Screening of Diabetic Retinopathy Using Teleophthalmology to Complement Human Resources for Eye Health: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Selection and Eligibility Criteria
2.2. Quality Assessment
2.3. Data Extraction
2.4. Data Analysis
2.5. Ethical Consideration
3. Results
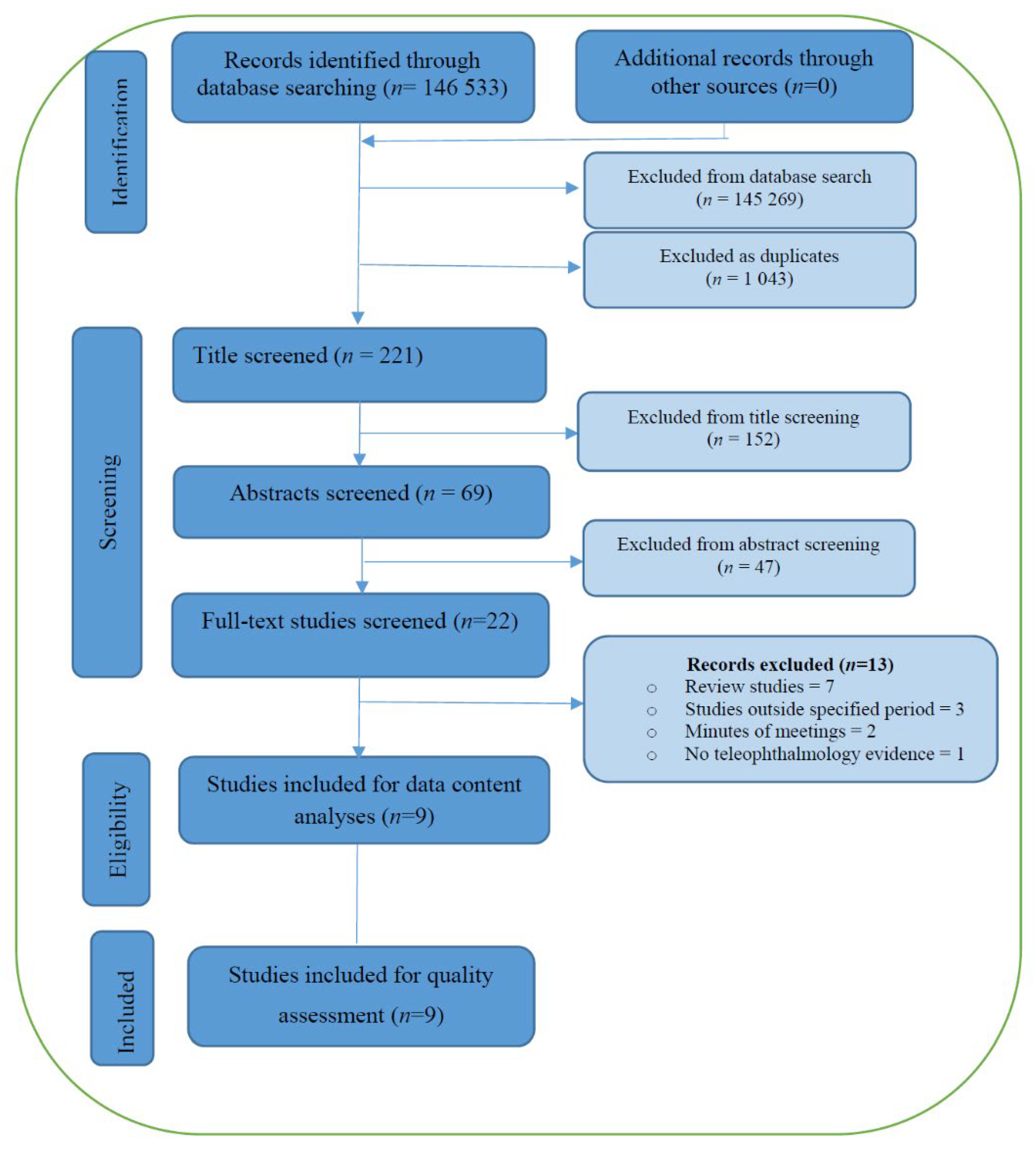
3.1. Search Results
3.2. Characteristics of Included Studies
- Evaluating the ability of trained imagers in using the ultra-wide field fundus camera (UWF) to determine the presence or absence of DR.
