The Efficacy of Regeneration Oil and Almond Oil on Split-Thickness Skin Graft Donor Sites: A Single-Blinded Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
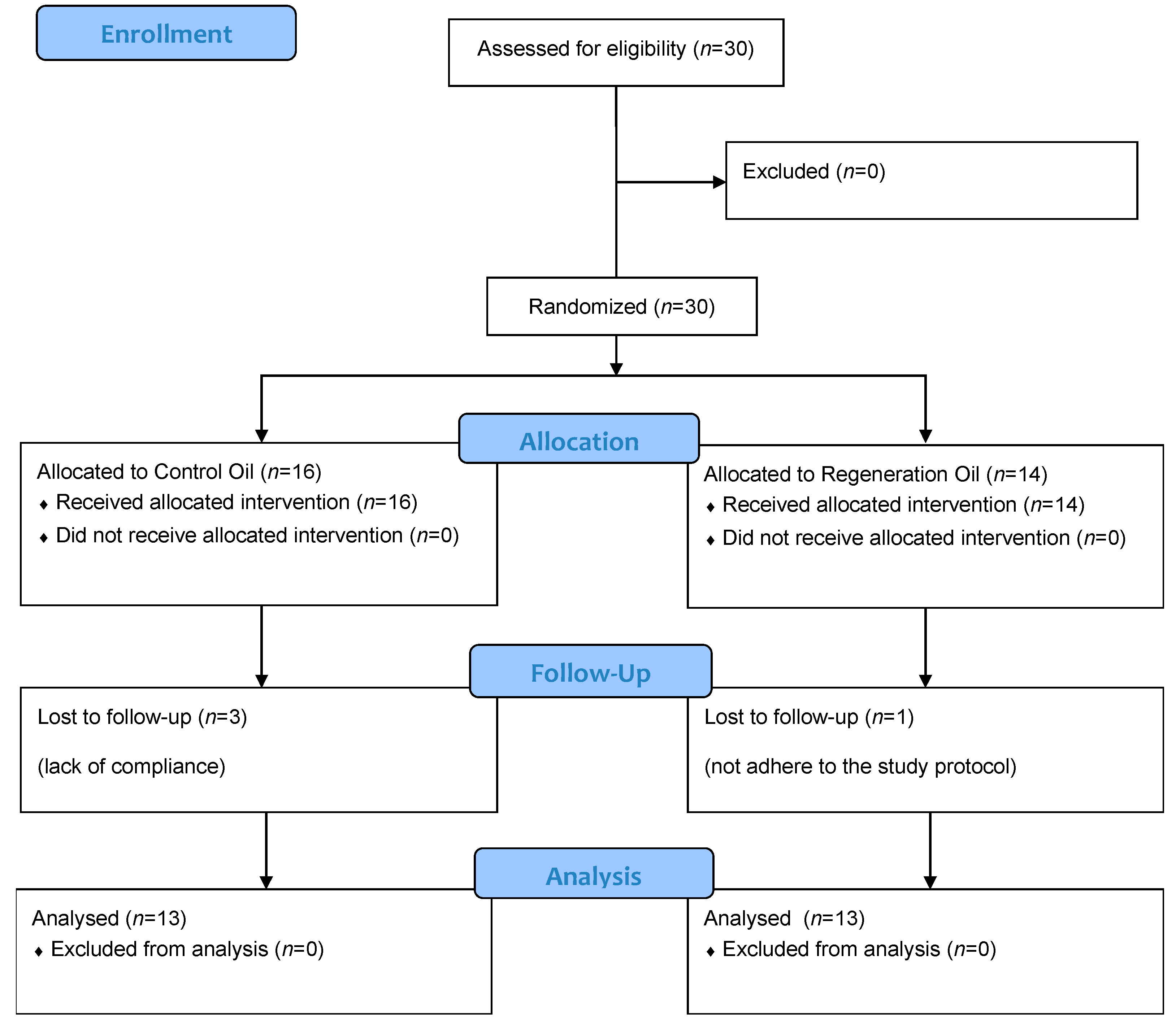
2.1. Trial Design and Participants
2.2. Procedure
2.3. Regeneration Oil/Control Oil
2.4. Outcome Measures
2.5. Statistical Analysis
3. Results
3.1. Primary Outcome: POSASOBS
3.2. Primary Outcome: POSASSELF
3.3. ITCH-Assessment-Scale
3.4. Colorimetry
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Farrar, A.J.; Farrar, F.C. Clinical Aromatherapy. Nurs. Clin. N. Am. 2020, 55, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Osaili, T.M.; Dhanasekaran, D.K.; Zeb, F.; Faris, M.E.; Naja, F.; Radwan, H.; Ismail, L.C.; Hasan, H.; Hashim, M.; Obaid, R.S.A. Status Review on Health-Promoting Properties and Global Regulation of Essential Oils. Molecules 2023, 28, 1809. [Google Scholar] [CrossRef]
- Cooke, B.; Ernst, E. Aromatherapy: A systematic review. Br. J. Gen. Pract. 2000, 50, 493–496. [Google Scholar]
- Maddocks-Jennings, W.; Wilkinson, J.M. Aromatherapy practice in nursing: Literature review. J. Adv. Nurs. 2004, 48, 93–103. [Google Scholar] [CrossRef]
- Lee, M.S.; Choi, J.; Posadzki, P.; Ernst, E. Aromatherapy for health care: An overview of systematic reviews. Maturitas 2012, 71, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Halm, M.; Baker, C.; Harshe, V. Effect of an Essential Oil Mixture on Skin Reactions in Women Undergoing Radiotherapy for Breast Cancer. J. Holist. Nurs. 2014, 32, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Barão Paixão, V.L.; Freire de Carvalho, J. Essential oil therapy in rheumatic diseases: A systematic review. Complement. Ther. Clin. Pract. 2021, 43, 101391. [Google Scholar] [CrossRef]
- Pereira, R.F.; Bártolo, P.J. Traditional Therapies for Skin Wound Healing. Adv. Wound Care 2016, 5, 208–229. [Google Scholar] [CrossRef]
- Draaijers, L.J.; Tempelman, F.R.H.; Botman, Y.A.M.; Tuinebreijer, W.E.; Middelkoop, E.; Kreis, R.W.; van Zuijlen, P.P.M. The Patient and Observer Scar Assessment Scale: A Reliable and Feasible Tool for Scar Evaluation. Plast. Reconstr. Surg. 2004, 113, 1960–1965. [Google Scholar] [CrossRef] [PubMed]
