Agricultural Systems Located in the Forest-Savanna Ecotone of the Venezuelan Amazonian. Are Organic Agroforestry Farms Sustainable?
Abstract
:1. Introduction
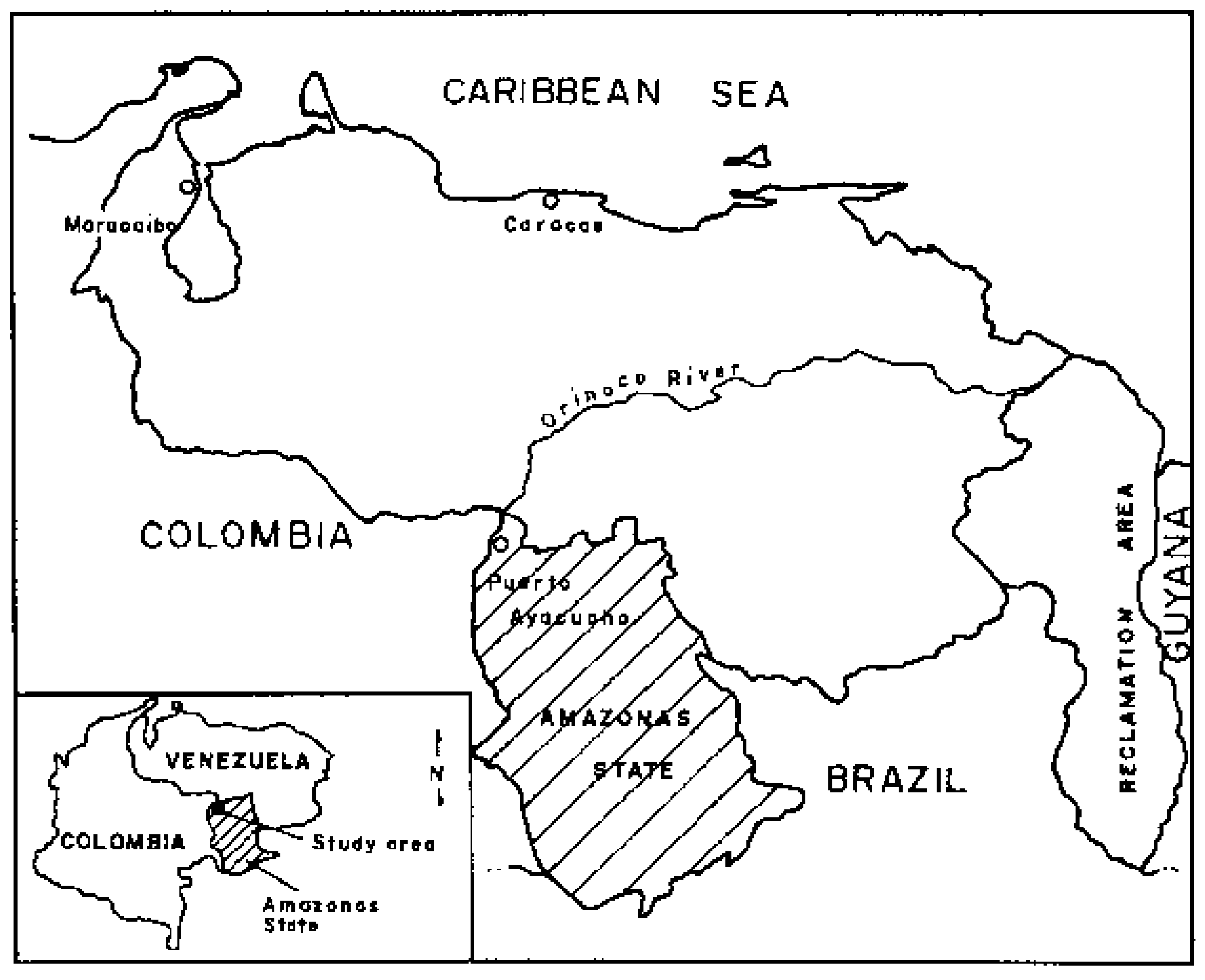
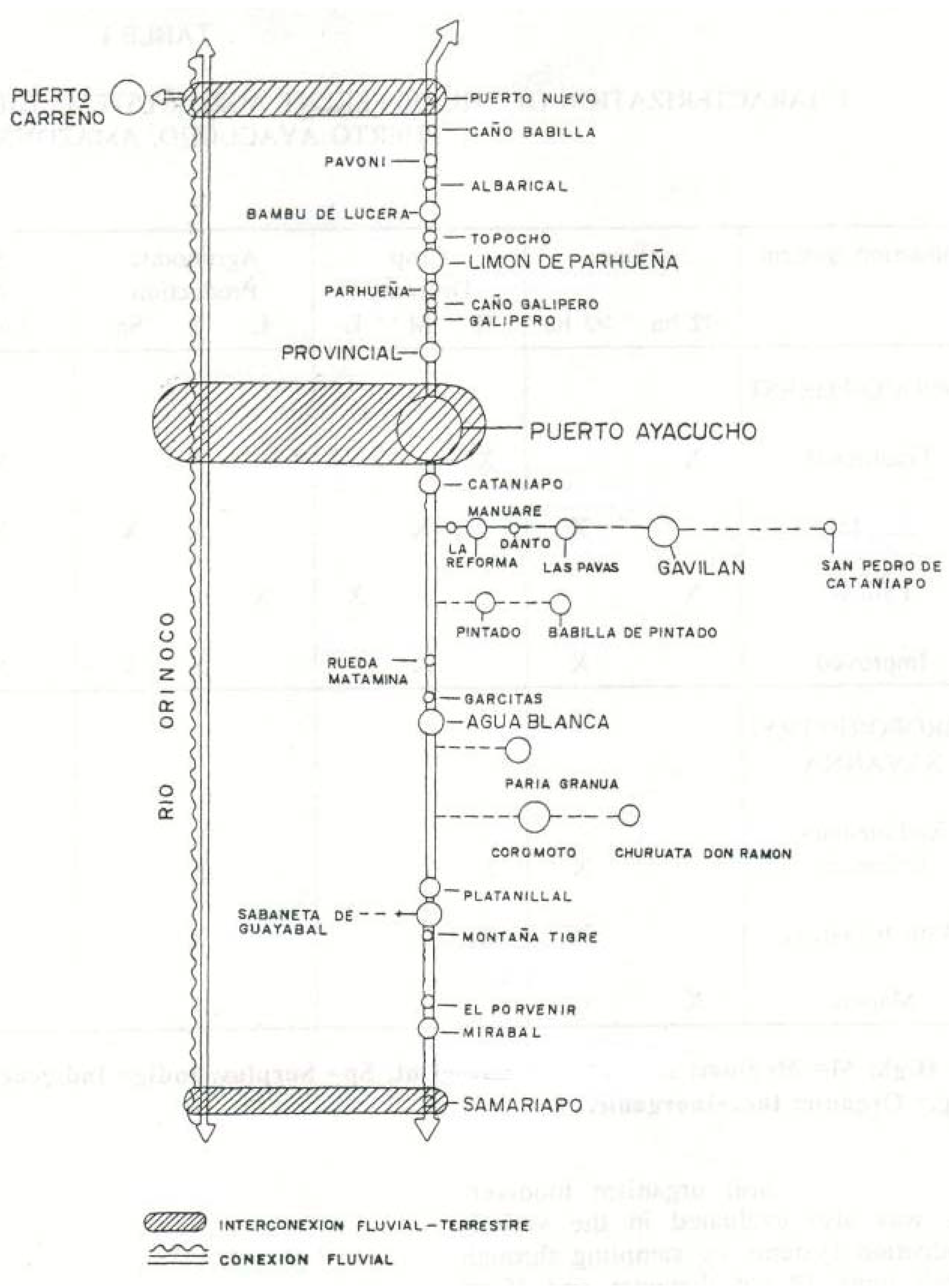
2. Materials and Methods
2.1. Site Description
2.2. Organic Agricultural Systems (OS) Assessed
2.2.1. Farm amended with cattle manure (CM)
2.2.2. Farm amended with chicken manure (CHM)
| Agricultural Systems | Type of Plantation | Surface | Type of fertiliser | Time of addition | Dose of Manure | Irrigation |
|---|---|---|---|---|---|---|
| Organic-CM | Year crops | < 2 ha | Cattle | >1 yr | 10 Mg ha-1 | No Irrigation |
| Organic-CHM | Agro-forestal | 5 ha | Chicken | 30 yr | 2 Mg ha-1 | Manual Irrigation |
| Organic-Comp | Agro-forestal | 5 ha | Chicken Compost | 30 yr | 2 Mg ha-1 | Sprinkles |
| Organic-GM | Yucca | < 2 ha | Green manure | >1 yr | Unknown | No Irrigation |
| Inorganic- I 1 | Brachiaria | 20 ha | Inorganic | 1 year | Unknown | No Irrigation |
| Inorganic-I 2 | Brachiaria, Leguminous | 0.4 ha | Inorganic | 2 years | 1 Mg ha-1 phosphate rock, 0.3 Mg ha-1 NPK | No Irrigation |
2.2.3. Farm amended with compost (Comp)
| OS | CM | CMH | Comp | |||
|---|---|---|---|---|---|---|
| Control | Treatment | Control | Treatment | Control | Treatment | |