- Evaluating the relative diagnostic value of the non-mydriatic fundus photographer (NMFP).
- Evaluating telemedicine screenings for DR among patients with type 1 or 2 diabetes in rural areas.
- Describing the telemedicine reporting of DR screening using UWF.
- Describing the results of DR screening programs implemented in primary care areas.
- Characterizing the prevalence of DR and DME.
- Exploring the feasibility of using telemedicine for DR screening.
- Identifying the prevalence and risk factors of DR and comparing DR identification and ungradable image rates between non-mydriatic NMFP and UWF.
3.3. Features of DR Screening
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Classification of Diabetes Mellitus; World Health Organization: Geneva, Switzerland, 2019.
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- WHO. World Report on Vision; World Health Organization: Geneva, Switzerland, 2019; pp. 1–180.
- Ghanchi, F.; Bailey, C.; Chakravarthy, U. Diabetic Retinopathy Guidelines; The Royal College of Ophthalmologists: London, UK, 2012; pp. 42–125. [Google Scholar]
- Steinmetz, J.D.; Bourne, R.R.; Briant, P.S.; Flaxman, S.R.; Taylor, H.R.; Jonas, J.B.; Abdoli, A.A.; Abrha, W.A.; Abualhasan, A.; Abu-Gharbieh, E.G.; et al. Causes of blindness and vision impairment in 2020 and trends over 30 years, and prevalence of avoidable blindness in relation to VISION 2020: The Right to Sight: An analysis for the Global Burden of Disease Study. Lancet Glob. Health 2021, 9, e144–e160. [Google Scholar] [CrossRef]
- Organization WHO. Strengthening Diagnosis and Treatment of Diabetic Retinopathy in SEA Region; World Health Organization: Geneva, Switzerland, 2020.
- Committee STDGE. SEMDSA 2017 guidelines for the management of type 2 diabetes mellitus. J. Endocr. Metab. Diabetes S. Afr. 2017, 22 (Suppl. 1), S1–S196. [Google Scholar]
- Memon, M.S.; Ahsan, S.; Fahadullah, M.; Parveen, K.; Salim, S.; Fahim, M.F. Diagnostic Accuracy of Direct Ophthalmoscopy and Non-Mydriatic Retinal Photography for Screening of Diabetic Retinopathy. Pak. J. Ophthalmol. 2020, 36, 120–124. [Google Scholar]
- Resnikoff, S.; Lansingh, V.C.; Washburn, L.; Felch, W.; Gauthier, T.-M.; Taylor, H.R.; Eckert, K.; Parke, D.; Wiedemann, P. Estimated number of ophthalmologists worldwide (International Council of Ophthalmology update): Will we meet the needs? Br. J. Ophthalmol. 2020, 104, 588–592. [Google Scholar] [CrossRef]
- Muqit, M. ICO Guidelines for Diabetic Eye Care 2017. B нoяб 2016 2016, 125, 1608–1622. [Google Scholar] [CrossRef] [Green Version]
- Coronado, A.; Singh, H.; Martin, J.; Costella, J.; Malvankar, M.; Hodge, W. Diagnostic accuracy of tele-ophthalmology alternatives for diabetic retinopathy screening: A meta-analysis. Investig. Ophthalmol. Vis. Sci. 2014, 55, 5330. [Google Scholar]
- Jin, K.; Lu, H.; Su, Z.; Cheng, C.; Ye, J.; Qian, D. Telemedicine screening of retinal diseases with a handheld portable non-mydriatic fundus camera. BMC Ophthalmol. 2017, 17, 89. [Google Scholar] [CrossRef]
- Peters, M.; Godfrey, C.; McInerney, P. Methodology for JBI scoping reviews: The Joanna Briggs lnstitute reviewers manual 2015. Adel. South Aust. Joanna Briggs Lnstitute 2015. Available online: https://repositorio.usp.br/item/002775594 (accessed on 6 May 2022).
- Taylor SB, H.; Das, T.; Farah, M.; Ferris, R.; Massin, P.; Mathenge, W.; Resnikoff, S.; Spivey, B.E.; Verdaguer TYW, J.; Zhao, P. ICO Guidelines for Diabetic Eye Care. Int. Counc. Ophthalmol. 2013, 1–25. Available online: https://www.worlddiabetesfoundation.org/sites/default/files/ICO%20Guidelines%20for%20Diabetic%20Eye%20Care_2014.pdf (accessed on 6 May 2022).
