Drugs Associated with Adverse Effects in Vulnerable Groups of Patients
Abstract
1. Introduction
2. Materials and Methods
3. Results. Case Studies in Different Pharmacological Classes
3.1. Antimicrobial Chemotherapy
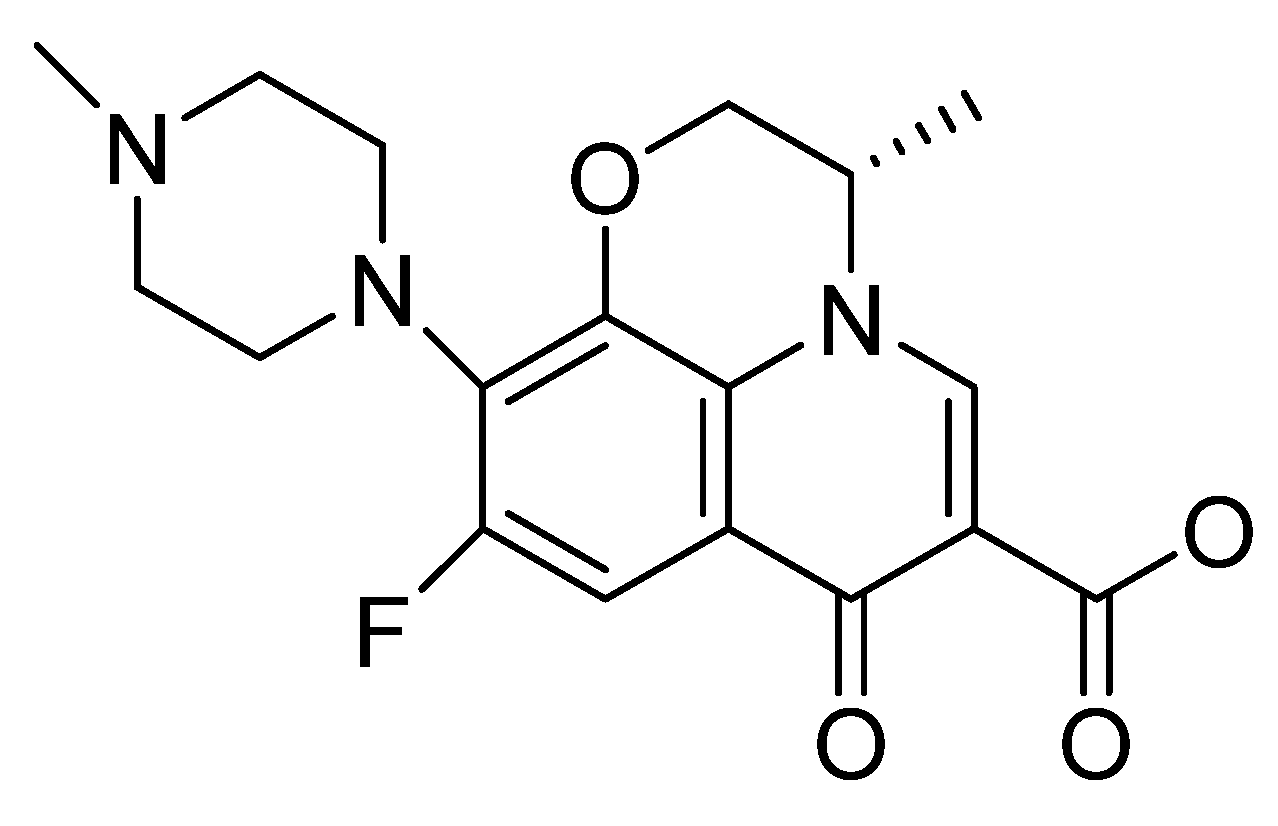
3.1.1. Fluoroquinolones
3.1.2. Aminoglycosides
3.2. Antiarrhythmics
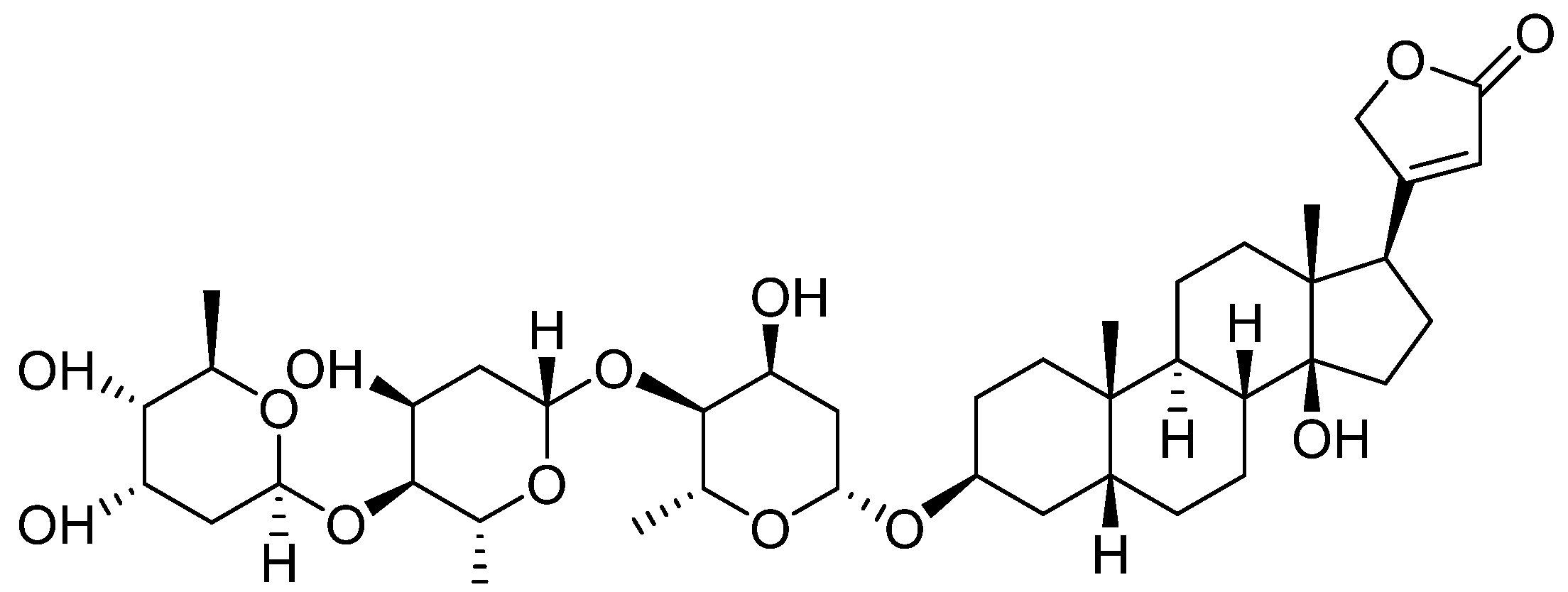
3.3. Cardiotonic Glycosides
3.4. Antihypertensives
3.4.1. Angiotensin-Converting Enzyme Inhibitors
3.4.2. Angiotensin II Receptor Blockers
3.5. Anti-Tumor Drugs
3.5.1. Bleomycin
3.5.2. Cisplatin
3.6. Drugs with Action on the Central Nervous System
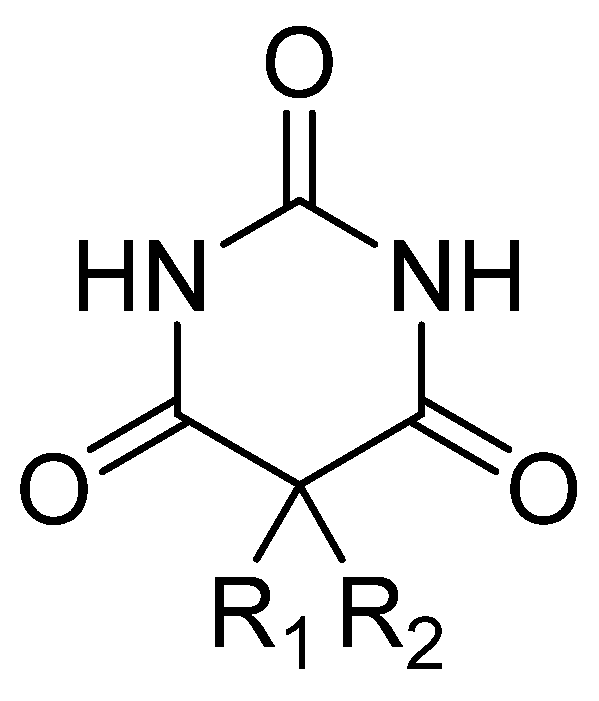
3.6.1. Hypnotic—Barbiturates
3.6.2. Antipsychotics
3.6.3. Antidepressants
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Qato, D.M.; Wilder, J.; Schumm, L.P.; Gillet, V.; Alexander, G.C. Changes in prescription and over-the-counter medication and dietary supplement use among older adults in the United States, 2005 vs. 2011. JAMA Intern. Med. 2016, 176, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Brookfield, K.F.; Su, F.; Elkomy, M.H.; Drover, D.R.; Lyell, D.J.; Carvalho, B. Pharmacokinetics and placental transfer of magnesium sulfate in pregnant women. Am. J. Obstet. Gynecol. 2016, 737, 737.e1–737.e9. [Google Scholar] [CrossRef]
- Pack, A.M.; Davis, A.R.; Kritzer, J.; Yoon, A.; Camus, A. Antiepileptic drugs: Are women aware of interactions with oral contraceptives and potential teratogenicity. Epilepsy Behav. 2009, 14, 640–644. [Google Scholar] [CrossRef] [PubMed]
- Duncombe, D.; Wertheim, E.H.; Skouteris, H.; Paxton, S.J.; Kelly, L. How well do women adapt to changes in their body size and shape across the course of pregnancy. J. Health Psychol. 2008, 13, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Halpern, D.G.; Weinberg, C.R.; Pinnelas, R.; Mehta-Lee, S.; Economy, K.E.; Valente, A.M. Use of Medication for Cardiovascular Disease During Pregnancy: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 457–476. [Google Scholar] [CrossRef] [PubMed]
