Real-Time (iOCT) Guided Epiretinal Membrane Surgery Using a Novel Forceps with Laser-Ablated Microstructure Tip Surface
Abstract
1. Introduction
2. Materials and Methods
2.1. Data and Statistical Analysis
2.2. Intraoperative OCT Analysis
3. Results
3.1. Demographics
3.2. Central Retinal Thickness
3.3. Vision
3.4. Intraoperative Image Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Swanson, E.A.; Lin, C.P.; Schuman, J.S.; Stinson, W.G.; Chang, W.; Hee, M.R.; Flotte, T.; Gregory, K.; Puliafito, C.A.; Fujimoto, J.G. Optical coherence tomography. Science 1991, 254, 1178–1181. [Google Scholar]
- Srinivasan, V.J.; Wojtkowski, M.; Witkin, A.J.; Duker, J.S.; Ko, T.H.; Carvalho, M.; Schuman, J.S.; Kowalczyk, A.; Fujimoto, J.G. High-definition and 3-dimensional imaging of macular pathologies with high-speed ultrahigh-resolution optical coherence tomography. Ophthalmology 2006, 113, 2054–2065.e3. [Google Scholar] [CrossRef] [PubMed]
- Binder, S.; Falkner-Radler, C.I.; Hauger, C.; Matz, H.; Glittenberg, C. Feasibility of intrasurgical spectral-domain optical coherence tomography. Retina 2011, 31, 1332–1336. [Google Scholar] [CrossRef] [PubMed]
- Dayani, P.N.; Maldonado, R.; Farsiu, S.; Toth, C.A. Intraoperative use of handheld spectral domain optical coherence tomography imaging in macular surgery. Retina 2009, 29, 1457–1468. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, J.P.; Kaiser, P.K.; Srivastava, S.K. Intraoperative optical coherence tomography using the RESCAN 700: Preliminary results from the DISCOVER study. Br. J. Ophthalmol. 2014, 98, 1329–1332. [Google Scholar] [CrossRef]
