Optimizing Multivariable Logistic Regression for Identifying Perioperative Risk Factors for Deep Brain Stimulator Explantation: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Data
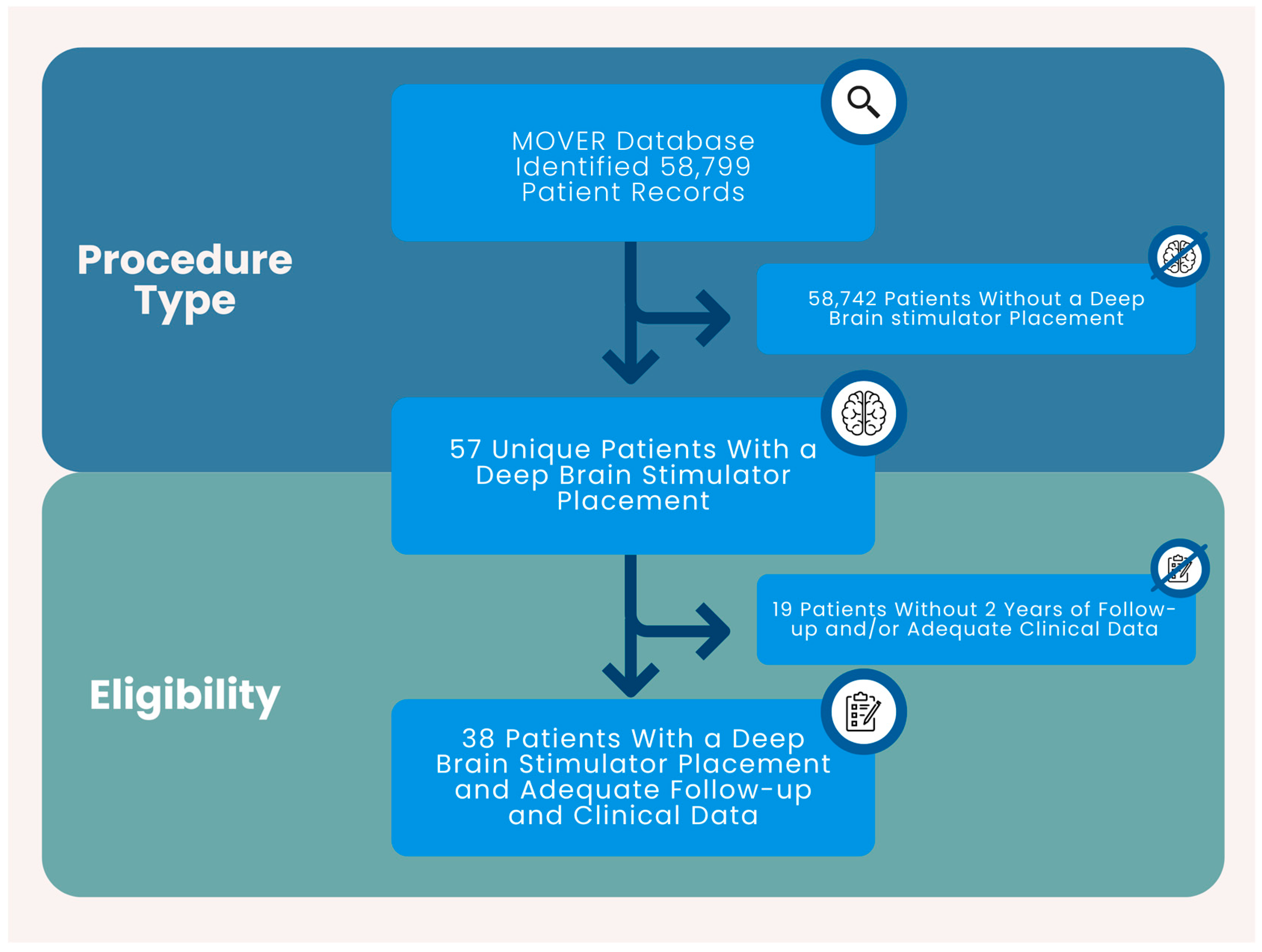
2.2. Participants
2.3. Data Preparation
2.4. Predictors
2.5. Sample Size
2.6. Missing Data
2.7. Analytical Methods
2.8. Class Imbalance
2.9. Fairness
2.10. Model Output
3. Results
3.1. Cohort Demographics
3.2. Multivariate Logistic Regression Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PD | Parkinson’s Disease |
| DBS | Deep brain stimulation |
| AUC | Area under the curve |
| RFECV | Recursive factor elimination with cross-validation |
| MOVER | Medical Informatics Operating Room Vitals and Events Repository |
| OR | Odds ratio |
| CI | 95% confidence interval |
| ASA | American Society of Anesthesiologists |
| BMI | Body mass index |
| ICD | International Classification of Diseases |
| SMOTE | Synthetic Minority Oversampling Technique |
| SD | Standard deviation |
| IQR | Interquartile range |
| ICU | Intensive care unit |
| LOS | Length of stay |
References
- Benabid, A.L.; Pollak, P.; Louveau, A.; Henry, S.; de Rougemont, J. Combined (thalamotomy and stimulation) stereotactic surgery of the VIM thalamic nucleus for bilateral Parkinson disease. Appl. Neurophysiol. 1987, 50, 344–346. [Google Scholar] [CrossRef] [PubMed]
- Servello, D.; Galbiati, T.F.; Iess, G.; Minafra, B.; Porta, M.; Pacchetti, C. Complications of deep brain stimulation in Parkinson’s disease: A single-center experience of 517 consecutive cases. Acta Neurochir. 2023, 165, 3385–3396. [Google Scholar] [CrossRef] [PubMed]
- Hacker, M.L.; Turchan, M.; Heusinkveld, L.E.; Currie, A.D.; Millan, S.H.; Molinari, A.L.; Konrad, P.E.; Davis, T.L.; Phibbs, F.T.; Hedera, P.; et al. Deep brain stimulation in early-stage Parkinson disease: Five-year outcomes. Neurology 2020, 95, e393–e401. [Google Scholar] [CrossRef] [PubMed]
- Figee, M.; Riva-Posse, P.; Choi, K.S.; Bederson, L.; Mayberg, H.S.; Kopell, B.H. Deep Brain Stimulation for Depression. Neurotherapeutics 2022, 19, 1229–1245. [Google Scholar] [CrossRef] [PubMed]
- Knotkova, H.; Hamani, C.; Sivanesan, E.; Le Beuffe, M.F.E.; Moon, J.Y.; Cohen, S.P.; Huntoon, M.A. Neuromodulation for chronic pain. Lancet 2021, 397, 2111–2124. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Fan, S.; Gan, Y.; Zhang, Y.; Gao, Y.; Xue, T.; Xie, H.; Ma, R.; Zhang, Q.; Zhao, B.; et al. Efficacy and safety of combined deep brain stimulation with capsulotomy for comorbid motor and psychiatric symptoms in Tourette’s syndrome: Experience and evidence. Asian J. Psychiatr. 2024, 94, 103960. [Google Scholar] [CrossRef] [PubMed]
