An Overview of Corneal Transplantation in the Past Decade
Abstract
1. Introduction
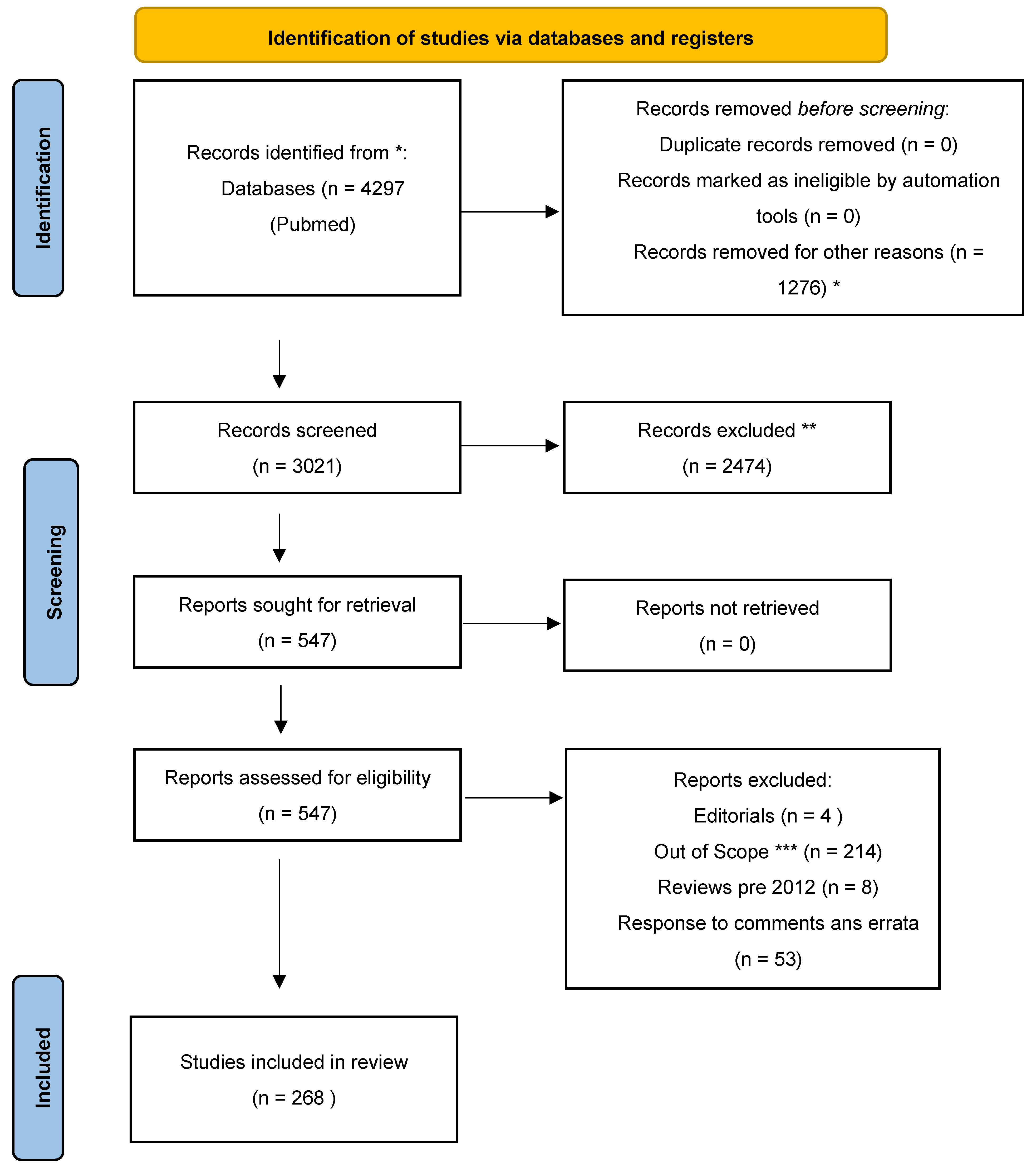
2. Materials and Methods
2.1. Corneal Transplant—Methodology, Indications, and Contraindications
2.2. Presurgical Protocols
2.3. Sources of Cornea Transplant Material
2.4. Wound Healing after Cornea Transplant—New Perspectives
2.5. Other Uses of Corneal Transplants
2.6. Preparation and Storage of Donor Corneas
2.7. The Effect of COVID-19 on Corneal Transplant Medicine and Services
2.8. Cornea Transplant in Developing Countries
2.9. Current Perspectives on CT
2.10. Keratoprostheses: A Game Changer
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rates, E.R.D.; Almeida, C.D.; Costa, E.D.P.F.; Farias, R.J.D.M.; Santos-Oliveira, R.; Alencar, L.M.R. Layer-by-layer investigation of ultrastructures and biomechanics of human cornea. Int. J. Mol. Sci. 2022, 23, 7833. [Google Scholar] [CrossRef] [PubMed]
- Musa, M.J.; Zeppieri, M. Principles and Technique of Fogging during Subjective Refraction; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Sridhar, M.S. Anatomy of cornea and ocular surface. Indian J. Ophthalmol. 2018, 66, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Rémont, L.; Duchesne, B.; La, C.; Rakic, J.M.; Hick, S. Updates in corneal transplantation. Rev. Med. de Liege 2014, 69, 490–496. [Google Scholar]
- Mota, S.H.-D.; Jacobo, M.P.; Revuelta, G.G.; Martínez, R.M.P. Corneal transplant in a second level hospital. A survival analysis. Gac. Med. De Mex. 2013, 149, 425–430. [Google Scholar]
- Belghmaidi, S.; Hajji, I.; Soummane, H.; Ennassiri, W.; Essafi, H.; Moutaouakil, A. Corneal transplantation: Study carried out at the Department of Ophthalmology, University Hospital Center Mohammed VI, Marrakech. Pan. Afr. Med. J. 2016, 23, 158. [Google Scholar]
- Studený, P.; Hamouz, J.; Kuchynka, P. Corneal transplantations in the Czech Republic in 2012. Czech Slovak Ophthalmol. 2014, 70, 224–227. [Google Scholar]
- Verdiguel-Sotelo, K.; Carrasco-Quiroz, A.; Rangel-Servín, J. Prognostic category of corneal transplantation in a referal center. Rev. Med. Del Inst. Mex. Del Seguro Soc. 2016, 54, 738–745. [Google Scholar]
- Sabater-Cruz, N.; Figueras-Roca, M.; Padró-Pitarch, L.; Tort, J.; Casaroli-Marano, R.P. Corneal transplantation activity in Catalonia, Spain, from 2011 to 2018: Evolution of indications and surgical techniques. PLoS ONE 2021, 16, e0249946. [Google Scholar] [CrossRef]
- Galvis, V.; Tello, A.; Gomez, A.J.; Rangel, C.M.; Prada, A.M.; Camacho, P.A. Corneal transplantation at an ophthalmological referral center in colombia: Indications and techniques (2004–2011). Open Ophthalmol. J. 2013, 7, 30–33. [Google Scholar] [CrossRef]
- Chen, M.C.; Kunselman, A.R.; Stetter, C.M.; Hannush, S.B.; Roberts, B.W. Corneal transplantation at Tenwek Hospital, Kenya, East Africa: Analysis of outcomes and associated patient socioeconomic characteristics. PLoS ONE 2017, 12, e0187026. [Google Scholar] [CrossRef]
- Hwang, S.; Chung, T.-Y.; Han, J.; Kim, K.; Lim, D.H. Corneal transplantation for keratoconus in South Korea. Sci. Rep. 2021, 11, 12580. [Google Scholar] [CrossRef] [PubMed]
- Sarnicola, C.; Sarnicola, E.; Cheung, A.Y.; Sarnicola, V. Deep anterior lamellar keratoplasty: Can all ruptures be fixed? Cornea 2023, 42, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, S.; Dohlman, T.H. Penetrating keratoplasty: Indications and graft survival by geographic region. Semin. Ophthalmol. 2022, 38, 31–43. [Google Scholar] [CrossRef]
- Sharma, N.; Mandal, S.; Asif, M.; Maharana, P.; Titiyal, J. A review of techniques and outcomes of endothelial keratoplasty in congenital hereditary endothelial dystrophy. Indian J. Ophthalmol. 2022, 70, 4108. [Google Scholar] [CrossRef]
- Goodman, C.F.B.; Mittal, R.B.; Awidi, A.; Vanner, E.A.; Han, E.; Daoud, Y.J.; Koo, E.H. Outcomes of secondary intraocular lens implantation and descemet stripping endothelial keratoplasty—Comparing staged versus combined surgical approach. Cornea, 2022; publish ahead. [Google Scholar] [CrossRef] [PubMed]
- Shahrudin, N.A.; Zahidin, A.Z.M.; Noh, U.K.; Halim, W.H.; Din, N. CMV endotheliitis: A cause for recurrent failed corneal transplant. GMS Ophthalmol. Cases 2017, 7, Doc31. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Montana, M.; Santosa, S.M.; Isjwara, I.D.; Huang, Y.-H.; Han, K.-Y.; O’Neil, C.; Wang, A.; Cortina, M.S.; de la Cruz, J.; et al. Angiogenesis and lymphangiogenesis in corneal transplantation–A review. Surv. Ophthalmol. 2018, 63, 453–479. [Google Scholar] [CrossRef]
