1. Introduction
One of the basic functions of soil microorganisms is the decomposition and transformation of organic materials, which are mostly derived from above and below-ground plant residues. Thus, soil microbial communities play a critical role in ecosystem processes, such as carbon cycling, nutrient turnover, or the production of trace gases. Soil microbial activities, populations and communities are governed by environmental variables and agricultural system, as conventional and organic system [
1,
2].
Conventional agriculture has an important role in improving food productivity to meet human demands. However, this system has been largely dependent on intensive chemical inputs. On the other hand, organic agriculture does not use synthetic fertilizers and pesticides and attempt to close nutrient cycle on their farms, protect environmental quality and enhance beneficial biological interactions and processes [
3]. Organic agriculture is gaining worldwide acceptance and has been expanding at annual rate of 20% in the last decade accounting for over 24 million hectares worldwide [
4]. In Brazil, organic production has intensified to meet market demands over the last years. This system avoid applications of synthetic fertilizers and pesticides, and use of organic inputs and recycling for nutrient supply, and emphasize cropping system design and biological processes for pest management, as defined by organic certification programs in Brazil [
2]. Also, it can reduce some negative effects attributed to conventional agriculture and has potential benefits in enhancing soil quality [
5].
Soil quality is the capacity of soil to maintain some key ecological functions, such as decomposition and formation of soil organic matter [
6]. Microbial processes are important for the management of farming system and improvement of soil quality. Microbial respiration of soil has received considerable attention because it can be used as a soil quality indicator [
7] and it is one important variable to quantify soil microbial activity [
8]. Soil microbial biomass carbon is considered the most dynamic and labile component of soil organic carbon. Thus, the pool of soil microbial carbon, its activity and composition are key parameters in soil processes [
7].
The number of studies comparing organic and conventional systems is limited, mainly on soil microbial processes. In Brazil, There is a long-term experiment aiming evaluate conventional and organic farming. The hypothesis for this study is that there would be changes in soil microbial activity by practices in organic farming due high input of organic C. In this paper, the objective was to investigate microbial activity and biomass of soil in plots under conventional and organic agriculture system. Here, there are results concerning basal respiration and soil microbial biomass of the second and third years after implantation of farming system.
2. Results
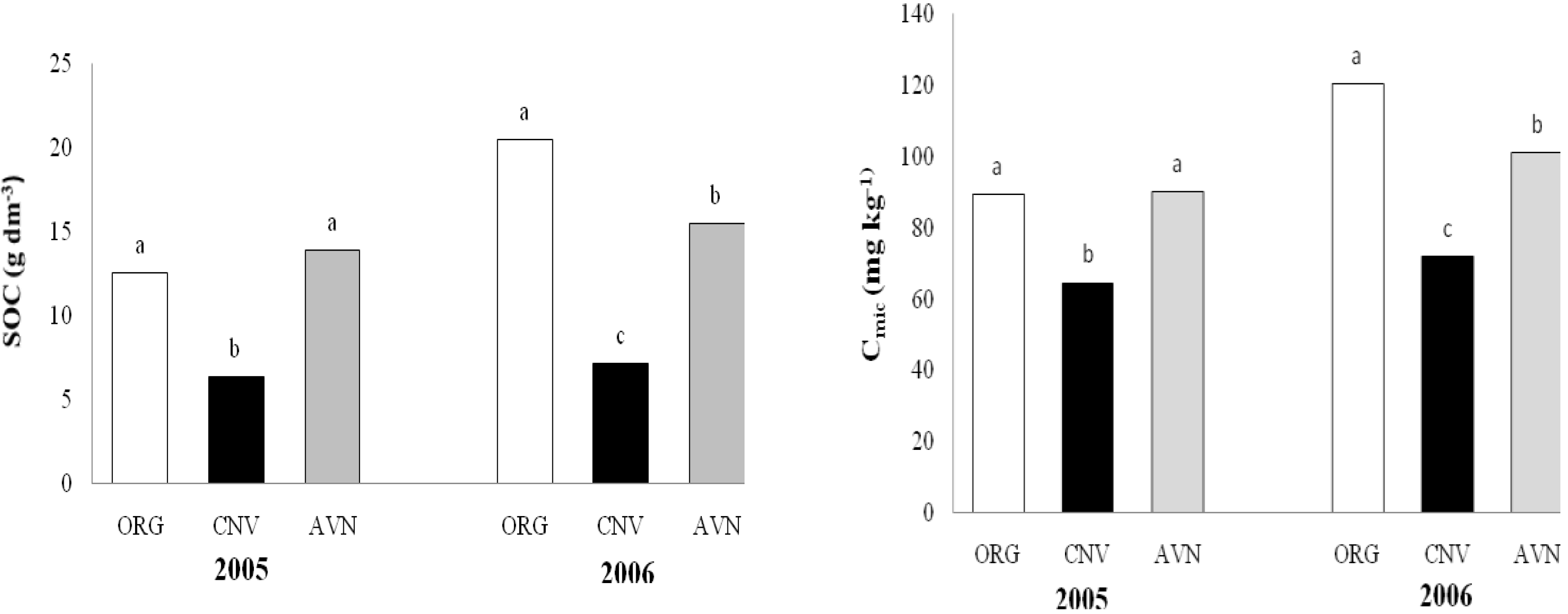
Soil organic carbon (SOC) was higher in the organic area (ORG) than in the conventional area (CNV) (
