PD-L1 Over-Expression Varies in Different Subtypes of Lung Cancer: Will This Affect Future Therapies?
Abstract
1. Introduction
Study Aims
- To assess the PD-L1 expression level in NSCLC, KRAS mutant adenocarcinoma, adenocarcinoma, and squamous cell carcinoma.
- To assess the PD-L1 expression level in SCLC and large cell neuroendocrine cancer (LCNEC).
- Role of PD-L1 positive and PD-L1 negative and effect on treatment.
2. Methods
2.1. Search Strategy
2.2. Exclusion Criteria
2.3. Data Review
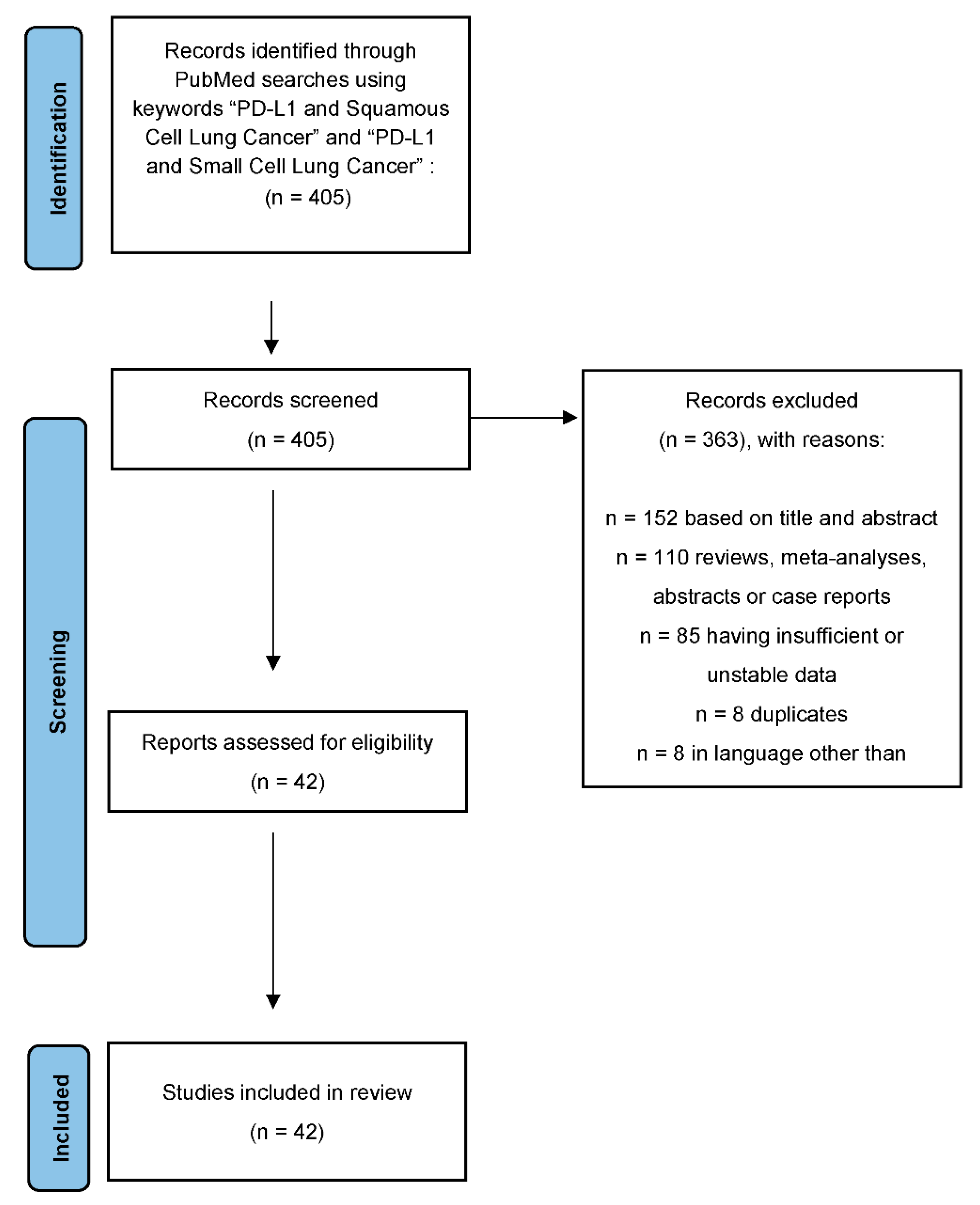
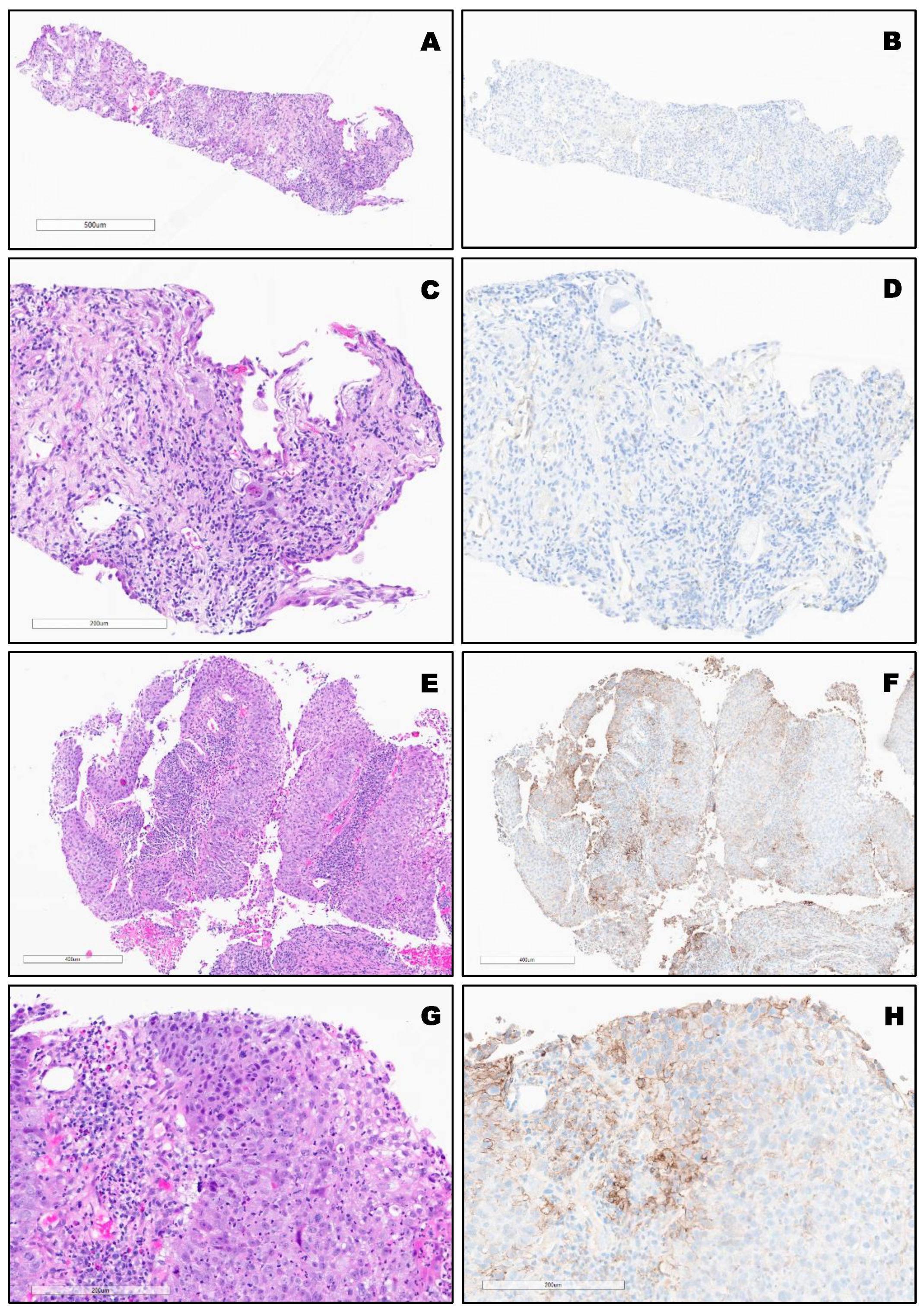
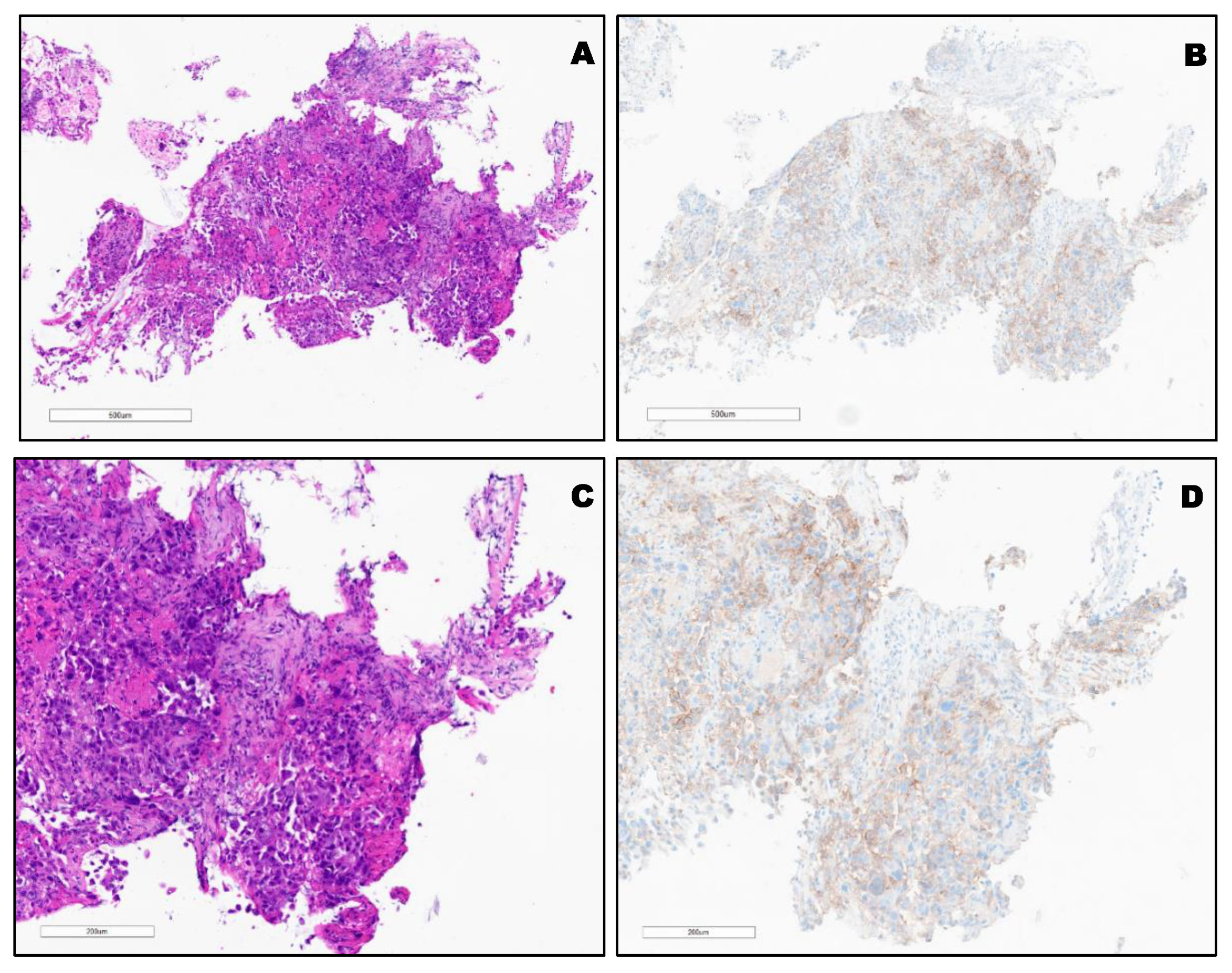
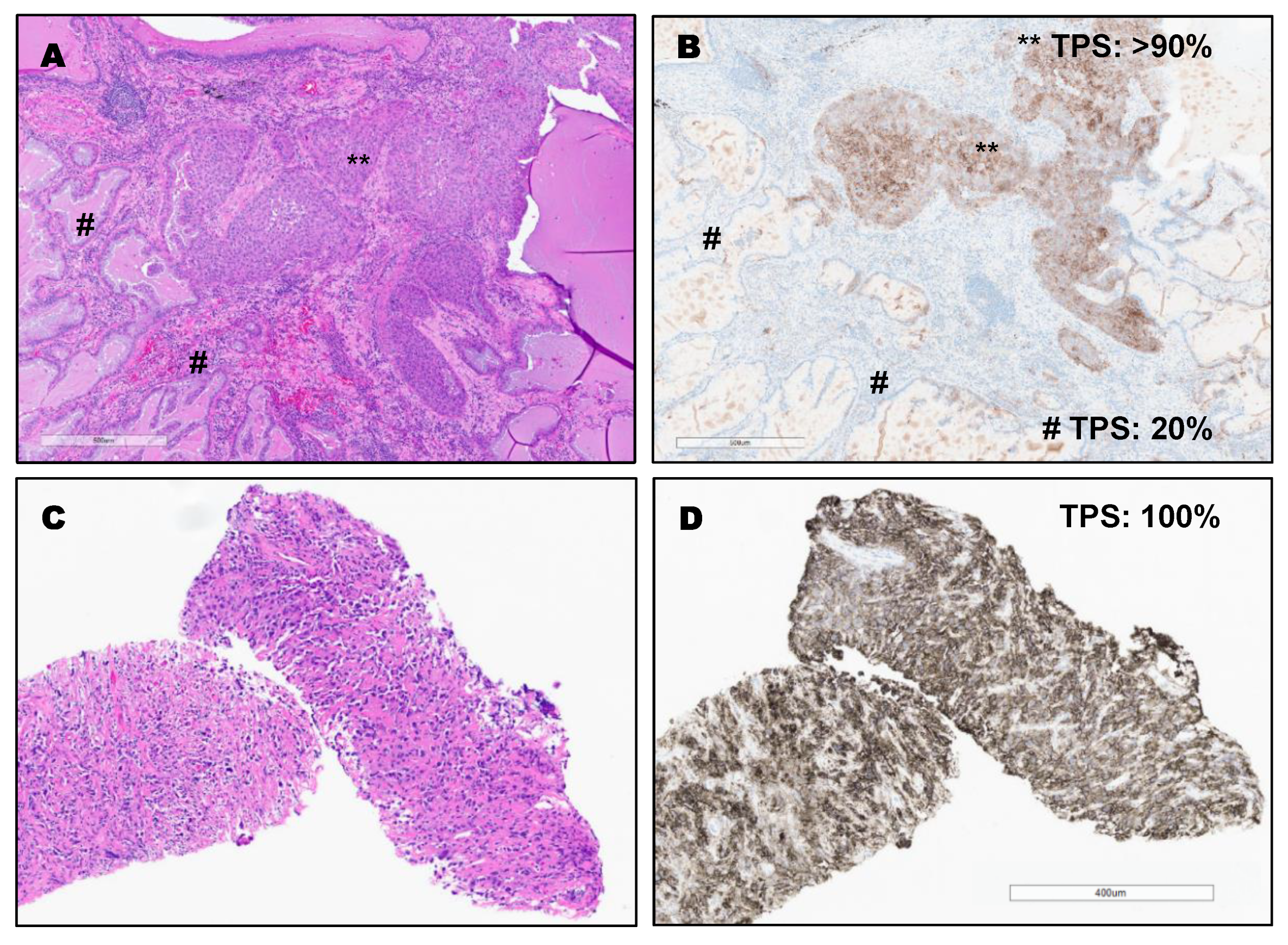
3. Results
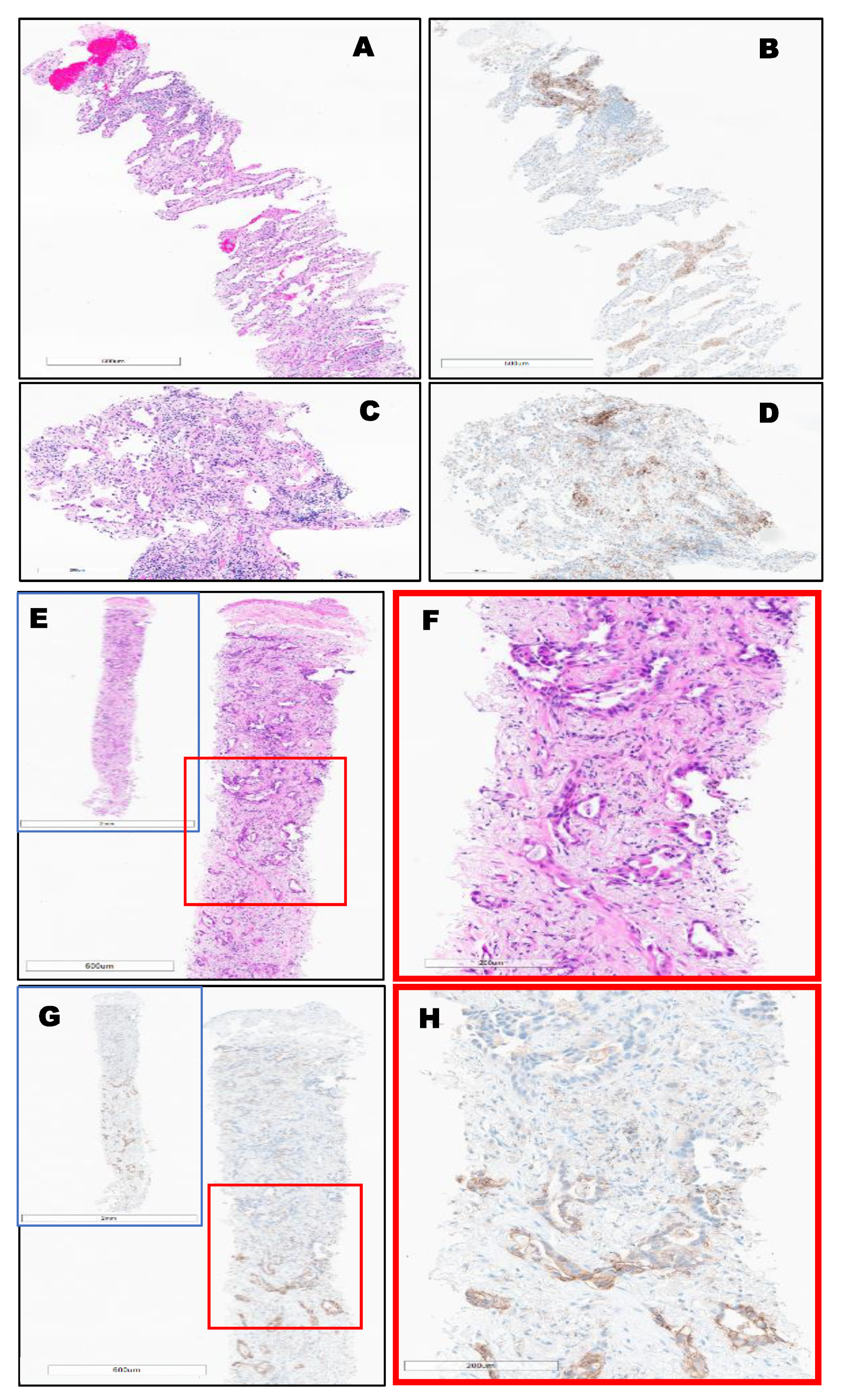
PD-L1 Clones and Hematoxylin and Eosin (H&E) Staining of Different Types of Lung Cancer
4. Discussion
4.1. Squamous Cell and Adenocarcinoma
4.2. KRAS Mutant Adenocarcinoma
4.3. Adenosquamous Carcinoma
4.4. Large Cell Carcinoma
4.5. Small Cell Carcinoma
4.6. Sarcomatoid Carcinoma
4.7. Large Cell Neuroendocrine Carcinoma
4.8. Biomarkers Associated with Immune Checkpoint Inhibitors (ICI) Sensitivity and Resistance
4.9. Ongoing Clinical Trials
4.10. Limitations
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Mcdermott, D.F.; Atkins, M.B. PD-1 as a potential target in cancer therapy. Cancer Med. 2013, 2, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Agata, Y.; Shibahara, K.; Honjo, T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 1992, 11, 3887–3895. [Google Scholar] [CrossRef] [PubMed]
- Agata, Y.; Kawasaki, A.; Nishimura, H.; Ishida, Y.; Tsubata, T.; Yagita, H.; Honjo, T. Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes. Int. Immunol. 1996, 8, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Ohaegbulam, K.C.; Assal, A.; Lazar-Molnar, E.; Yao, Y.; Zang, X. Human cancer immunotherapy with antibodies to the PD-1 and PD-L1 pathway. Trends Mol. Med. 2015, 21, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Ghiotto, M.; Gauthier, L.; Serriari, N.; Pastor, S.; Truneh, A.; Nunès, J.A.; Olive, D. PD-L1 and PD-L2 differ in their molecular mechanisms of interaction with PD-1. Int. Immunol. 2010, 22, 651–660. [Google Scholar] [CrossRef]
- Brahmer, J.R.; Drake, C.G.; Wollner, I.; Powderly, J.D.; Picus, J.; Sharfman, W.H.; Topalian, S.L. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: Safety, clinical activity, pharmacodynamics, and immunologic correlates. J. Clin. Oncol. 2010, 28, 3167–3175. [Google Scholar] [CrossRef]
- Zheng, P.; Zhou, Z. Human cancer immunotherapy with PD-1/PD-L1 blockade. Biomark. Cancer 2015, 7 (Suppl. S2), BIC.S29325. [Google Scholar] [CrossRef]