- Verdier, D.D.; Sugar, A.; Baratz, K.; Beck, R.; Dontchev, M.; Dunn, S.; Gal, R.L.; Holland, E.J.; Kollman, C.; Lass, J.H.; et al. Corneal thickness as a predictor of corneal transplant outcome. Cornea 2013, 32, 729–736. [Google Scholar] [CrossRef]
- Zhou, H.W.; Xie, L.X. Effects of cataract surgery on endothelium in transplanted corneal grafts: Comparison of extracapsular cataract extraction and phacoemulsification for complicated cataract after penetrating keratoplasty. Chin. Med. J. 2016, 129, 2096–2101. [Google Scholar] [CrossRef]
- Steger, B.; Kaye, S.B.; Romano, V. Corneal transplantation: The fine line between donor shortage and tissue quality. BMJ Open Ophthalmol. 2022, 7, e001046. [Google Scholar] [CrossRef]
- Niederkorn, J.Y. Corneal transplantation and immune privilege. Int. Rev. Immunol. 2013, 32, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Shaharuddin, B.; Ahmad, S.; Meeson, A.; Ali, S. Concise review: Immunological properties of ocular surface and importance of limbal stem cells for transplantation. Stem Cells Transl. Med. 2013, 2, 614–624. [Google Scholar] [CrossRef] [PubMed]
- Armitage, W.J.; Goodchild, C.; Griffin, M.D.; Gunn, D.J.; Hjortdal, J.; Lohan, P.; Murphy, C.C.; Pleyer, U.; Ritter, T.; Tole, D.M.; et al. High-risk corneal transplantation: Recent developments and future possibilities. Transplantation 2019, 103, 2468–2478. [Google Scholar] [CrossRef] [PubMed]
- Major, J.; Foroncewicz, B.; Szaflik, J.P.; Mucha, K. Immunology and Donor-Specific Antibodies in Corneal Transplantation. Arch. Immunol. Et Ther. Exp. 2021, 69, 32. [Google Scholar] [CrossRef] [PubMed]
- Boynton, G.E.; Woodward, M.A. Eye-bank preparation of endothelial tissue. Curr. Opin. Ophthalmol. 2014, 25, 319–324. [Google Scholar] [CrossRef]
- Van Rooij, J.; Lucas, E.H.; Geerards, A.J.; Remeijer, L.; Wubbels, R. Corneal transplantation for Fuchs’ endothelial dystrophy: A comparison of three surgical techniques concerning 10 year graft survival and visual function. PLoS ONE 2018, 13, e0203993. [Google Scholar] [CrossRef] [PubMed]
- Akpek, E.K.; Alkharashi, M.; Hwang, F.; Ng, S.M.; Lindsley, K. Artificial corneas versus donor corneas for repeat corneal transplants. Cochrane Database Syst. Rev. 2014, 11, CD009561. [Google Scholar] [CrossRef]
- Arya, S.K.; Raj, A.; Deswal, J.; Kohli, P.; Rai, R. Donor demographics and factors affecting corneal utilisation in Eye Bank of North India. Int. Ophthalmol. 2021, 41, 1773–1781. [Google Scholar] [CrossRef]
- Fu, L.; Hollick, E.J. Artificial Cornea Transplantation; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- AlMutlak, M.; Li, J.Y.; Bin Helayel, H.; Fairaq, R. Future of corneal donation and transplantation: Insights from the COVID-19 pandemic. Cornea 2020, 40, 274–276. [Google Scholar] [CrossRef]
- Gupta, N.; Dhasmana, R.; Maitreya, A.; Badahur, H. Glycerol-preserved corneal tissue in emergency corneal transplantation: An alternative for fresh corneal tissue in COVID-19 crisis. Indian J. Ophthalmol. 2020, 68, 1412–1416. [Google Scholar] [CrossRef]
- Islam, M.M.; Buznyk, O.; Reddy, J.C.; Pasyechnikova, N.; Alarcon, E.I.; Hayes, S.; Lewis, P.; Fagerholm, P.; He, C.; Iakymenko, S.; et al. Biomaterials-enabled cornea regeneration in patients at high risk for re-jection of donor tissue transplantation. NPJ Regen. Med. 2018, 3, 2. [Google Scholar] [CrossRef] [PubMed]
- Iglicki, M.; Loewenstein, A.; Manera, M.; Castro, C.; Busch, C.; Zur, D.; Negri, H.P. Autologous corneal transplant from an enucleated fellow eye for choroidal melanoma: A case report. Case Rep. Ophthalmol. 2020, 11, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Chun, Y.S.; Kim, J.C. Autologous tragal perichondrium transplantation: A novel approach for the management of painful bullous keratopathy. Korean J. Ophthalmol. 2013, 27, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Parekh, M.; Ferrari, S.; Sheridan, C.; Kaye, S.; Ahmad, S. Concise review: An update on the culture of human corneal en-dothelial cells for transplantation. Stem Cells Transl. Med. 2016, 5, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, N.; Qidwai, U.S.A.; Hussain, M.; Kazi, A. Efficacy of topical bevacizumab in high-risk corneal transplant survival. Pak. J. Med. Sci. 2013, 29, 519–522. [Google Scholar] [CrossRef]
