1. Introduction
Pancreatic adenocarcinoma is a leading cause of cancer-related mortality worldwide [
1,
2]. Over half of the patients have metastases on presentation, whereas only 15% of patients have resectable disease [
3]. Although complete surgical resection is the only curative treatment option for this cancer entity, even after radical surgery prognosis remains poor [
4].
Cancer surgery is often associated with systemic release of tumor cells, particularly during the vulnerable perioperative period [
5,
6]. Hereby, three factors of stress response are considered to impair cellular immunity: the response to surgical trauma, to general anesthesia, and to opioid analgesia [
7]. Furthermore, general anesthesia itself is known to impair various immune components such as neutrophil, macrophage, dendritic cell, T-cell, and NK-cell functions [
8,
9,
10,
11,
12]. In addition, opioids given during anesthesia and for postoperative pain control inhibit humoral and cellular immune response, natural killer cell activity, cytokine expression and phagocytic activity [
12,
13].
Beneficial effects of regional anesthesia have been demonstrated on the mentioned factors suppressing immune function. Neuroaxial anesthesia decreases the neuroendocrine stress response to surgical tissue injury by blocking afferent noxious input as well as efferent sympathetic transmission [
14,
15]. Thus, it reduces the need for general anesthesia, minimizes perioperative opioid requirement [
16] and improves the short-term perioperative outcome [
17,
18,
19,
20].
Accordingly, it has been hypothesized that the incidence of cancer recurrence is decreased after surgery with regional anesthesia and analgesia compared to surgery with general anesthesia and opioid analgesia alone. To date, beneficial effects of regional anesthesia on oncological outcome have been associated with other tumor entities such as prostatic [
21], breast [
22], bladder [
23], colorectal [
24] and gastric cancer [
25]. Whereas these trials have suggested a positive effect of regional anesthesia on cancer recurrence, others have not found any significant improvement in studies including patients with varying abdominal cancer surgery [
26,
27]. However, the tumor entities of prostatic and breast cancer, which have mainly been shown to be associated with a reduced recurrence when regional anesthesia was applied [
21,
22], are characterized by a comparably slow tumor progression, frequently depending on hormone receptor status. To date, the prognostic effect of PDA on pancreatic cancer has not been investigated.
An improved understanding of the effects of perioperative management could lead to better oncological outcomes. Therefore, we hypothesized that the administration of PDA may also improve overall survival in pancreatic adenocarcinoma. 127 consecutive patients with pancreatic ductal adenocarcinoma that had undergone radical tumor resection within five years at our institution were analyzed for overall survival in a retrospective observational study.
2. Materials and Methods
All collected data adhered to the guidelines established by the Declaration of Helsinki and has been approved by the ethics committee of the Medical Faculty, Heinrich-Heine-University, Düsseldorf, Germany (2020-848).
This is an observational retrospective study to analyse overall survival as a primary endpoint after a median follow-up time of 17.26 months. Clinical data were collected from patients’ medical records, compiled into an Excel-file database and analyzed retrospectively. The following data were collected: demographic parameters (age, gender, ASA-score), tumor characteristics (T-stage, N-stage, grading), treatment characteristics (surgical procedures, type of anesthesia, operating time, blood transfusion).
2.1. Anesthesia
All patients underwent surgery under general anesthesia (GA). GA was induced with intravenous sufentanil (0.1–0.5 µg kg−1), thiopental (3–7 mg kg−1), cisatracurium (0.1 mg kg−1) and maintained with sevoflurane (endtidal concentration 1.2–2.8 Vol.%). Additional boli of sufentanil were administered as required.
In addition to GA, thoracic peridural anesthesia (PDA) is usually recommended for patients undergoing complex viscero-surgical procedures at our institution. Patients who did not give their consent, patients with coagulopathies, local infections or other contraindications, such as a high probability to undergo complex vascular reconstruction with consecutive anticoagulation, were excluded from PDA. Prior to induction of GA a peridural catheter was inserted at the thoracic level (T6–T10). Typically, a bolus of 15–150 mg of ropivacaine (0.375–0.75%) was given initially. During surgery PDA was maintained by a continuous infusion of ropivacaine (7.5–50 mg/h, 0.375%), supplemented by peridural sufentanil (total dose 15–135 µg) at the discretion of the attending anesthesiologist.