- Lesterhuis, W.J.; Steer, H.; Lake, R.A. PD-L2 is predominantly expressed by Th2 cells. Mol. Immunol. 2011, 49, 1–3. [Google Scholar] [CrossRef]
- Youngnak, P.; Kozono, Y.; Kozono, H.; Iwai, H.; Otsuki, N.; Jin, H.; Omura, K.; Yagita, H.; Pardoll, D.M.; Chen, L.; et al. Differential binding properties of B7-H1 and B7-DC to programmed death-1. Biochem. Biophys. Res. Commun. 2003, 307, 672–677. [Google Scholar] [CrossRef]
- Fife, B.T.; Pauken, K.E.; Eagar, T.N.; Obu, T.; Wu, J.; Tang, Q.; Azuma, M.; Krummel, M.F.; Bluestone, J.A. Interactions between PD-1 and PD-L1 promote tolerance by blocking the TCR-induced stop signal. Nat. Immunol. 2009, 10, 1185–1192. [Google Scholar] [CrossRef]
- Keir, M.E.; Liang, S.C.; Guleria, I.; Latchman, Y.E.; Qipo, A.; Albacker, L.A.; Koulmanda, M.; Freeman, G.J.; Sayegh, M.H.; Sharpe, A.H. Tissue expression of PD-L1 mediates peripheral T cell tolerance. J. Exp. Med. 2006, 203, 883–895. [Google Scholar] [CrossRef] [PubMed]
- Latchman, Y.E.; Liang, S.C.; Wu, Y.; Chernova, T.; Sobel, R.A.; Klemm, M.; Kuchroo, V.K.; Freeman, G.; Sharpe, A.H. PD-L1-deficient mice show that PD-L1 on T cells, antigen-presenting cells, and host tissues negatively regulates T cells. Proc. Natl. Acad. Sci. USA 2004, 101, 10691–10696. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, T.; Akiba, H.; Iwai, H.; Matsuda, H.; Aoki, M.; Tanno, Y.; Shin, T.; Tsuchiya, H.; Pardoll, D.M.; Okumura, K.; et al. Expression of programmed death 1 ligands by murine T cells and APC. J. Immunol. 2002, 169, 5538–5545. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Chen, S.; Yang, L.; Li, Y. The role of PD-1 and PD-L1 in T-cell immune suppression in patients with hematological malignancies. J. Hematol. Oncol. 2013, 6, 74. [Google Scholar] [CrossRef]
- Chapon, M.; Randriamampita, C.; Maubec, E.; Badoual, C.; Fouquet, S.; Wang, S.-F.; Marinho, E.; Farhi, D.; Garcette, M.; Jacobelli, S.; et al. Progressive upregulation of PD-1 in primary and metastatic melanomas associated with Blunted TCR signaling in infiltrating T lymphocytes. J. Investig. Dermatol. 2011, 131, 1300–1307. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, S.; Gong, D.; Qin, Y.; Shen, Q. Programmed death-1 upregulation is correlated with dysfunction of tumor-infiltrating CD8+ T lymphocytes in human non-small cell lung cancer. Cell. Mol. Immunol. 2010, 7, 389–395. [Google Scholar] [CrossRef]
- Muenst, S.; Soysal, S.D.; Gao, F.; Obermann, E.C.; Oertli, D.; Gillanders, W.E. The presence of programmed death 1 (PD-1)-positive tumor-infiltrating lymphocytes is associated with poor prognosis in human breast cancer. Breast Cancer Res. Treat. 2013, 139, 667–676. [Google Scholar] [CrossRef]