- Bhatti, N.; Qidwai, U.; Hussain, M.; Kazi, A. Efficacy of sub-conjunctival and topical bevacizumab in high-risk corneal transplant survival. J. Pak. Med. Assoc. 2013, 63, 1256–1259. [Google Scholar]
- Hattori, T.; Saban, D.R.; Emami-Naeini, P.; Chauhan, S.K.; Funaki, T.; Ueno, H.; Dana, R. Donor-derived, tolerogenic dendritic cells suppress immune rejection in the indirect allosensitization-dominant setting of corneal transplantation. J. Leukoc. Biol. 2012, 91, 621–627. [Google Scholar] [CrossRef]
- Dohlman, T.; Di Zazzo, A.; Omoto, M.; Hua, J.; Ding, J.; Hamrah, P.; Chauhan, S.K.; Dana, R. E-selectin mediates immune cell trafficking in corneal transplantation. Transplantation 2016, 100, 772–780. [Google Scholar] [CrossRef]
- Guo, Y.-Y.; Yin, C.-J.; Zhao, M.; Guo, L.-T.; Su, R.-F.; Fu, X.-X.; Dong, W.-L.; Tan, X.-B. Effect of RMT1-10 on the immunological characteristics of dendritic cells cultured in vitro and corneal transplantation in vivo. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 9150–9162. [Google Scholar]
- Sachdeva, G.S.; Cabada, J.P.; Karim, S.S.; Kahandawa, D.L.; Thomas, K.A.; Kumar, A.; Barry, R.J.; Butt, G.F. Effectiveness of matching human leukocyte antigens (HLA) in corneal transplantation: A systematic review protocol. Syst. Rev. 2021, 10, 150. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, L.; Zhang, L. Cyclosporine nanomicelle eye drop: A novel medication for corneal graft transplantation treatment. Biol. Pharm. Bull. 2015, 38, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Gupta, N.; Vanathi, M.; Tandon, R. Corneal transplantation in the modern era. Indian J. Med. Res. 2019, 150, 7–22. [Google Scholar]
- Jafarinasab, M.R.; Feizi, S.; Esfandiari, H.; Kheiri, B.; Feizi, M. Traumatic wound dehiscence following corneal transplanta-tion. J. Ophthalmic Vis. Res. 2012, 7, 214–218. [Google Scholar]
- Doğan, C.; Arslan, O. Outcomes of therapeutic and tectonic penetrating keratoplasty in eyes with perforated infectious corneal ulcer. Turk. J. Ophthalmol. 2019, 49, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Linke, S.J.; Eddy, M.-T.; Bednarz, J.; Fricke, O.H.; Wulff, B.; Schröder, A.-S.; Hassenstein, A.; Klemm, M.; Püschel, K.; Richard, G.; et al. Thirty years of cornea cultivation: Long-term experience in a single eye bank. Acta Ophthalmol. 2012, 91, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Nathawat, R.; Parihar, J.S. Policy framework for advancing eye banking and cornea transplantation. Indian J. Ophthalmol. 2021, 69, 2563. [Google Scholar] [CrossRef]
- Agarwal, R.; Shakarwal, C.; Sharma, N.; Titiyal, J. Intraoperative optical coherence tomography-guided donor corneal tissue assessment and preparation. Indian J. Ophthalmol. 2022, 70, 3496. [Google Scholar] [CrossRef]
- Modis, L., Jr.; Langenbucher, A.; Seitz, B. Corneal endothelial cell density and pachymetry measured by contact and noncontact specular microscopy. J. Cataract. Refract. Surg. 2002, 28, 1763–1769. [Google Scholar] [CrossRef]
- Bonci, P.; Della Valle, V.; Lia, A. Preparation of donor corneas: A study of the endothelial population. Eur. J. Ophthalmol. 2008, 18, 341–344. [Google Scholar] [CrossRef]
- Hagenah, M.; Winter, R. Disinfection of human donor corneas during preparation and preservation. Klin. Monbl. Augenheilkd. 1996, 209, 1–6. [Google Scholar] [CrossRef]
- Wykrota, A.A.; Weinstein, I.; Hamon, L.; Daas, L.; Flockerzi, E.; Suffo, S.; Seitz, B. Approval rates for corneal donation and the origin of donor tissue for transplantation at a university-based tertiary referral center with corneal subspecialization hosting a LIONS Eye Bank. BMC Ophthalmol. 2022, 22, 17. [Google Scholar]
- Solley, K.D.; Berges, A.; Diaz, C.; Ostrander, B.T.; Ding, A.S.; Larson, S.A.; Frank, K.; Lee, D.; Guerrero, J.; deCarvalho, T.; et al. Evaluation of efficacy, efficiency, and cell viability of a novel descemet membrane endothelial keratoplasty graft preparation device, DescePrep, in nondiabetic and diabetic human donor corneas. Cornea 2022, 41, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Gupta, R.K. Quantitative & qualitative analysis of endothelial cells of donor cornea before & after penetrating keratoplasty in different pathological conditions. Indian J. Med. Res. 2016, 143, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Lass, J.H.; Sugar, A.; Benetz, B.A.; Beck, R.W.; Dontchev, M.; Gal, R.L.; Kollman, C.; Gross, R.; Heck, E.; Holland, E.J.; et al. Endothelial cell density to predict endothelial graft failure after penetrating keratoplasty. Arch. Ophthalmol. 2010, 128, 63–69. [Google Scholar] [CrossRef]
