraction (91), oxidation reaction (72), catalysts (72), separation methods (70), chemical stability (52), aprotic solvents (37), polar solvents (35), chemical engineering (35), solvent mixtures (28), toxicity (19), biodegradation (19), petroleum (18), liquid-liquid equilibrium (18), corrosion (9), etc. Furthermore, 189 patents and patent applications that relate to the synthesis of sulfolane, its purification and use as a solvent or extractor, are mostly owned by Philips Petroleum Company (24), Lloyd (15), Berg (15) and Mobil Oil Corporation (10), respectively.
A natural question appears, what actually triggers an increasing interests in sulfolane that fades after some years? Surprisingly, the waves of interest superpose on the fluctuations in average annual crude oil prices (
$/barrel) provided by the Organization of the Petroleum Exporting Countries (OPEC) [
37,
38]. Generally, the constant growth in number of publications related to sulfolane is observed throughout the years with a peak reached in 2016. Roughly speaking, the revealed ‘wavy’ trends in sulfolane interests provide an illustration of the partial correlations with oil prices as shown in
Figure 2. In the period of the relative oil prices stability (1987–1999) the amount of sulfolane-related papers was more or less constant, while the growth of the oil prices induced noticeable increase of the scientific activity, as observed between 1973 and 1985 or in a new millennium as well. Obviously, sulfolane is used in the aromatic extraction in the oil industry, where price per barrel put pressure on the economy of the refining processes.
Briefly, in the aromatic extraction process sulfolane is initially mixed with the petroleum feedstock to dissolve aromatics in the extractor column. A subsequent stripper column removes any remaining non-aromatics from the aromatic-laden sulfolane. Then, the sulfolane/aromatics mixture is fed through a recovery column, where aromatics are vaporized and captured. Sulfolane is recycled for reuse. A simplified flow diagram of this process with a typical extractor-stripper-regenerator configuration is shown in
Figure 3.
Sulfolane is vital in the processing technology offered by Shell for the (pre)treatment of natural gases. Combination of sulfolane (40–60%) with alkanolamines (30–45% of diisopropanolamine (DIPA) or 30–45% of methyl diethanolamine (MDEA)) and water (5–15%) is used as a physical solvent in the Sulfinol
® gas sweetening process to remove toxic levels of sour gases (e.g., H
2S, CO
2) from raw natural gas condensate [
39]. The Sulfinol
® process is a liquid–liquid extraction, blending liquid gas feed into liquid solvent feed to dissolve the sulfur compounds of interest, where sulfolane flows counter-current to the flow of the feed stream in the extractor column. Sulfur-containing compounds are separated from the solvent by heating, subsequently condensed and recovered. Sulfolane is regenerated to remove impurities and recycled for repeated usage.
2.2. Physicochemical Properties
Dichotomic (lipo/hydro-philic) nature of sulfolane, directly related to its chemical structure where the five-membered ring allows the sulfonyl moiety to be exposed at the interface (see
Figure 1a), makes it broadly used industrially, due to its advantageous combination of physicochemical properties [
13]. In fact, a strong solvation of cations by the oxygen atoms in the sulfone group, possibly increases nucleophilicity of the corresponding, less solvated anions. As a dipolar aprotic substance, sulfolane is completely miscible with a wide range of different liquids, e.g., water, acetone, glycerol, toluene and other aromatic solvents, but it does not dissolve in alkanes. Sulfolane, in its pure form, is almost odorless, colorless and weakly to moderately basic; it forms a globular molecule with a specific gravity of 1.26 g/cm
3 and a high Hildebrand solubility parameter (δ = 27.2 [MPa]
1/2) which translates into high solvency power for reactions containing polarizable intermediates [
20]. Basically, sulfolane is thermally stable, therefore it is the solvent of choice for different acid-catalyzed reactions at elevated temperatures, but at 220 °C, it decomposes slowly with the formation of sulfur dioxide and a polymeric material [
11]. With its high stability against strong acids and bases, sulfolane can operate within a wide range of reaction types/conditions including formation of various fluoroaromatic compounds, oxidations, nitrations, rearrangements, phosphonylations and condensation reactions, respectively. Some of its other more important physicochemical properties are provided in
Table 1.
