1. Introduction
In the area of fine chemicals synthesis, research and development aiming at efficient catalytic procedures is one of the main topics in academia and industry [
1]. Heterogeneously catalyzed hydrogenation reactions fulfill the criteria of
Green Chemistry in terms of atom economy and waste formation [
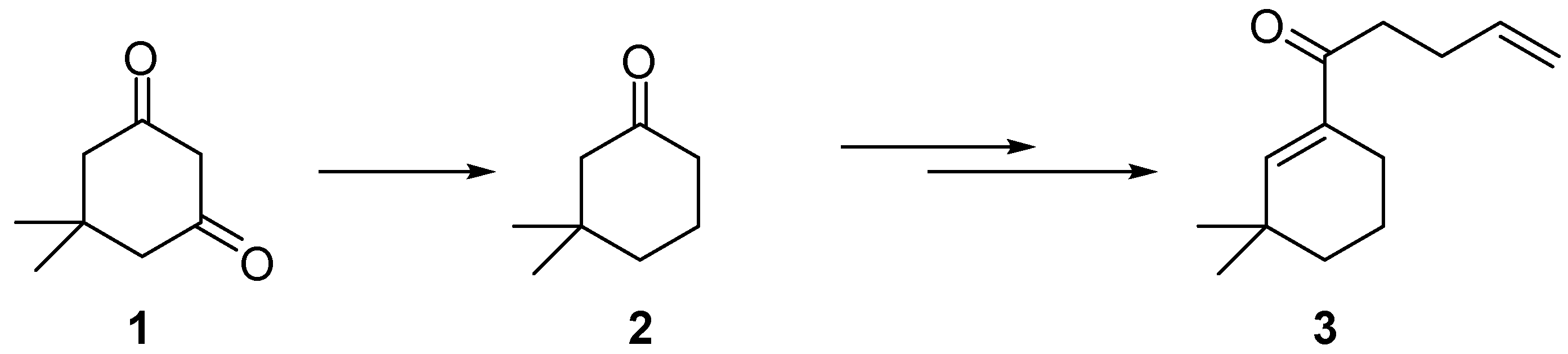
2]. An important hydrogenation reaction in the synthesis of intermediates for the flavors and fragrance industry is the selective hydrogenation of dimedone (
1) to 3,3-dimethylcyclohexanone (
2). The ketone
2 can be used for the synthesis of 1-(3,3-dimethyl-1-cyclohexen-1-yl)-4-penten-1-one (
3), which has a woody, green odor and is widely used as perfuming and/or odor-modifying agent as well as flavoring and/or taste-modifying ingredient (
Scheme 1) [
3,
4].
Scheme 1.
Selective reduction of dimedone (1) for the synthesis of 1-(3,3-dimethyl-1-cyclohexen-1-yl)-4-penten-1-one (3).
Scheme 1.
Selective reduction of dimedone (1) for the synthesis of 1-(3,3-dimethyl-1-cyclohexen-1-yl)-4-penten-1-one (3).
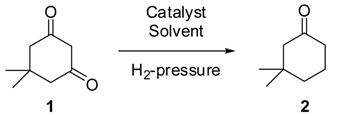
For the hydrogenation of dimedone (
1) to 3,3-dimethylcyclohexanone (
2) several two-step protocols are described in literature, for example the transformation of dimedone to an enol triflate followed by hydrogenation over PtO
2 [
5]. The preparation of the triflate is very expensive, requires corrosive conditions and is not atom economic at all. The reduction of
1 over nickel alloy to the saturated alcohol, followed by subsequent oxidation to
2 with dichromate in sulphuric acid as described by Champagne
et al. is also not applicable at industrial scale due to the highly toxic and corrosive conditions [
6].
The only one-step reduction of dimedone (
1) to
2 was reported by Cormier. He obtained 3,3-dimethylcyclohexanone (
2) in 69-73% yield (98-99% purity) by Pd-catalyzed medium-pressure hydrogenation of dimedone (
1) in a solvent mixture of concentrated H
2SO
4 and propionic acid [
7].