| Water retention capacity (%) | 6.44 a ± 0.4 | 9.75 b ± 0.5 | 7.99 a ± 0.4 | 8.98 b ± 0.6 | 6.44 a ± 0.6 | 12.97 b ± 2.2 |
| Bulk density (Mg m -3) | 1.76 a ± 0.03 | 1.46 b ± 0.02 | 1.66 a ± 0.03 | 1.54 b ± 0.03 | 1.76 a ± 0.04 | 1.47 b ± 0.02 |
| Sand (%) | 95.9 a ± 1.1 | 96.9 a ± 1.1 | 95.5 a ± 1.2 | 95.0 a ± 1.1 | 95.9 a ± 1.2 | 93.4 a ± 2.2 |
| Silt (%) + clay (%) | 4.1 a ± 1.1 | 3.1 a ± 1.3 | 4.5 a ± 1.1 | 5.0 a ± 1.2 | 4.1 a ± 1.3 | 6.6 a ± 2.2 |
2.2.4. Site amended with green manure (GM)
2.3. Inorganic Agricultural Systems (IS) Assessed
2.3.1. Traditional cattle ranch (Hato llanero) cropped with Brachiaria (I 1)
2.3.2. Association of grass and leguminous inorganic fertilised (I 2)
2.4. Soil Sampling
2.4.1. Microbial biomass and enzymatic activities
2.4.2. Pedofauna sampling
2.5. Statistics
3. Results
3.1. Soil Physical Changes of Organic Agricultural Systems (OS)
3.2. Soil Chemical Changes of Organic Agricultural Systems (OS)
| OS | CM | CHM | Comp | GM | ||||
|---|---|---|---|---|---|---|---|---|
| T | C | T | C | T | C | T | C | |
| pH | 7.1 a ± 0.02 | 5.6 b ± 0.12 | 5.9 a ± 0.03 | 5.4 b ± 0.05 | 5.5 a ± 0.10 | 5.3 a ± 0.04 | 5.3 a ± 0.00 | 5.4 a ± 0.01 |
| TOC (g kg-1) | 33050 a ± 12476 | 4475 b ± 1276 | 12530 a ± 4674 | 6950 b ± 463 | 11575 a ± 3445 | 8178 a ± 793 | 6525 a ± 496 | 5328 a ± 1026 |
| N (g kg-1) | 2107 a ± 1172 | 422 b ± 116 | 557 a ± 29 | 296 a ± 72 | 1078 a ± 123 | 249 b ± 63 | 471 a ± 157 | 278 a ± 27 |