- Aranda, J.V.; Beharry, J.V. Pharmacokinetics, pharmacodynamics and metabolism of caffeine in newborns. Semin. Fetal Neonatal Med. 2020, 25, 101183. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.A.; Thee, S.; Jacobs, K.; Ebrahim, A.; Hesseling, A.C.; Schaaf, H.S. Hearing loss in children treated for multidrugresistant tuberculosis. J. Infect. 2013, 66, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Nam, H.; Park, S.; Chae, J.-H.; Kim, T.-S. Decreased cardiovascular death in schizophrenia patients treated with antipsychotics: A Korean national cohort study. Schizophr. Res. 2021, 228, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.S.; Schneeweiss, S.; Avorn, J.; Fischer, M.A.; Mogun, H.; Solomon, D.H.; Brookhart, M.A. Risk of Death in Elderly Users of Conventional vs. Atypical Antipsychotic Medications. N. Engl. J. Med. 2005, 353, 2335–2341. [Google Scholar] [CrossRef]
- Berger, S.J.; Hofer, A. Sicherheitsaspekte bei der Behandlung mit Clozapin. Neuropsychiatrie 2023, 37, 122–129. [Google Scholar] [CrossRef]
- Zito, M.F.; Marder, S.R. Rethinking the risks and benefits of long-term maintenance in schizophrenia. Schizophr. Res. 2020, 225, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Peccatori, P.A.; Azim, H.A., Jr. Pregnancy in breast cancer survivors: A need for proper counseling. Breast 2009, 18, 337–338. [Google Scholar] [CrossRef] [PubMed]
- Yabe, K.; Goto, K.; Jindo, T.; Sekiguchi, M.; Furuhama, K. Structure–phototoxicity relationship in Balb/c mice treated with fluoroquinolone derivatives, followed by ultraviolet-A irradiation. Toxicol. Lett. 2005, 157, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.E.; Kennelley, G.E.; Amaye-Obu, T.; Jowdy, P.F.; Ghadersohi, S.; Nasir-Moin, M.; Paragh, G.; Berman, H.A.; Huss, W.J. The phenomenon of phototoxicity and long-term risks of commonly prescribed and structurally diverse drugs. J. Photochem. Photobiol. 2024, 19, 100221. [Google Scholar] [CrossRef] [PubMed]