- Ehlers, J.P.; McNutt, S.; Dar, S.; Tao, Y.K.; Srivastava, S.K. Visualisation of contrast-enhanced intraoperative optical coherence tomography with indocyanine green. Br. J. Ophthalmol. 2014, 98, 1588–1591. [Google Scholar] [CrossRef]
- Ehlers, J.P.; McNutt, S.A.; Kaiser, P.K.; Srivastava, S.K. Contrast-enhanced intraoperative optical coherence tomography. Br. J. Ophthalmol. 2013, 97, 1384–1386. [Google Scholar] [CrossRef]
- Ehlers, J.P.; Srivastava, S.K.; Feiler, D.; Noonan, A.I.; Rollins, A.M.; Tao, Y.K. Integrative advances for OCT-guided ophthalmic surgery and intraoperative OCT: Microscope integration, surgical instrumentation, and heads-up display surgeon feedback. PLoS ONE 2014, 9, e105224. [Google Scholar] [CrossRef]
- Ehlers, J.P.; Tao, Y.K.; Farsiu, S.; Maldonado, R.; Izatt, J.A.; Toth, C.A. Integration of a spectral domain optical coherence tomography system into a surgical microscope for intraoperative imaging. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3153–3159. [Google Scholar] [CrossRef]
- Falkner-Radler, C.I.; Glittenberg, C.; Gabriel, M.; Binder, S. Intrasurgical Microscope-Integrated Spectral Domain Optical Coherence Tomography-Assisted Membrane Peeling. Retina 2015, 35, 2100–2106. [Google Scholar] [CrossRef]
- Hahn, P.; Migacz, J.; O’Connell, R.; Day, S.; Lee, A.; Lin, P.; Vann, R.; Kuo, A.; Fekrat, S.; Mruthyunjaya, P.; et al. Preclinical evaluation and intraoperative human retinal imaging with a high-resolution microscope-integrated spectral domain optical coherence tomography device. Retina 2013, 33, 1328–1337. [Google Scholar] [CrossRef] [PubMed]