Owning to its combination of chemical and/or physical properties, sulfolane has been employed in a variety of new applications as an extraction distillation solvent, polymer solvent, polymer plasticizer, polymerization solvent as well as in the electronic/electrical applications.
2.3. Sulfolane-Induced Corrosion
The liquid-liquid extraction of benzene/toluene/xylenes (BTX) from naphtha and gasoline fractions using sulfolane is usually achieved at temperatures in the range of 180–200 °C in a typical, extractor-stripper-regenerator configuration (see
Figure 3). Under standard operating conditions pure sulfolane is considered to be a stable compound (decomposition rate is 0.009%/h) [
11] and non-aggressive towards carbon/stainless steel, but at temperatures about 200 °C process of sulfolane decomposition spikes, followed by generation of SO
2 and formation of corrosive H
2SO
3 according to the generic Equation (2).
As a matter of fact, sulfolane should be stored under a nitrogen blanket and out of contact with oxygen and strong oxidizing agents such as chlorates, nitrates and peroxides, that can cause degradation. The presence of impurities such as oxygen and/or chlorides or water can accelerate sulfolane ‘breakdown’ process as well. In fact, some general correlations between oxygen/chloride and water concentrations are described, leading to enhanced sulfolane corrosivity, that causes noticeable issues of the refining installation as illustrated schematically in
Figure 3. Moreover, oxygen-degraded sulfolane has a lower pH with lower extractive power, a higher acid number and a darker color compared to pure sulfolane as shown in
Figure 4.
Chlorides react with organic acidic molecules and possibly sulfolane itself to create precipitates. Unfortunately, there is a ‘gap’ in the detailed and quantitative evaluation of individual impurities, their limits and interactions on corrosion rate of carbon and alloyed steels [
25]. Electrochemical corrosion monitoring techniques such as Linear Polarization Resistance (LPR), Electrochemical Noise (ECN) or Harmonic Analysis are broadly recognized for their responsiveness and accuracy for specification of instantaneous corrosion rate; however, application field has been basically limited by the necessity for a conductive/aqueous process environment. This limitation stems from the fact, that electrochemical systems cannot provide reliable readings in media at conductance levels less than 10μS/cm, due to significant impact of solution resistance on measured values of Polarization Resistance (PR). To overcome this limitation a noticeable progress in multi-technique electrochemical systems, involving low frequency impedance and harmonic distortion analysis, has been achieved. Corrosion control and monitoring in sulfolane units does not find prominent presence in published literature and what little is available focuses on indirect, qualitative assessments, e.g., color of process sulfolane, acid number or pH, rather than direct measurement of corrosion rates. Evidently, those qualitative approaches provide an approximately cumulative picture of sulfolane physicochemical quality, that does not necessarily assist in quantifying propensity for sulfolane corrosion in real systems.
Electrochemical techniques such as Linear Polarization Resistance (LPR)—capable for corrosion measurement in terms of minutes/hours may not work efficiently in a very low-conductive (3–10 μS/cm) environment of sulfolane-hydrocarbon mixture due to high IR-drop between electrodes. Other techniques, including electrochemical impedance spectroscopy (EIS) or Potentiodynamic/Galvanodynamic Polarization, are mostly suited for laboratory application and cannot be utilized for field operation. More functional space can be given to systems incorporating multi-electrochemical approach by combining low frequency impedance measurements (LFI), harmonic distortion analysis (HDA) and electrochemical noise (ECN), that may reduce, to a reasonable degree, the impact of IR-drop. With proper adjustment of electrode surface area and maintenance of close proximity between electrodes, it is possible to accurately measure corrosion rates, even at very low conductance levels (5 μS/cm or less) [
25]. The mechanism for detecting the result of corrosion is to monitor the equipment in contact with the solvent as described in the experimental (case study) section.