- Eljaaly, K.; Alkhalaf, A.; Alhifany, A.A.; Alshibani, M. Photosensitivity induced by lomefloxacin versus other fluoroquinolones: A meta-analysis. J. Infect. Chemother. 2020, 26, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Van der Linden, P.D.; Sturkenboom, M.C.J.M.; Herings, R.M.C.; Leufkens, H.G.M.; Stricker, B.C. Fluoroquinolones and risk of Achilles tendon disorders: Case-control study. Br. Med. J. 2002, 324, 1306–1307. [Google Scholar] [CrossRef] [PubMed]
- Khaliq, Y.; Zhanel, G.G. Fluoroquinolone-associated tendinopathy: A critical review of the literature. Clin. Infect. Dis. 2003, 36, 1404–1410. [Google Scholar] [CrossRef] [PubMed]
- Baik, S.; Lau, J.; Huser, V.; McDonald, C.J. Association between tendon ruptures and use of fluoroquinolone, and other oral antibiotics: A 10-year retrospective study of 1 million US senior Medicare beneficiaries. BMJ 2020, 10, e034844. [Google Scholar] [CrossRef] [PubMed]
- Shu, Y.; Zhang, Q.; He, X.; Liu, Y.; Wu, P.; Chen, L. Fluoroquinolone-associated suspected tendonitis and tendon rupture: A pharmacovigilance analysis from 2016 to 2021 based on the FAERS database. Pharmacoepidemiology 2022, 13, 990241. [Google Scholar] [CrossRef]
- Fernández-Cuadros, M.E.; Casique-Bocanegra, L.O.; Albaladejo-Florín, M.J.; Gómez-Dueñas, S.; Ramos-Gonzalez, C.; Pérez-Moro, O.S. Bilateral Levofloxacin-Induced Achilles Tendon Rupture: An Uncommon Case Report and Review of the Literature. Clin. Med. Insights Arthritis Musculoskelet. Disord. 2019, 12, 1179544119835222. [Google Scholar] [CrossRef]
- Kawtharani, F.; Masrouha, K.Z.; Afeiche, N. Bilateral Achilles Tendon Ruptures Associated with Ciprofloxacin Use in the Setting of Minimal Change Disease: Case Report and Review of the Literature. J. Foot Ankle Surg. 2016, 55, P276–P278. [Google Scholar] [CrossRef] [PubMed]
- Manen, S.; Bost-Bru, C.; Wroblewski, I.; De Crescenzo, M.; Mortamet, G. Aminoglycoside prescription: Compliance with national guidelines in a pediatric hospital. Arch. Pédiatrie 2021, 28, 583–586. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.K.; Shivani, S.; Patel, A.; Shivani; Kaur, M. Aminoglycoside induced kidney injury and its pharmacological treatment–overview. J. Popul. Ther. Clin. Pharmacol. 2023, 30, 685–690. [Google Scholar] [CrossRef]
