Perinatal Outcomes in Premature Placental Calcification and the Association of a Color Doppler Study: Report from a Tertiary Care Hospital in Eastern India
Abstract
:1. Introduction
Objective
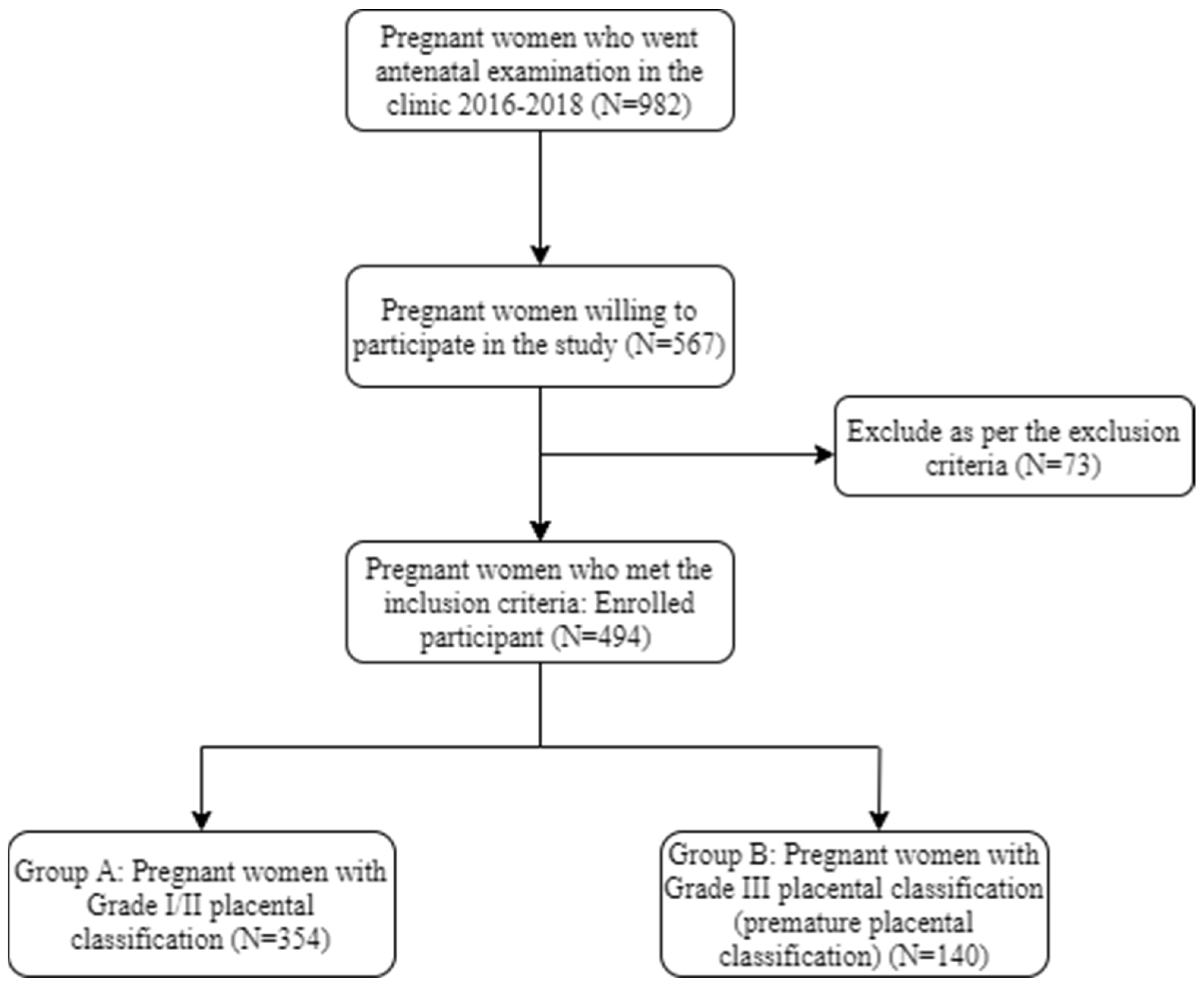
2. Materials and Methods
2.1. Study Design
2.2. Statistical Analysis
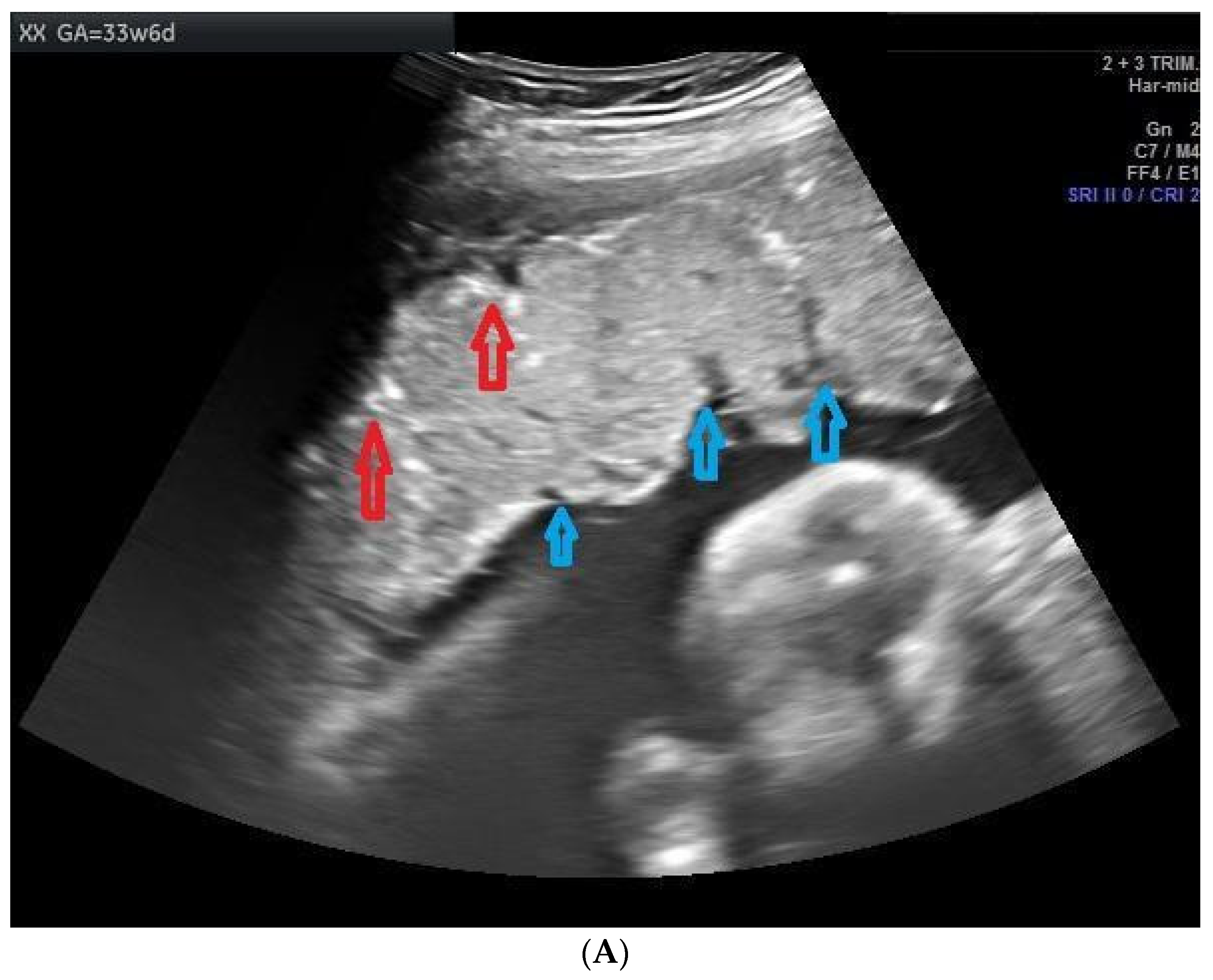
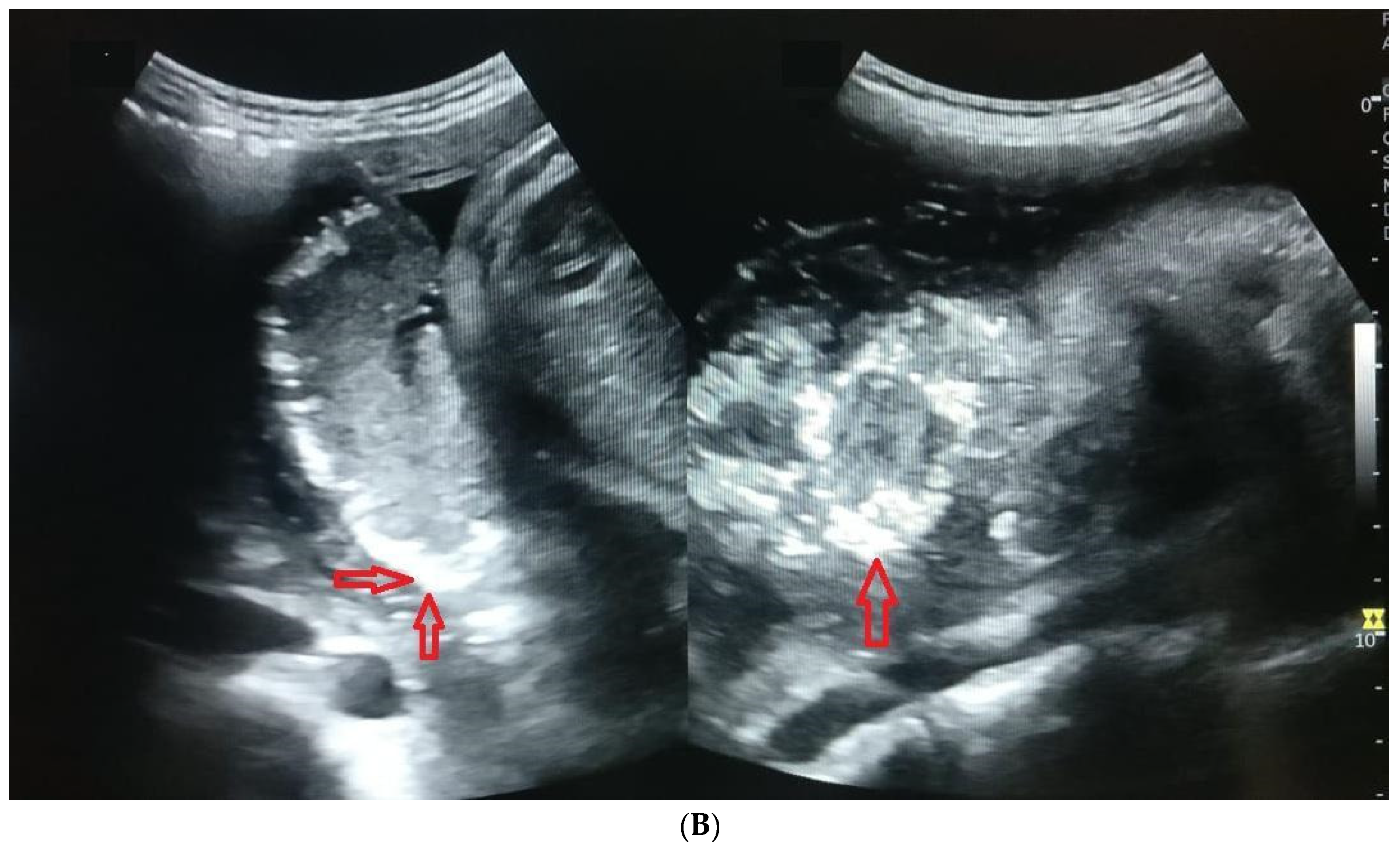
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Harris, R.D.; Alexander, R.D. Ultrasound of the placenta and umbilical cord. In Ultrasonography in Obstetrics and Gynaecology; Callen, P.W., Ed.; W.B. Saunders: Philadelphia, PA, USA, 2000; pp. 602–604. [Google Scholar]
- Nolan, R.L. The placenta, membranes, umbilical cord, and amniotic fluid. In A Practical Guide to Ultrasound in Obstetrics and Gynaecology; Sauerbrei, E.E., Nguyen, K.T., Nolan, R.L., Eds.; LippincottRaven: Philadelphia, PA, USA, 1998; pp. 438–439. [Google Scholar]
- Winsberg, F. Echographic changes with placental ageing. J. Clin. Ultrasound 1973, 1, 52–55. [Google Scholar] [CrossRef]
- Grannum, P.A.; Berkowitz, R.L.; Hobbins, J.C. The ultrasonic changes in the maturing placenta and their relation to fetal pulmonic maturity. Am. J. Obstet. Gynecol. 1979, 133, 915–922. [Google Scholar] [CrossRef]
- Spirt, B.A.; Gorden, L.P. Sonography of the placenta. In Sonography in Obstetrics and Gynecology: Principles and Practice; Fleischer, A.C., Manning, F.A., Jeanty, P., Romero, R., Eds.; McGraw-Hill: New York, NY, USA, 2001; pp. 195–197. [Google Scholar]
- McKenna, D.; Tharmaratnam, S.; Mahsud, S.; Dornan, J. Ultrasonic evidence of placental calcification at 36 weeks’ gestation: Maternal and fetal outcomes. Acta Obstet. Gynecol. Scand. 2005, 84, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Chitlange, S.; Hazari, K.; Joshi, J.; Shah, R.; Mehta, A. Ultrasonographically observed preterm grade III placenta and perinatal outcome. Int. J. Gynecol. Obstet. 1990, 31, 325–328. [Google Scholar] [CrossRef]