- Krauss, J.K.; Lipsman, N.; Aziz, T.; Boutet, A.; Brown, P.; Chang, J.W.; Davidson, B.; Grill, W.M.; Hariz, M.I.; Horn, A.; et al. Technology of deep brain stimulation: Current status and future directions. Nat. Rev. Neurol. 2021, 17, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Yue, J.K.; Wang, D.D. Trends in safety and cost of deep brain stimulation for treatment of movement disorders in the United States: 2002–2014. Br. J. Neurosurg. 2021, 35, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Reich, M.M.; Hsu, J.; Ferguson, M.; Schaper, F.; Joutsa, J.; Roothans, J.; Nickl, R.C.; Frankemolle-Gilbert, A.; Alberts, J.; Volkmann, J.; et al. A brain network for deep brain stimulation induced cognitive decline in Parkinson’s disease. Brain 2022, 145, 1410–1421. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.; Ahmed, M.; Markosian, C.; Ezike, J.Z.; Agrawal, R.; Randhawa, K.; Liang, Z.; Abraham, M.; Paskhover, B.; Mammis, A. Complications associated with deep brain stimulation for Parkinson’s disease: A MAUDE study. Br. J. Neurosurg. 2021, 35, 625–628. [Google Scholar] [CrossRef] [PubMed]
- Olson, M.C.; Shill, H.; Ponce, F.; Aslam, S. Deep brain stimulation in PD: Risk of complications, morbidity, and hospitalizations: A systematic review. Front. Aging Neurosci. 2023, 15, 1258190. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Mirzadeh, Z.; Lambert, M.; Gonzalez, O.; Moran, A.; Shetter, A.G.; Ponce, F.A. Cost of Deep Brain Stimulation Infection Resulting in Explantation. Stereotact. Funct. Neurosurg. 2017, 95, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Jung, I.H.; Chang, K.W.; Park, S.H.; Chang, W.S.; Jung, H.H.; Chang, J.W. Complications After Deep Brain Stimulation: A 21-Year Experience in 426 Patients. Front. Aging Neurosci. 2022, 14, 819730. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Yang, H.J.; Kwon, J.H.; Kim, M.H.; Lee, J.; Jeon, B. Mortality of Deep Brain Stimulation and Risk Factors in Patients With Parkinson’s Disease: A National Cohort Study in Korea. J. Korean Med. Sci. 2023, 38, e10. [Google Scholar] [CrossRef] [PubMed]
- Samad, M.; Angel, M.; Rinehart, J.; Kanomata, Y.; Baldi, P.; Cannesson, M. Medical Informatics Operating Room Vitals and Events Repository (MOVER): A public-access operating room database. JAMIA Open 2023, 6, ooad084. [Google Scholar] [CrossRef] [PubMed]
- McKinney, W. Data Structures for Statistical Computing in Python. In Proceedings of the 9th Python in Science Conference, Austin, TX, USA, 28 June–3 July 2010; pp. 51–56. [Google Scholar]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- Hunter, J.D. Matplotlib: A 2D graphics environment. Comput. Sci. Eng. 2007, 9, 90–95. [Google Scholar] [CrossRef]