- Beaulieu-Jones, B.R.; Lin, B.; Phillips, A.M.; Haime, M.; Quin, J.A. Postoperative Atrial Fibrillation After Surgical Aortic Valve Replacement: Amiodarone and Warfarin Use. J. Surg. Res. 2023, 291, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Maraffa, J.M. Amiodarona, Encyclopedia of Toxicology, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2024; Volume 1, pp. 399–402. [Google Scholar] [CrossRef]
- Pérez-Ruiz, T.; Martínez-Lozano, C.; García-Martínez, M.D. Simultaneous determination of amiodarone and its metabolite desethylamiodarone by high-performance liquid chromatography with chemiluminescent detection. Anal. Chim. Acta 2008, 623, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Erven, L.; Schalij, M.J. Amiodarone: An effective antiarrhythmic drug with unusual side effects. Educ. Heart Clin. Pharmacol. 2010, 96, 1593–1600. [Google Scholar] [CrossRef] [PubMed]
- Papiris, S.A.; Triantafillidou, C.; Kolilekas, L.; Markoulaki, D.; Manali, E.D. Amiodarone: Review of pulmonary effects and toxicity. Drug Saf. 2010, 33, 539–558. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.A.; Khan, A.; Zaka, M. A Review of Structure Activity Relationship of Amiodarone and Its Derivatives. Open J. Med. Chem. 2016, 6, 37–42. [Google Scholar] [CrossRef][Green Version]
- Ganesan, K.; Bradley, B.; Jones, D.W.; Solomon, S. A Case Report on Type 2 Amiodarone Induced Thyrotoxicosis and Hypercalcemia. Am. J. Med. Sci. 2021, 362, 308–313. [Google Scholar] [CrossRef]
- Shah, M.; Rogers, L.; Tribuls, K.; Sorresso, D.J. Acute amiodarone-induced pulmonary toxicity. Chest 2023, 164, A4608–A4609. [Google Scholar] [CrossRef]
- Camus, P.; Fanton, A.; Bonniaud, P.; Camus, C.; Foucher, P. Interstitial lung disease induced by drugs and radiation. Respiration 2004, 71, 301–326. [Google Scholar] [CrossRef] [PubMed]