- Health Professions Council of South Africa. General Ethical Guidelines for Good Practice in Telemedicine; HPCSA: Pretoria, South Africa, 2014. [Google Scholar]
- Hong, Q.N.; Fàbregues, S.; Bartlett, G.; Boardman, F.; Cargo, M.; Dagenais, P.; Marie-Pierre, G.; Frances, G.; Belinda, N.; Alicia, O.; et al. The Mixed Methods Appraisal Tool (MMAT) version 2018 for information professionals and researchers. Educ. Inf. 2018, 34, 285–291. [Google Scholar] [CrossRef] [Green Version]
- Pluye, P.; Robert, E.; Cargo, M.; Bartlett, G.; O’Cathain, A.; Griffiths, F.; Boardman, F.; Gagnon, M.P.; Rousseau, M.C. Proposal: A Mixed Methods Appraisal Tool for Systematic Mixed Studies (MMAT)—Version 2011; McGill University: Montreal, QC, Canada, 2011. [Google Scholar]
- Sedgwick, P. Meta-analyses: What is heterogeneity? BMJ 2015, 350, h1435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huedo-Medina, T.; Sanchez-Meca, J.; Marín-Martínez, F.; Botella, J. Assessing heterogeneity in meta-analysis: Q statistic or I 2 Index? Psychol. Methods 2006, 11, 193–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furuya-Kanamori, L.; Barendregt, J.; Doi, S. A new improved graphical and quantitative method for detecting bias in meta-analysis. Int. J. Evid.-Based Healthc. 2018, 16, 195–203. [Google Scholar] [CrossRef]
- Pareja-Ríos, A.; Bonaque-González, S.; Serrano-García, M.; Cabrera-López, F.; Abreu-Reyes, P.; Marrero-Saavedra, M.D. Tele-ophthalmology for diabetic retinopathy: 8 years of experience. Arch. Soc. Española Oftalmol. 2017, 92, 63–70. [Google Scholar] [CrossRef]
- Silva, P.S.; Cavallerano, J.D.; Tolson, A.M.; Rodriguez, J.; Rodriguez, S.; Ajlan, R.; Tolls, D.; Patel, B.; Sehizadeh, M.; Thakore, K.; et al. Real-Time Ultrawide Field Image Evaluation of Retinopathy in a Diabetes Telemedicine Program. Diabetes Care 2015, 38, 1643–1649. [Google Scholar] [CrossRef] [Green Version]
- Paulo, S. Silva mHDC, Drew G Lewis. Identification of diabetic retinopathy and ungradable image rate with Ultrawide field imaging in a national teleophthalmology program. Am. Acad. Ophthalmol. 2016, 123, 1360–1367. [Google Scholar]
- Bursell, S.E.; Fonda, S.J.; Lewis, D.G.; Horton, M.B. Prevalence of diabetic retinopathy and diabetic macular edema in a primary care based teleophthalmology program for American Indians and Alaskan Natives. PLoS ONE. 2018, 13, e0198551. [Google Scholar] [CrossRef] [Green Version]
- Villa, S.R.; Álvarez, C.A.; Del Valle, R.D.D.; Méndez, R.S.; García, M.C.; García, M.R.; Martín, M.C.; Vazquez, M.R. Five-year experience of tele-ophthalmology for diabetic retinopathy screening in a rural population. Arch. Soc. Española Oftalmol. 2016, 91, 428–430. [Google Scholar]