- Morris, V.; Murphy, L.M.; Rosenberg, M.; Rosenberg, L.; Holzer, C.E.; Meyer, W.J. Itch Assessment Scale for the Pediatric Burn Survivor. J. Burn Care Res. 2012, 33, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Piault, E.; Doshi, S.; Brandt, B.A.; Angün, Ç.; Evans, C.J.; Bergqvist, A.; Trocio, J. Linguistic validation of translation of the self-assessment goal achievement (saga) questionnaire from English. Health Qual. Life Outcomes 2012, 10, 40. [Google Scholar] [CrossRef] [PubMed]
- Ault, P.; Plaza, A.; Paratz, J. Scar massage for hypertrophic burns scarring—A systematic review. Burns 2018, 44, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Chan, A.-W.; Belley-Côté, E.P.; Drucker, A.M. Noninferiority Randomized Controlled Trials. J. Investig. Derm. 2022, 142, 1773–1777. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0022202X22003700 (accessed on 24 January 2023). [CrossRef] [PubMed]
- Bond, J.S.; Duncan, J.A.L.; Sattar, A.; Boanas, A.; Mason, T.; Oʼkane, S.; Ferguson, M.W.J. Maturation of the Human Scar: An Observational Study. Plast Reconstr. Surg. 2008, 121, 1650–1658. [Google Scholar] [CrossRef] [PubMed]
| Comorbidities | Control Oil (n = 16) | Regeneration Oil (n = 14) | |
|---|---|---|---|
| Cardiovascular | n (%) | 4 (25%) | 4 (28.6%) |
| Psychiatric | n (%) | 4 (25%) | 1 (7.1%) |
| Mild kidney disease | n (%) | 2 (12.5%) | 0 (0%) |
| Endocrinological | n (%) | 0 (%) | 2 (14.2%) |
| Rheumatological | n (%) | 2 (12.5%) | 4 (28.6%) |
| Neurological | n (%) | 0 (%) | 2 (14.2%) |
| Pulmonary | n (%) | 1 (6.3%) | 2 (14.2%) |
| Neoplastic | n (%) | 1 (6.3%) | 2 (14.2%) |
| Metabolic | n (%) | 4 (25%) | 4 (28.6%) |
| Outcome | Baseline | 4 Weeks | 12 Weeks | 24 Weeks |
|---|---|---|---|---|
| POSASOBS score Mean (SD) | ||||
| Control oil | 49.4 (±4.2) | 27.2 (±2.8) | 18.2 (±2.9) | 13.6 (±3.1) |
| Regeneration oil | 46.2 (±4.0) | 23.9 (±2.4) | 17.0 (±2.3) | 12.2 (±2.1) |
| p | 0.786 | 0.378 | 0.826 | 0.711 |