2.4. Biotransformation and Biodegradation
There is no routine environmental monitoring protocols and sufficient data available, that investigate the environmental fate including adsorption, (bio)transformation and excretion of sulfolane. In fact, sulfolane has been detected in contaminated groundwater samples and identified in the edible plant tissues, taken from nearby gardens surrounding industrial installations [
12]. Unfortunately, very little information is accessible on the toxicity and possible irritancy of sulfolane. In general, this substance appeared to be completely free from skin irritating due to low skin penetration (skin permeability of only 0.2 mg/(m
2/h)) and sensitizing properties with only very mild irritancy to the eyes [
11]. Although the human health impact is unknown, toxicity effects were examined extensively in rats exposed to sulfolane [
50], e.g., influence of sulfolane on behavioral and autonomic thermoregulation was studied in rats (and also in mice) [
51]. Aerosolized sulfolane is known to be acutely toxic at higher doses (over 200 mg/kg) with convulsions, vomiting, leukopenia and death in exposed guinea pigs, squirrel monkeys and dogs reported - none of these toxic effects were observed at exposures to lower concentrations (20 mg×m
-3) [
52]. Moreover, sulfolane produces toxic signs indicative of central nervous system (CNS) stimulation or dose-dependent depression at acute concentrations in mammals [
53,
54]. Available values of lethal dose, at which 50% percentage of subjects will die (LD
50) for various animals species are summarized in
Table 2.
The profound analysis of sulfolane toxicity is not the primary objective of this paper, but additional hazard data are presented in
Supplementary Materials (
Table S1).
In the last decades, the indirect impact of air/water/soil contamination on human health and the surrounding environment has been extensively scrutinized and is better understood now [
3,
57]. Traditionally, the industrial waste/sludge have been treated by physicochemical methods, which are costly, use lots of chemicals and generate waste products. On the other hand, the biopurification of the contaminated air/water/soil is gaining particular attention due to high cost-effectiveness (low investment and operating expenses), treatment efficiency and environmental acceptability. Microbiological methods are becoming attractive alternatives to the conventional physicochemical removal/cleaning procedures as they are based on the natural ability of microorganisms to degrade odorous/toxic contaminants from industrial/municipal airstreams and operate under mild conditions (at around ambient temperature and atmospheric pressure) [
1,
2]. In other words, organic contaminants can be attenuated by microbial degradation; however the rates of volatilization, diffusion and leaching are basically dependent on the sorption mechanisms (sorbate-sorbent interactions) of organic pollutants onto soil constituents [
22]. In fact, these microbial ‘predators’ use the pollutants as nutritional source to draw energy and carbon for growth (biomass production) and to maintain biological activity under aerobic or anaerobic conditions, whereas other nutrients (minerals and trace elements) need to be supplied. In a broader sense, a single/mixed microbial population or consortium might be regarded as the biocatalysts in the degradation process, which continually evolve to meet the changes in pollutant feed and environmental conditions transforming pollutants into biomass and harmless (by-)products [
7].
Since natural processes are not effective in sulfolane removal, several treatment methods have been suggested, including adsorption on biologically activated carbon [
58] and aerobic or anaerobic microcosm procedures to assess the biological and abiotic sulfolane degradation potential in the contaminated ground water and sediment. The mixed population of microbes was used to bioremediate the sulfolane-containing wastewater from refinery under aerobic conditions [
59]. The biodegradation potential of sulfolane by planktonic microbes residing in groundwater alone was examined as well [
51]. Several important factors limiting sulfolane biodegradation in the contaminated subarctic aquifer substrate were scrutinized extensively, including the impact of the nutrient limitations (nitrate, sulphate or iron-reduced conditions) as well as the hydrocarbon co-contamination on the sulfolane-contaminated samples under the oxic and anoxic conditions, respectively. Obviously, oxygen was specified as an important biodegradation stimulator, since anaerobic conditions do not appear to support sulfolane bioremediation. Basically, in situ nutrient amendments (mineral or organic) and hydrocarbon co-contamination are likely to naturally control the biodegradation rate, especially at the bacterial incubation stage. In order to augment the biodegradation of sulfolane in soil, the addition of nitrogen and, to lesser extent phosphate, is valid as was suggested by Greene et al. [
56,
57,
58,
59,
60,
61,
62].