2.2. Surgical Procedures
Surgery always aimed at complete tumor resection without microscopic residues. Surgical procedures included partial pancreatoduodenectomy, and distal and total pancreatectomy. Partial pancreatoduodenectomy was usually performed as a pylorus-preserving procedure with a three-loop reconstruction. Lymphadenectomy routinely included clearance of the peripancreatic, hepatoduodenal, celiac and interaortocaval lymph nodes. All procedures were carried out by a transverse laparotomy.
2.3. Postoperative Pain Management
Postoperative analgesia was monitored by an interdisciplinary team of surgeons and anesthesiologists. All patients, except those with an established allergic history or intolerance, received metamizole at a rate of 1 g every 6 h. As a routine, patients with peridural catheters received continuous peridural ropivacaine 0.2% with infusion rates of 4–12 mL/h. In case of insufficient analgesia for mobilisation or respiratory physiotherapy patients received intravenous piritramide up to 30 mg per day. Patients without peridural catheters were treated with intravenous piritramide through patient-controlled analgesia or per physician’s order in case of poor compliance. Furthermore, all patients received intravenous paracetamol up to 4 g per day and/or etoricoxib 60 to 120 mg per day, if required. Peridural catheters were removed in case of inadequate function, suspected infection, if no longer required by the patient or at the latest on POD 7.
2.4. Follow-Up
Follow-up was performed every 3 months for the first two years after surgery, every 6 months until 5 years after surgery and every year thereafter. Follow-up examinations included a physical examination and medical history, an abdominal ultrasound, a complete laboratory work-up, and a chest and abdominal computed tomography. If patients were not followed at our institution, a study nurse contacted the referring physician at the same time intervals mentioned above to collect the data.
2.5. Statistical Analysis
Descriptive statistical analysis and graphing were performed using MS Excel from Microsoft (Redmond, Washington, DC, USA) and JMP 14.1 from SAS Institute Inc. (Cary, NC, USA). All results were expressed as mean ± standard deviation. Statistical significance was determined by Student’s t-test and the χ2 test for two-by-two frequency tables.
A Cox’s proportional hazard regression model was applied to investigate the association between overall survival and perioperative administration of PDA, adjusted by the following covariates: age, vascular invasion, sex, operation time, T-stage, N-stage, grading, administration of adjuvant chemotherapy and ASA-score. To achieve a parsimonious model, a model selection procedure was established by choosing the regression model with minimum AIC (Akaike Information Criterion) [
28]. The model class runs from the model including all covariates to the model with a constant term. Second order interactions were further analyzed between the selected variables in the model. The stability of this model selection process was validated by taking 500 bootstrap samples and by repeating the full selection process in each sample [
29]. Results of the model are presented in terms of hazard ratios and their 95% confidence intervals calculated from the Cox’s regression model. We tested the crucial assumption that the hazards are proportional over time by using the Schoenfeld residuals.
We performed a retrospective sample size determination for a two-sided log-rank test, which is equivalent to the test calculated from the proportional hazard Cox’s regression. We took a significance level of 5% two-sided and corrected by Bonferroni for sub-group analysis, i.e., the final significance level for the sample size determination was 2.5%. We utilized a power of 80% and expected HR = 0.40. Then, when the sample size in each group is 60, with a total number of events required, E, of 45, a 0.025 level two-sided log-rank test for equality of survival curves will have 80% power to detect the difference between two curves with a constant hazard ratio of 0.4. In our data we have a total of n = 96 patients, 24 less patients than required, and E = 51 events, 6 more events as indicated in the sample size determination. Clearly, this is a retrospective explorative study, but the number of events and the number of patients is close to the required number by the sample size determination.
The statistical analysis was performed using the statistical software R version 4.0.2 (R Core Team, Vienna, Austria, 2020) [
30]. We used reporting tools based on the standards of replicable research using the R’s package “knitr” [
31]. The analysis based on the proportional hazard Cox’s regression and the estimation of the C-statistics was performed with the R’s package “survival” [
32].