| C/N | 15.7 | 10.6 | 22.5 | 23.5 | 10.7 | 32.8 | 13.9 | 19.2 |
| Avail. P (g kg-1) | 113.0 a ± 8.7 | 0.5 b ± 0,5 | 96.0 a ± 11.2 | 2.2 b ± 2.2 | 20.0 a ± 0.5 | 0.6 b ± 0.1 | 0.9 a ± 0.8 | 0.2 b ± 0.1 |
| Organic agricultural system | CM | CHM | Comp | |||
|---|---|---|---|---|---|---|
| Control | Treatment | Control | Treatment | Control | Treatment | |
| Magnesium (µg/g) | 26 b ± 1.5 | 811 a ± 13 | 30 b ± 1.2 | 349 a ± 5 | 16 b ± 0.2 | 145 a ± 11 |
| Calcium (µg/g) | 162 b ± 10 | 1555 a ± 17 | 175 b ± 13 | 1954 a ± 25 | 60 b ± 1.4 | 864 a ± 67 |
| Potassium (µg/g) | 60 b ± 6 | 232 a ± 0.0 | 68 b ± 2 | 167 a ± 5 | 42 b ± 1 | 108 a ± 3 |
3.3. Microbial C, N and P in Organic Agricultural Systems (OS)
| OS | CM | CHM | Comp | GM | ||||
|---|---|---|---|---|---|---|---|---|
| T | C | T | C | T | C | T | C | |
| Mineralisable N (µg g-1) | 49.8 a ± 29.7 | 16.1 b ± 2.4 | 13.7 a ± 4.4 | 11.0 a ± 1.2 | 23.4 a ± 0.6 | 16.2 b ± 2.1 | 17.7 a ± 4.0 | 12.1 a ± 1.1 |
| C-MB (µg g-1) | 122 a ± 59 | 98 a ± 33 | 154 a ± 23 | 54 b ± 22 | 106 a ± 50 | 54 b ± 25 | 81 a ± 44 | 50 a ± 14 |
| Microbial quotient (%) | 0.37 | 2.19 | 1.23 | 0.78 | 0.92 | 0.66 | 1.24 | 0.94 |
| N-MB (µg g-1) | 59 a ±38.6 | 8 b ± 2.8 | 19 a ± 2.5 | 12 a ±2.9 | 22 a ± 2.0 | 12 a ± 1.2 | 11 a ± 4.0 | 13 a ± 5.2 |