Figure 1). Compared with soil in native vegetation (AVN), SOC content was higher in ORG only in 2006. At the end of the third year of the implantation of farming systems (2006), SOC content were 20.5, 15.5 and 7.2 g kg
-3 in the ORG, AVN and CNV, respectively.
A similar pattern was observed for soil microbial biomass C (C
mic) where there was a difference between soil under organic and conventional agriculture system (
Figure 1). In 2005, C
mic contents were 89.4, 64.5 and 90 mg kg
-3 in the ORG, CNV and AVN, respectively, whilein 2006, C
mic contents were 120.5, 72 and 101 mg kg
-3 in ORG, CNV and AVN, respectively.
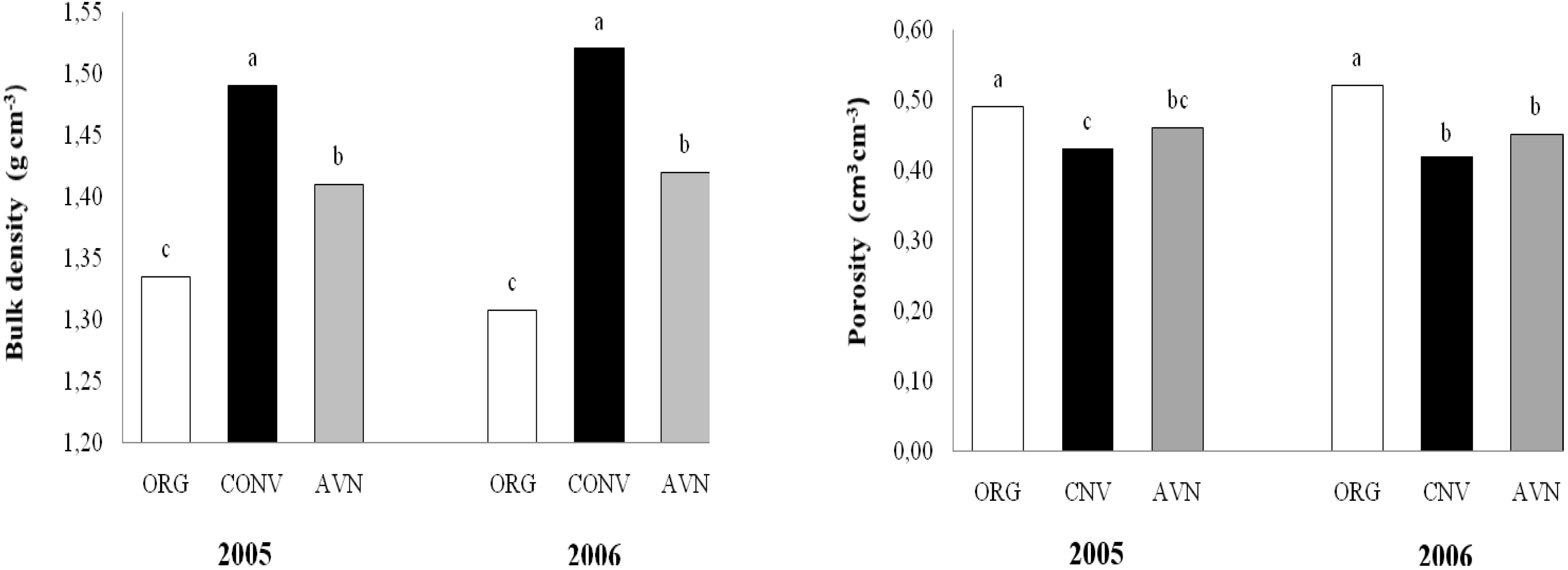
Soil bulk density and porosity were lower and higher, respectively, in the ORG area than in the CNV and AVN areas after three years of management in organic and conventional farming (
Figure 2).
Figure 1.
Soil microbial biomass (Cmic) and organic carbon (SOC) in plots under conventional and organic farming and native vegetation.
Figure 1.
Soil microbial biomass (Cmic) and organic carbon (SOC) in plots under conventional and organic farming and native vegetation.
Figure 2.
Soil bulk density and soil porosity in plots under conventional and organic farming and native vegetation.
Figure 2.
Soil bulk density and soil porosity in plots under conventional and organic farming and native vegetation.
At the end of the third year, the average of soil bulk density was 1.31, 1.42 and 1.52 g cm-3 in the ORG, AVN and CNV, respectively. Contrasting to SOC content, the soil bulk density decreased significantly (P < 0.05) following organic practices.
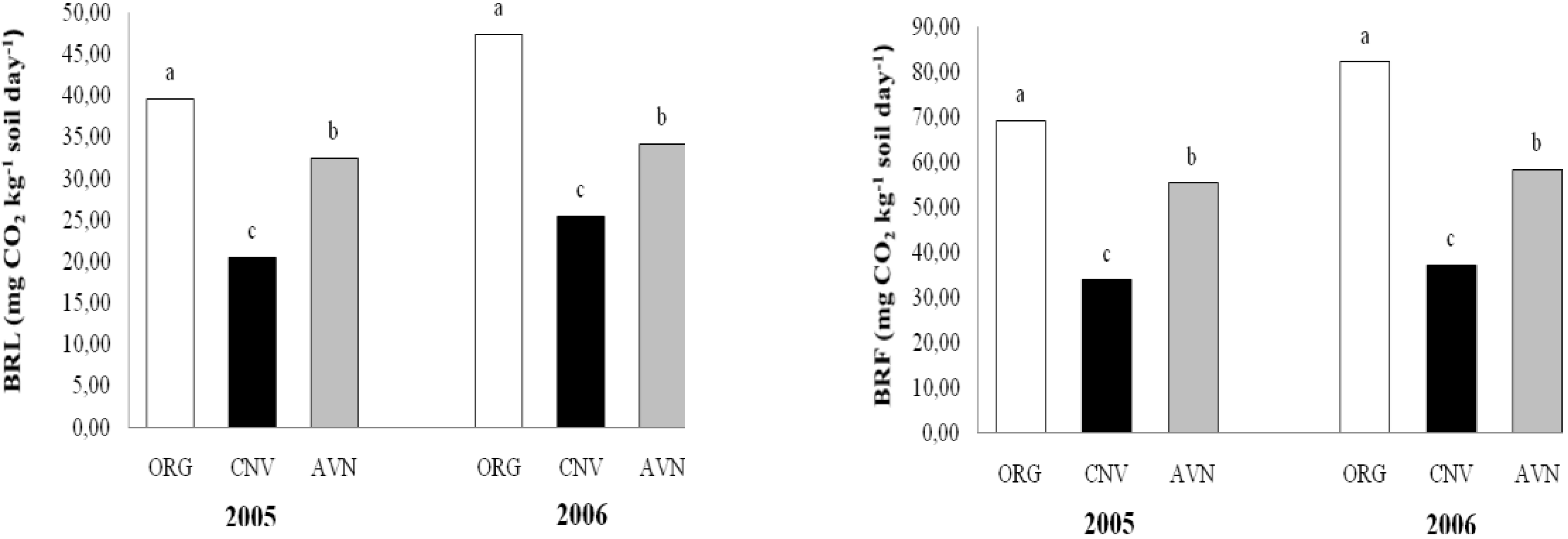
Figure 3.