- Wei, F.; Zhong, S.; Ma, Z.; Kong, H.; Medvec, A.; Ahmed, R.; Freeman, G.J.; Krogsgaard, M.; Riley, J.L. Strength of PD-1 signaling differentially affects T-cell effector functions. Proc. Natl. Acad. Sci. USA 2013, 110, E2480–E2489. [Google Scholar] [CrossRef]
- Yamamoto, R.; Nishikori, M.; Kitawaki, T.; Sakai, T.; Hishizawa, M.; Tashima, M.; Kondo, T.; Ohmori, K.; Kurata, M.; Hayashi, T.; et al. PD-1 PD-1 ligand interaction contributes to immunosuppressive microenvironment of Hodgkin lymphoma. Blood 2008, 111, 3220–3224. [Google Scholar] [CrossRef]
- Ameratunga, M.; Asadi, K.; Lin, X.; Walkiewicz, M.; Murone, C.; Knight, S.; Mitchell, P.; Boutros, P.; John, T. PD-L1 and tumor infiltrating lymphocytes as prognostic markers in resected NSCLC. PLoS ONE 2016, 11, e0153954. [Google Scholar] [CrossRef]
- Boland, J.M.; Kwon, E.D.; Harrington, S.M.; Wampfler, J.A.; Tang, H.; Yang, P.; Aubry, M.C. Tumor B7-H1 and B7-H3 expression in squamous cell carcinoma of the lung. Clin. Lung Cancer 2013, 14, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, D.; Clavé, S.; Taus, Á.; Hardy-Werbin, M.; Rocha, P.; Lorenzo, M.; Menéndez, S.; Salido, M.; Albanell, J.; Pijuan, L.; et al. Heterogeneity of tumor and immune cell PD-L1 expression and lymphocyte counts in surgical NSCLC samples. Clin. Lung Cancer 2017, 18, 682–691.e5. [Google Scholar] [CrossRef] [PubMed]
- Abdel Karim, N.; Sendilnathan, A.; Eldessouki, I.; Orr-Asman, M.; Xie, C.; Wang, J.; Elnakat, H. Immune checkpoint markers in lung large cell neuroendocrine carcinomas (L-LCNEC). J. Thorac. Oncol. 2017, 12, S1583–S1584. [Google Scholar] [CrossRef]
- D’Incecco, A.; Andreozzi, M.; Ludovini, V.; Rossi, E.; Capodanno, A.; Landi, L.; Tibaldi, C.; Minuti, G.; Salvini, J.; Coppi, E.; et al. PD-1 and PD-L1 expression in molecularly selected non-small-cell lung cancer patients. Br. J. Cancer 2015, 112, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Dolled-Filhart, M.; Locke, D.; Murphy, T.; Lynch, F.; Yearley, J.H.; Frisman, D.; Pierce, R.; Weiner, R.; Wu, D.; Emancipator, K. Development of a prototype immunohistochemistry assay to measure programmed death ligand-1 expression in tumor tissue. Arch. Pathol. Lab. Med. 2016, 140, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, J.; Stinchcombe, T.E. Treatment of KRAS-mutant non–small cell lung cancer. JAMA 2017, 317, 1835. [Google Scholar] [CrossRef] [PubMed]
- Silva AP, S.; Coelho, P.V.; Anazetti, M.; Simioni, P.U. Targeted therapies for the treatment of non-small-cell lung cancer: Monoclonal antibodies and biological inhibitors. Hum. Vaccines Immunother. 2017, 13, 843–853. [Google Scholar] [CrossRef]