- Jamal, A.; Moshfeghi, M.; Moshfeghi, S.; Mohammadi, N.; Zarean, E.; Jahangiri, N. Is preterm placental calcification related to adverse maternal and foetal outcome? J. Obstet. Gynaecol. 2017, 37, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Emmrich, P. Pathology of the placenta. X. Syncytial proliferation, calcification, cysts, pigments and metabolic disorders. Zent. Pathol. 1992, 138, 77–84. (In German) [Google Scholar]
- Ernst, L.; Parkash, V. Placental Pathology in Fetal Bartter Syndrome. Pediatr. Dev. Pathol. 2002, 5, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Dane, B.; Dane, C.; Aksoy, F.; Cetin, A.; Yayla, M. Antenatal Bartter syndrome: Analysis of two cases with placental findings. Fetal Pediatr. Pathol. 2010, 29, 121–126. [Google Scholar] [CrossRef]
- Klaritsch, P.; Haeusler, M.; Karpf, E.; Schlembach, D.; Lang, U. Spontaneous intrauterine umbilical artery thrombosis leading to severe fetal growth restriction. Placenta 2008, 29, 374–377. [Google Scholar] [CrossRef]
- Cunningham, F.G.; Leveno, K.J.; Bloom, S.L.; Hauth, J.C.; Rouse, D.J.; Spong, C.Y. Parturition. In Williams Obstetrics; Cunningham, F.G., Ed.; McGraw-Hill: New York, NY, USA, 2010; pp. 146–147. [Google Scholar]
- Kasznica, J.M.; Petcu, E.B. Placenta calcium pump: Clinical-based evidence. Pediatr. Pathol. Mol. Med. 2003, 22, 223–227. [Google Scholar] [CrossRef]
- Agababov, R.M.; Abashina, T.N.; Suzina, N.E.; Vainshtein, M.B.; Schwartsburd, P.M. Link between the early calcium deposition in placenta and nanobacterial-like infection. J. Biosci. 2007, 32, 1163–1168. [Google Scholar] [CrossRef] [PubMed]