- Hussain, N.; Edraki, M.; Tahhan, R.; Sanalkumar, N.; Kenz, S.; Akasha, N.K.; Mtemererwa, B.; Mohammed, N. Telemedicine for diabetic retinopathy screening using an ultra-widefield fundus camera. Clin. Ophthalmol. 2017, 11, 1477–1482. [Google Scholar] [CrossRef] [Green Version]
- Chin, E.K.; Ventura, B.V.; See, K.Y.; Seibles, J.; Park, S.S. Nonmydriatic fundus photography for teleophthalmology diabetic retinopathy screening in rural and urban clinics. Telemed. e-Health 2014, 20, 102–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pooja, D.; Jani, L.F. Evaluation of diabetic retinal screening and factors for Ophthalmology referral in telemedicine network. Am. Med. Assoc. 2017, 135, 706–714. [Google Scholar]
- Modi, K.D.; Chandwani, R.; Kumar, K.H.; Ahmed, I.; Senthil, T. Use of telemedicine in remote screening for retinopathy in type 2 diabetes. Apollo Med. 2015, 12, 260–263. [Google Scholar] [CrossRef]
- Yau, J.W.; Rogers, S.L.; Kawasaki, R.; Lamoureux, E.L.; Kowalski, J.W.; Bek, T.; Chen, S.-J.; Dekker, J.M.; Fletcher, A.; Grauslund, J.; et al. Global Prevalence and Major Risk Factors of Diabetic Retinopathy. Diabetes Care 2012, 35, 556–564. [Google Scholar] [CrossRef] [Green Version]
- Thomas, R.L.; Halim, S.; Gurudas, S.; Sivaprasad, S.; Owens, D.R. IDF Diabetes Atlas: A review of studies utilising retinal photography on the global prevalence of diabetes related retinopathy between 2015 and 2018. Diabetes Res. Clin. Pract. 2019, 157, 107840. [Google Scholar] [CrossRef]
- Olafsdottir, E.; Andersson, D.K.; Dedorsson, I.; Svärdsudd, K.; Jansson, S.P.; Stefánsson, E. Early detection of type 2 diabetes mellitus and screening for retinopathy are associated with reduced prevalence and severity of retinopathy. Acta Ophthalmol. 2016, 94, 232–239. [Google Scholar] [CrossRef] [Green Version]
- Palmer, J.J.; Chinanayi, F.; Gilbert, A.; Pillay, D.; Fox, S.; Jaggernath, J.; Naidoo, K.; Graham, R.; Patel, D.; Blanchet, K. Mapping human resources for eye health in 21 countries of sub-Saharan Africa: Current progress towards VISION 2020. Hum. Resour. Health 2014, 12, 44. [Google Scholar] [CrossRef] [Green Version]
- Burgess, P.I.; Maccormick, I.; Harding, S.P.; Bastawrous, A.; Beare, N.; Garner, P. Epidemiology of diabetic retinopathy and maculopathy in Africa: A systematic review. Diabet. Med. A J. Br. Diabet. Assoc. 2012, 30, 399–412. [Google Scholar] [CrossRef] [Green Version]
- Cleland, C.R.; Burton, M.J.; Hall, C.; Hall, A.; Courtright, P.; Makupa, W.U.; Philippin, H. Diabetic retinopathy in Tanzania: Prevalence and risk factors at entry into a regional screening programme. Trop. Med. Int. Health 2016, 21, 417–426. [Google Scholar] [CrossRef] [Green Version]