- Karim, N.; Bahassi, E.-M.; Gaber, O.; Sadraei, N.H.; Morris, J. P2.03a-027 a phase I study of the non-receptor tyrsine kinase inhibitor (NKI) bosutinib in combination with pemetrexed in patients with advanced solid tumors. J. Thorac. Oncol. 2017, 12, S904. [Google Scholar] [CrossRef]
- Karim, N.A.; Schuster, J.; Eldessouki, I.; Gaber, O.; Namad, T.; Wang, J.; Xie, C.; Morris, J.C. Pulmonary sarcomatoid carcinoma: University of cincinnati experience. Oncotarget 2018, 9, 4102. [Google Scholar] [CrossRef]
- Karim, N.; Eldessouki, I.; Yellu, M.; Namad, T.; Wang, J.; Gaber, O. A case study in advanced lung cancer patients with vimentin over expression. Clin. Lab. 2017, 63, 1575–1579. [Google Scholar] [CrossRef]
- Hassan, R.; Gulati, S.; Mahender, Y.; Eldessouki, I.; Siddiqi, N.I.; Xie, C.; Pruemer, J.; Karim, N.A. Impact of low molecular weight heparin on overall survival in patients with advanced lung cancer: A retrospective study. Am. J. Clin. Exp. Med. 2017, 5, 173–175. [Google Scholar] [CrossRef][Green Version]
- Scapin, G.; Yang, X.; Prosise, W.W.; McCoy, M.; Reichert, P.; Johnston, J.M.; Kashi, R.S.; Strickland, C. Structure of full-length human anti-PD1 therapeutic IgG4 antibody pembrolizumab. Nat. Struct. Mol. Biol. 2015, 22, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Thudium, K.B.; Han, M.; Wang, X.-T.; Huang, H.; Feingersh, D.; Garcia, C.; Wu, Y.; Kuhne, M.; Srinivasan, M.; et al. In Vitro characterization of the anti-PD-1 antibody nivolumab, BMS-936558, and in vivo toxicology in non-human primates. Cancer Immunol. Res. 2014, 2, 846–856. [Google Scholar] [CrossRef] [PubMed]
- Boyerinas, B.; Jochems, C.; Fantini, M.; Heery, C.R.; Gulley, J.L.; Tsang, K.Y.; Schlom, J. Antibody-dependent cellular cytotoxicity activity of a novel anti-PD-L1 antibody avelumab (MSB0010718C) on human tumor cells. Cancer Immunol. Res. 2015, 3, 1148–1157. [Google Scholar] [CrossRef]
- Garon, E.B.; Rizvi, N.A.; Hui, R.; Leighl, N.; Balmanoukian, A.S.; Eder, J.P.; Patnaik, A.; Aggarwal, C.; Gubens, M.; Horn, L.; et al. Pembrolizumab for the treatment of non–small-cell lung cancer. N. Engl. J. Med. 2015, 372, 2018–2028. [Google Scholar] [CrossRef]
- Horn, L.; Brahmer, J.; Reck, M.; Borghaei, H.; Spigel, D.; Steins, M.; Ready, N.; Chow, L.Q.; Vokes, E.E.; Felip, E.; et al. Phase III, randomized trial (CheckMate 057) of nivolumab (NIVO) versus docetaxel (DOC) in advanced non-squamous cell (non-SQ) non-small cell lung cancer (NSCLC). Eur. J. Cancer 2015, 51 (Suppl. S3), abstr 3010. [Google Scholar]