- Garcin, T.; Gauthier, A.-S.; Crouzet, E.; He, Z.; Herbepin, P.; Perrache, C.; Acquart, S.; Cognasse, F.; Forest, F.; Thuret, G.; et al. Innovative corneal active storage machine for long-term eye banking. Am. J. Transplant. 2018, 19, 1641–1651. [Google Scholar] [CrossRef]
- Garcin, T.; Gauthier, A.-S.; Crouzet, E.; He, Z.; Herbepin, P.; Perrache, C.; Acquart, S.; Cognasse, F.; Forest, F.; Gain, P.; et al. Three-month storage of human corneas in an active storage machine. Transplantation 2020, 104, 1159–1165. [Google Scholar] [CrossRef]
- Amouzegar, A.; Chauhan, S.K.; Dana, R. Alloimmunity and tolerance in corneal transplantation. J. Immunol. 2016, 196, 3983–3991. [Google Scholar] [CrossRef]
- Martheswaran, T.; Desautels, J.D.; Moshirfar, M.; Shmunes, K.M.; Ronquillo, Y.; Hoopes, P.C. A contemporary risk analysis of iatrogenic transmission of Creutzfeldt-Jakob Disease (CJD) via corneal transplantation in the United States. Ophthalmol. Ther. 2020, 9, 465–483. [Google Scholar] [CrossRef]
- Soda, R.; Fukuoka, H.; Sotozono, C. A case of recurrent fungal keratitis post-amniotic membrane transplantation for corneal perforation. Case Rep. Ophthalmol. 2022, 13, 147–153. [Google Scholar] [CrossRef]
- Chow, K.M.; Alias, R. A 10-Year retrospective case series on wound dehiscence following corneal transplant. Cureus 2022, 14, e25184. [Google Scholar] [CrossRef]
- Gómez-Benlloch, A.; Montesel, A.; Pareja-Aricò, L.; Mingo-Botín, D.; Michael, R.; Barraquer, R.I.; Alió, J. Causes of corneal transplant failure: A multicentric study. Acta Ophthalmol. 2021, 99, e922–e928. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, S.K.; Jurkunas, U.; Funaki, T.; Dastjerdi, M.; Dana, R. Quantification of allospecific and nonspecific corneal endo-thelial cell damage after corneal transplantation. Eye 2015, 29, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Gellert, A.; Unterlauft, J.D.; Rehak, M.; Girbardt, C. Descemet membrane endothelial keratoplasty (DMEK) improves vi-sion-related quality of life. Graefes Arch. Clin. Exp. Ophthalmol. 2022, 260, 3639–3645. [Google Scholar] [CrossRef]
- Eleiwa, T.; Elsawy, A.; Ozcan, E.; Chase, C.; Feuer, W.; Yoo, S.H.; Perez, V.L.; Shousha, M.F.A. Prediction of corneal graft rejection using central endothelium/Descemet’s membrane complex thickness in high-risk corneal transplants. Sci. Rep. 2021, 11, 14542. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Higa, K.; Tsubota, K.; Shimazaki, J. Elevation of preoperative recipient aqueous cytokine levels in eyes with primary graft failure after corneal transplantation. Mol. Vis. 2018, 24, 613–620. [Google Scholar] [PubMed]
- Fodor, M.; Petrovski, G.; Pásztor, D.; Gogolák, P.; Rajnavölgyi, É.; Berta, A. Effects of Awakening and the Use of Topical Dexamethasone and Levofloxacin on the Cytokine Levels in Tears Following Corneal Transplantation. J. Immunol. Res. 2014, 2014, 570685. [Google Scholar] [CrossRef]
- Yuan, J.; Zhai, J.-J.; Huang, X.; Zhou, S.-Y.; Chen, J.-Q. Ocular Safety and Pharmacokinetics Study of FK506 Suspension Eye Drops After Corneal Transplantation. J. Ocul. Pharmacol. Ther. 2012, 28, 153–158. [Google Scholar] [CrossRef]
- Khattak, A.; An-Nakhli, F. Incidence and quantification of corneal haze by Pentacam Scheimpflug densitometry following photorefractive keratectomy for myopia in virgin and post corneal transplant eyes with dark irides. Saudi J. Ophthalmol. 2020, 34, 8–12. [Google Scholar] [CrossRef]
- Stevenson, L.J.; Abell, R.G.; McGuinness, M.B.; Vajpayee, R.B. Comparative evaluation of clinical characteristics and visual outcomes of traumatic and non-traumatic graft dehiscence following corneal transplantation surgery. Clin. Ophthalmol. 2019, ume 13, 2243–2249. [Google Scholar] [CrossRef]
- Learned, D.L.; Gupta, C.K.; Stec, L.A.; Heidemann, D.G. Perioperative corneal transplant wound dehiscence. Anesthesiology 2016, 124, 185. [Google Scholar] [CrossRef]