- Pasquinelli, G.; Papadopulos, F.; Nigro, M. Nanobacteria and psammoma bodies: Ultrastructural observations in a case of pathological placental calcification. Ultrastruct. Pathol. 2010, 34, 344–350. [Google Scholar] [CrossRef]
- Hills, D.; Irwin, G.; Tuck, S.; Baim, R. Distribution of placental grade in high-risk gravidas. Am. J. Roentgenol. 1984, 143, 1011–1013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patterson, R.M.; Hayashi, R.H.; Cavazos, D. Ultrasonographically observed early placental maturation and perinatal outcome. Am. J. Obstet. Gynecol. 1983, 147, 773–777. [Google Scholar] [CrossRef]
- Valenzuela, A.B.; Mendez, A.G. Premature aging of the placenta. Ultrasonic diagnosis. Ginecol. Obs. Mex. 1995, 63, 287–292. [Google Scholar]
- Proud, J.; Grant, A.M. Third trimester placental grading by ultrasonography as a test of fetal wellbeing. BMJ 1987, 294, 1641–1644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, L.-Y.; Yu, Y.-H.; Hu, M.-L. Association between ultrasonographic signs of placental premature aging and pregnancy outcome. Di 1 Jun Yi Da Xue Xue Bao = Acad. J. First Med. Coll. PLA 2005, 25, 318–320. (In Chinese) [Google Scholar]
- Kazzi, G.M.; Gross, T.L.; Rosen, M.G.; Jaatoul-Kazzi, N.Y. The relationship of placental grade, fetal lung maturity, and neonatal outcome in normal and complicated pregnancies. Am. J. Obstet. Gynecol. 1984, 148, 54–58. [Google Scholar] [CrossRef]
- Miller JMJr Brown, H.L.; Kissling, G.A.; Gabert, H.A. The relationship of placental grade to fetal size and growth at term. Am. J. Perinatol. 1988, 5, 19–21. [Google Scholar] [CrossRef]