- Joos, K.M.; Shen, J.H. Miniature real-time intraoperative forward-imaging optical coherence tomography probe. Biomed. Opt. Express 2013, 4, 1342–1350. [Google Scholar] [CrossRef] [PubMed]
- Pichi, F.; Alkabes, M.; Nucci, P.; Ciardella, A.P. Intraoperative SD-OCT in macular surgery. Ophthalmic Surg. Lasers Imaging Retina 2012, 43, S54–S60. [Google Scholar] [CrossRef] [PubMed]
- Ray, R.; Barañano, D.E.; Fortun, J.A.; Schwent, B.J.; Cribbs, B.E.; Bergstrom, C.S.; Hubbard, G.B., III; Srivastava, S.K. Intraoperative microscope-mounted spectral domain optical coherence tomography for evaluation of retinal anatomy during macular surgery. Ophthalmology 2011, 118, 2212–2217. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.K.; Ehlers, J.P.; Toth, C.A.; Izatt, J.A. Intraoperative spectral domain optical coherence tomography for vitreoretinal surgery. Opt. Lett. 2010, 35, 3315–3317. [Google Scholar] [CrossRef]
- Diaz, R.I.; Randolph, J.C.; Sigler, E.J.; Calzada, J.I. Intraoperative grasp site correlation with morphologic changes in retinal nerve fiber layer after internal limiting membrane peeling. Ophthalmic Surg. Lasers Imaging Retina 2014, 45, 45–49. [Google Scholar] [CrossRef]
- Ehlers, J.P.; Han, J.; Petkovsek, D.; Kaiser, P.K.; Singh, R.P.; Srivastava, S.K. Membrane Peeling-Induced Retinal Alterations on Intraoperative OCT in Vitreomacular Interface Disorders From the PIONEER Study. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7324–7330. [Google Scholar] [CrossRef]
- Leung, E.H.; Flynn, H.W., Jr.; Rosenfeld, P.J. Crescent-Shaped Retinal Defects Associated With Membrane Peeling With a Diamond-Dusted Membrane Scraper. Ophthalmic Surg. Lasers Imaging Retina 2016, 47, 90–93. [Google Scholar] [CrossRef]