Since there have been some studies reporting on the biodegradation of sulfolane in water, there is some knowledge ‘gap’ that hampers the proper understanding and assessment of the advanced oxidative processed (AOPs), employed in the treatment of sulfolane in an aqueous medium [
12]. Almost four decades ago, the vacuum UV photolysis of sulfolane was proposed with SO
2, cyclobutane, 1-butene and ethylene as the reaction products [
63]; however preliminary experiments on sulfolane degradation using H
2O
2/UV in the contaminated groundwaters were reported in 2005 [
53]. The application of UV-based AOPs to treat sulfolane in an aqueous medium (synthetic water and ‘real’ sulfolane-containing groundwater) with suitable photoactive oxidants (H
2O
2/TiO
2/O
3) was reported recently, revealing a ‘synergetic effect’ observed in the combined (H
2O
2/O
3) photolysis [
12]. Moreover, the effective method for mineralization of sulfolane-contaminated (ground)water by calcium peroxide (CaO
2) and calcium oxide (CaO) conjugated with ozone was proposed recently as well [
23]. Hence, the laboratory-scale experimental program was prepared to test the sulfolane bioremediation rate and specify the suitable working conditions [
54]. It was shown, that combination of high pH and CaO/CaO
2 system seems to enhance the sulfolane mineralization effectiveness. In fact, Fenton’s catalyst with the mixture of nitrilotriacetic acid (NTA), Fe(III) and H
2O
2 was also successfully employed for sulfolane degradation in groundwater [
26].
Removal of sulfur-based molecules from waste gases or groundwater is necessary for reasons of health, safety and also anti-corrosion protection due to corrosive potential of these compounds to mild steel or concrete during emission. Luckily, most odorous inorganic compounds are readily degraded in the desulfurization process by sulfur-oxidizing bacterial populations or microbial consortia [
64].
5. Conclusions
A worldwide rise of atmospheric pollution due to the expansion of industrial/agricultural areas and urban settlements is a peculiar ‘landmark’ of the modern civilization. The global ecosystem is confronted with volatile organic compounds (VOCs) as well as inorganic odorous compounds (VICs), that pose hazards to health of human beings and plants vegetation, forming a significant part of indoors/outdoors pollution. The sulfur-containing derivatives and their metabolites, regarded as ‘old devils of green’ chemistry, constitute a relevant class of air/water/soil contaminants with increasingly detrimental impact on human health following long-term exposure. In over-polluted world modern chemistry plays invariably pivotal role in development sustainable, atom-economical and operationally simple protocols to provide eco-friendly (by-)products; however some industrially-engineered solvents have become environmentally unfavorable. Pollutants emitted from certain solids or liquids originate from anthropogenic activity or the biogenic emissions of certain reactive hydrocarbon derivatives formed as chemical (by-)products of natural transformations, respectively. The