- Waskom, M.L. seaborn: Statistical data visualization. J. Open Source Softw. 2021, 6, 3021. [Google Scholar] [CrossRef]
- Abode-Iyamah, K.O.; Chiang, H.Y.; Woodroffe, R.W.; Park, B.; Jareczek, F.J.; Nagahama, Y.; Winslow, N.; Herwaldt, L.A.; Greenlee, J.D.W. Deep brain stimulation hardware-related infections: 10-year experience at a single institution. J. Neurosurg. 2019, 130, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Deeb, W.; Leentjens, A.F.G.; Mogilner, A.Y.; Servello, D.; Meng, F.; Zhang, J.; Galbiati, T.F.; Okun, M.S. Deep brain stimulation lead removal in Tourette syndrome. Park. Relat. Disord. 2020, 77, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Scelzo, E.; Beghi, E.; Rosa, M.; Angrisano, S.; Antonini, A.; Bagella, C.; Bianchi, E.; Caputo, E.; Lena, F.; Lopiano, L.; et al. Deep brain stimulation in Parkinson’s disease: A multicentric, long-term, observational pilot study. J. Neurol. Sci. 2019, 405, 116411. [Google Scholar] [CrossRef] [PubMed]
- Farrokhi, F.R.; Marsans, M.T.; Sikora, M.; Monsell, S.E.; Wright, A.K.; Palmer, M.; Hoefer, A.; McLeod, P.; Mark, J.; Carlson, J. Pre-operative smoking history increases risk of infection in deep brain stimulation surgery. J. Clin. Neurosci. 2019, 69, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, J.E.; Kashyap, S.; Ray, K.; Ananda, A. Infections in Deep Brain Stimulator Surgery. Cureus 2019, 11, e5440. [Google Scholar] [CrossRef] [PubMed]
- Bjerknes, S.; Skogseid, I.M.; Sæhle, T.; Dietrichs, E.; Toft, M. Surgical site infections after deep brain stimulation surgery: Frequency, characteristics and management in a 10-year period. PLoS ONE 2014, 9, e105288. [Google Scholar] [CrossRef] [PubMed]
- Tiefenbach, J.; Kuvliev, E., Jr.; Dullur, P.; Mandava, N.; Hogue, O.; Kondylis, E.; Sharma, A.; Rammo, R.; Nagel, S.; Machado, A.G. The Rate and Risk Factors of Deep Brain Stimulation-Associated Complications: A Single-Center Experience. Oper. Neurosurg. 2024, 28, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Murin, P.J.; Murin, P.J.; Lima de Mendonca, Y.; Martins, Y.C. Identification of Perioperative Risk Factors for Early Sacral Nerve Stimulator Explantation: A Single-Center Retrospective Cohort Study. J. Clin. Med. 2025, 14, 2363. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, N.; Shaheen, A.; Elgendy, A.; Bezchlibnyk, Y.B.; Zesiewicz, T.; Dalm, B.; Jain, J.; Green, A.L.; Aziz, T.Z.; Flouty, O. Deep brain stimulation for chronic pain: A systematic review and meta-analysis. Front. Hum. Neurosci. 2023, 17, 1297894. [Google Scholar] [CrossRef] [PubMed]