- Ito, Y.; Terasaki, H.; Takahashi, A.; Yamakoshi, T.; Kondo, M.; Nakamura, M. Dissociated optic nerve fiber layer appearance after internal limiting membrane peeling for idiopathic macular holes. Ophthalmology 2005, 112, 1415–1420. [Google Scholar] [CrossRef]
- Tadayoni, R.; Paques, M.; Massin, P.; Mouki-Benani, S.; Mikol, J.; Gaudric, A. Dissociated optic nerve fiber layer appearance of the fundus after idiopathic epiretinal membrane removal. Ophthalmology 2001, 108, 2279–2283. [Google Scholar] [CrossRef]
- Uchida, A.; Srivastava, S.K.; Ehlers, J.P. Analysis of Retinal Architectural Changes Using Intraoperative OCT Following Surgical Manipulations With Membrane Flex Loop in the DISCOVER Study. Investig. Ophthalmol. Vis. Sci. 2017, 58, 3440–3444. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, J.P.; Itoh, Y.; Xu, L.T.; Kaiser, P.K.; Singh, R.P.; Srivastava, S.K. Factors associated with persistent subfoveal fluid and complete macular hole closure in the PIONEER study. Investig. Ophthalmol. Vis. Sci. 2014, 56, 1141–1146. [Google Scholar] [CrossRef] [PubMed]
- Steel, D.H.; Dinah, C.; Habib, M.; White, K. ILM peeling technique influences the degree of a dissociated optic nerve fibre layer appearance after macular hole surgery. Graefes Arch. Clin. Exp. Ophthalmol. 2015, 253, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Karacorlu, M.; Karacorlu, S.; Ozdemir, H. Iatrogenic punctate chorioretinopathy after internal limiting membrane peeling. Am. J. Ophthalmol. 2003, 135, 178–182. [Google Scholar] [CrossRef]
