Do Patient-Reported Quality-of-Life (QoL) Scales Provide an Adequate Assessment of Patients with Cryptoglandular Anal Fistulae? A Systematic Review of Measurement Instruments and Their Content Validity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Scope
2.2. Search Strategy
Study Selection
- The study aim was to assess the content validity of PROMs assessing QoL or
- Studies describing the development of patient-reported QoL measurement instruments
- More than 50% of the study sample had an anal fistula of cryptoglandular aetiology
- Published as a full-text article
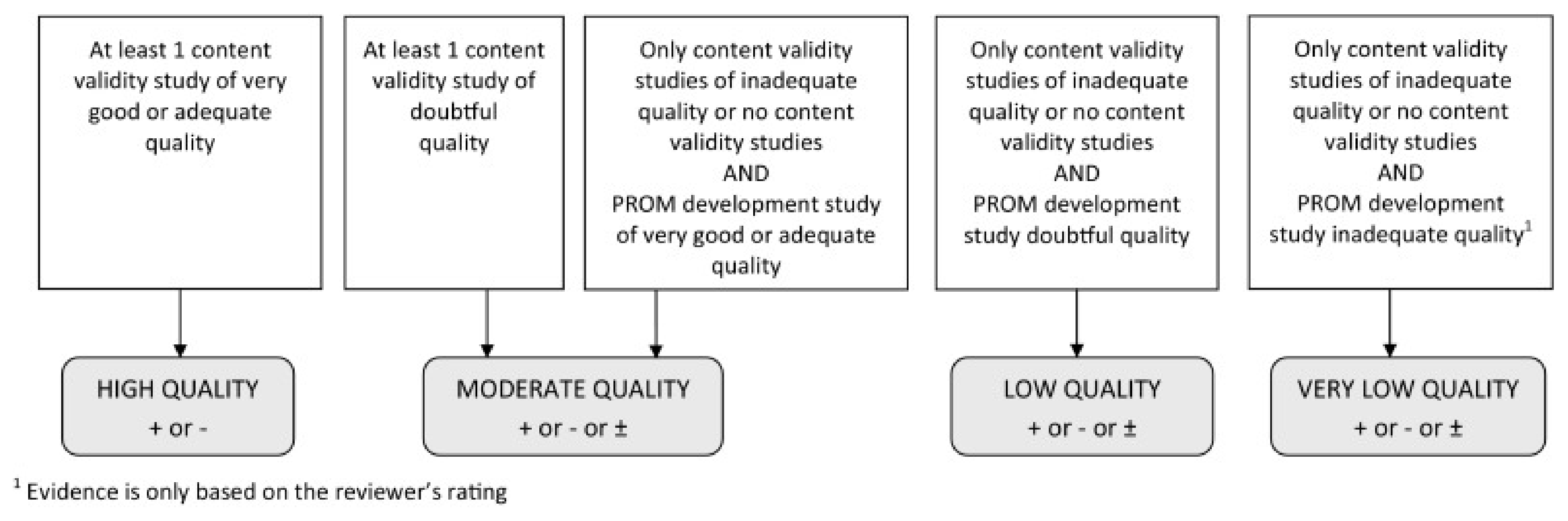
2.3. Quality Assessment and Data Extraction
3. Results
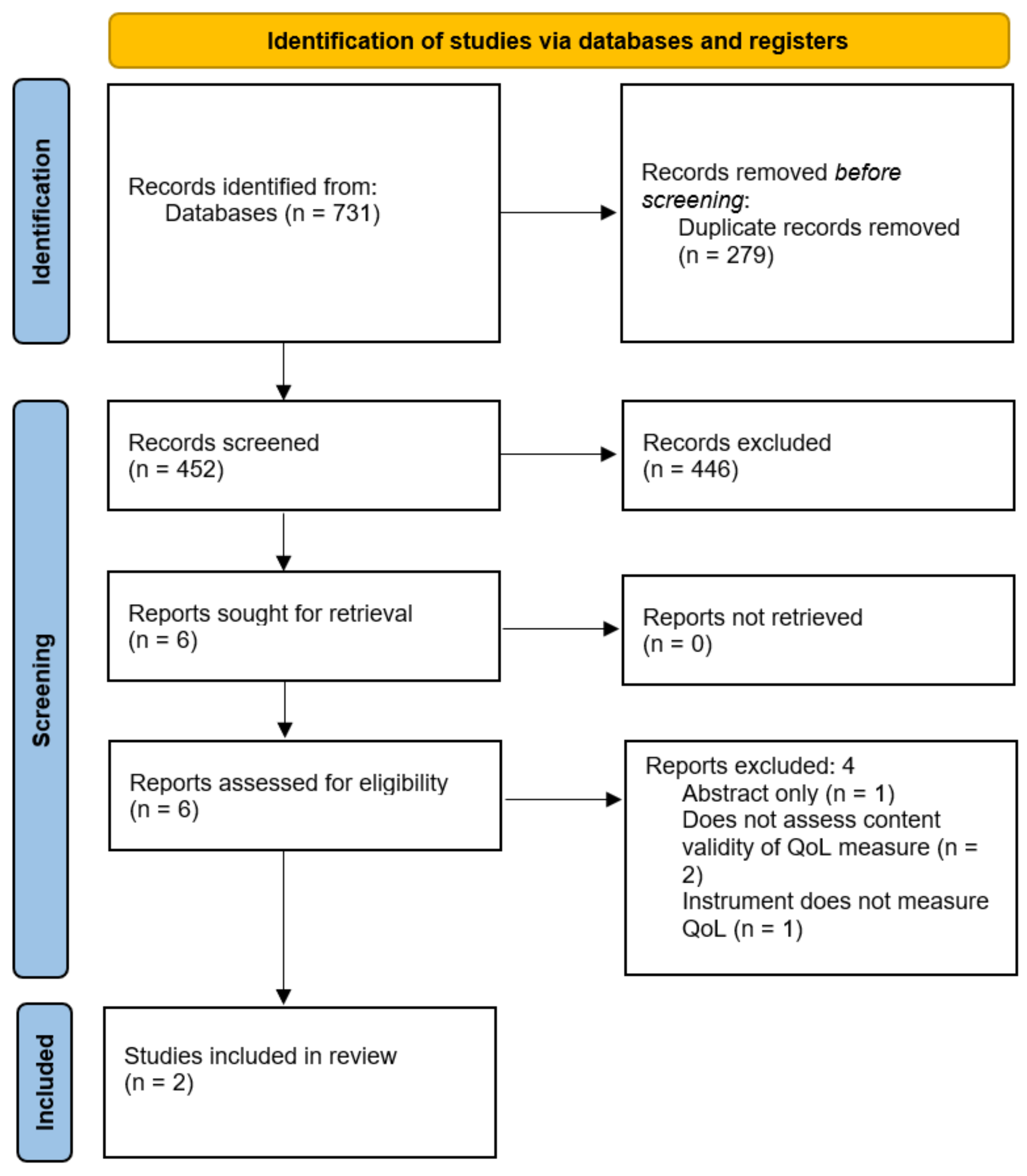
Search Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dudukgian, H.; Abcarian, H. Why do we have so much trouble treating anal fistula? World J. Gastroenterol. 2011, 17, 3292–3296. [Google Scholar] [CrossRef]
- Parks, A.G. Pathogenesis and treatment of fistula-in-Ano. Br. Med. J. 1961, 1, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Zanotti, C.; Martinez-Puente, C.; Pascual, I.; Pascual, M.; Herreros, D.; Olmo, D.G. An assessment of the incidence of fistula-in-Ano in four countries of the European Union. Int. J. Colorectal Dis. 2007, 22, 1459–1462. [Google Scholar] [CrossRef]
- Iqbal, N.; Machielsen, A.J.H.M.; Breukink, S.O.; Woodcock, R.; Kane, G.; Alrubaiy, L.; Grossi, U.; Kimman, M.L.; Tozer, P.J. Living with cryptoglandular anal fistula: A qualitative investigation of the patient’s experience through semi-structured patient interviews. Qual. Life Res. 2022, 31, 2505–2518. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Machielsen, A.J.H.M.; Kimman, M.L.; Kane, G.; Woodcock, R.; Grossi, U.; Tozer, P.J.; Breukink, S.O.; AFCOS Consensus Meeting Collaborators. AFCOS: The Development of a Cryptoglandular Anal Fistula Core Outcome Set. Ann. Surg. 2022. [Google Scholar] [CrossRef] [PubMed]
- Machielsen, A.J.H.M.; Iqbal, N.; Kimman, M.L.; Sahnan, K.; Adegbola, S.O.; Kane, G.; Woodcock, R.; Kleijnen, J.; Grossi, U.; Breukink, S.O.; et al. Heterogeneity in outcome selection, definition and measurement in studies assessing the treatment of cryptoglandular anal fistula: Findings from a systematic review. Tech. Coloproctol. 2021, 25, 761–830. [Google Scholar] [CrossRef] [PubMed]
- Prinsen, C.A.C.; Mokkink, L.B.; Bouter, L.M.; Alonso, J.; Patrick, D.L.; de Vet, H.C.W.; Terwee, C.B. COSMIN guideline for systematic reviews of patient-reported outcome measures. Qual. Life Res. 2018, 27, 1147–1157. [Google Scholar] [CrossRef] [PubMed]