| C-MB/ N-MB | 2.07 | 12.25 | 8.11 | 4.50 | 4.81 | 4.50 | 7.36 | 3.85 |
| P-MB (µg g-1) | n.d. | 0.2 ± 0.05 | n.d. | 0.2 ± 0.1 | n.d. | 0.2 ± 0.01 | n.d. | 0.5 ± 0.2 |
| UA (µg g-1h-1) | 70 a ± 12 | 39 b ± 13 | 4 a ± 1 | 9 a ± 3 | 12.1 a ± 10 | 9.2 a ± 7 | 30 a ±23 | 32 a ± 3 |
| PA (µg g-1h-1) | 37.0 a ± 1.7 | 10.0 b ± 2.4 | 27.9 a ± 4.7 | 15.0 b ± 3.3 | 49.6 a ± 1.5 | 10.7 b ± 7.5 | 7.2 a ± 1.5 | 3.2 a ± 1.5 |
3.4. Enzymatic Activities in Organic Agricultural Systems (OS)
3.5. Soil Chemical Analysis of Inorganic Agricultural Systems (IS) in Savannas
| IS | Brachiaria-I 1 | Brachiaria-Leguminous-I 2 | ||
|---|---|---|---|---|
| T | C | T | C | |
| pH | 4.9 a ± 0.02 | 4.9 a ± 0.1 | 5.3 a ± 0.02 | 5.3 a ± 0.01 |
| TOC (g kg-1) | 4,500 a ± 242 | 5,050 a ± 739 | 4,350 a ± 172 | 5,328 a ± 1026 |
| N (g kg-1) | 294 a ± 66 | 298 a ± 83 | 376 a ± 88 | 278 a ± 27 |
| C/N | 15.3 | 16.9 | 11.6 | 19.2 |
| Avail. P (g kg-1) | 1.0 a ± 0.04 | 0.1 b ± 0.2 | 0.34 b ± 0.2 | 0.23 b ± 0.1 |
3.6. Microbial C, N and P and Enzymatic Activities in Inorganic Agricultural Systems (IS)
| IS | Brachiaria-I 1 | Brachiaria-Leguminous-I 2 | ||
|---|---|---|---|---|
| T | C | T | C | |
| Mineralisable N (µg g-1) | 13.6 a ± 10.9 | 9.9 a ±0.5 | 18.5 a ± 3.9 | 12.1a ± 1.0 |
| C-MB (µg g-1) | 78 a ± 35 | 47 a ± 39 | 74 a ± 38 | 50 a ± 14 |
| Microbial quotient (%) | 1.73 | 0.93 | 1.70 | 0.94 |