For a potential industrial application it was necessary to find suitable reaction conditions for the reduction of dimedone (1) to 3,3-dimethylcyclohexanone (2) with high selectivity in one step with no or low waste formation and in the absence of any strongly oxidative (or corrosive) reagents and halides. In the present communication we describe the highly selective and efficient synthesis of 2 by hydrogenation of 1 using heterogeneous catalysis over palladium supported on the ion exchange resin Amberlyst 15® in order to circumvent the described drawbacks.
2. Results and Discussion
In an extensive screening of the hydrogenation of
1 to
2 we compared palladium as catalyst on various supports in various solvents. Selected results are summarized in
Table 1.
Table 1.
Selected screening results of the hydrogenation of 1 to 2.
Table 1.
Selected screening results of the hydrogenation of 1 to 2.
| Entry | Solvent | Catalyst | p [bar] | Yield of 2
[GC wt%] | Σ∗
By-products |
|---|
| 1 | Propionic acid | 5% Pd/C | 10 | 89 | 7.4 |
| 2 | Acetic acid+ 5% H2SO4 | 5% Pd/C | 2 | 87 | 5.2 |
| 3 | Isopropanol | 5% Pd/C | 2 | 89 | 11 |
| 4 | Isopropanol +5% H2SO4 | 5% Pd/C | 2 | 88 | 6 |
| 5 | Methanol | 5% Pd/C | 2 | 83 | 15 |
| 6 | Isopropanol + 5% H2SO4 | 5%Pd/Al2O3 | 2 | 70 | 12 |
| 7 | Isopropanol | 5% Pd/Al2O3 | 2 | 14 | 45 |
| 8 | Isopropanol + 5% H2SO4 | 5% Pd/SiO2 | 2 | 84 | 8 |
| 9 | Isopropanol | 5% Pd/SiO2 | 2 | 7 | 58 |
| 10 | Isopropanol | 0.2-1% Pd/Amberlyst 15® | 2 | 96 | 1.8 |
| 11 | Methanol | 0.2-1% Pd/Amberlyst 15® | 2 | 97 | 1.3 |
| 12** | Methanol | 0.2-1% Pd/Amberlyst 15® | 2 | 97 | 1.2 |
In the beginning, we used the conditions described by Cormier [
7]: Propionic acid and acetic acid as solvents applied in the hydrogenation over palladium on charcoal led to an acceptable yield of
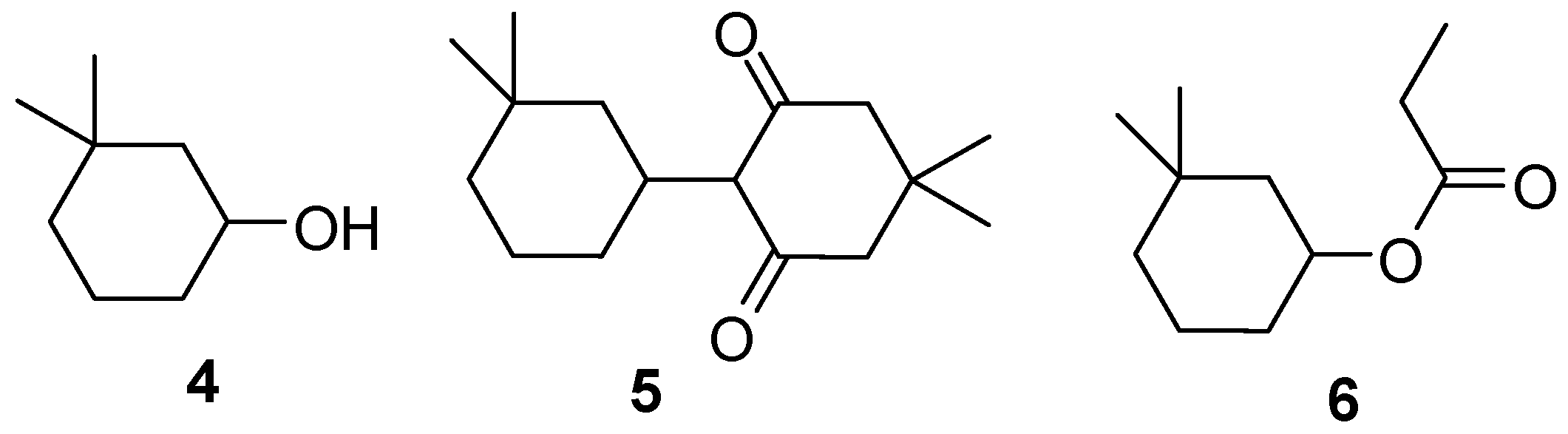
2 of almost 90% (entries 1 and 2). However, several by-products were identified (
Figure 1): the over-reduced alcohol
4, various aldol condensation products, e.g.