- Terwee, C.B.; Prinsen, C.A.C.; Chiarotto, A.; Westerman, M.J.; Patrick, D.L.; Alonso, J.; Bouter, L.M.; de Vet, H.C.W.; Mokkink, L.B. COSMIN methodology for evaluating the content validity of patient-reported outcome measures: A Delphi study. Qual. Life Res. 2018, 27, 1159–1170. [Google Scholar] [CrossRef] [PubMed]
- Prinsen, C.A.C.; Vohra, S.; Rose, M.R.; Boers, M.; Tugwell, P.; Clarke, M.; Williamson, P.R.; Terwee, C.B. How to select outcome measurement instruments for outcomes included in a “Core Outcome Set”—A practical guideline. Trials 2016, 17, 449. [Google Scholar] [CrossRef] [PubMed]
- Terwee, C.B.; Jansma, E.; Riphagen, I.I.; De Vet, H.C.W. Development of a methodological PubMed search filter for finding studies on measurement properties of measurement instruments. Qual. Life Res. 2009, 18, 1115–1123. [Google Scholar] [CrossRef]
- Tabassum, A.; Zahid, A.; Byrne, C.M.; Young, C.J. Development of Perianal Sepsis Index (PASI) for assessing quality of life in chronic perianal sepsis. Eur. Surg. 2018, 50, 270–277. [Google Scholar] [CrossRef]
- García-Aguilar, J.; Davey, C.S.; Le, C.T.; Lowry, A.C.; Rothenberger, D.A. Patient satisfaction after surgical treatment for fistula-in-Ano. Dis. Colon Rectum 2000, 43, 1206–1212. [Google Scholar] [CrossRef]
- Cavanaugh, M.; Hyman, N.; Osler, T. Fecal incontinence severity index after fistulotomy: A predictor of quality of life. Dis. Colon Rectum 2002, 45, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Ferrer-Márquez, M.; Espínola-Cortés, N.; Reina-Duarte, A.; Granero-Molina, J.; Fernández-Sola, C.; Hernández-Padilla, J.M. Analysis and Description of Disease-Specific Quality of Life in Patients with Anal Fistula. Cir. Esp. (Engl. Ed.) 2018, 96, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Ferrer-Márquez, M.; Espínola-Cortés, N.; Reina-Duarte, A.; Granero-Molina, J.; Fernández-Sola, C.; Hernández-Padilla, J.M. Design and psychometric evaluation of the quality of life in patients with anal fistula questionnaire. Dis. Colon Rectum 2017, 60, 1083–1091. [Google Scholar] [CrossRef]
- Irvine, E. Usual therapy improves perianal Crohn’s disease as measured by a new disease activity index. J. Clin. Gastroenterol. 1995, 20, 27–32. [Google Scholar]