| N-MB (µg g-1) | 20 a ± 3.4 | 13 a ± 0.02 | 20 a ± 7.8 | 13 a ± 5.3 |
| C-MB/ N-MB | 3.9 | 3.6 | 3.7 | 3.8 |
| P-MB (µg g-1) | n.d | n.d | n.d. | 0.5 ± 0.2 |
| UA (µg g-1h-1) | 73 a ± 0.6 | 43 b ± 30 | 46 a ± 34 | 32 a ± 3 |
| PA (µg g-1h-1) | 9 b ± 4.4 | 18 a ± 2.0 | 10 a ± 1.1 | 3 b ± 1.5 |
3.7. Community Structure and Abundance of Macro Arthropods
| Groups | Comp | CHM | CM | NS |
|---|---|---|---|---|
| Formicidae | 780.4 ± 357.9 | 400.0 ±152.0 | 2660.0 ± 1078.9 | 305.4 ±160.7 |
| Coleoptera | 191 ± 51.8 | 54.3 ± 13.2 | 663.3 ± 214.2 | 42.9 ± 26.5 |
| Hemiptera | 8.9 | 16.1 | 336.7 | 5.4 |
| Aphidoidea | 19.6 | 7.1 | 6.7 | |
| Homoptera | 8.9 | 7.1 | 23.3 | 7.1 |
| Orthoptera Mantodea Gryllidae Gryllotalpidae | 71.4 | 30.4 | 130.0 | 32.1 |
| Blattodea | 85.7 | 5.4 | 3.3 | 1.8 |
| Psocoptera Thysanoptera | 1.8 | 3.3 | ||
| Plecoptera Dermaptera | 10.0 | 12.5 | ||
| Isoptera | 7.1 | 1.8 | ||
| Araneida | 28.6 | 7.1 | 36.7 | 23.2 |
| Diplopoda | 19.6 | 21.4 | 13.3 | |
| Chilopoda | 1.8 | 1.8 | 3.3 | |
| Isopoda Scorpiones | 41.1 | 1.8 | ||
| Total | 1266.0 ± 387.9 | 562.5 ± 178.4 | 3830.0 ±1134.7 | 432.1 ± 140.5 |
4. Discussion
5. Conclusions
Acknowledgments
References
- Oliveira, P.S.; Marquis, R.J. (Eds.) The Cerrados of Brazil: ecology and natural history of a neotropical savanna; Columbia University Press: New York, NY, USA, 2002.