- Vosmar, M.B.J.G.; Jongsma, H.W.; Dongen, P.W.J. The value of ultrasonic placental grading: No correlation with intra-uterine growth retardation or with maternal smoking. J. Périnat. Med. 1989, 17, 137–143. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.Y.; Yu, Y.H.; Hu, M.L. The relationship between placental aging, Doppler indices of umbilical artery and fetal prognosis. J. Pract. Obstet. Gynecol. 2005, 5, 315–318. [Google Scholar]
- Richardus, J.H.; Graafmans, W.C.; Bruin, K.M.V.D.P.-D.; Amelink-Verburg, M.P.; Verloove-Vanhorick, S.P.; MacKenbach, J.P. An European concerted action investigating the validity of perinatal mortality as an outcome indicator for the quality of antenatal and perinatal care. J. Périnat. Med. 1997, 25, 313–324. [Google Scholar] [PubMed]
- Mirza, F.G.; Ghulmiyyah, L.M.; Tamim, H.M.; Makki, M.; Jeha, D.; Nassar, A.H. To Ignore or Not To Ignore Placental Calcifications on Prenatal Ultrasound: A Systematic Review and Meta-analysis. J. Matern. Neonatal Med. 2017, 31, 1–21. [Google Scholar] [CrossRef]
- Agarwal, R.; Tiwari, A.; Wadhwa, N.; Radhakrishnan, G.; Bhatt, S.; Batra, P. Abnormal umbilical artery Doppler velocimetry and placental histopathological correlation in fetal growth restriction. S. Afr. J. Obstet. Gynaecol. 2017, 23, 12. [Google Scholar] [CrossRef] [Green Version]
| Characteristics | Group A (Those without Premature Placental Calcification) n = 354 | Group B (Those with Premature Placental Calcification) n = 140 | p-Value |
|---|---|---|---|
| Maternal age (y) | 26.79 ± 2.05 | 26.36 ± 2.34 | >0.05 |
| Body mass index (kg/m2) | 23.9 ± 2.05 | 24.3 ± 1.95 | >0.05 |
| Socio-Economic status | |||
| Lower Lower middle Upper middle | 82 (23.1%) 192 (54.2%) 80 (22.7%) | 36 (25.7%) 71 (50.7%) 24 (23.6%) | >0.05 |
| Parity | |||
| Primigravida Multigravida | 148 (41.8%) 206 (58.2%) | 48 (34.2%) 92 (65.8%) | >0.05 |