- Cameron, L.J.; Wales, K.; Casey, A.; Pike, S.; Jolliffe, L.; Schneider, E.J.; Christie, L.J.; Ratcliffe, J.; Lannin, N.A. Self-reported quality of life following stroke: A systematic review of instruments with a focus on their psychometric properties. Qual. Life Res. 2021, 31, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Wan, E.Y.F.; Choi, E.P.H.; Yu, E.Y.T.; Chin, W.Y.; Fung, C.S.C.; Chan, A.K.C.; Lam, C.L.K. Evaluation of the internal and external responsiveness of Short Form-12 Health Survey version 2 (SF-12v2) in patients with type 2 diabetes mellitus. Qual. Life Res. 2018, 27, 2459–2469. [Google Scholar] [CrossRef] [PubMed]
- Bahall, M.; LeGall, G.; Khan, K. Quality of life among patients with cardiac disease: The impact of comorbid depression. Health Qual. Life Outcomes 2020, 18, 189. [Google Scholar] [CrossRef] [PubMed]
- Owen, H.A.; Buchanan, G.; Schizas, A.; Emmanuel, A.; Cohen, R.; Williams, A.B. Quality of life following fistulotomy—Short term follow-up. Colorectal Dis. 2017, 19, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Owen, H.A.; Buchanan, G.; Schizas, A.; Cohen, R.; Williams, A.B. Quality of life with anal fistula. Ann. R. Coll. Surg. Engl. 2016, 98, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Lara, F.J.P.; Serrano, A.M.; Moreno, J.U.; Carmona, J.H.; Marquez, M.F.; Pérez, L.R.; Moreno, A.D.R.; Muñoz, H.O. Platelet-rich fibrin sealant as a treatment for complex perianal fistulas: A multicentre study. J. Gastrointest. Surg. 2014, 19, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Olmo, D.; Herreros, D.; Pascual, I.; Pascual, J.A.; Del-Valle, E.; Zorrilla, J.; De-La-Quintana, P.; Garcia-Arranz, M.; Pascual, M. Expanded adipose-derived stem cells for the treatment of complex perianal fistula: A phase II clinical trial. Dis. Colon Rectum 2009, 52, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Jarrar, A.; Church, J. Advancement flap repair: A good option for complex anorectal fistulas. Dis. Colon Rectum 2011, 54, 1537–1541. [Google Scholar] [CrossRef]
- Madbouly, K.M.; El Shazly, W.; Abbas, K.S.; Hussein, A.M. Ligation of intersphincteric fistula tract versus mucosal advancement flap in patients with high transsphincteric fistula-in-Ano: A prospective randomized trial. Dis. Colon Rectum 2014, 57, 1202–1208. [Google Scholar] [CrossRef] [PubMed]
- Van Koperen, P.J.; Bemelman, W.A.; Gerhards, M.F.; Janssen, L.W.; van Tets, W.F.; van Dalsen, A.D.; Slors, J.F.M. The anal fistula plug treatment compared with the mucosal advancement flap for cryptoglandular high transsphincteric perianal fistula: A double-blinded multicenter randomized trial. Dis. Colon Rectum 2011, 54, 387–393. [Google Scholar] [CrossRef]