| POSASSELF scores Mean (SD) | ||||
| Control oil | 39.2 (±4.0) | 19.7 (±2.7) | 14.9 (±2.1) | 12.1 (±1.6) |
| Regeneration oil | 34.8 (±4.0) | 17.5 (±1.7) | 14.2 (±2.0) | 8.9 (±1.1) |
| p | 0.766 | 0.489 | 0.835 | 0.134 |
| ITCH rate, in % | ||||
| Control oil | 62.5 | 85.7 | 64.3 | 21.4 |
| Regeneration oil | 69.2 | 69.2 | 30.8 | 15.3 |
| p | 1.000 | 0.385 | 0.128 | 1.000 |
| Colorimetry scores Mean [confidence interval] | ||||
| Control oil | −24.4 [−45.9 to −2.9] | −12.0 [−31.2 to 7.2] | −5.0 [−17.8 to 7.8] | |
| Regeneration oil | −24.3 [−42.5 to −6.1] | −13.6 [−27.0 to −0.2] | −5.1 [−16.9 to 6.7] | |
| p | 0.385 | 0.128 | 1.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riedler, K.; Hecker, A.; Bauer, B.; Tax, C.; Gmainer, D.G.; Pignet, A.-L.; Kamolz, L.-P.; Lumenta, D.B. The Efficacy of Regeneration Oil and Almond Oil on Split-Thickness Skin Graft Donor Sites: A Single-Blinded Randomized Controlled Trial. Clin. Pract. 2023, 13, 648-655. https://doi.org/10.3390/clinpract13030059
Riedler K, Hecker A, Bauer B, Tax C, Gmainer DG, Pignet A-L, Kamolz L-P, Lumenta DB. The Efficacy of Regeneration Oil and Almond Oil on Split-Thickness Skin Graft Donor Sites: A Single-Blinded Randomized Controlled Trial. Clinics and Practice. 2023; 13(3):648-655. https://doi.org/10.3390/clinpract13030059
Chicago/Turabian StyleRiedler, Karoline, Andrzej Hecker, Birgit Bauer, Christa Tax, Daniel Georg Gmainer, Anna-Lisa Pignet, Lars-Peter Kamolz, and David Benjamin Lumenta. 2023. "The Efficacy of Regeneration Oil and Almond Oil on Split-Thickness Skin Graft Donor Sites: A Single-Blinded Randomized Controlled Trial" Clinics and Practice 13, no. 3: 648-655. https://doi.org/10.3390/clinpract13030059
APA StyleRiedler, K., Hecker, A., Bauer, B., Tax, C., Gmainer, D. G., Pignet, A.-L., Kamolz, L.-P., & Lumenta, D. B. (2023). The Efficacy of Regeneration Oil and Almond Oil on Split-Thickness Skin Graft Donor Sites: A Single-Blinded Randomized Controlled Trial. Clinics and Practice, 13(3), 648-655. https://doi.org/10.3390/clinpract13030059