- Kwok, A.K.; Lai, T.Y.; Man-Chan, W.; Woo, D.C. Indocyanine green assisted retinal internal limiting membrane removal in stage 3 or 4 macular hole surgery. Br. J. Ophthalmol. 2003, 87, 71–74. [Google Scholar] [CrossRef]
- Pavlidis, M.; Georgalas, I.; Körber, N. Determination of a New Parameter, Elevated Epiretinal Membrane, by En Face OCT as a Prognostic Factor for Pars Plana Vitrectomy and Safer Epiretinal Membrane Peeling. J. Ophthalmol. 2015, 2015, 838646. [Google Scholar] [CrossRef]
- Chatziralli, I.P.; Theodossiadis, P.G.; Steel, D.H.W. Internal Limiting Membrane Peeling in Macular Hole Surgery; Why, When, and How? Retina 2018, 38, 870–882. [Google Scholar] [CrossRef]

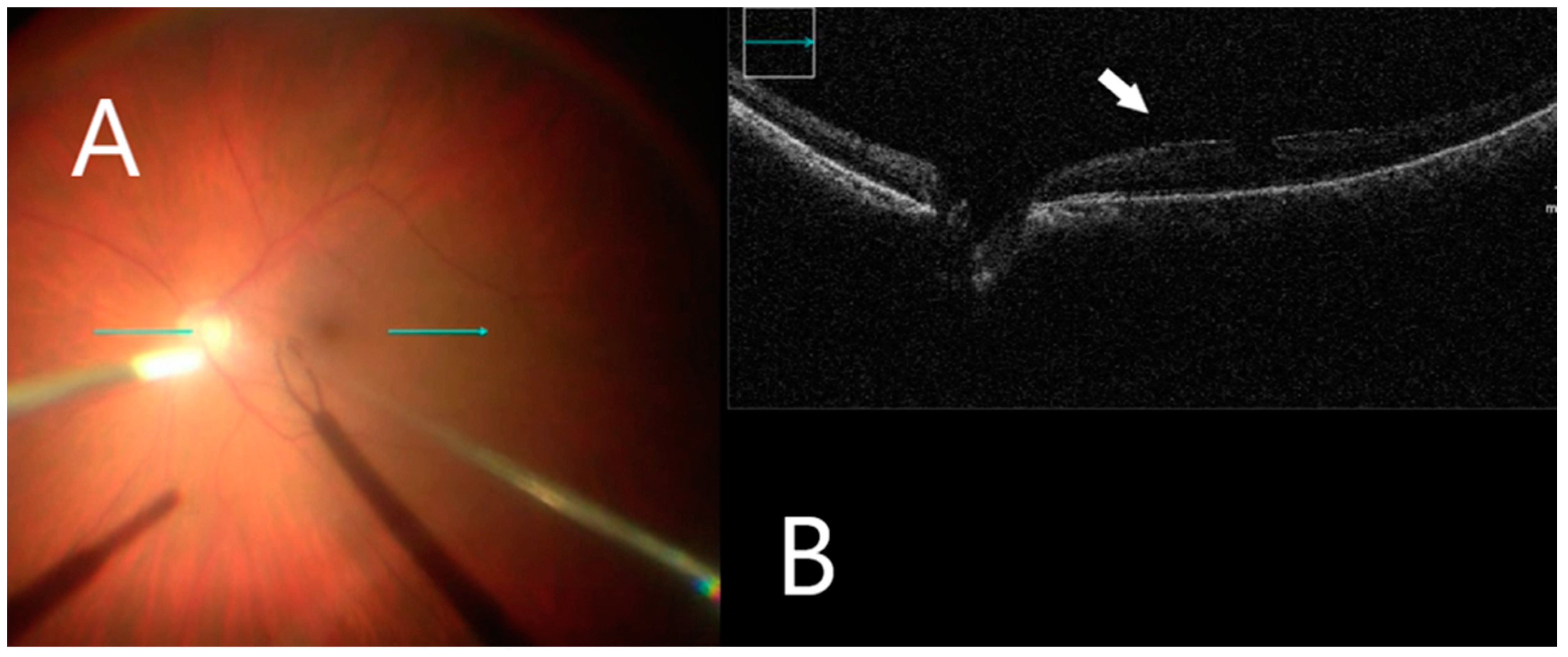
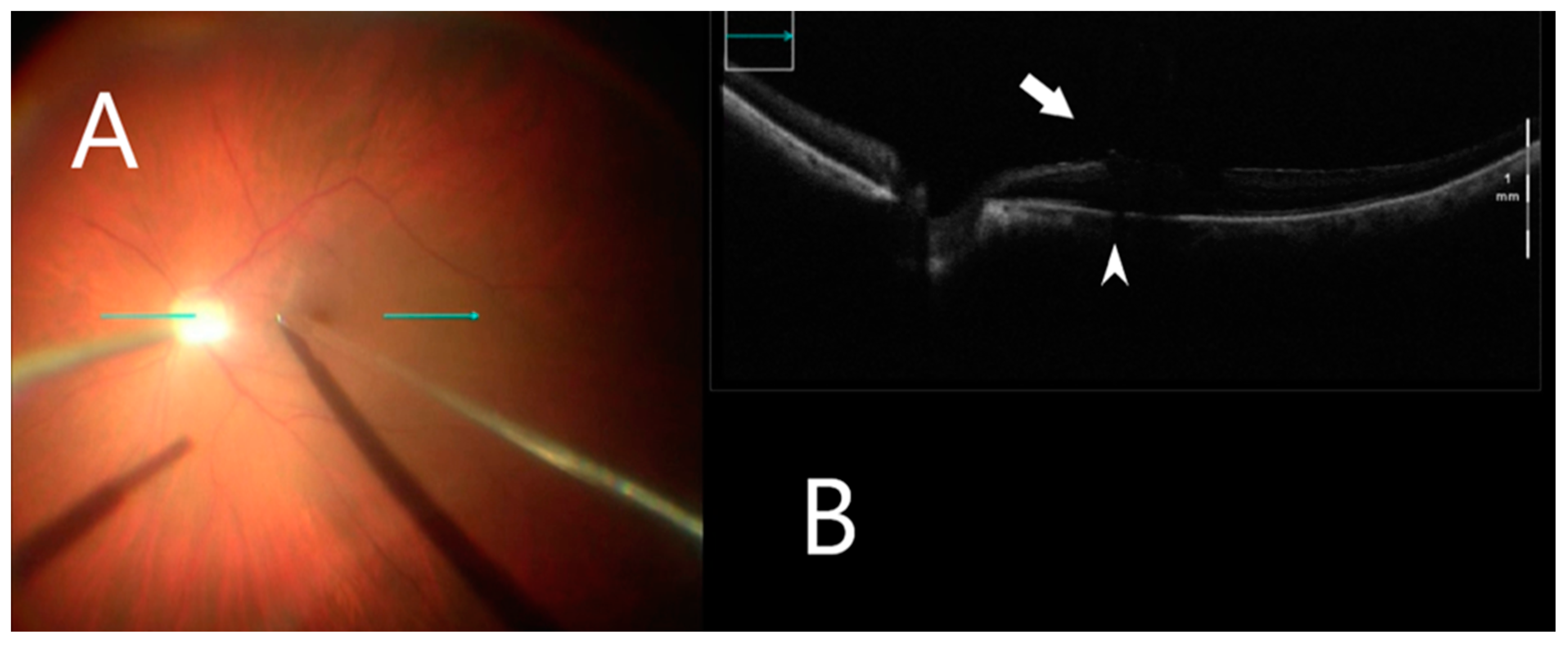
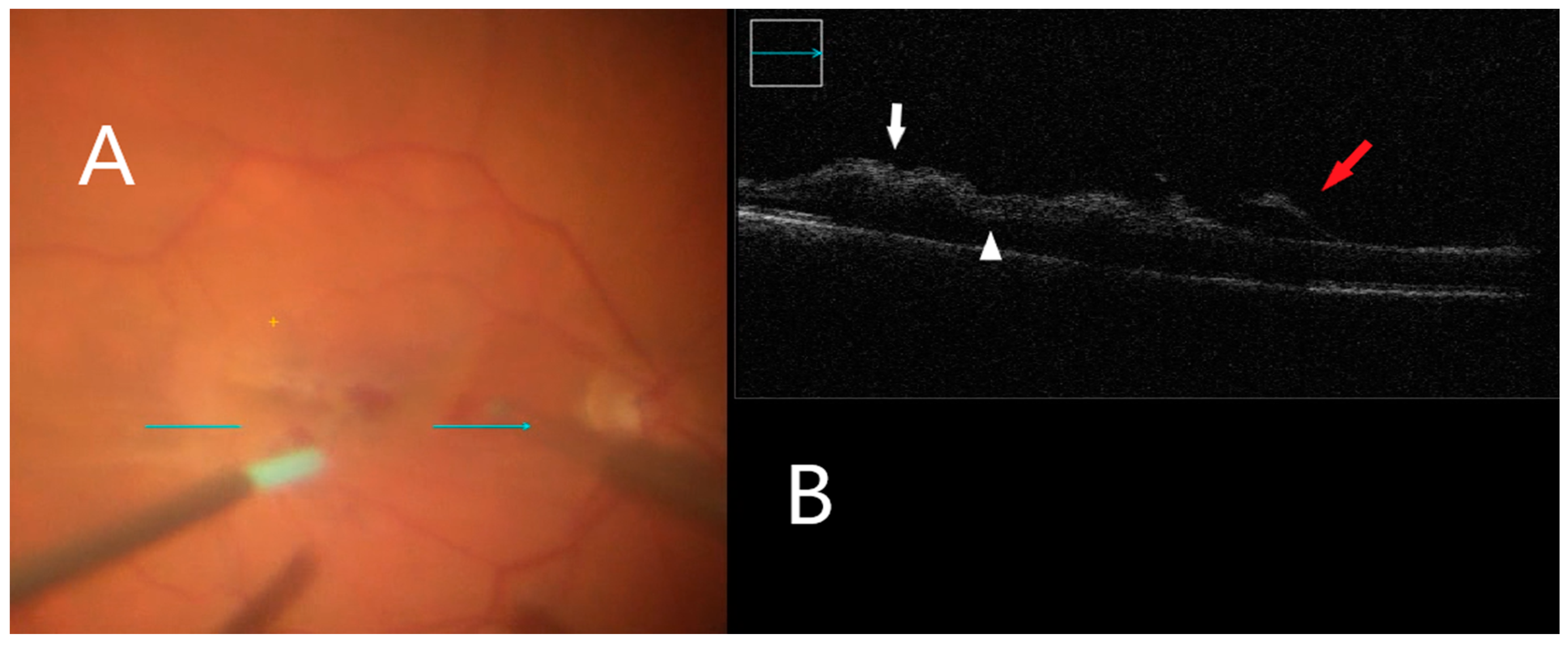
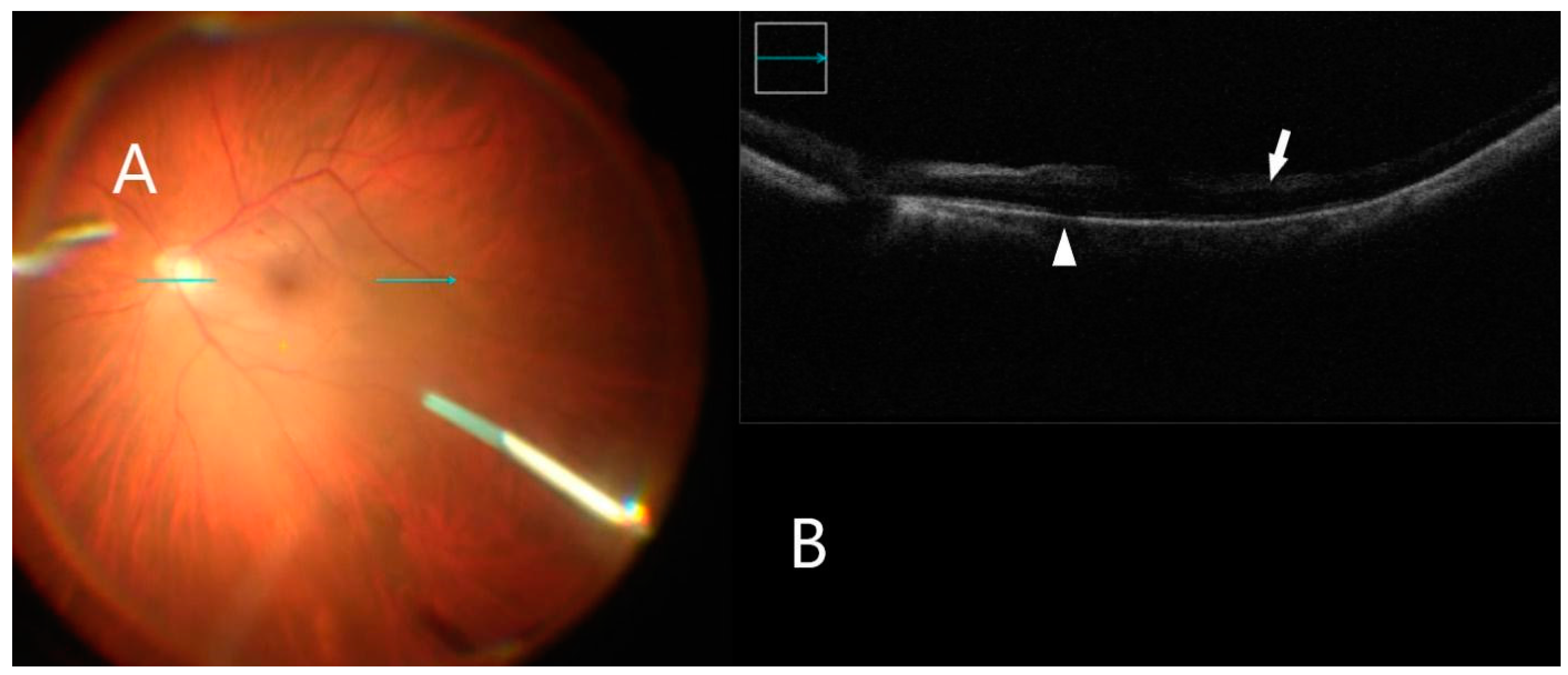
| Diagnosis Group | ERM, n = 11 | FTMH, n = 7 | VMT, n = 2 | ERM after RD, n = 3 | All, n = 23 |
|---|---|---|---|---|---|
| Sex, n | |||||
| Men | 6 (55%) | 4 (57%) | 1 (50%) | 3 (100%) | 14 (61%) |
| Women | 5 (45%) | 3 (43%) | 1 (50%) | 0 | 9 (39%) |
| Age, years | 71 | 74 | 80 | 73 | 75 |
| Laterality of the eye, n | |||||
| Right | 7 (64%) | 4 (57%) | 1 (50%) | 2 (67%) | 14 (61%) |
| Left | 4 (36%) | 3 (43%) | 1 (50%) | 1 (33%) | 9 (39%) |
| Lens status, n | |||||
| Phakic | 0 | 0 | 0 | 0 | 0 |
| Pseudophakic | 11 (100%) | 7 (100%) | 2 (100%) | 3 (100%) | 23 (100%) |
| Diagnosis Group | ERM, n = 11 | FTMH, n = 7 | VMT, n = 2 | ERM after RD, n = 3 | All, n = 23 |
|---|---|---|---|---|---|
| BRVA logMAR, mean ± SD before Peeling | 0.52 ± 0.23 | 0.69 ± 0.4 | 0.3 ± 0.14 | 0.8 ± 0.26 | 0.5 ± 0.34 |
| BRVA logMAR, mean ± SD after Peeling | 0.27 ± 0.14 | 0.43 ± 0.56 | 0.6 ± 0.57 | 0.13 ± 0.15 | 0.33 ± 0.36 |