5, and the propyl ester
6.
Figure 1.
Frequently encountered by-products in the reduction of 1 to 2 in propionic acid.
Figure 1.
Frequently encountered by-products in the reduction of 1 to 2 in propionic acid.
The saturated alcohol 4 was the major by-product. We found out that the purification of 2 by distillation is difficult in the presence of alcohol 4, as both compounds have very similar boiling points [bp (2) 180°C; bp (4) 185°C at 1 bar]. Moreover, carrying out the experiments in the carboxylic acid solution in a steel autoclave led to visible corrosion of the reactor walls.
The hydrogenation over Pd/C in isopropanol without addition of acid gave a similar yield of 89%, but again also a substantial amount of the saturated alcohol 4 (entry 3). When acid was added to the reaction over Pd/C in an alcoholic solvent, the amount of 4 as by-product decreased from 11 to 6%, but the conversion of the starting material was not complete (entry 4).
When palladium on supports like silica or alumina was used, the hydrogenation of 1 in isopropanol with H2SO4 led to lower yields, compared to the experiments with Pd/C (entries 6 and 8 vs. entry 4). In the absence of acid the reactions over Pd/SiO2 and Pd/Al2O3 led to even lower yields (entries 7 and 9). The experiments show clearly that the presence of acid is beneficial for the hydrogenation of 1 to 2 in terms of selectivity and yield of 2. However, as already discussed, the use of strongly acidic conditions rules out the application of the reaction in standard steel equipment, requires glass lined reactors and is expensive on an industrial scale.
We therefore turned our attention to the use of a solid acid, e.g. ion exchange resins like Amberlyst 15®. The catalytic system of palladium and Amberlyst 15® provides a perfectly suitable combination for heterogeneous hydrogenation under acidic conditions. Amberlyst 15® is a strongly acidic macroreticular cation exchange resin used as a heterogeneous acid catalyst and suitable for non-aqueous catalysis. The hydrogenation of 1 in isopropanol over Amberlyst 15® and palladium furnished a very high yield of 2 and less than 2% of by-products (entry 10). The replacement of isopropanol by methanol delivered even higher yield and only 1.3% of by-products (entry 11). We applied this protocol to a reaction at larger scale (scale-up factor 100) and were pleased to find out, that the reaction is easily scalable (entry 12).