- Hussain, N.; Weaver, T. Response to the Letter to the Editor Regarding: “Identifying Predictors for Early Percutaneous Spinal Cord Stimulator Explant at One and Two Years: A Retrospective Database Analysis”. Neuromodulation 2023, 26, 710. [Google Scholar] [CrossRef] [PubMed]


| DBS | Explant | No Explant | p-Value | |
|---|---|---|---|---|
| Number of Patients | 38 | 5 (13.2%) | 33 (86.8%) | 0.1440 |
| Sex | 0.1440 | |||
| Male n (%) | 25 (65.8%) | 5 (100.0%) | 20 (60.6%) | 0.4416 |
| Female n (%) | 13 (34.2%) | 0 (0.0%) | 13 (39.4%) | |
| Age (years +/− SD) | 64.8 +/− 11.6 | 62.4 +/− 6.2 | 65.2 +/− 12.2 | 0.1316 |
| Anesthesia Type | 0.1316 | |||
| General Anesthesia n (%) | 37 (97.4%) | 4 (80.0%) | 33 (100.0%) | >0.9999 |
| Monitored Airway n (%) | 1 (2.6%) | 1 (20.0%) | 0 (0.0%) | 0.8107 |
| ASA Score (median, IQR) | 3.0 (3.0–3.0) | 3.0 (3.0–3.0) | 3.0 (3.0–3.0) | 0.2489 |
| Length of Stay (median, IQR) | 1.0 (1.0–1.3) | 1.0 (1.0–1.5) | 1.0 (1.0–1.5) | |
| ICU Admission n (%) | 36 (94.73%) | 4 (80.0%) | 32 (97.0%) | >0.9999 |
| Indication for DBS ** | >0.9999 | |||
| Primary PD n (%) | 30 (78.94%) | 4 (80.0%) | 26 (78.8%) | 0.1195 |
| Secondary PD n (%) | 1 (2.63%) | 0 (0.0%) | 1 (3.0%) | 0.3121 |
| Essential Tremor n (%) | 5 (13.15%) | 2 (40.0%) | 3 (9.1%) | >0.9999 |
| Dystonia n (%) | 9 (23.68%) | 0 (0.0%) | 9 (27.3%) | |
| Spasticity n (%) | 1(2.63%) | 0 (0.0%) | 1 (3.0%) | 0.2440 |
| Surgical Approach | 0.2440 | |||
| Percutaneous n (%) | 29 (76.31%) | 3 (60.0%) | 26 (83.9%) | |
| Open n (%) | 7 (18.42%) | 2 (40.0%) | 5 (16.1%) | 0.1316 |
| Medical Comorbidities | 0.4456 | |||
| Epilepsy (%) | 1 (2.63%) | 1 (20.0%) | 0 (0.0%) | >0.9999 |
| Neuropathy (%) | 4 (10.52%) | 1 (20.0%) | 3 (9.1%) | 0.0108 * |
| Acute Postoperative Pain (%) | 2 (5.26%) | 0 (0.0%) | 2 (6.1%) | 0.1690 |
| Chronic Pain (%) | 5 (13.15%) | 3 (60.0%) | 2 (6.1%) | |
| Dysautonomia (%) | 6 (15.78%) | 2 (40.0%) | 4 (12.1%) | >0.9999 |
| Chronic Fatigue (%) | 0.0% | 0.0% | 0.0% | 0.1316 |
| Cognitive Impairment (%) | 5 (13.15%) | 0 (0.0%) | 5 (15.2%) | >0.9999 |
| Restless Leg Syndrome (%) | 1 (2.63%) | 1 (20.0%) | 0 (0.0%) | 0.1195 |
| Cerebrovascular Disease (%) | 1 (2.63%) | 0 (0.0%) | 1 (3.0%) | 0.5701 |
| Sleep Apnea (%) | 5 (13.15%) | 2 (40.0%) | 3 (9.1%) | 0.4456 |
| Sleep Disorder, Any (%) | 6 (15.78%) | 0 (0.0%) | 6 (18.2%) | >0.9999 |
| Chronic Obstructive Pulmonary Disease (%) | 4 (10.52%) | 1 (20.0%) | 3 (9.1%) | 0.2227 |
| Hypertension (%) | 17 (44.73%) | 2 (40.0%) | 15 (45.5%) | >0.9999 |