- Rizvi, N.A.; Brahmer, J.R.; Ou, S.H.I.; Segal, N.H.; Khleif, S.; Hwu, W.; Gutierrez, M.; Schoffski, P.; Hamid, O.; Weiss, J.; et al. Safety and clinical activity of MEDI4736, an anti-programmed cell death-ligand 1 (PD-L1) antibody, in patients with non-small cell lung cancer (NSCLC). J. Clin. Oncol. 2015, 33, 8032. [Google Scholar] [CrossRef]
- Fehrenbacher, L.; Spira, A.; Ballinger, M.; Kowanetz, M.; Vansteenkiste, J.; Mazieres, J.; Park, K.; Smith, D.; Artal-Cortes, A.; Lewanski, C.; et al. Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): A multicentre, open-label, phase 2 randomised controlled trial. Lancet 2016, 387, 1837–1846. [Google Scholar] [CrossRef]
- Spira, A.I.; Park, K.; Mazières, J.; Vansteenkiste, J.F.; Rittmeyer, A.; Ballinger, M.; Waterkamp, D.; Kowanetz, M.; Mokatrin, A.; Fehrenbacher, L. Efficacy, safety and predictive biomarker results from a randomized phase II study comparing MPDL3280A vs docetaxel in 2L/3L NSCLC (POPLAR). J. Clin. Oncol. 2015, 33, 8010. [Google Scholar] [CrossRef]
- Chatterjee, M. Systematic evaluation of pembrolizumab dosing in patients with advanced non-small-cell lung cancer. Ann. Oncol. 2016, 27, 1291–1298. Available online: https://www.medscape.com/medline/abstract/27117531 (accessed on 1 June 2022). [CrossRef]
- Phase II Study of Maintenance Pembrolizumab in Small Cell Lung Cancer. Available online: http://www.oncologytube.com/video/phase-ii-study-of-maintenance-pembrolizumab-in-small-cell-lung-cancer/10001673 (accessed on 15 April 2018).
- Pawelczyk, K.; Piotrowska, A.; Ciesielska, U.; Jablonska, K.; Glatzel-Plucinska, N.; Grzegrzolka, J.; Podhorska-Okolow, M.; Dziegiel, P.; Nowinska, K. Role of PD-L1 expression in non-small cell lung cancer and their prognostic significance according to clinicopathological factors and diagnostic markers. Int. J. Mol. Sci. 2019, 20, 824. [Google Scholar] [CrossRef] [PubMed]
- Gadgeel, S.M.; Ventimiglia, J.; Kalemkerian, G.P.; Fidler, M.J.; Chen, W.; Sukari, A.; Halmos, B.; Boerner, J.; Wozniak, A.J.; Galasso, C.; et al. Phase II study of maintenance pembrolizumab (pembro) in extensive stage small cell lung cancer (ES-SCLC) patients (pts). J. Clin. Oncol. 2017, 35, 8504. [Google Scholar] [CrossRef]
- Shi, X.; Wu, S.; Sun, J.; Liu, Y.; Zeng, X.; Liang, Z. PD-L1 expression in lung adenosquamous carcinomas compared with the more common variants of non-small cell lung cancer. Sci. Rep. 2017, 7, 46209. [Google Scholar] [CrossRef] [PubMed]
- Co, M. KEYTRUDA (Pembrolizumab) Prescribing Information. 2014. Available online: https://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf (accessed on 1 June 2022).