- Terzo, F.; Ricci, A.; D’Ascanio, M.; Raffa, S.; Mariotta, S. Amiodarone-induced pulmonary toxicity with an excellent response to treatment: A case report. Respir. Med. Case Rep. 2019, 29, 100974. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rivera, A.A.; Melendez, D.A.C.; Sultan, S.; Goyal, A.; Sharma, B. Amiodarone-induced pulmonary toxicity leading to acute respiratory distress syndrome. Chest 2023, 164, A1541–A1542. [Google Scholar] [CrossRef]
- Bhusare, B.P.; John, C.K.; Bhatt, V.P.; Nikam, T.D. In vitro propagation of Digitalis lanata Ehrh. through direct shoot regeneration—A source of cardiotonic glycoside. Ind. Crops Prod. 2018, 121, 313–319. [Google Scholar] [CrossRef]
- Ziff, O.J.; Kotecha, D. Digoxin: The good and the bad. Trends Cardiovasc. Med. 2016, 26, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Abhiraj, R.; Aggarwal, P. Digoxin toxicity and a rare tachycardia. Vis. J. Emerg. Med. 2023, 33, 101795. [Google Scholar] [CrossRef]
- Luciano, K.S.; Bogo, V.S.; Schulze, M.L.; Ronsoni, R.d.M. Bidirectional Ventricular Tachycardia Due to Digitalis Intoxication. J. Card. Arrythmias 2021, 34, 100–104. [Google Scholar] [CrossRef]
- Pita-Fernández, S.; Lombardía-Cortiña, M.; Orozco-Veltran, D.; Gil-Guillén, V. Clinical manifestations of elderly patients with digitalis intoxication in the emergency department. Arch. Gerontol. Geriatr. 2011, 53, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Doherty, J.E. The Clinical Pharmacology of Digitalis Glycosides: A Review. Am. J. Med. Sci. 2010, 339, 462–481. [Google Scholar] [CrossRef]
- Atalay, E.; Özdemir, M.T.; Çiğsar, G.; Omurca, F.; Aslan, N.; Yildiz, M.; Gey, Z.B. Angiotensin Converting Enzyme Inhibitor-related Angioedema: A Case of an Unexpected Death. Iran. J. Allergy Asthma Immunol. 2015, 14, 642–645. Available online: https://pubmed.ncbi.nlm.nih.gov/26725563/#:~:text=expand-,PMID%3A%2026725563,-Free%20article (accessed on 20 February 2024).
- Shrim, A.; Berger, H.; Kingdom, J.; Hamoudi, A.; Shah, P.S.; Koren, G. Prolonged exposure to angiotensin-converting enzyme inhibitors during pregnancy. Can. Fam. Physician 2005, 51, 1335–1337. Available online: https://pubmed.ncbi.nlm.nih.gov/16250418 (accessed on 15 January 2024).