Soil basal respiration in laboratory (BRL) and field (BRF) in plots under conventional and organic farming and native vegetation.
Figure 3.
Soil basal respiration in laboratory (BRL) and field (BRF) in plots under conventional and organic farming and native vegetation.
Soil basal respiration in laboratory (BRL) and field (BRF) assays were significantly (P < 0.05) enhanced by the organic practices in both years as compared to the CNV and AVN (
Figure 3). In ORG, the basal respiration rates statistically significantly increased in the laboratory and field, respectively, from 39.6 and 69.2 mg CO
2 kg
-1 day
-1 in 2005 to 47.4 and 82.1 mg CO
2 kg
-1 day
-1 in 2006.
On the other hand, in both years, the lower values for soil basal respiration rates were observed in conventional farming system (CNV). There was a significant correlation between soil respiration estimated in laboratory and in field (
Table 1), indicating that the both methods are suitable for soil basal respiration estimation.
Table 1.
Linear correlation among soil organic carbon (SOC), bulk density (SBD), total porosity (TP) and soil basal respiration in field (BRF) and laboratory (BRL).
Table 1.
Linear correlation among soil organic carbon (SOC), bulk density (SBD), total porosity (TP) and soil basal respiration in field (BRF) and laboratory (BRL).
| | SBD | TP | SOC | BRF | BRL |
|---|
| SBD | 1 | - 0.721* | - 0.628* | - 0.651* | - 0.509 |
| TP | | 1 | 0.693* | 0.702* | 0.435 |
| SOC | | | 1 | 0.663* | 0.520 |
| SRF | | | | 1 | 0.702* |
| SRL | | | | | 1 |
3. Discussion
The results indicate that organic practices related with permanents C inputs (annually 50 Mg ha
-1 of cow compost) can significantly impact SOC and C
mic content. The increase in SOC content is important for semi-arid region, as Piauí state, due to the low levels of organic matter observed for our soils. The high SOC is important for sustainability since that organic matter influence soil physical, chemical and biological properties [
9]. These results are in accordance with Glover
et al [
10] and Melero
et al. [
1] who observed that organic farming system has been shown to maintain soil organic matter at high levels than conventional system.
Additionally, the increase of C
mic in organic plots is, probably, also due to the microbial biomass contained in the organic amendments. The results are in agreement with the findings of Melero
et al. [
1] and Tu
et al. [
11] in soils under organic farming systems. According to Fließbach and Mader [
12], over the long term, microbial biomass C is significantly affected by the long-term management as well as by its intensity.
The lower bulk density observed in ORG is due to permanent addition of compost that contributes to increase organic matter input, decreasing soil bulk density [
13]. The results concerning soil bulk density and total porosity indicated that the increase of soil porosity was accomplished by decrease in soil bulk density, as reported by Werner [
14] in organic systems. This effect under organic farming is important and may have implications for plant root growth and nutrient availability to crops. Similar results were mentioned by Swezey
et al. [
15] and Glover
et al. [
10] in comparison between organic and conventional apple orchards plots.
The higher soil respiration in the organic farming system indicates a higher soil microbial activity due to the permanent and continuous addition of an exogenous source of labile organic matter to the soil and the consequent stimulation of heterotrophic microorganisms [
16].