- Brossard, M.; López-Hernández, D. Des indicateurs d´évolution du milieu et des sols pour rendre durable l´usage des savanes d´Amerique du Sud. Natures Sciences et Sociétés 2005, 13, 266–278. [Google Scholar] [CrossRef]
- López-Hernández, D.; Hernández-Hernández, R.M.; Brossard, M. Historia del uso reciente de tierras de las sabanas de América del Sur. Interciencia 2005, 30, 623–630. [Google Scholar]
- Rondón, M.A.; Acevedo, D.; Hernández-Hernández, R.M.; Pubiano, Y.; Rivera, M.; Amézquita, E.; Romero, M.; Sarmiento, L.; Ayarza, M.; Barrios, E.; Rao, I. Carbon sequestration potential of the Neotropical savanna of Colombia and Venezuela. In Carbon sequestration in soils of Latin America; Lal, R., Cerri, C., Bernoux, M., Etchevers, J., Pellegrino, C., Eds.; Haworth Press Inc.: New York, NY, USA, 2006; pp. 213–243. [Google Scholar]
- Liebig, M.A.; Doran, J.W. Impact of organic production practices on soil quality indicators. J. Environ. Qual. 1999, 28, 1601–1609. [Google Scholar] [CrossRef]
- García, P. Los Suelos del Estado Amazonas: sus Potencialidades Agrícolas. Venesuelos 1994, 2, 59–66. [Google Scholar]
- López-Contreras, A.Y.; Hernández-Valencia, I.; López-Hernández, D. Fractionation of soil phosphorus in organic amended farms located on sandy soils of Venezuelan Amazonian. Biol. Fert. Soils 2007, 43, 771–777. [Google Scholar] [CrossRef]
- López-Hernández, D.; García-Guadilla, M.P.; Torres, F.; Chacón, P.; Paoletti, M.G. Identification, characterization and preliminary evaluation of Venezuelan Amazonian production systems in Puerto Ayacucho savanna-forest ecotone. Interciencia 1997, 22, 307–314. [Google Scholar]
- Netuzhilin, Y.; Chacón, P.; Cerda, H.; López-Hernández, D.; Torres, F.; Paoletti, M. Biodiversity tools to evaluate sustainability in savanna-forest ecotone in the Amazon (Venezuela). In Management of tropical agroecosystems and benefical soil biota; Reddy, M.V., Ed.; Science Publishers Inc.: Enfield, NH, USA, 1999; pp. 291–352. [Google Scholar]
- Hafez, A.A.R. Comparative changes in soil-physical properties induced by admixtures of manures from various domestic animals. Soil Sci. 1974, 118, 53–59. [Google Scholar] [CrossRef]
- Schtønning, P.; Christensen, B.T.; Carstensen, B. Physical and chemical properties of a sandy loam receiving animal manure, mineral fertilizer or no fertilizer for 90 years. Eur. J Soil Sci. 1994, 45, 257–268. [Google Scholar] [CrossRef]
- López-Hernández, D.; Araujo, Y.; López, A.Y.; Hernández-Valencia, I.; Hernández, C. Changes in the soil properties and in earthworm populations as induced by organic long-term fertilization in the Venezuelan Amazonia. Soil Sci. 2004, 169, 188–194. [Google Scholar] [CrossRef]
- Bulluck, L.R.; Brosius, M.; Evanylo, G.K.; Ristaino, J.B. Organic and synthetic fertility amendments influence soil microbial, physical and chemical properties on organic and conventional farms. App. Soil Ecol. 2002, 19, 147–160. [Google Scholar] [CrossRef]
- Cookson, W.R.; Marschner, P.; Clark, I.M.; Milton, N.; Smirk, M.N.; Murphy, D.V.; Osman, M.; Stockdale, E.A.; Hirsch, P.R. The influence of season, agricultural management, and soil properties on gross nitrogen transformations and bacterial community structure. Austr. J. Soil Research 2006, 44, 453–465. [Google Scholar]
- Hati, K.M.; Swarup, A.; Singh, D.; Misra, A.K.; Ghosh, P.K. Long-term continuous cropping, fertilization, and manuring effects on physical properties and organic carbon content of a sandy loam soil. Austr. J. Soil Research 2006, 44, 487–495. [Google Scholar] [CrossRef]