| Total n = 494 | Group A (Those without Premature Placental Calcification) n = 354 | Group B (Those with Premature Placental Calcification) n = 140 | p-Value | Relative Risk (95% CI) | |
|---|---|---|---|---|---|
| Pre-eclampsia | 99 (20.0) | 59 (16.7) | 40 (28.6) | 0.002 | 1.71 (1.20 to 2.43) |
| GDM | 58 (11.7) | 40 (11.3) | 18 (12.8) | 0.626 | 1.13 (0.67 to 1.91) |
| At least one abnormal Doppler index (uterine or umbilical or fetal middle cerebral arterial index) * | 124 (25.1) | 74 (20.9) | 50 (35.7) | 0.001 | 1.70 (1.26 to 2.30) |
| PPROM | 147 (29.7) | 97 (27.4) | 50 (35.7) | 0.063 | 1.30 (0.98 to 1.72) |
| ANEMIA | 25 (5.1) | 15 (4.2) | 10 (7.1) | 0.1871 | 1.68 (0.77 to 3.66) |
| Obstetrics cholestasis | 44 (8.9) | 24 (6.8) | 20 (14.3) | 0.009 | 2.1 (1.20 to 3.68) |
| FGR | 69 (14.0) | 39 (11.0) | 30 (21.4) | 0.002 | 1.94 (1.26 to 3.00) |
| Hemoglobinopathy | 15 (03.0) | 10 (2.8) | 5 (3.6) | 0.663 | 1.26 (0.44 to 3.63) |
| Placental abruption | 53 (9.3) | 29 (8.1) | 24 (17.1) | 0.004 | 2.09 (1.26 to 3.46) |
| PPH | 45 (9.1) | 31 (8.75) | 14 (10) | 0.664 | 1.14 (0.62 to 2.08) |
| Maternal transfer to ICU | 78 (15.7) | 49 (13.8) | 29 (20.7) | 0.057 | 1.49 (0.98 to 2.26) |
| Total (n = 494) | Group A (Those without Prematureplacental Calcification) n = 354 | Group B (Those with Premature Placental Calcification) n = 140 | p-Value | Relative Risk (95% CI) | |
|---|---|---|---|---|---|
| Preterm birth | 70 (14.2) | 45 (12.7) | 25 (17.9) | 0.139 | 1.40 (0.89 to 2.19) |
| Low APGAR | 53 (08.9) | 29 (6.8) | 24 (14.3) | 0.004 | 2.10 (1.26 to 3.46) |
| NICU Admission | 33 (05.1) | 18 (4.2) | 15 (7.1) | 0.026 | 2.10 (1.09 to 4.06) |
| Abnormal CTG | 65 (13.15) | 39 (11.0) | 26 (18.5) | 0.024 | 1.68 (1.06 to 2.65) |