- Wu, Y.; Chen, W.; Xu, Z.P.; Gu, W. PD-L1 distribution and perspective for cancer immunotherapy-blockade, knockdown, or inhibition. Front. Immunol. 2019, 10, 2022. [Google Scholar] [CrossRef] [PubMed]
- McDermott, D.F.; Sosman, J.A.; Sznol, M.; Massard, C.; Gordon, M.S.; Hamid, O.; Powderly, J.D.; Infante, J.R.; Fassò, M.; Wang, Y.V.; et al. Atezolizumab, an anti–programmed death-ligand 1 antibody, in metastatic renal cell carcinoma: Long-term safety, clinical activity, and immune correlates from a phase la study. J. Clin. Oncol. 2016, 34, 833–842. [Google Scholar] [CrossRef]
- Rittmeyer, A.; Barlesi, F.; Waterkamp, D.; Park, K.; Ciardiello, F.; von Pawel, J.; Gadgeel, S.M.; Hida, T.; Kowalski, D.M.; Dols, M.C.; et al. Atezolizumab versus docetaxel in patients with previously treated nonsmall-cell lung cancer (OAK): A phase 3, open-label, multicentre randomized controlled trial. Lancet 2017, 389, 255–265. [Google Scholar] [CrossRef]
- Gandini, S.; Massi, D.; Mandala, M. PD-L1 expression in cancer patients receiving anti PD-1/PD-L1 antibodies: A systematic review and meta-analysis. Crit. Rev. Oncol. Hemat. 2016, 100, 88–98. [Google Scholar] [CrossRef]
- Kintsler, S.; Cassataro, M.A.; Drosch, M.; Holenya, P.; Knuechel, R.; Braunschweig, T. Expression of programmed death ligand (PD-L1) in different tumors. Comparison of several current available antibody clones and antibody profiling. Ann. Diagn. Pathol. 2019, 41, 24–37. [Google Scholar] [CrossRef]
- Brunnström, H.; Johansson, A.; Westbom-Fremer, S.; Backman, M.; Djureinovic, D.; Patthey, A.; Isaksson-Mettävainio, M.; Gulyas, M.; Micke, P. PD-L1 immunohistochemistry in clinical diagnostics of lung cancer: Inter-pathologist variability is higher than assay variability. Mod. Pathol. 2017, 30, 1411–1421. [Google Scholar] [CrossRef]
- Fang, W.; Hong, S.; Chen, N.; He, X.; Zhan, J.; Qin, T.; Zhou, T.; Hu, Z.; Ma, Y.; Zhao, Y.; et al. PD-L1 is remarkably over-expressed in EBV-associated pulmonary lymphoepithelioma-like carcinoma and related to poor disease-free survival. Oncotarget 2015, 6, 33019–33032. [Google Scholar] [CrossRef]
- Kerr, E.M.; Martins, C.P. Metabolic rewiring in mutant Kras lung cancer. FEBS J. 2018, 285, 28–41. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodriguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csoszi, T.; Fulop, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N. Engl. J. Med. 2016, 375, 1823–1833. [Google Scholar] [CrossRef] [PubMed]
- Falk, A.T.; Yazbeck, N.; Guibert, N.; Chamorey, E.; Paquet, A.; Ribeyre, L.; Bence, C.; Zahaf, K.; Leroy, S.; Marquette, C.; et al. Effect of mutant variants of the KRAS gene on PD-L1 expression and on the immune microenvironment and association with clinical outcome in lung adenocarcinoma patients. Lung Cancer 2018, 121, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.H.; Melloni, G.E.M.; Gulhan, D.C.; Park, P.J.; Haigis, K.M. The origins and genetic interactions of KRAS mutations are allele- and tissue-specific. Nat. Commun. 2021, 12, 1808. [Google Scholar] [CrossRef] [PubMed]
- Nottegar, A.; Tabbò, F.; Luchini, C.; Brunelli, M.; Bria, E.; Veronese, N.; Santo, A.; Cingarlini, S.; Gilioli, E.; Ogliosi, C.; et al. Pulmonary adenocarcinoma with enteric differentiation: Immunohistochemistry and molecular morphology. Appl. Immunohistochem. Mol. Morphol. 2018, 26, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zheng, S.; Jin, R.; Wang, X.; Wang, F.; Zang, R.; Xu, H.; Lu, Z.; Huang, J.; Lei, Y.; et al. The superior efficacy of anti-PD-1/PD-L1 immunotherapy in KRAS-mutant non-small cell lung cancer that correlates with an inflammatory phenotype and increased immunogenicity. Cancer Lett. 2020, 470, 95–105. [Google Scholar] [CrossRef]
- Uramoto, H.; Yamada, S.; Hanagiri, T. Clinicopathological characteristics of resected adenosquamous cell carcinoma of the lung: Risk of coexistent double cancer. J. Cardiothorac. Surg. 2010, 5, 92. [Google Scholar] [CrossRef]
- Hsia, J.Y.; Chen, C.Y.; Hsu, C.P.; Shai, S.E.; Wang, P.Y. Adenosquamous carcinoma of the lung. surgical results compared with squamous cell and adenocarcinoma. Scand. Cardiovasc. J. 1999, 33, 29–32. [Google Scholar]
- Song, Z.; Lin, B.; Shao, L.; Zhang, Y. Therapeutic efficacy of gefitinib and erlotinib in patients with advanced lung adenosquamous carcinoma. J. Chin. Med. Assoc. 2013, 76, 481–485. [Google Scholar] [CrossRef][Green Version]
- Cooper, W.A.; Tran, T.; Vilain, R.E.; Madore, J.; Selinger, C.I.; Kohonen-Corish, M.; Yip, P.; Yu, B.; O’Toole, S.A.; McCaughan, B.C.; et al. PD-L1 expression is a favorable prognostic factor in early-stage non-small cell carcinoma. Lung Cancer 2015, 89, 181–188. [Google Scholar] [CrossRef]
- Samstein, R.M.; Lee, C.H.; Shoushtari, A.N.; Hellmann, M.D.; Shen, R.; Janjigian, Y.Y.; Barron, D.A.; Zehir, A.; Jordan, E.J.; Omuro, A.; et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat. Genet. 2019, 51, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Zhang, Y.; Yuan, Y.; Wang, J.; Liu, K.; Yu, B.; Xie, L.; Ou-Yang, C.; Wu, L.; Ye, X. The comprehensive analyses of genomic variations and assessment of TMB and PD-L1 expression in chinese lung adenosquamous carcinoma. Front. Genet. 2021, 11, 609405. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, H.; Sanchez-Vega, F.; La, K.; Chatila, W.; Jonsson, P.; Halpenny, D.; Plodkowski, A.; Long, N.; Sauter, J.L.; Rekhtman, N.; et al. Molecular determinants of response to anti–programmed cell death (PD)-1 and anti–programmed death-ligand 1 (PD-L1) blockade in patients with non–small-cell lung cancer profiled with targeted next-generation sequencing. J. Clin. Oncol. 2018, 36, 633–641. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Jia, M.; Li, Y.; Sun, P.; Gao, H. Differential expression of PD-L1 in central and peripheral and TTF1-positive and -negative small-cell lung cancer. Front. Med. 2020, 7, 621838. [Google Scholar] [CrossRef] [PubMed]