| Subretinal hyporeflectance | 2 (18%) | 0 | 1 (50%) | 1 (33%) | 4 (17%) |
| Retinal break | 0 | 0 | 0 | 0 | 0 |
| Increased inner retinal hyper-reflectivity associated with retinal hemorrhage | 2 (18%) | 1 (14%) | 1 (50%) | 1 (33%) | 5 (22%) |
| Increased inner retinal hyper-reflectivity not associated with retinal hemorrhage | 6 (55%) | 5 (71%) | 1 (50%) | 2 (66%) | 14 (61%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashurov, A.; Chronopoulos, A.; Heim, J.; Schutz, J.S.; Arndt, C.; Hattenbach, L.-O. Real-Time (iOCT) Guided Epiretinal Membrane Surgery Using a Novel Forceps with Laser-Ablated Microstructure Tip Surface. Clin. Pract. 2022, 12, 818-825. https://doi.org/10.3390/clinpract12050086
Ashurov A, Chronopoulos A, Heim J, Schutz JS, Arndt C, Hattenbach L-O. Real-Time (iOCT) Guided Epiretinal Membrane Surgery Using a Novel Forceps with Laser-Ablated Microstructure Tip Surface. Clinics and Practice. 2022; 12(5):818-825. https://doi.org/10.3390/clinpract12050086
Chicago/Turabian StyleAshurov, Agharza, Argyrios Chronopoulos, Julia Heim, James Scott Schutz, Carl Arndt, and Lars-Olof Hattenbach. 2022. "Real-Time (iOCT) Guided Epiretinal Membrane Surgery Using a Novel Forceps with Laser-Ablated Microstructure Tip Surface" Clinics and Practice 12, no. 5: 818-825. https://doi.org/10.3390/clinpract12050086
APA StyleAshurov, A., Chronopoulos, A., Heim, J., Schutz, J. S., Arndt, C., & Hattenbach, L.-O. (2022). Real-Time (iOCT) Guided Epiretinal Membrane Surgery Using a Novel Forceps with Laser-Ablated Microstructure Tip Surface. Clinics and Practice, 12(5), 818-825. https://doi.org/10.3390/clinpract12050086