| Meconium-stained liquor | 56 (11.3) | 31 (8.7) | 25 (17.8) | 0.004 | 2.03 (1.25 to 3.32) |
| LBW | 124 (25.1) | 85 (24.01) | 46 (32.8) | 0.040 | 1.36 (1.01 to 1.84) |
| Birth weigt (g) | 2714.25 + 496.62 | 2575.04 + 606.75 | 0.008 | ||
| Gestation at delivery | 38 wk 3 d + 1 wk 5 d | 38 wk 0 d + 1 wk 6 d | 0.056 | ||
| Delivery Mode (LSCS) | 172 (34.8) | 118 (33.3) | 54 (38.5) | 0.263 | 1.11 (0.89 to 1.49) |
| Neonatal death | 15 (03.0) | 10 (2.8) | 5 (3.6) | 0.663 | 1.26 (0.44 to 3.63) |
| Doppler Characteristics | Group A (Those without Premature Placental Calcification) n = 354 (%) | Group B (Those with Premature Placental Calcification n = 140 (%) | p-Value |
|---|---|---|---|
| UMB PI ≥ 95th, | 32 (9.03) | 27 (19.2) | 0.003 |
| MCA PI ≤ 5th, | 29 (8.19) | 25 (17.8) | 0.003 |
| CPR ≤ 5th, | 27 (7.62) | 20 (14.2) | 0.027 |
| Mean uterine PI ≥ 95th | 26 (7.34) | 18 (12.8) | 0.078 |
| Unilateral notch in uterine artery. | 35 (9.88) | 20 (14.2) | 0.203 |
| bilateral notch in uterine artery. | 22 (6.21) | 9 (6.4) | 1.000 |
| AREDF | 15 (2.82) | 13 (5.71) | 0.049 |
| At least one abnormal Doppler index (uterine or umbilical or fetal middle cerebral arterial index) * | 74 (20.9) | 50 (35.7) | 0.001 |
| Indications | Group A (Those without Premature Placental Calcification) n = 354 (%) | Group B (Those with Premature Placental Calcification n = 140 (%) |
|---|---|---|
| CPD | 25 (7.06) | 12 (8.57) |
| Fetal Distress | 29 (8.19) | 16 (11.4) |
| CDMR | 24 (6.77) | 10 (7.14) |
| Antepartum hemorrhage | 10 (2.82) | 4 (2.85) |
| Obstructed Labour | 6 (1.69) | 2 (1.42) |
| Severe preeclampsia | 24 (6.77) | 10 (7.14) |
| Sensitivity (%) | Specificity (%) | Positive Predictive Value (%) | Negative Predictive Value (%) | |
|---|---|---|---|---|
| preeclampsia | 40.4 | 74.7 | 28.6 | 83.3 |