- Jayne, D.G.; Scholefield, J.; Tolan, D.; Gray, R.; Edlin, R.; Hulme, C.T.; Sutton, A.J.; Handley, K.; Hewitt, C.A.; Kaur, M.; et al. Anal fistula plug versus surgeon’s preference for surgery for trans-sphincteric anal fistula: The FIAT RCT. Health Technol. Assess. 2019, 23, 1–76. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.-H.; Liu, H.-L.; Li, Z.; Xiao, Y.-H.; Li, A.-J.; Chang, Y.; Zhang, Y.; Lv, L.; Lin, M.-B. Video-Assisted Anal Fistula Treatment (VAAFT) for Complex anal Fistula: A Preliminary Evaluation in China. Med. Sci. Monit. 2017, 23, 2065–2071. [Google Scholar] [CrossRef] [PubMed]
- Pescatori, M.; Ayabaca, S.; Caputo, D. Can anal manometry predict anal incontinence after fistulectomy in males? Colorectal Dis. 2004, 6, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Seneviratne, S.; Samarasekera, D.N.; Kotalawala, W. Quality of life following surgery for recurrent fistula-in-Ano. Technol. Coloproctol. 2009, 13, 215–217. [Google Scholar] [CrossRef]
- Roig, J.V.; Garcia-Armengol, J.; Jordan, J.C.; Moro, D.; Garcia-Granero, E.; Alos, R. Fistulectomy and sphincteric reconstruction for complex cryptoglandular fistulas. Colorectal Dis. 2010, 12, e145-52. [Google Scholar] [CrossRef] [PubMed]
- Jayarajah, U.; Wickramasinghe, D.P.; Samarasekera, D.N. Anal incontinence and quality of life following operative treatment of simple cryptoglandular fistula-in-Ano: A prospective study. BMC Res. Notes 2017, 10, 572. [Google Scholar] [CrossRef]
- Eypasch, E.; Williams, J.I.; Wood-Dauphinee, S.; Ure, B.M.; Schmulling, C.; Neugebauer, E.; Troidl, H. Gastrointestinal quality of life index: Development, validation and application of a new instrument. Br. J. Surg. 1995, 82, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Sailer, M.; Bussen, D.; Debus, E.S.; Fuchs, K.H.; Thiede, A. Quality of life in patients with benign anorectal disorders. Br. J. Surg. 1998, 85, 1716–1719. [Google Scholar] [CrossRef] [PubMed]
- Rockwood, T.H.; Church, J.M.; Fleshman, J.W.; Kane, R.L.; Mavrantonis, C.; Thorson, A.G.; Wexner, S.D.; Bliss, D.; Lowry, A.C. Fecal incontinence quality of life scale. Dis. Colon Rectum 2000, 43, 9–16. [Google Scholar] [CrossRef]
| COSMIN Standards | Peri-Anal Sepsis Index (PASI) [8] | Quality of Life Anal Fistula Questionnaire (QoLAF-Q) [12] |
|---|---|---|
| General design requirements | ||
| Is a clear description provided of the construct to be measured? | Inadequate | Inadequate |