| Hyperlipidemia (%) | 7 (18.42%) | 2 (40.0%) | 5 (15.2%) | 0.1195 |
| Atrial Fibrillation (%) | 2 (5.26%) | 0 (0.0%) | 2 (6.1%) | 0.5272 |
| Diabetes Mellitus (%) | 5 (13.15%) | 2 (40.0%) | 3 (9.1%) | 0.2489 |
| Chronic Kidney Disease (%) | 5 (13.15%) | 1 (20.0%) | 4 (12.1%) | >0.9999 |
| Fibromyalgia (%) | 2 (5.26%) | 1 (20.0%) | 1 (3.0%) | >0.9999 |
| Irritable Bowel Syndrome (%) | 1 (2.63%) | 0 (0.0%) | 1 (3.0%) | 0.1690 |
| Underweight/Cachexia (%) | 4 (10.52%) | 0 (0.0%) | 4 (12.1%) | >0.9999 |
| Obesity (%) | 6 (15.78%) | 2 (40.0%) | 4 (12.1%) | >0.9999 |
| Migraine (%) | 1 (2.63%) | 0 (0.0%) | 1 (3.0%) | 0.5701 |
| Urinary Incontinence (%) | 3 (7.89%) | 0 (0.0%) | 3 (9.1%) | |
| Malignancy (%) | 6 (15.78%) | 0 (0.0%) | 6 (18.2%) | |
| Bowel Incontinence (%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0.2489 |
| Substance Use | >0.9999 | |||
| Opioid Use (%) | 2 (5.26%) | 1 (20.0%) | 1 (3.0%) | 0.0026 * |
| Substance Use (%) | 1 (2.63%) | 0 (0.0%) | 1 (3.0%) | |
| Tobacco Use (%) | 13 (34.21%) | 5 (100.0%) | 8 (24.2%) | >0.9999 |
| Psychiatric Comorbidities | >0.9999 | |||
| Anxiety (%) | 8 (21.05%) | 1 (20.0%) | 7 (21.2%) | |
| MDD (%) | 6 (15.78%) | 1 (20.0%) | 5 (15.2%) | |
| ADHD (%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | |
| ETOH (%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | |
| OCD (%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | |
| PTSD (%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murin, P.J.; Prabhune, A.S.; Martins, Y.C. Optimizing Multivariable Logistic Regression for Identifying Perioperative Risk Factors for Deep Brain Stimulator Explantation: A Pilot Study. Clin. Pract. 2025, 15, 132. https://doi.org/10.3390/clinpract15070132
Murin PJ, Prabhune AS, Martins YC. Optimizing Multivariable Logistic Regression for Identifying Perioperative Risk Factors for Deep Brain Stimulator Explantation: A Pilot Study. Clinics and Practice. 2025; 15(7):132. https://doi.org/10.3390/clinpract15070132
Chicago/Turabian StyleMurin, Peyton J., Anagha S. Prabhune, and Yuri Chaves Martins. 2025. "Optimizing Multivariable Logistic Regression for Identifying Perioperative Risk Factors for Deep Brain Stimulator Explantation: A Pilot Study" Clinics and Practice 15, no. 7: 132. https://doi.org/10.3390/clinpract15070132
APA StyleMurin, P. J., Prabhune, A. S., & Martins, Y. C. (2025). Optimizing Multivariable Logistic Regression for Identifying Perioperative Risk Factors for Deep Brain Stimulator Explantation: A Pilot Study. Clinics and Practice, 15(7), 132. https://doi.org/10.3390/clinpract15070132