- Diamond, M.A.; Chan, S.W.S.; Zhou, X.; Glinka, Y.; Girard, E.; Yucel, Y.; Gupta, N. Lymphatic vessels identified in failed corneal transplants with neovascularisation. Br. J. Ophthalmol. 2018, 103, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Le, V.N.H.; Schneider, A.-C.; Scholz, R.; Bock, F.; Cursiefen, C. Fine Needle-Diathermy Regresses Pathological Corneal (Lymph)Angiogenesis and Promotes High-Risk Corneal Transplant Survival. Sci. Rep. 2018, 8, 5707. [Google Scholar] [CrossRef]
- Bourges, J.L. When cataracts lead to a corneal transplant. Med. Sci. 2020, 36, 747–751. [Google Scholar]
- Oh, J.Y.; Lee, D.S.; Heo, J.W.; Choi, H.J.; Kim, M.K.; Wee, W.R. Combined corneal allotransplantation and vitreoretinal surgery using an Eckardt temporary keratoprosthesis: Analysis for factors determining corneal allograft survival. Clin. Ophthalmol. 2014, 8, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Vicente, A.; Byström, B.; Lindström, M.; Stenevi, U.; Domellöf, F.P. Aniridia-related keratopathy: Structural changes in naïve and transplanted corneal buttons. PLoS ONE 2018, 13, e0198822. [Google Scholar] [CrossRef]
- Moshirfar, M.; Basharat, N.F.; Seitz, T.S.; Ply, B.K.; Ronquillo, Y.C.; Hoopes, P.C. Corneal Transplant Rejections in Patients Receiving Immune Checkpoint Inhibitors. J. Clin. Med. 2022, 11, 5647. [Google Scholar] [CrossRef]
- Almulhim, A.; Alnaim, A.F.; Abdulrazek, A.; Alotaibi, H.A. Incidence and outcomes of transplant of infected donor corneal tissues in a tertiary hospital in Saudi Arabia. Cureus 2022, 14, e25514. [Google Scholar] [CrossRef]
- Cabra-Rodríguez, R.; Tenorio-Abreu, A.; Breval, I.Z.-Y. Candida parapsilosis infection in a corneal transplant patient. Rev. Esp. Quim. 2018, 31, 285–287. [Google Scholar]
- Sati, A.; Wagh, S.; Mishra, S.; Kumar, S.; Kumar, P. Post-corneal transplant Candida keratitis—Incidence and outcome. Indian J. Ophthalmol. 2022, 70, 536. [Google Scholar] [CrossRef]
- Griffin, B.; Walkden, A.; Okonkwo, A.; Au, L.; Brahma, A.; Carley, F. Microbial keratitis in corneal transplants: A 12-year analysis. Clin. Ophthalmol. 2020, ume 14, 3591–3597. [Google Scholar] [CrossRef]
- van der Wekken-Pas, L.; de Haas, P.; Wisse, R.; Rados, M.; van der Elst, K. Candida keratitis and endopthalmitis after corneal transplantation; two case reports, a novel regimen and literature overview of therapeutic options. Eur. J. Ophthalmol. 2021. [Google Scholar] [CrossRef]
- Raffa, L. Rhino-orbito-cerebral mucormycosis following penetrating keratoplasty. J. Surg. Case Rep. 2019, 2019, rjz314. [Google Scholar] [CrossRef]
- Giurgola, L.; Gatto, C.; Honisch, C.; Rossi, O.; Ragazzi, E.; Tothova, J.D. Killing efficacy of a new hypothermic corneal storage medium against the micro-organisms frequently found in human donor cornea intended for transplantation. BMJ Open Ophthalmol. 2021, 6, e000833. [Google Scholar] [CrossRef]
- Oh, J.Y.; Lee, R.H.; Yu, J.M.; Ko, J.H.; Lee, H.J.; Ko, A.Y.; Roddy, G.W.; Prockop, D.J. Intravenous mesenchymal stem cells prevented rejection of allogeneic corneal transplants by aborting the early inflammatory response. Mol. Ther. 2012, 20, 2143–2152. [Google Scholar] [CrossRef]
- Jarstad, A.; Chou, J.; Garg, S. Cardiac arrest during cornea transplant surgery. BMJ Case Rep. 2020, 13, e235272. [Google Scholar] [CrossRef] [PubMed]
- Kornmann, H.L.; Gedde, S.J. Glaucoma management after corneal transplantation surgeries. Curr. Opin. Ophthalmol. 2016, 27, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Al-Mahmood, A.M.; Al-Swailem, S.A.; Edward, D.P. Glaucoma and Corneal Transplant Procedures. J. Ophthalmol. 2012, 2012, 576394. [Google Scholar] [CrossRef] [PubMed]
- Nobacht, S.; Kusters, B.; Breukink, M.B.; Rongen, G.A.; Cruysberg, J.R.M. Recurrence of paraproteinemic crystalline ker-atopathy after corneal transplantation: A case of monoclonal gammopathy of ocular significance. Am. J. Ophthalmol. Case Rep. 2020, 19, 100803. [Google Scholar] [CrossRef]
- Paniz-Mondolfi, A.E.; Agemy, S.; Canete-Gibas, C.; Gitman, M.R.; Iacob, C.E.; Necula, I.; Wang, C.Y.; Delgado Noguera, L.A.; Sanders, C.; Wiederhold, N.P.; et al. First report of human infection caused by Colletotrichum chlo-rophyti occurring in a post-corneal transplant patient with endophthalmitis. Med. Mycol. Case Rep. 2021, 32, 73–76. [Google Scholar] [CrossRef]