increasing public awareness of necessity for environmental protection is the main driving force for the increasingly stringent regulations governing release of hazardous pollutants and reduced sulfur compounds. Hence, environmental legislations are constantly pushing industry for developing/optimizing of cost-effective ‘green’ manufacturing technologies, that impose less burden on the ecosystem An attractive alternative to commonly used industrial extractive liquids is sulfolane (C4H8SO2), a versatile dipolar aprotic solvent. In the present review we mainly focus on the sulfolane synthesis, application of sulfolane as an extractive solvent due to its ‘unique’ physicochemical properties as well as the potential of sulfolane to cause equipment corrosion and subsequent spills. For the purpose of the proposed investigations a dedicated testing vessel was designed and constructed. The potential risk for groundwater contamination, danger for human health and ways of sulfolane biodegradation were reviewed briefly as well. Moreover, the primary goal of the presented case study was to verify applicability of industrial, multi-electrochemical technique for reliable detection of corrosion in low conductive process fluids. Several aspects of corrosion measurement of general and localized corrosion modes were investigated. Incipient attempts to quantify impact of process parameters (temperature) and impurities (oxygen and chlorides) on stainless steel corrosion in pure sulfolane provided reasonably meaningful. A number of experiments to assess the effect of various process parameters on the corrosion behavior of AISI 304L steel in sulfolane were carried out. To summarize the experimental section:
general corrosion rate during all experiments did not exceed of 1 mpy, which is expected behavior of austenitic stainless steel;
clear impact of temperature on corrosion rate was observed with its maximum at 230 °C;
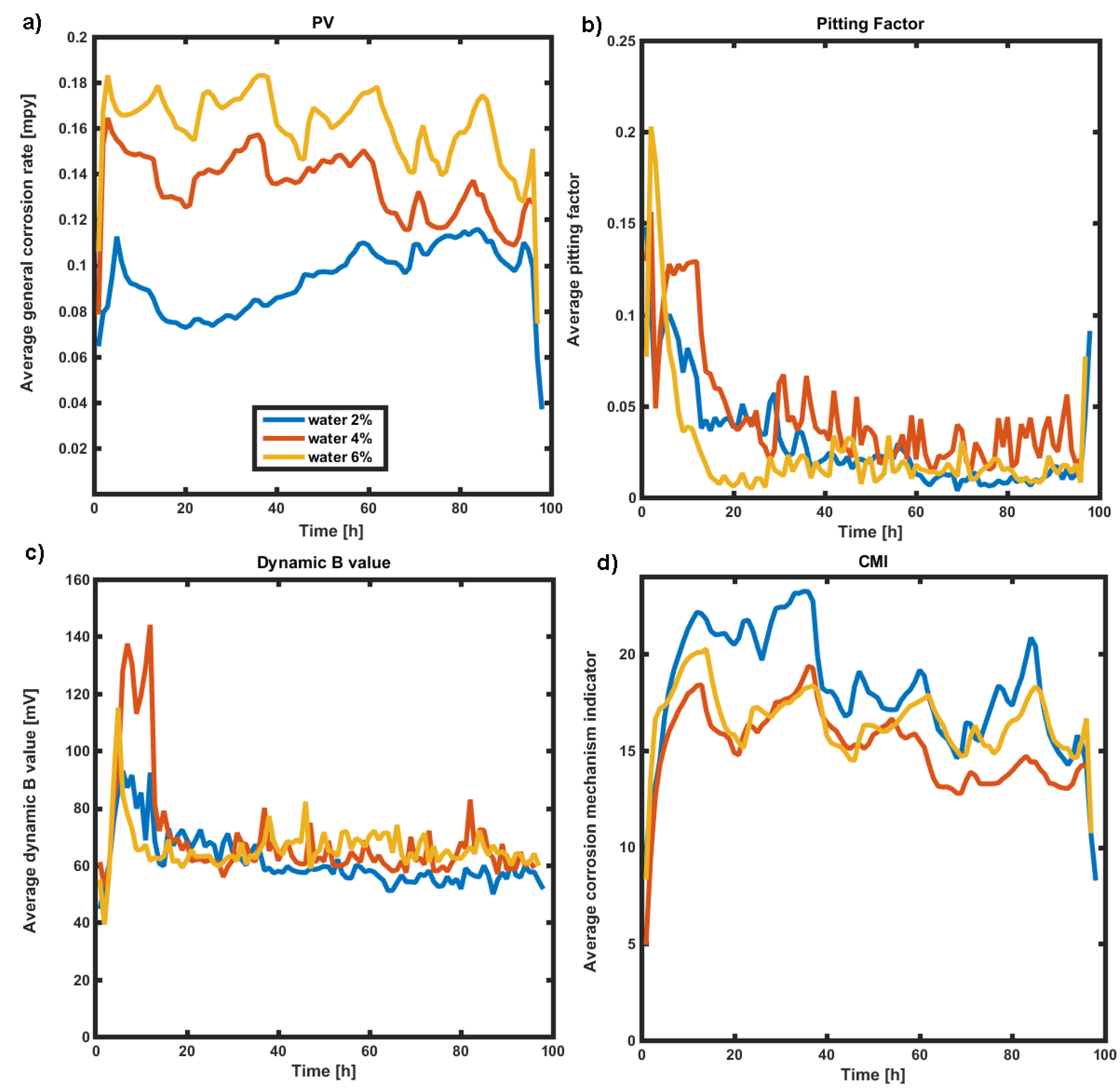
general (uniform) degradation mode was dominant as confirmed by both Pitting Factor values (<0.1) and surface microscopic examination;
strong impact of water (even at low temperature) on sulfolane corrosivity was observed—increase of water concentration accelerates sulfolane degradation as indicated by elevated corrosion rate and increase of suspended dark deposits;
no impact of oxygen on general corrosion and localized corrosion potential was revealed;
chlorides generate the most severe corrosive environment (at test temperature of 95 °C) in terms of general corrosion, but do not enhance localized corrosion phenomenon;
no significant deposition was observed during all tests—CMI parameter was below 100 units.
It should be emphasized, that there is a clear absence of detailed and quantitative evaluation of individual impurities, their limits and interactions on corrosion rate of carbon and alloyed steels; therefore more detailed control of experiments, a wider range of solution conditions, a wider range of temperatures, and with this the corresponding surface analysis of the electrodes must be performed in the future.