| Is the origin of the construct clear? | Doubtful | Doubtful |
| Is a clear description provided of the target population for which the PROM was developed? | Very Good | Very Good |
| Is a clear description provided of the context of use? | Very Good | Very Good |
| Was the PROM development study performed in a sample representing the target population for which the PROM was developed? | Very Good | Inadequate |
| Concept elicitation | ||
| Was an appropriate qualitative data collection method used to identify relevant items for a new PROM? | Doubtful | Not assessed |
| Were skilled group moderators/interviewers used? | Doubtful | |
| Were the group meetings or interviews based on an appropriate topic or interview guide? | Doubtful | |
| Were the group meetings or interviews recorded and transcribed verbatim? | Doubtful | |
| Was an appropriate approach used to analyse the data? | Doubtful | |
| Was at least part of the data coded independently? | Doubtful | |
| Was data collection continued until saturation was reached? | Doubtful | |
| For quantitative studies: was the sample size appropriate? | Inadequate/Not applicable |
| COSMIN Standards for Asking Patients about Comprehensibility of a PROM | Quality of Life Anal Fistula Questionnaire (QoLAF-Q) [12] |
|---|---|
| Was an appropriate qualitative method used for assessing the comprehensibility of the PROM instructions, items, response options, and recall period? | Doubtful |
| Was each item tested in an appropriate number of patients? | Very Good |
| Were skilled group moderators/interviewers used? | Doubtful |
| Were the group meetings or interviews based on an appropriate topic or interview guide? | Doubtful |
| Were the group meetings or interviews recorded and transcribed verbatim? | Doubtful |
| Was an appropriate approach used to analyse the data? | Doubtful |
| Were at least two researchers involved in the analysis? | Doubtful |
| PROM (Subscale) | PROM Development Study | Rating of Reviewers | OVERALL RATINGS PER PROM | QUALITY OF EVIDENCE | PROM Development Study | Content Validity Study | Rating of Reviewers | OVERALL RATINGS PER PROM | QUALITY OF EVIDENCE | |
|---|---|---|---|---|---|---|---|---|---|---|
| PASI | QoLAF-Q | |||||||||
| Relevance | ||||||||||
| 1 | Are the included items relevant for the construct of interest? | ? | + | ? | + | |||||
| 2 | Are the included items relevant for the target population of interest? | + | + | + | + | |||||