Other comparisons of conventional and organic systems have also reported increased soil microbial respiration under organic management [
10,
17]. Additionally, higher soil respiration, in the organic farming system, is an indicative of high biological activity, suggesting rapid decomposition of organic residues that make nutrients available for plant growth.
In accordance with Franzluebber and Arshad [
18], CO
2 production in laboratory assay was strongly correlated with CO
2 production in field assay. The soil bulk density was negatively correlated with total porosity and SOC (
Table 1). The soil respiration in field was significantly correlated with soil porosity and bulk density, indicating that the soil respiration is influenced by these factors. The measurement of soil bulk density provides a relative value of soil compaction and reflects significant changes in their structure. The increase in soil bulk density promotes lower porosity and WFPS, by reduction in the size and number of macropores, decreasing soil aeration and, consequently, soil microbial activity [
19]. The increase in soil porosity is important for soil respiration by increase in aeration necessary for biological process.
4. Experimental Section
The field study was carried out at the Irrigation District of Piauí, Brazil (03o05’ S; 41o47’ W; 46 m). The climate is rather dry with a mean precipitation of 1,000 mm yr-1, with seasonal fluctuation during the year, and an annual mean temperature of 30oC. The soil is a Typic Quartzipsamment (according to the USDA soil classification) and showed 920, 50 and 30 g kg-1 of sand, silt and clay contents, respectively.
Table 2.
Annual inputs of fertilizers (N-P-K) on the conventionally and organically managed plots.
Table 2.
Annual inputs of fertilizers (N-P-K) on the conventionally and organically managed plots.
| Conventional system | Organic system |
| 01/2005 | 01/2004 |
| 100 kg N ha-1 (Urea) | Green manure (11 Mg ha-1 of fresh biomass) |
| 80 kg P2O5 ha-1 (Superphosphate) | 0.5 Mg ha-1 rock phosphate (24% P) |
| 40 kg K2O ha-1 (KCl) | 1.2 Mg ha-1 “MB4” |
| 07/2005 | 04/2004 |
| 100 kg N ha-1 (Urea) | 50 Mg ha-1 cow compost |
| 40 kg K2O ha-1 (KCl) | 0.5 Mg ha-1 rock phosphate (24% P) |
| | 1.2 Mg ha-1 “MB4” |
| | 50 Mg ha-1 carnauba straw. |
| 01/2006 | 04/2005 |
| 100 kg N ha-1 (Urea) | 50 Mg ha-1 cow compost |
| 80 kg P2O5 ha-1 (Superphosphate) | 0.5 Mg ha-1 rock phosphate |
| 40 kg K2O ha-1 (KCl) | 1.2 Mg ha-1 “MB4” |
| | 50 Mg ha-1 carnauba straw. |
Three different areas were evaluated and showed the following management: a) Conventional farming with
Malpighia glabra (CNV); b) Organic farming system with
Malpighia glabra (ORG); Native vegetation (AVN) used as reference system. The size of each area varied between 0.2 and 0.5 hectare. The area in conventional farming is managed since 1999 (with annual cowpea/watermelon crop rotation). The “acerola” plants were sowed in 2004 and the management includes annual applications of chemical and organic fertilizers (
Table 2).
The area under organic farming was implanted after the withdrawal of all conventional practices and was managed since 2003. Initially, the area in organic system received inputs from green manure (
Crotalaria juncea, Vigna unguiculata, Cajanus cajan) that contributed with approximately 400 kg ha
-1 N fixed (Araújo, unpublished). The green manure was incorporated in soil at 0-20 cm depth. Additionally, there was the application of 0.5 t ha
-1 of rock phosphate and 1.2 t ha
-1 of lime (“MB4”). Additional information about these systems is in Araújo
et al. [
2]. The
Table 3 shows the chemical characteristics of composted cow manure used as organic input.
Table 3.
Some chemical characteristics of the composted cow manure added to organic plots.