- Powlson, D.S.; Jenkinson, D.S. A comparison of the organic matter, biomass adenosine triphosphate and mineralizable nitrogen contents of ploughed and direct-drilled soils. J. Agric. Sci. 1981, 97, 713–721. [Google Scholar] [CrossRef]
- Ŝimek, M.; Hopkins, D.W.; Kalčïk, J.; Picek, T.; Šantrůčková, H.; Staňa, J.; Trávník, K. Biological and chemical properties of arable soils affected by long-term organic and inorganic fertilizer applications. Biol. Fert. Soils 1999, 29, 300–308. [Google Scholar] [CrossRef]
- Nannipieri, P.; Kandeler, E.; Ruggiero, P. Enzyme activities and microbiological and biochemical processes in soil. In Enzymes in the Environment; Burns, R.G., Dick, W.A., Eds.; Marcel Dekker: New York, USA, 2002; pp. 1–33. [Google Scholar]
- Larking, R.P.; Honeycutt, C.W.; Griffin, T.S. Effect of swine and dairy manure amendments on microbial communities in three soils as influenced by environmental conditions. Biol. Fert. Soils 2006, 43, 51–61. [Google Scholar] [CrossRef]
- Lalande, R.; Gagnon, B.; Simard, R.R.; Côté, D. Soil microbial biomass and enzyme activity following liquid hog manure application in a long-term field trial. Can. J. Soil. 2000, 80, 263–269. [Google Scholar] [CrossRef]
- Lockeretz, W.; Shearer, G.; Kohl, D.H. Organic farming in the Corn Belt. Science (Washington, DC) 1981, 211, 540–547. [Google Scholar] [CrossRef]
- Conant, R.; Paustian, K.; Elliot, E. Grassland management and conversion into grassland: effects on soil carbon. Ecol. Appl. 2001, 11, 343–355. [Google Scholar] [CrossRef]
- Hernández-Hernández, R.M.; López-Hernández, D. Microbial biomass, mineral nitrogen and carbon content in savanna soil aggregates under conventional and no-tillage. Soil Biol. Biochem. 2002, 34, 199–207. [Google Scholar]
- Doran, J.W.; Parkin, T.B. Defining and assessing soil quality. In Defining soil quality for a sustainable environment; Dorian, J.W., Jones, A.J., Eds.; Soil Sci. Soc. Am.; Special Publication 35. ASA and SSSA: Madison, WI, USA, 1994; pp. 3–21. [Google Scholar]
- Gil-Sotres, F.; Trasar-Cepeda, C.; Leirós, M.C.; Seoane, S. Different approaches to evaluating soil quality using biochemical properties. Soil Biol. Biochem. 2005, 37, 877–887. [Google Scholar] [CrossRef]
- Dormaar, J.F.; Chang, C. Effect of 20 annual applications of excess feedlot manure on labile soil phosphorus. Can. J. Soil Sci. 1995, 75, 507–512. [Google Scholar] [CrossRef]
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Anderson, J.M.; Ingram, J.S.I. Tropical soil biology and fertility: a handbook of methods, 2nd Ed. ed; CAB International: Wallingford, UK, 1993; pp. 16–18. [Google Scholar]
- Stanford, G.; Smith, S.J. Nitrogen mineralization potentials of soils. Soil Sci. Soc. Amer. Proc. 1972, 36, 465–472. [Google Scholar] [CrossRef]
- Olsen, S.R.; Sommers, L.E. Phosphorus Methods of Soil Analysis Part. 2: Chemical and microbiological properties. Page, A.L., Miller, R.H., Keeney, D.R., Eds.; In Soil Sci. Amer. J.; Agronomy Series 9; 1986; Madison, Wiscosin, USA; pp. 403–430. [Google Scholar]
- Murphy, J.; Riley, J. A modified single solution method for the determination of phosphate in natural waters. Anal. Chem. Acta. 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Casanova, E.F. Introducción a la Ciencia del Suelo; Editorial Litopar: Caracas, Venezuela, 1991; p. 393. [Google Scholar]
- Brookes, P.C.; Landman, A.; Pruden, G.; Jenkinson, P.D. Chloroform fumigation and the release of soil nitrogen: A rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol. Biochem. 1985, 17, 837–842. [Google Scholar] [CrossRef]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Hedley, M.J.; Stewart, J.W.; Chauhan, B.S. Changes in inorganic and organic soil phosphorus fractions induced by cultivation practices and by laboratory incubations. Soil Sci. Soc. Am. J. 1982, 46, 970–976. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Use of p- nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol. Biochem. 1969, 1, 301–307. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Assay of urease activity in soils. Soil Biol. Biochem. 1972, 4, 479–487. [Google Scholar] [CrossRef]