- Machingura, P.I.; Macheka, B.; Mukona, M.; Mateveke, K.; Okwanga, P.N.; Gomo, E. Prevalence and risk factors associated with retinopathy in diabetic patients at Parirenyatwa Hospital outpatients’ clinic in Harare, Zimbabwe. Arch. Med. Biomed. Res. 2017, 3, 104–111. [Google Scholar] [CrossRef]
- Burgess, P.I.; Msukwa, G.; Beare, N.A. Diabetic retinopathy in sub-Saharan Africa: Meeting the challenges of an emerging epidemic. BMC Med. 2013, 11, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sreelatha, O.K.; Ramesh, S.V. Teleophthalmology: Improving patient outcomes? Clin. Ophthalmol. 2016, 10, 285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bozzani, F.M.; Griffiths, U.K.; Blanchet, K.; Schmidt, E. Health systems analysis of eye care services in Zambia: Evaluating progress towards VISION 2020 goals. BMC Health Serv. Res. 2014, 14, 94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, S.; Ramulu, P.; Lamoureux, E.L.; Sabanayagam, C. Addressing risk factors, screening, and preventative treatment for diabetic retinopathy in developing countries: A review. Clin. Exp. Ophthalmol. 2016, 44, 300–320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bouskill, K.; Smith-Morris, C.; Bresnick, G.; Cuadros, J.; Pedersen, E.R. Blind spots in telemedicine: A qualitative study of staff workarounds to resolve gaps in diabetes management. BMC Health Serv. Res. 2018, 18, 617. [Google Scholar] [CrossRef] [PubMed]
- Schmid, K.L.; Swann, P.G.; Pedersen, C.; Schmid, L.M. The detection of diabetic retinopathy by Australian optometrists. Clin. Exp. Optom. 2002, 85, 221–228. [Google Scholar] [CrossRef]
- Galiero, R.; Pafundi, P.C.; Nevola, R.; Rinaldi, L.; Acierno, C.; Caturano, A.; Salvatore, T.; Adinolfi, L.E.; Costagliola, C.; Sasso, F.C. The Importance of Telemedicine during COVID-19 Pandemic: A Focus on Diabetic Retinopathy. J. Diabetes Res. 2020, 2020, 9036847. [Google Scholar] [CrossRef]
| Population | Diabetic Patients, Ocular Manifestations of Diabetes, Ocular Complications of Diabetes, Diabetic Retinopathy, Retinal Complications of Diabetes |
|---|---|
| Concept | Tele-ophthalmology or Telemedicine |
| Context | All available studies (globally) |
| Author, Year | Country, Economical Profiling | Study Design | Level of Care (PHC/Other) | Type of Health Provider Taking Image | Professionals Grading Images | Device Used, Fixed/Portable | Mydriatic Used [Yes/No] | Sensitivity Level |
|---|---|---|---|---|---|---|---|---|
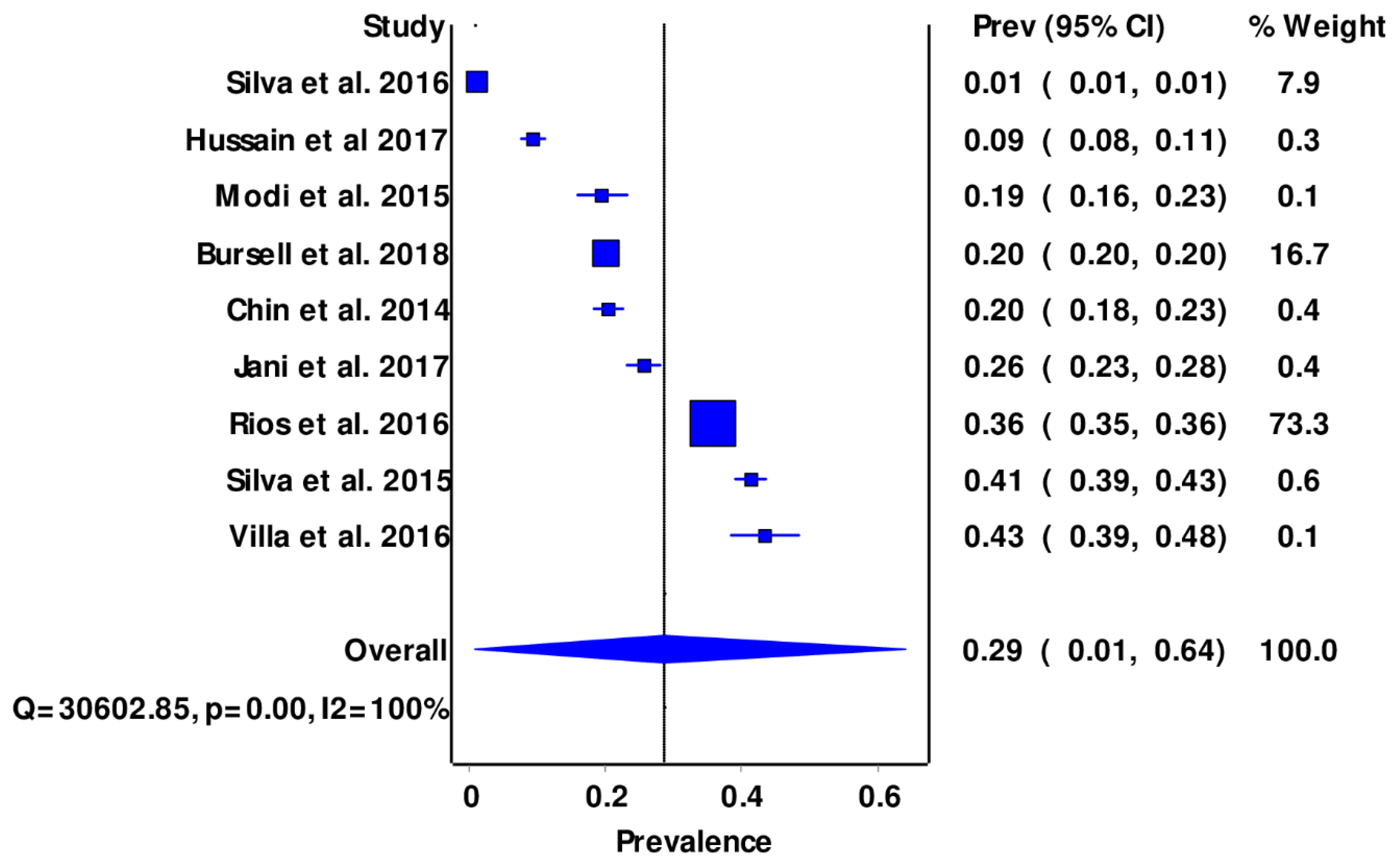