Table 3.
Some chemical characteristics of the composted cow manure added to organic plots.
| Moisture (g kg-1) | 198 |
| pH (1:2.5) | 6.9 |
| EC (1:2.5) | 1.8 |
| Organic Matter (g kg-1) | 281.6 |
| Total nitrogen (g kg-1) | 8.32 |
| C/N | 19.1 |
| P-available (g kg-1) | 4.1 |
| K-exchangeable (g kg-1) | 3.5 |
The organic amendments are applied in superficial form in the plant canopies. In the disease and insect control in organic management system are used alternative methods, as plant extract with “nin” (Azadiractha indica) and biocontrol with Baccilus and Beuveria bassiana.
Soil samples were collected at August 2005 and September 2006. The areas under ORG, CNV and AVN systems were divided in four sub-areas with about 300 m2 (four randomized replicates). In each sub-area, soil samples were taken at 0-20 cm and were selected using a grid pattern at 20 points (under the plant canopy). The soil samples from each point were bulked and thoroughly mixed with in a plastic bag, and a composite sample was taken. The composite samples for each sub-areas and depth were air-dried and sieved at 2 mm.
Soil pH was measured in a 1:2.5 (soil:water ratio). Exchangeable calcium (Ca
2+) and magnesium (Mg
2+) were determined using 1N potassium chloride. Soil organic carbon (SOC) was quantified by wet combustion using a mixture of potassium dichromate and sulfuric acid under heating [
20]. Exchangeable K
+ and available P were extracted by method described by Tedesco
et al. [
21]. The
Table 4 shows soil chemical properties in all areas evaluated.
Table 4.
Soil chemical properties of plots under conventional and organic farming and native vegetation.
Table 4.
Soil chemical properties of plots under conventional and organic farming and native vegetation.
| | pH | P | K | Ca | Mg | CEC |
|---|
| | | mg dm-3 | ---------------- cmolc dm-3 ------------------ |
|---|
| | | | | 2005 | | |
| ORG | 7.0 | 98.7 | 0.1 | 2.3 | 0.8 | 3.7 |
| CNV | 7.1 | 103.2 | 0.3 | 1.8 | 0.6 | 3.1 |
| AVN | 6.8 | 99.1 | 0.2 | 2.1 | 0.8 | 3.5 |
| | | | | 2006 | | |
| ORG | 7.2 | 96.5 | 0.6 | 2.4 | 0.9 | 4.2 |
| CNV | 7.0 | 71.1 | 0.1 | 2.4 | 1.1 | 4.1 |
| AVN | 6.9 | 77.4 | 0.2 | 2.2 | 1.0 | 3.9 |
Bulk density was measured using intact soil cores (50 mm deep and 70 mm diameter insert in all points described before. for each sub-area). Total porosity was determined according to USDA [
22].
Soil respiration evaluation in laboratory was based on the estimation of CO
2-C evolution by an incubation-alkaline absorption method [
8]. The same points located for soil samples were used for evaluation of soil respiration in field. The CO
2-C was determined using an alkali absorption technique [
23]. The values were corrected to a 24 h basis and adjusted to equivalent at 60% WFPS through the equation described by Parkin
et al. [
24]. The values in both soil respiration methods were expressed as mg CO
2 kg
-1 soil day
-1. Soil microbial biomass carbon (C
mic) was determined according to Vance
et al. [
25] with extraction of organic carbon (C) from fumigated and unfumigated soils by 1 M K
2SO
4. Organic C was measured using dichromate digestion and an extraction efficiency coefficient of 0.38 was used to convert the difference in soluble C between the fumigated and the unfumigated soil in microbial C
mic.
A split-plot ANOVA with whole areas in a randomized design treatment as the whole area factor and year as the subplot factor was performed. If the treatment main effect was significant and interactions were non-significant mean separation was carried out using the LSD procedure on main effect means for treatments. If treatment and year (or sampling date) interaction was significant, the LSD was applied to treatment means separately for each year (or each date).