- Acheampong, E.; Abed, A.; Morici, M.; Bowyer, S.; Amanuel, B.; Lin, W.; Millward, M.; Gray, E.S. Tumour PD-L1 expression in small-cell lung cancer: A systematic review and meta-analysis. Cells 2020, 9, 2393. [Google Scholar] [CrossRef]
- Wang, A.; Wang, H.; Liu, Y.; Zhao, M.; Zhang, H.; Lu, Z.; Fang, Y.; Chen, X.; Liu, G. The prognostic value of PD-L1 expression for non-small cell lung cancer patients: A meta-analysis. Eur. J. Surg. Oncol. EJSO 2015, 41, 450–456. [Google Scholar] [CrossRef]
- Sharma, J.; Borczuk, A.; Liu, H.; Li, S.; Perez-Soler, R.; Halmos, B.; Cheng, H. P2. 01-056 distinct PD-L1 expression in different components of pulmonary sarcomatoid carcinoma and its association with MET mutation: Topic: Immune mechanisms in thoracic cancer and targeted therapy. J. Thorac. Oncol. 2017, 12, S819–S820. [Google Scholar] [CrossRef][Green Version]
- Domblides, C.; Leroy, K.; Monnet, I.; Mazières, J.; Barlesi, F.; Gounant, V.; Baldacci, S.; Mennecier, B.; Toffart, A.C.; Audigier-Valette, C.; et al. Efficacy of immune checkpoint inhibitors in lung sarcomatoid carcinoma. J. Thorac. Oncol. 2020, 15, 860–866. [Google Scholar] [CrossRef]
- Tsuruoka, K.; Horinouchi, H.; Goto, Y.; Kanda, S.; Fujiwara, Y.; Nokihara, H.; Yamamoto, N.; Asakura, K.; Nakagawa, K.; Sakurai, H.; et al. PD-L1 expression in neuroendocrine tumors of the lung. Lung Cancer 2017, 108, 115–120. [Google Scholar] [CrossRef]
- Shirasawa, M.; Yoshida, T.; Takayanagi, D.; Shiraishi, K.; Yagishita, S.; Sekine, K.; Kanda, S.; Matsumoto, Y.; Masuda, K.; Shinno, Y.; et al. Activity and immune correlates of programmed death-1 blockade therapy in patients with advanced large cell neuroendocrine carcinoma. Clin. Lung Cancer 2021, 22, 282–291.e6. [Google Scholar] [CrossRef]
- Green, M.R.; Monti, S.; Rodig, S.J.; Juszczynski, P.; Currie, T.; O’Donnell, E.; Chapuy, B.; Takeyama, K.; Neuberg, D.; Golub, T.R.; et al. Integrative analysis reveals selective 9p24.1 amplification, increased PD-1 ligand expression, and further induction via JAK2 in nodular sclerosing Hodgkin lymphoma and primary mediastinal large B-cell lymphoma. Blood 2010, 116, 3268–3277. [Google Scholar] [CrossRef] [PubMed]
- Sucker, A.; Zhao, F.; Pieper, N.; Heeke, C.; Maltaner, R.; Stadtler, N.; Real, B.; Bielefeld, N.; Howe, S.; Weide, B.; et al. Acquired IFNγ resistance impairs anti-tumor immunity and gives rise to T-cell-resistant melanoma lesions. Nat. Commun. 2017, 8, 15440. [Google Scholar] [CrossRef] [PubMed]
- Zaretsky, J.M.; Garcia-Diaz, A.; Shin, D.S.; Escuin-Ordinas, H.; Hugo, W.; Hu-Lieskovan, S.; Torrejon, D.Y.; Abril-Rodriguez, G.; Sandoval, S.; Barthly, L.; et al. Mutations associated with acquired resistance to PD-1 blockade in melanoma. N. Engl. J. Med. 2016, 375, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Kalbasi, A.; Tariveranmoshabad, M.; Hakimi, K.; Kremer, S.; Campbell, K.M.; Funes, J.M.; Vega-Crespo, A.; Parisi, G.; Champekar, A.; Nguyen, C.; et al. Uncoupling interferon signaling and antigen presentation to overcome immunotherapy resistance due to JAK1 loss in melanoma. Sci. Transl. Med. 2020, 12, eabb0152. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Vanderbilt, C.M.; Cotzia, P.; Stella, J.A., III; Chang, J.C.; Chen, Y.; Tang, L.H.; DeLair, D.F.; Yao, J.; Ladanyi, M.; et al. JAK2, PD-L1, and PD-L2 (9p24.1) amplification in metastatic mucosal and cutaneous melanomas with durable response to immunotherapy. Hum. Pathol. 2019, 88, 87–91. [Google Scholar] [CrossRef]
- Shi, Y.; Lei, Y.; Liu, L.; Zhang, S.; Wang, W.; Zhao, J.; Zhao, S.; Dong, X.; Yao, M.; Wang, K.; et al. Integration of comprehensive genomic profiling, tumor mutational burden, and PD-L1 expression to identify novel biomarkers of immunotherapy in non-small cell lung cancer. Cancer Med. 2021, 10, 2216–2231. [Google Scholar] [CrossRef]
- Horn, S.; Leonardelli, S.; Sucker, A.; Schadendorf, D.; Griewank, K.G.; Paschen, A. Tumor CDKN2A-associated JAK2 loss and susceptibility to immunotherapy resistance. J. Natl. Cancer Inst. 2018, 110, 677–681. [Google Scholar] [CrossRef]
- Aredo, J.V.; Mambetsariev, I.; Hellyer, J.A.; Amini, A.; Neal, J.W.; Padda, S.K.; McCoach, C.E.; Riess, J.W.; Cabebe, E.C.; Naidoo, J.; et al. Durvalumab for stage III EGFR-mutated NSCLC after definitive chemoradiotherapy. J. Thorac. Oncol. 2021, 16, 1030–1041. [Google Scholar] [CrossRef]
- Wang, C.; Wang, H.N.; Wang, L. Biomarkers for predicting the efficacy of immune checkpoint inhibitors. J. Cancer 2022, 13, 481–495. [Google Scholar] [CrossRef]
- Kato, S.; Goodman, A.; Walavalkar, V.; Barkauskas, D.A.; Sharabi, A.; Kurzrock, R. Hyperprogressors after immunotherapy: Analysis of genomic alterations associated with accelerated growth rate. Clin. Cancer Res. 2017, 23, 4242–4250. [Google Scholar] [CrossRef]
- Saâda-Bouzid, E.; Defaucheux, C.; Karabajakian, A.; Coloma, V.P.; Servois, V.; Paoletti, X.; Even, C.; Fayette, J.; Guigay, J.; Loirat, D.; et al. Hyperprogression during anti-PD-1/PD-L1 therapy in patients with recurrent and/or metastatic head and neck squamous cell carcinoma. Ann. Oncol. 2017, 28, 1605–1611. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Zhou, H.; Shen, J.; Li, J.; Zhang, Y.; Hong, S.; Zhang, L. MDM2/4 amplification predicts poor response to immune checkpoint inhibitors: A pan-cancer analysis. ESMO Open 2020, 5, e000614. [Google Scholar] [CrossRef] [PubMed]