- Villanacci, V.; Del Sordo, R. Angiotensin II receptor antagonist (Olmesartan) associated gastro-entero-colopathy. The multiform expressions of damage due to this class of drugs. Dig. Liver Dis. 2021, 53, P1260–P1261. [Google Scholar] [CrossRef] [PubMed]
- Shibata, S.; Fujita, T. 22-Renin Angiotensin Aldosterone System Blockers, Hypertension, 4th ed.; A Companion to Braunwald’s Heart Disease; Elsevier: Amsterdam, The Netherlands, 2024; pp. 258–273. [Google Scholar] [CrossRef]
- Velázquez-Armenta, E.Y.; Han, J.Y.; Choi, J.S.; Yang, K.M.; Nava-Ocampo, A.A. Angiotensin II receptor blockers in pregnancy: A case report and systematic review of the literature. Hypertens. Pregnancy 2007, 26, 51–66. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.R.; Khalil, R.A. Regulation of vascular angiotensin II type 1 and type 2 receptor and angiotensin-(1–7)/MasR signaling in normal and hypertensive pregnancy. Biochem. Pharmacol. 2024, 220, 115963. [Google Scholar] [CrossRef] [PubMed]
- Gersak, K.; Cvijic, M.; Cerar, L.K. Angiotensin II receptor blockers in pregnancy: A report of five cases. Reprod. Toxicol. 2009, 28, 109–112. [Google Scholar] [CrossRef]
- Sani, S.M.S.; Sahranavard, M.; Yazdanabad, M.J.; Shamsi, M.S.; Elyasi, S.; Mohammadpour, A.H.; Sathyapalan, T.; Arasteh, O.; Ghavami, V.; Sahebkar, A. The effect of concomitant use of Colony-Stimulating factors on bleomycin pulmonary toxicity–A systematic review and meta-analysis. Int. Immunopharmacol. 2022, 112, 109227. [Google Scholar] [CrossRef]
- Keijzer, A.; Kuenen, B. Fatal pulmonary toxicity in testis cancer with bleomycin-containing chemotherapy. J. Clin. Oncol. 2007, 25, 3543–3544. [Google Scholar] [CrossRef]
- Hu, S.; Palmer, L.B. Unilateral pulmonary fibrosis in setting of bleomycin toxicity and left hemidiaphragm paralysis. Chest 2023, 164, A3168. [Google Scholar] [CrossRef]
- Arif, S.; Jafri, F.; Tahir, O. Spontaneous pneumomediastinum and multiple pneumothoraces: An unusual presentation of bleomycin-related lung toxicity. Chest 2020, 158, A1147. [Google Scholar] [CrossRef]
- Aldossary, S.A. Review on Pharmacology of Cisplatin: Clinical use, toxicity and mechanism of resistance of cisplatin. Biomed. -Pharmacol. J. 2019, 12, 7–15. [Google Scholar] [CrossRef]
- Quintanilha, J.C.F.; Saavedra, K.F.; Visacri, M.B.; Moriel, P.; Salazar, L.A. Role of epigenetic mechanisms in cisplatin-induced toxicity. Crit. Rev. Oncol./Hematol. 2019, 137, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Cardinaal, R.M.; Huizing, E.H.; Veldman, J.E.; Smoorenburg, G.F. Cisplatin-induced ototoxicity: Morphological evidence of spontaneous outer hair cell recovery in albino guinea pigs? Hear. Res. 2000, 144, 147–156. [Google Scholar] [CrossRef]
- Yoo, K.H.; Tang, J.J.; Rashid, M.A.; Cho, C.H.; Corujo-Ramirez, A.; Choi, J.; Bae, M.G.; Brogren, D.; Hawse, J.R.; Hou, X.; et al. Nicotinamide Mononucleotide Prevents Cisplatin-Induced Cognitive Impairments. Found. Cancer J. Driv. Transform. Sci. 2021, 81, 3727–3737. [Google Scholar] [CrossRef] [PubMed]
- Moke, D.J.; Luo, C.; Millstein, J.; Knight, K.R.; Rassekh, S.R.; Brooks, B.; Orgel, E. Prevalence and risk factors for cisplatin-induced hearing loss in children, adolescents, and young adults: A multi-institutional North American cohort study. Lancet Child Adolesc. Health 2021, 5, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Truong, M.T.; Winzelberg, J.; Chang, K.W. Recovery from cisplatin-induced ototoxicity: A case report and review. Int. J. Pediatr. Otorhinolaryngol. 2007, 71, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Suddock, J.T.; Cain, M.D. Barbiturate Toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://pubmed.ncbi.nlm.nih.gov/29763050 (accessed on 15 January 2024).