- Konidaris, V.; Mersinoglou, A.; Vyzantiadis, T.-A.; Papadopoulou, D.; Boboridis, K.G.; Ekonomidis, P. Corneal Transplant Infection due to Alternaria alternata: A Case Report. Case Rep. Ophthalmol. Med. 2013, 2013, 589620. [Google Scholar] [CrossRef]
- Martinez, J.D.; Galor, A.; Perez, V.L.; Karp, C.L.; Yoo, S.H.; Alfonso, E.C. Endothelial graft failure after contralateral autologous corneal transplantation. Cornea 2013, 32, 745–750. [Google Scholar] [CrossRef]
- Aboshiha, J.; Jones, M.N.A.; Hopkinson, C.L.; Larkin, D.F.P. Differential survival of penetrating and lamellar transplants in management of failed corneal grafts. JAMA Ophthalmol. 2018, 136, 859. [Google Scholar] [CrossRef]
- Tan, D.; Ang, M.; Arundhati, A.; Khor, W.-B. Development of selective lamellar keratoplasty within an asian corneal transplant program: The singapore corneal transplant study (an American ophthalmological society thesis). Am. J. Ophthalmol. 2015, 113, T10. [Google Scholar]
- Masmoudi, J.; Feki, I.; Trigui, D.; Baati, I.; Jaoua, A. Complex visual hallucinations secondary to corneal transplant: The Charles Bonnet syndrome. La Tunis. medicale 2015, 93, 396–397. [Google Scholar]
- Desautels, J.D.; Moshirfar, M.; Martheswaran, T.; Shmunes, K.M.; Ronquillo, Y. Risks Posed to Corneal Transplant Recipients by COVID-19-Affected Donors. Ophthalmol. Ther. 2020, 9, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, J.; Jeria, S.; Thakkar, A. A review of corneal transplantation: An insight on the overall global post-COVID-19 impact. Cureus 2022, 14, e29160. [Google Scholar] [CrossRef]
- Anonymous. Correction: Corneal transplantation in the aftermath of the COVID-19 pandemic: An international perspective. Br. J. Ophthalmol. 2022, 106, e1. [Google Scholar] [CrossRef] [PubMed]
- Tanyildiz, B.; Oklar, M.; Gunaydin, N.T.; Kandemir, B. Changing trends in the corneal transplantation and the impact of the COVID-19 pandemic on corneal transplant recipient selection. Saudi J. Ophthalmol. 2022, 36, 95–101. [Google Scholar]
- Aiello, F.; Besso, F.G.; Pocobelli, G.; Afflitto, G.G.; Gisoldi, R.A.M.C.; Nucci, C.; Ponzin, D.; Bonci, P.; Calabrò, G.; Ceccuzzi, R.; et al. Corneal transplant during COVID-19 pandemic: The Italian Eye Bank national report. Cell Tissue Bank. 2021, 22, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Mencucci, R.; Cennamo, M.; Ponzin, D.; Besso, F.G.; Pocobelli, G.; Buzzi, M.; Nucci, C.; Aiello, F. Impact of the COVID-19 pandemic on corneal transplantation: A report from the Italian association of eye banks. Front. Med. 2022, 9, 844601. [Google Scholar] [CrossRef] [PubMed]
- Ang, M.; Moriyama, A.; Colby, K.; Sutton, G.; Liang, L.; Sharma, N.; Hjortdal, J.; Lam, D.S.C.; Williams, G.P.; Armitage, J.; et al. Corneal transplantation in the aftermath of the COVID-19 pandemic: An international perspective. Br. J. Ophthalmol. 2020, 104, 1477–1481. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Duan, F.; Luo, C.; Liu, Q.; Qu, X.; Liang, L.; Wu, K. Characteristics of ocular findings of patients with coronavirus disease 2019 (COVID-19) in Hubei Province, China. JAMA Ophthalmol. 2020, 138, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Mudgil, N.; Wadgave, Y.; Mishra, S.S. Corneal transplantation during COVID-19 pandemic: Need for special considerations—A live review. AIMS Public Health 2021, 8, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Franch, A.; Fasolo, A.; Carraro, P.; Favarato, M.; Birattari, F.; Leon, P.E.; Ponzin, D. Corneal transplantation during the COVID-19 pandemic: An operational guide. Eur. J. Ophthalmol. 2021, 32, 842–847. [Google Scholar] [CrossRef]
- Siddharthan, K.S.; Agrawal, A.; Reddy, J.K. Four in one: Four recepients with a single donor tissue—A novel concept for eye transplantation surgery post-COVID-19. Indian J. Ophthalmol. 2020, 68, 2471–2474. [Google Scholar] [CrossRef] [PubMed]
- Sati, A.; Shankar, S.; Jha, A.; Gurunadh, V.S. Customised component corneal transplantation: A blessing for three patients. BMJ Case Rep. 2014, 2014, bcr2014205579. [Google Scholar] [CrossRef]
- Bhalerao, S.; Vuyyuru, S.; Mohamed, A.; Gogri, P.; Garg, P.; Mallipudi, R. Impact of COVID-19 related national lockdown on care of corneal transplantation patients at a tertiary eye care centre in India. Indian J. Ophthalmol. 2021, 69, 1569–1574. [Google Scholar] [CrossRef]