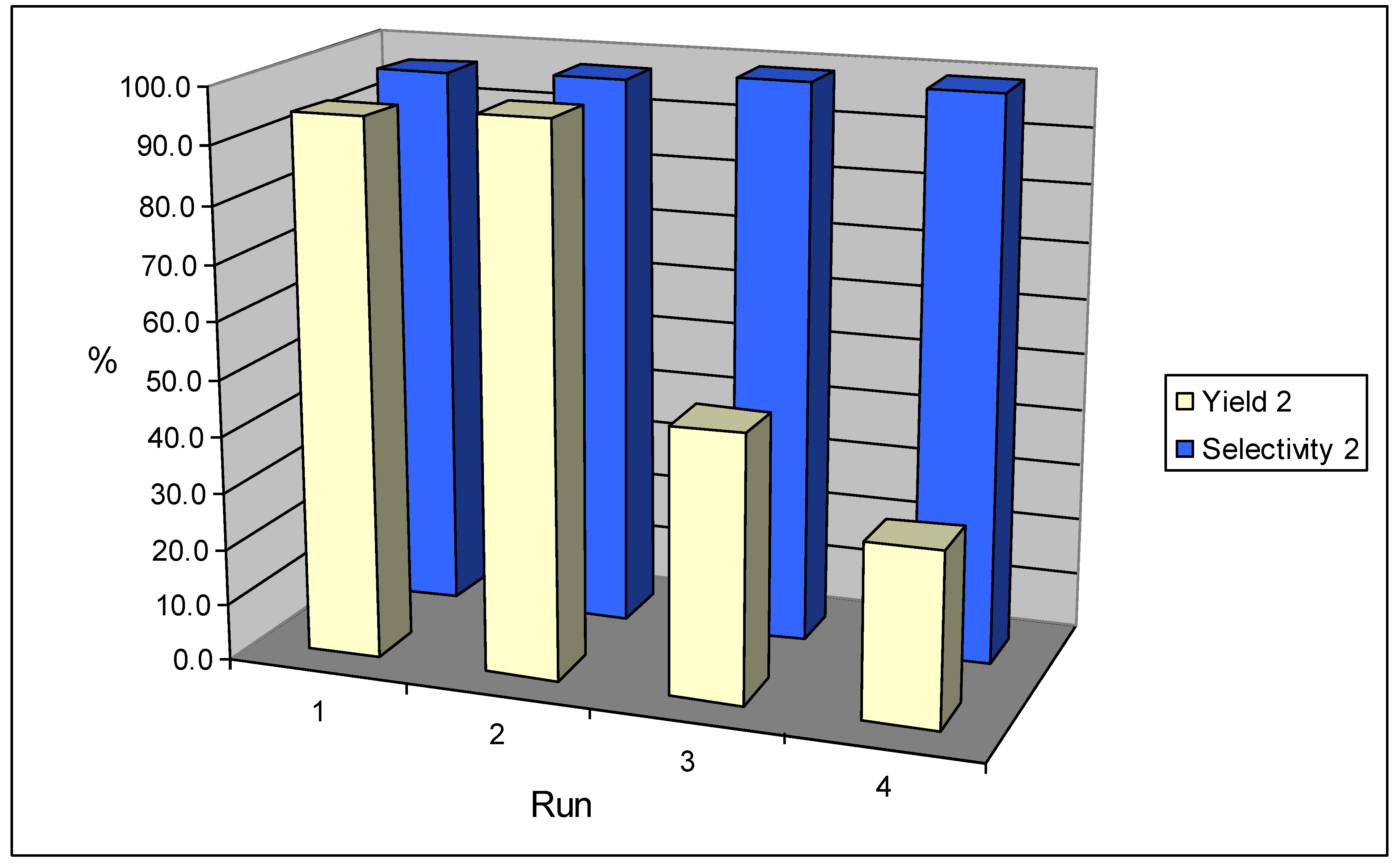
We then tested if the catalyst-system can be recycled. After an experiment, the Amberlyst 15
® with the palladium was filtered off, rinsed once with methanol and used for the next run. The results are illustrated in
Figure 2.
Figure 2.
Yield and selectivity of 2 during recycling experiments.
Figure 2.
Yield and selectivity of 2 during recycling experiments.
During the first two runs, the yield of 2 remained high. The third and the fourth run delivered a decreased yield, but the selectivity remained exceptionally high. We suspected that the activity of the catalyst might have dropped due to leaching of palladium from the support into the solution. Therefore, samples of the final reaction mixture were analysed after filtration of the catalyst. Elementary analysis showed that 0.01% of palladium had leached into the solution. We do not think that the decrease in activity is due to this marginal amount of leaching. The reasons for the decrease in activity could therefore be clogging of catalyst pores, or loss of acidity. These topics are currently under investigation.
Compared to the literature procedures (
Table 1, entries 1, 2) and to the hydrogenation in solvents containing catalytic amounts of acid, the hydrogenation of dimedone (
1) over palladium on Amberlyst 15
® in methanol leads to high yields and high selectivities of
2. The reaction was easily scaled up. The catalyst was recycled up to four times. The third and the fourth run led to decreased conversion and yield, but the selectivity always remained higher than 95%. Further investigations are ongoing to clarify the reaction mechanism.