| Chin, 2014 (21) | United States of America (USA) HIC | Retrospective, Cross-sectional (C/S) study | Primary Health Care (PHC) | Non-Ophthalmologist (NON) [Trained medical personnel] | Ophthalmologist (Retinal Specialist) | Non mydriatic fundus photography (NMFP), fixed | No | 20.40% |
| Modi, 2015 (22) | India LMIC | Quantitative, C/S study | PHC | Not specified | Ophthalmologist (Retinal specialist) | NMFP, fixed | No | 19.40% |
| Silva, 2015 (23) | USA HIC | Quantitative, C/S study | PHC | NON (Trained imagers) | NON (Optometrist) | Ultra-wide Field photographer (UWF), fixed | No | 41.30% |
| Silva, 2016 (24) | USA HIC | Retrospective, C/S study | PHC | NON (Trained imagers) | NON (Optometrist) | NMFP and UWF, fixed | No | 0.90% |
| Rios, 2016 (25) | Spain HIC | Retrospective, C/S study | PHC | NON (Nurses and Physicians) | Ophthalmologist | NMFP | Yes | 35.60% |
| Villa, 2016 (26) | Spain HIC | C/S study | PHC | NON Trained Nurses | NON (PHC Physicians) | NMFP, portable | No | 43.45% |
| Jani, 2017 (27) | USA HIC | C/S study | PHC | NON Trained Nurses and Clinic Technician | Ophthalmologist | NMFP, fixed | No | 25.50% |
| Bursell, 2017 (28) | USA HIC | C/S study | PHC | NON (Trained imagers) | NON (Optometrist) | NMFP, fixed | No | 20.00% |
| Hussain, 2017 (29) | United Arab Emirates HIC | C/S study | Multi-Specialty | NON (Trained Nurses) | Ophthalmologist (Retinal specialist) | UWF, fixed | No | 9.20% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zikhali, T.; Kalinda, C.; Xulu-Kasaba, Z.N. Screening of Diabetic Retinopathy Using Teleophthalmology to Complement Human Resources for Eye Health: A Systematic Review and Meta-Analysis. Clin. Pract. 2022, 12, 457-467. https://doi.org/10.3390/clinpract12040050
Zikhali T, Kalinda C, Xulu-Kasaba ZN. Screening of Diabetic Retinopathy Using Teleophthalmology to Complement Human Resources for Eye Health: A Systematic Review and Meta-Analysis. Clinics and Practice. 2022; 12(4):457-467. https://doi.org/10.3390/clinpract12040050
Chicago/Turabian StyleZikhali, Thembile, Chester Kalinda, and Zamadonda Nokuthula Xulu-Kasaba. 2022. "Screening of Diabetic Retinopathy Using Teleophthalmology to Complement Human Resources for Eye Health: A Systematic Review and Meta-Analysis" Clinics and Practice 12, no. 4: 457-467. https://doi.org/10.3390/clinpract12040050
APA StyleZikhali, T., Kalinda, C., & Xulu-Kasaba, Z. N. (2022). Screening of Diabetic Retinopathy Using Teleophthalmology to Complement Human Resources for Eye Health: A Systematic Review and Meta-Analysis. Clinics and Practice, 12(4), 457-467. https://doi.org/10.3390/clinpract12040050