- Garcia, A.M.G.; de Sousa, L.B.; Shiguematsu, A.I. Impacts of COVID-19 pandemic and public policies on corneal transplantations in Brazil. Arq. Bras. de Oftalmol. 2023, 86, 277–285. [Google Scholar] [CrossRef]
- Bhat, S.; Joshi, P.; Balasubramaniam, A. Impact of COVID-19 pandemic on patients with corneal transplant. Indian J. Ophthalmol. 2021, 69, 1967–1968. [Google Scholar] [CrossRef]
- Thuret, G.; Courrier, E.; Poinard, S.; Gain, P.; Baud’Huin, M.; Martinache, I.; Cursiefen, C.; Maier, P.; Hjortdal, J.; Ibanez, J.S.; et al. One threat, different answers: The impact of COVID-19 pandemic on cornea donation and donor selection across Europe. Br. J. Ophthalmol. 2020, 106, 312–318. [Google Scholar] [CrossRef]
- York, N.J.; Tinley, C. Corneal donations in South Africa: A 15-year review. S. Afr. Med. J. 2017, 107, 697–701. [Google Scholar] [CrossRef]
- Eze, B.I.; Okoye, O.; Eze, J.N. Knowledge and attitudes regarding eye donation and corneal transplant: Medical versus non-medical university students in a developing country in Africa. Exp. Clin. Transpl. 2014, 12, 454–461. [Google Scholar]
- Williams, A.M.; Muir, K.W. Awareness and attitudes toward corneal donation: Challenges and opportunities. Clin. Ophthalmol. 2018, ume 12, 1049–1059. [Google Scholar] [CrossRef]
- Wang, X.; Jin, L.; Wang, J.; Garrett, E.H.; Shuman, J.; Yang, K.; Schottman, T.; Chen, T.; Wang, J.; Wang, C.; et al. Attitudes and knowledge concerning corneal donation in a population-based sample of urban Chinese adults. Cornea 2016, 35, 1362–1367. [Google Scholar] [CrossRef] [PubMed]
- Roba, A.A.; Tilahun, Y.; Bekele, S. Life with another cornea: Impact of corneal transplantation and eye banking in Ethiopia. Ethiop. Med. J. 2013, 51, 67–72. [Google Scholar] [PubMed]
- Hussen, M.S.; Gebreselassie, K.L.; Woredekal, A.T.; Adimassu, N.F. Willingness to donate eyes and its associated factors among adults in Gondar town, North West Ethiopia. BMC Ophthalmol. 2017, 17, 178. [Google Scholar] [CrossRef]
- Gain, P.; Jullienne, R.; He, Z.; Aldossary, M.; Acquart, S.; Cognasse, F.; Thuret, G. Global survey of corneal transplantation and eye banking. JAMA Ophthalmol. 2016, 134, 167–173. [Google Scholar] [CrossRef]
- Kim, M.K.; Hara, H. Current status of corneal xenotransplantation. Int. J. Surg. 2015, 23, 255–260. [Google Scholar] [CrossRef]
- Le, R.; Yucel, N.; Khattak, S.; Yucel, Y.H.; Prud’Homme, G.J.; Gupta, N. Current indications and surgical approaches to corneal transplants at the University of Toronto: A clinical-pathological study. Can. J. Ophthalmol. 2016, 52, 74–79. [Google Scholar] [CrossRef]
- Liu, S.; Wong, Y.L.; Walkden, A. Current perspectives on corneal transplantation. Clin. Ophthalmol. 2022, 16, 631–646. [Google Scholar] [CrossRef]
- Wong, Y.L.; Liu, S.; Walkden, A. Current perspectives on corneal transplantation (Part 2). Clin. Ophthalmol. 2022, 16, 647–659. [Google Scholar] [CrossRef] [PubMed]
- Young, A.L.; Kam, K.W.; Jhanji, V.; Cheng, L.L.; Rao, S.K. A new era in corneal transplantation: Paradigm shift and evolution of techniques. Hong Kong Med. J. 2012, 18, 509–516. [Google Scholar]
- Grzybowski, A.; Patryn, R. Current state of corneal transplants in Poland. Acta Ophthalmol. 2017, 96, e93–e94. [Google Scholar] [CrossRef]
- Amagai, Y.; Karasawa, K.; Kyungsook, J.; Matsuda, A.; Kojima, M.; Watanabe, J.; Hibi, T.; Matsuda, H.; Tanaka, A. Development of a novel carrier optimized for cell sheet transplantation. Biomatter 2015, 5, e1027846. [Google Scholar] [CrossRef]
- Rossi, F.; Canovetti, A.; Malandrini, A.; Lenzetti, I.; Pini, R.; Menabuoni, L. An “all-laser” endothelial transplant. J. Vis. Exp. 2015, 101, e52939. [Google Scholar]
- Zheng, X.; Zhang, D.; Li, S.; Zhang, J.; Zheng, J.; Du, L.; Gao, J. An Experimental Study of Femto-Laser in Assisting Xenograft Acellular Cornea Matrix Lens Transplantation. Experiment 2018, 24, 5208–5215. [Google Scholar] [CrossRef]
- Dragnea, D.C.; Birbal, R.S.; Ham, L.; Dapena, I.; Oellerich, S.; van Dijk, K.; Melles, G.R.J. Bowman layer transplantation in the treatment of keratoconus. Eye Vis. 2018, 5, 24. [Google Scholar] [CrossRef]
- Muijzer, M.B.; Kroes, H.Y.; Hasselt, P.M.; Wisse, R.P.L. Bilateral posterior lamellar corneal transplant surgery in an infant of 17 weeks old: Surgical challenges and the added value of intraoperative optical coherence tomography. Clin. Case Rep. 2022, 10, e05637. [Google Scholar] [CrossRef]