| FGR | 43.5 | 74.1 | 21.4 | 89 |
| PLACENTAL ABRUPTION | 45.3 | 73.7 | 17.1 | 91.5 |
| Low APGAR | 45.3 | 73.7 | 17.1 | 91.5 |
| NICU admission | 45.5 | 72.9 | 10.7 | 94.9 |
| Abnormal CTG | 40 | 73.4 | 18.6 | 89 |
| Meconium-stained liquor | 44.6 | 73.7 | 17.9 | 91.7 |
| LBW | 35.1 | 74.1 | 32.9 | 76 |
| Abnormal Doppler parameters | 40.3 | 75.7 | 35.7 | 79.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dash, S.; Das, B.; Panda, S.R.; Rajguru, M.; Jena, P.; Mishra, A.; Rath, S.K. Perinatal Outcomes in Premature Placental Calcification and the Association of a Color Doppler Study: Report from a Tertiary Care Hospital in Eastern India. Clin. Pract. 2021, 11, 841-849. https://doi.org/10.3390/clinpract11040099
Dash S, Das B, Panda SR, Rajguru M, Jena P, Mishra A, Rath SK. Perinatal Outcomes in Premature Placental Calcification and the Association of a Color Doppler Study: Report from a Tertiary Care Hospital in Eastern India. Clinics and Practice. 2021; 11(4):841-849. https://doi.org/10.3390/clinpract11040099
Chicago/Turabian StyleDash, Sudarshan, Banya Das, Soumya Ranjan Panda, Monalisa Rajguru, Pramila Jena, Abheepsa Mishra, and Sudhanshu Kumar Rath. 2021. "Perinatal Outcomes in Premature Placental Calcification and the Association of a Color Doppler Study: Report from a Tertiary Care Hospital in Eastern India" Clinics and Practice 11, no. 4: 841-849. https://doi.org/10.3390/clinpract11040099
APA StyleDash, S., Das, B., Panda, S. R., Rajguru, M., Jena, P., Mishra, A., & Rath, S. K. (2021). Perinatal Outcomes in Premature Placental Calcification and the Association of a Color Doppler Study: Report from a Tertiary Care Hospital in Eastern India. Clinics and Practice, 11(4), 841-849. https://doi.org/10.3390/clinpract11040099