- Van den Hondel, K.E. The rise of suicides using a deadly dose of barbiturates in Amsterdam and Rotterdam, the Netherlands, between 2006 and 2017. J. Forensic Leg. Med. 2020, 70, 101916. [Google Scholar] [CrossRef] [PubMed]
- Hassanian-Moghaddam, H.; Ghadiri, F.; Shojaei, M.; Zamani, N. Phenobarbital overdose presenting with status epilepticus: A case report. Seizure 2016, 40, 57–58. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.; Bentur, H.; Dolan, C.; Bugembe, T.; Gill, A.; Appleton, R. Outpatient anti-epileptic drug prescribing errors in a Children’s Hospital: An audit and literature review. Seizure 2014, 23, 786–791. [Google Scholar] [CrossRef] [PubMed]
- Koenig, M.; McCollum, B.; Spivey, J.K.; Coleman, J.K.; Shelton, C.H.A.R.L.E.S.; Cotes, R.O.; De Leon, J.O.S.E. Four cases of myocarditis in US hospitals possibly associated with impaired metabolism of clozapine and a comparison with previously published cases. Neuropsychopharmacol. Hung. 2022, 24, 29–41. [Google Scholar]
- DuBois, D. Toxicology and overdose of atypical antipsychotic medications in children: Does new necessarily mean safer? Curr. Opin. Pediatr. 2005, 17, 227–233. [Google Scholar] [CrossRef]
- Kumra, S.; Oberstar, J.V.; Sikich, L.; Findling, R.L.; McClellan, J.M.; Vinogradov, S.; Schulz, S.C. Efficacy and tolerability of second-generation antipsychotics in children and adolescents with schizophrenia. Schizophr. Bull. 2008, 34, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Shnoda, M.; Sagalov, A.; Patel, H.; Siddique, M.; Hegde, S. Clozapine-induced myocarditis: A rare case of myocarditis with life-threatening implications. J. Am. Coll. Cardiol. 2023, 81, 3781. [Google Scholar] [CrossRef]
- Tan, H.H.; Hoppe, J.; Heard, K. A systematic review of cardiovascular effects following atypical antipsychotic overdose. Am. J. Emerg. Med. 2009, 27, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Saad, M.A.; El-Sahar, A.E.; Sayed, R.H.; Elbaz, E.M.; Helmy, H.S.; Senousy, M.A. Venlafaxine Mitigates Depressive-Like Behavior in Ovariectomized Rats by Activating the EPO/EPOR/JAK2 Signaling Pathway and Increasing the Serum Estradiol Level. Neurotherapeutics 2019, 16, 404–415. [Google Scholar] [CrossRef]
- Singh, D.; Saadabadi, A. Venlafaxine. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Pan, J.J.; Shen, W.W. Serotonin Syndrome Induced by Low-Dose Venlafaxine. Ann. Pharmacother. 2003, 37, 209–211. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ștefan, C.S.; Nechita, A.; Dragostin, O.-M.; Fulga, A.; Lisă, E.-L.; Vatcu, R.; Dragostin, I.; Velicescu, C.; Fulga, I. Drugs Associated with Adverse Effects in Vulnerable Groups of Patients. Clin. Pract. 2024, 14, 1010-1020. https://doi.org/10.3390/clinpract14030080
Ștefan CS, Nechita A, Dragostin O-M, Fulga A, Lisă E-L, Vatcu R, Dragostin I, Velicescu C, Fulga I. Drugs Associated with Adverse Effects in Vulnerable Groups of Patients. Clinics and Practice. 2024; 14(3):1010-1020. https://doi.org/10.3390/clinpract14030080
Chicago/Turabian StyleȘtefan, Claudia Simona, Aurel Nechita, Oana-Maria Dragostin, Ana Fulga, Elena-Lăcrămioara Lisă, Rodica Vatcu, Ionut Dragostin, Cristian Velicescu, and Iuliu Fulga. 2024. "Drugs Associated with Adverse Effects in Vulnerable Groups of Patients" Clinics and Practice 14, no. 3: 1010-1020. https://doi.org/10.3390/clinpract14030080
APA StyleȘtefan, C. S., Nechita, A., Dragostin, O.-M., Fulga, A., Lisă, E.-L., Vatcu, R., Dragostin, I., Velicescu, C., & Fulga, I. (2024). Drugs Associated with Adverse Effects in Vulnerable Groups of Patients. Clinics and Practice, 14(3), 1010-1020. https://doi.org/10.3390/clinpract14030080