- Jonuscheit, S.; Doughty, M.J.; Ramaesh, K. Assessment of a variable frame (polygonal) method to estimate corneal endothelial cell counts after corneal transplantation. Eye 2012, 26, 803–809. [Google Scholar] [CrossRef]
- Medina, C.A.; Rowe, A.; Yun, H.; Knickelbein, J.E.; Lathrop, K.L.; Hendricks, R.L. Azithromycin treatment increases survival of high-risk corneal allotransplants. Cornea 2013, 32, 658–666. [Google Scholar] [CrossRef]
- Capozzi, P.; Petroni, S.; Buzzonetti, L. Combined HLA matched limbal stem cells allograft with amniotic membrane trans-plantation as a prophylactic surgical procedure to prevent corneal graft rejection after penetrating keratoplasty: Case report. Ann. Ist. Super. Sanita 2014, 50, 298–300. [Google Scholar] [PubMed]
- Zhang, L.; Li, G.; Sessa, R.; Kang, G.J.; Shi, M.; Ge, S.; Gong, A.J.; Wen, Y.; Chintharlapalli, S.; Chen, L. Angiopoietin-2 blockade promotes survival of corneal transplants. Investig. Opthalmol. Vis. Sci. 2017, 58, 79–86. [Google Scholar] [CrossRef]
- Poinard, S.; Garcin, T.; Trone, M.C.; Mentek, M.; Lambert, C.; Bonjean, P.; Renault, D.; Thuret, G.; Gain, P.; Gauthier, A.S. Ob-jective measurement of adherence to topical steroid medication after penetrating keratoplasty using an electronic monitoring aid: A pilot study. Digit Health 2022, 8, 20552076221121155. [Google Scholar] [PubMed]
- Trone, M.C.; Poinard, S.; Crouzet, E.; Garcin, T.; Mentek, M.; Forest, F.; Matray, M.; Thuret, G.; Gain, P. Dropless penetrating keratoplasty using a subconjunctival dexamethasone implant: Safety pilot study. Br. J. Ophthalmol. 2021, 107, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, X.; Li, W.; Jiu, X.; Tian, M. Systematic review and meta-analysis of femtosecond laser–enabled keratoplasty versus conventional penetrating keratoplasty. Eur. J. Ophthalmol. 2020, 31, 976–987. [Google Scholar] [CrossRef] [PubMed]
- Hos, D.; Matthaei, M.; Bock, F.; Maruyama, K.; Notara, M.; Clahsen, T.; Hou, Y.; Le, V.N.H.; Salabarria, A.C.; Horstmann, J.; et al. Immune reactions after modern lamellar (DALK, DSAEK, DMEK) versus conventional pene-trating corneal transplantation. Prog. Retin. Eye. Res. 2019, 73, 100768. [Google Scholar] [CrossRef]
- Heinzelmann, S.; Böhringer, D.; Eberwein, P.; Reinhard, T.; Maier, P. Outcomes of Descemet membrane endothelial keratoplasty, Descemet stripping automated endothelial keratoplasty and penetrating keratoplasty from a single centre study. Graefe’s Arch. Clin. Exp. Ophthalmol. 2016, 254, 515–522. [Google Scholar] [CrossRef]
- Dohlman, C. The boston keratoprosthesis—The first 50 years: Some reminiscences. Annu. Rev. Vis. Sci. 2022, 8, 1–32. [Google Scholar] [CrossRef]
- Avadhanam, V.; Smith, H.; Liu, C. Keratoprostheses for corneal blindness: A review of contemporary devices. Clin. Ophthalmol. 2015, ume 9, 697–720. [Google Scholar] [CrossRef]
- Omari, A.M.; Khan, N.B.; Tausif, H.M.; Nyalakonda, R.B.; Williams, G.A.M.; Gupta, C.M. Temporary Keratoprosthesis with Penetrating Keratoplasty in Conjunction with Pars Plana Vitrectomy for Repair of Retinal Detachments in Patients with Anterior and Posterior Segment Pathology. Retin. Cases Brief Rep. 2021; publish ahead. [Google Scholar] [CrossRef]
- Cisiecki, S.M.; Bonińska, K.M.; Bednarski, M.M. Temporary keratoprosthesis combined with vitrectomy for severe globe injury. Retin. Cases Brief Rep. 2022; publish ahead. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Musa, M.; Zeppieri, M.; Enaholo, E.S.; Chukwuyem, E.; Salati, C. An Overview of Corneal Transplantation in the Past Decade. Clin. Pract. 2023, 13, 264-279. https://doi.org/10.3390/clinpract13010024
Musa M, Zeppieri M, Enaholo ES, Chukwuyem E, Salati C. An Overview of Corneal Transplantation in the Past Decade. Clinics and Practice. 2023; 13(1):264-279. https://doi.org/10.3390/clinpract13010024
Chicago/Turabian StyleMusa, Mutali, Marco Zeppieri, Ehimare S. Enaholo, Ekele Chukwuyem, and Carlo Salati. 2023. "An Overview of Corneal Transplantation in the Past Decade" Clinics and Practice 13, no. 1: 264-279. https://doi.org/10.3390/clinpract13010024
APA StyleMusa, M., Zeppieri, M., Enaholo, E. S., Chukwuyem, E., & Salati, C. (2023). An Overview of Corneal Transplantation in the Past Decade. Clinics and Practice, 13(1), 264-279. https://doi.org/10.3390/clinpract13010024