- Ricciuti, B.; Arbour, K.C.; Lin, J.J.; Vajdi, A.; Vokes, N.; Hong, L.; Zhang, J.; Tolstorukov, M.Y.; Li, Y.Y.; Spurr, L.F.; et al. Diminished efficacy of programmed death-(ligand)1 inhibition in STK11- and KEAP1-mutant lung adenocarcinoma is affected by KRAS mutation status. J. Thorac. Oncol. 2022, 17, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Nagata, A.; Ono, Y.; Yamada, T. Complete nucleotide sequence of the 16S rRNA gene of Mycobacterium bovis BCG. J. Bacteriol. 1988, 170, 2886–2889. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Chen, A.X.; Gartrell, R.D.; Silverman, A.M.; Aparicio, L.; Chu, T.; Bordbar, D.; Shan, D.; Samanamud, J.; Mahajan, A.; et al. Immune and genomic correlates of response to anti-PD-1 immunotherapy in glioblastoma [published correction appears. Nat. Med. 2019, 25, 462–469. [Google Scholar] [CrossRef]
- Chen, X.; Su, C.; Ren, S.; Zhou, C.; Jiang, T. Pan-cancer analysis of KEAP1 mutations as biomarkers for immunotherapy outcomes. Ann. Transl. Med. 2020, 8, 141. [Google Scholar] [CrossRef]
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.R.; Steins, M.; Ready, N.E.; Chow, L.Q.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [Google Scholar] [CrossRef]
- Yang, Y.; Shen, S.; Sun, Y.; Husain, H.; Zhou, H.; Lu, S.; Li, Z. The relationship between different subtypes of KRAS and PD-L1 & tumor mutation burden (TMB) based on next-generation sequencing (NGS) detection in Chinese lung cancer patients. Transl. Lung Cancer Res. 2022, 11, 213–223. [Google Scholar]
- Galan-Cobo, A.; Sitthideatphaiboon, P.; Qu, X.; Poteete, A.; Pisegna, M.A.; Tong, P.; Chen, P.H.; Boroughs, L.K.; Rodriguez, M.L.; Zhang, W.; et al. LKB1 and KEAP1/NRF2 pathways cooperatively promote metabolic reprogramming with enhanced glutamine dependence in KRAS-mutant lung adenocarcinoma. Cancer Res. 2019, 79, 3251–3267. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Ciuleanu, T.E.; Pluzanski, A.; Lee, J.S.; Otterson, G.A.; Audigier-Valette, C.; Minenza, E.; Linardou, H.; Burgers, S.; Salman, P.; et al. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N. Engl. J. Med. 2018, 378, 2093–2104. [Google Scholar] [CrossRef]
- Cheng, B.; Yuan, W.; Su, J.; Liu, Y.; Chen, J. Recent advances in small molecule based cancer immunotherapy. Eur. J. Med. Chem. 2018, 157, 582–598. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, L.; Rodriguez-Abreu, D.; Gadgeel, S.; Esteban, E.; Felip, E.; De Angelis, F.; Domine, M.; Clingan, P.; Hochmair, M.J.; Powell, S.F.; et al. Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N. Engl. J. Med. 2018, 378, 2078–2092. [Google Scholar] [CrossRef] [PubMed]
- Kogure, Y.; Hashimoto, H.; Oki, M. A randomized phase III study of pembrolizumab versus pembrolizumab-carboplatinpemetrexed for locally advanced or metastatic nonsquamous non-small-cell lung cancer with PD-L1 50% or more (LAPLACE50): Study protocol. Clin. Lung Cancer 2021, 5, 19. [Google Scholar]
| Lung Cancer Subtype | PD-L1 ≥ 50% | PD-L1 1–49% | PD-L1 >1% | PD-L1 <1% | References |
|---|---|---|---|---|---|
| All NSCLC | 540/4063 (13.29%) | 1356/3184 (42.59%) | 1763/4761 (37.03%) | 2912/4613 (63.13%) | |
| Squamous Cell Carcinoma | 284/1766 (16.08%) | 569/1189 (47.86%) | 743/1810 (41.05%) | 1067/1810 (58.95%) | [22,25,35,40,41] |
| Adenocarcinoma | 179/1919 (9.33%) | 712/1507 (47.25%) | 826/2379 (34.72%) | 1553/2379 (65.28%) | [22,25,35,40,41] |
| KRAS Mutant Adenocarcinoma | - | 37/136 (27.21%) | - | - | [40] |
| Adenosquamous Carcinoma | - | 21/54 (38.89%) | 3/7 (42.86%) | 4/7 (57.14%) | [42] |
| Large Cell Carcinoma | - | 10/35 (28.75%) | 31/88 (35.23%) | 52/77 (67.53%) | [42] |
| Large Cell Neuroendocrine Carcinoma | - | - | 1/23 (4.35%) | 22/23 (95.65%) | [43] |
| Small Cell Lung Cancer | - | 73/194 (37.63%) | - | - | [25] |
| Sarcomatoid carcinoma | 78% | 22% | - | - | [44] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ullah, A.; Pulliam, S.; Karki, N.R.; Khan, J.; Jogezai, S.; Sultan, S.; Muhammad, L.; Khan, M.; Jamil, N.; Waheed, A.; et al. PD-L1 Over-Expression Varies in Different Subtypes of Lung Cancer: Will This Affect Future Therapies? Clin. Pract. 2022, 12, 653-671. https://doi.org/10.3390/clinpract12050068
Ullah A, Pulliam S, Karki NR, Khan J, Jogezai S, Sultan S, Muhammad L, Khan M, Jamil N, Waheed A, et al. PD-L1 Over-Expression Varies in Different Subtypes of Lung Cancer: Will This Affect Future Therapies? Clinics and Practice. 2022; 12(5):653-671. https://doi.org/10.3390/clinpract12050068
Chicago/Turabian StyleUllah, Asad, Steven Pulliam, Nabin Raj Karki, Jaffar Khan, Sana Jogezai, Sandresh Sultan, Lal Muhammad, Marjan Khan, Nimra Jamil, Abdul Waheed, and et al. 2022. "PD-L1 Over-Expression Varies in Different Subtypes of Lung Cancer: Will This Affect Future Therapies?" Clinics and Practice 12, no. 5: 653-671. https://doi.org/10.3390/clinpract12050068
APA StyleUllah, A., Pulliam, S., Karki, N. R., Khan, J., Jogezai, S., Sultan, S., Muhammad, L., Khan, M., Jamil, N., Waheed, A., Belakhlef, S., Ghleilib, I., Vail, E., Heneidi, S., & Karim, N. A. (2022). PD-L1 Over-Expression Varies in Different Subtypes of Lung Cancer: Will This Affect Future Therapies? Clinics and Practice, 12(5), 653-671. https://doi.org/10.3390/clinpract12050068