| 3 | Are the included items relevant for the context of use of interest? | + | + | + | + | |||||
| 4 | Are the response options appropriate? | ? | + | ? | + | |||||
| 5 | Is the recall period appropriate? | ? | ? | ? | ? | |||||
| RELEVANCE RATING (+/−/±/?) | ? | + | + | Very low | ? | + | + | Very low | ||
| Comprehensiveness | ||||||||||
| 6 | Are all key concepts included? | ? | - | ? | - | |||||
| COMPREHENSIVENESS RATING (+/−/±/?) | ? | - | - | Very low | ? | - | - | Very low | ||
| Comprehensibility | ||||||||||
| 7 | Are the PROM instructions understood by the population of interest as intended? | ? | + | ? | ? | - | ||||
| 8 | Are the PROM items and response options understood by the population of interest as intended? | ? | + | ? | ? | + | ||||
| 9 | Are the PROM items appropriately worded? | |||||||||
| 10 | Do the response options match the question? | |||||||||
| COMPREHENSIBILITY RATING (+/−/±/?) | ? | + | + | Very low | ? | ? | +/− | +/− | Very low | |
| CONTENT VALIDITY RATING (+/−/±/?) | ? | +/− | +/− | Very low | ? | ? | +/− | +/− | Very low | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iqbal, N.; Shah, R.; Alrubaiy, L.; Tozer, P. Do Patient-Reported Quality-of-Life (QoL) Scales Provide an Adequate Assessment of Patients with Cryptoglandular Anal Fistulae? A Systematic Review of Measurement Instruments and Their Content Validity. Clin. Pract. 2022, 12, 628-639. https://doi.org/10.3390/clinpract12040066
Iqbal N, Shah R, Alrubaiy L, Tozer P. Do Patient-Reported Quality-of-Life (QoL) Scales Provide an Adequate Assessment of Patients with Cryptoglandular Anal Fistulae? A Systematic Review of Measurement Instruments and Their Content Validity. Clinics and Practice. 2022; 12(4):628-639. https://doi.org/10.3390/clinpract12040066
Chicago/Turabian StyleIqbal, Nusrat, Rishi Shah, Laith Alrubaiy, and Phil Tozer. 2022. "Do Patient-Reported Quality-of-Life (QoL) Scales Provide an Adequate Assessment of Patients with Cryptoglandular Anal Fistulae? A Systematic Review of Measurement Instruments and Their Content Validity" Clinics and Practice 12, no. 4: 628-639. https://doi.org/10.3390/clinpract12040066
APA StyleIqbal, N., Shah, R., Alrubaiy, L., & Tozer, P. (2022). Do Patient-Reported Quality-of-Life (QoL) Scales Provide an Adequate Assessment of Patients with Cryptoglandular Anal Fistulae? A Systematic Review of Measurement Instruments and Their Content Validity. Clinics and Practice, 12(4), 628-639. https://doi.org/10.3390/clinpract12040066