- Araujo, Y.; López-Hernández, D. Earthworm populations in a savanna agroforestry system of Venezuelan Amazonia. Biol. Fert. Soils. 1999, 29, 413–418. [Google Scholar] [CrossRef]
- Thiarks, A.E.; Mazurak, A.P.; Chesmin, L. Physical and chemical properties of soil associated with heavy applications of manure from cattle feed lots. Soil Sci. Soc. Am. Proc. 1974, 38, 826–830. [Google Scholar] [CrossRef]
- San Jose, J.J.; Montes, R.; Rocha, C. Neotropical savanna converted to food cropping and cattle feeding system: soil carbon and nitrogen changes over 30 years. Forest Ecol. Manage. 2003, 184, 17–32. [Google Scholar] [CrossRef]
- López-Hernández, D.; Burnham, C.P. The covariance of phosphate sorption with other soil properties in some British and Tropical soils. J. Soil Sci. 1974, 25, 196–206. [Google Scholar] [CrossRef]
- Nardoto, G.B.; Bustamante, M.M.C. Effects of fire on soil nitrogen dynamics and microbial biomass of Central Brazil. Pesquisa Agr. Brasil. 2003, 38, 955–962. [Google Scholar]
- Pascual, J.A.; García, C.; Hernández, T. Lasting microbiological and biochemical effects of the addition of municipal solid waste to an arid soil. Biol. Fert. Soils 1999, 30, 1–6. [Google Scholar] [CrossRef]
- Schloter, M.; Dilly, O.; Munch, J.C. Indicators for evaluating soil quality. Agric. Ecosyst. Environ. 2003, 98, 255–262. [Google Scholar] [CrossRef]
- Potter, R.L.; Jordan, C.F.; Guedes, R.M.; Batmanian, J.G.; Han, V.G. Assessment of phosphorus fractionation method for soils: problems for further investigation. Agric. Ecosyst. Environ. 1991, 34, 453–463. [Google Scholar] [CrossRef]
- Paoletti, M.G. Bioindicators of sustainability and biodiversity in agroecosystems. In Socio-Economic and policy issues for sustainable farming systems; Paoletti, M.G., Napiet, T., Ferro, O., Stinner, B., Stinner, D., Eds.; Cooperativa Amicizia: Padova, Italy, 1993; pp. 21–36. [Google Scholar]
- López-Hernández, D.; Ojeda, A.D. Alternativas en el manejo agroecológico de los suelos de las sabanas del norte de Suramérica. Ecotropicos 1996, 9, 99–115. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
López-Hernández, D.; Hernández, C.L.; Netuzhilin, I.; López-Contreras, A.Y. Agricultural Systems Located in the Forest-Savanna Ecotone of the Venezuelan Amazonian. Are Organic Agroforestry Farms Sustainable? Sustainability 2009, 1, 215-233. https://doi.org/10.3390/su1020215
López-Hernández D, Hernández CL, Netuzhilin I, López-Contreras AY. Agricultural Systems Located in the Forest-Savanna Ecotone of the Venezuelan Amazonian. Are Organic Agroforestry Farms Sustainable? Sustainability. 2009; 1(2):215-233. https://doi.org/10.3390/su1020215
Chicago/Turabian StyleLópez-Hernández, Danilo, Carmen Leonor Hernández, Igor Netuzhilin, and Ana Yamila López-Contreras. 2009. "Agricultural Systems Located in the Forest-Savanna Ecotone of the Venezuelan Amazonian. Are Organic Agroforestry Farms Sustainable?" Sustainability 1, no. 2: 215-233. https://doi.org/10.3390/su1020215
APA StyleLópez-Hernández, D., Hernández, C. L., Netuzhilin, I., & López-Contreras, A. Y. (2009). Agricultural Systems Located in the Forest-Savanna Ecotone of the Venezuelan Amazonian. Are Organic Agroforestry Farms Sustainable? Sustainability, 1(2), 215-233. https://doi.org/10.3390/su1020215