3. Results
Medical records of a total of 127 consecutive patients with pancreatic ductal adenocarcinoma that had undergone radical tumor resection at our institution were analyzed in a retrospective observational study. 19 patients (15%) with residual tumor (R1 or R2 resections) as well as 9 patients (6.3%) who died perioperatively (60 days) were excluded. One of them died perioperatively after R1 resection. Accordingly, 100 patients remained in the study for further analyses.
During follow-up, two patients died for other reasons than tumor recurrence and were excluded from the prognostic analysis, which is based on the remaining 98 patients.
Our patient collective consisted of 48 (49%) male and 50 (51%) female patients, the median age was 65 years (range 41–85 years).
Patient and tumor characteristics are summarized in
Table 1.
The majority of the patients (62.2%) had a reduced physical health status with an ASA score of 3 or more. Most patients were diagnosed at an advanced tumor stage. 89.8% presented with large primary tumors (pT3 or pT4) whereas 76.5% had lymph node involvement (pN1). 53.1% of the patients had poorly differentiated primary tumors.
3.1. Surgical Procedure
89 patients (90.8%) underwent partial pancreatoduodenectomy, 5 patients (5.1%) had distal pancreatectomy, and 4 patients (4.1%) required total pancreatectomy (
Table 2).
In 26 patients (26.5%) infiltration of neighbouring large vessels—usually the portal vein or the superior mesenteric vein, respectively—was found. In order to achieve clear resection margins, vascular resection with subsequent reconstruction was performed in all of these cases. One patient received an additional reconstruction of the celiac trunk, and another patient had a segmental resection of the common hepatic artery, which was reconstructed by termino-terminal anastomosis. The mean duration of surgery was 413 min with a range of 210–755 min and a standard deviation of 103 min. For statistical analysis patients were divided into two groups with duration of surgery of either <420 min (59 patients) or >420 min (39 patients). The majority of patients (90%) showed a limited intraoperative blood loss with a maximum transfusion of 4 packed red blood cell concentrates. Four patients received 5–8 red blood cell concentrates and four patients needed more than 8 transfusions of packed red blood cell concentrates during surgery (
Table 2).
3.2. Anesthesia
In 28 (28.6%) of the patients, surgery was performed under GA, whereas 70 (71.4%) received GA + PDA. All patients received intravenous sufentanil during the procedure. If required to attain sufficient analgesia, additional boli of sufentanil were given peridurally. There was a wide range of overall intraoperative dosages of opioids varying between 15 and 150 µg (data not shown).
3.3. Adjuvant Therapy
All patients were assessed by a multidisciplinary tumor board consisting of oncologic surgeons, oncologists, radiotherapists, pathologists and radiologists. According to the recommendation of the tumor board, 79 patients (80.6%) received adjuvant chemotherapy (
Table 2).
3.4. Survival Data
Mean follow-up time after pancreatic resection was 17.26 months, ranging from 3 to 66 months. Overall 1-, 3- and 5-year survival rates after resection were 72.29 months and 17%, respectively, without differences between groups (GA only vs. PDA + GA).
3.5. Prognostic Parameters
The assessment of parameters with potential impact on prognosis included T-stage, N-stage, grading, operating time, age, ASA classification and amount of red blood cell concentrates transfused. With respect to clinic-pathological parameters, our results did not show any statistical significance between the two groups (PDA + GA vs. GA only) (
Table 3).
Survival data analyzed with respect to prognostic data are summarized in
Table 4. Grading was found to be of prognostic significance at univariate analysis. Patients with well-differentiated primary tumors (G
1–2) showed a mean overall survival of 35.15 months compared to 22.86 months in patients with poor tumor differentiation (
p = 0.030). Tumor involvement of the resected lymph nodes (pN1) was also associated with worse prognosis, but the difference in overall survival time (38.48 vs. 23.25 months) did not reach statistical significance (
p = 0.057).
In this analysis the application of PDA in addition to GA was not found to be a significant prognostic parameter concerning overall survival (
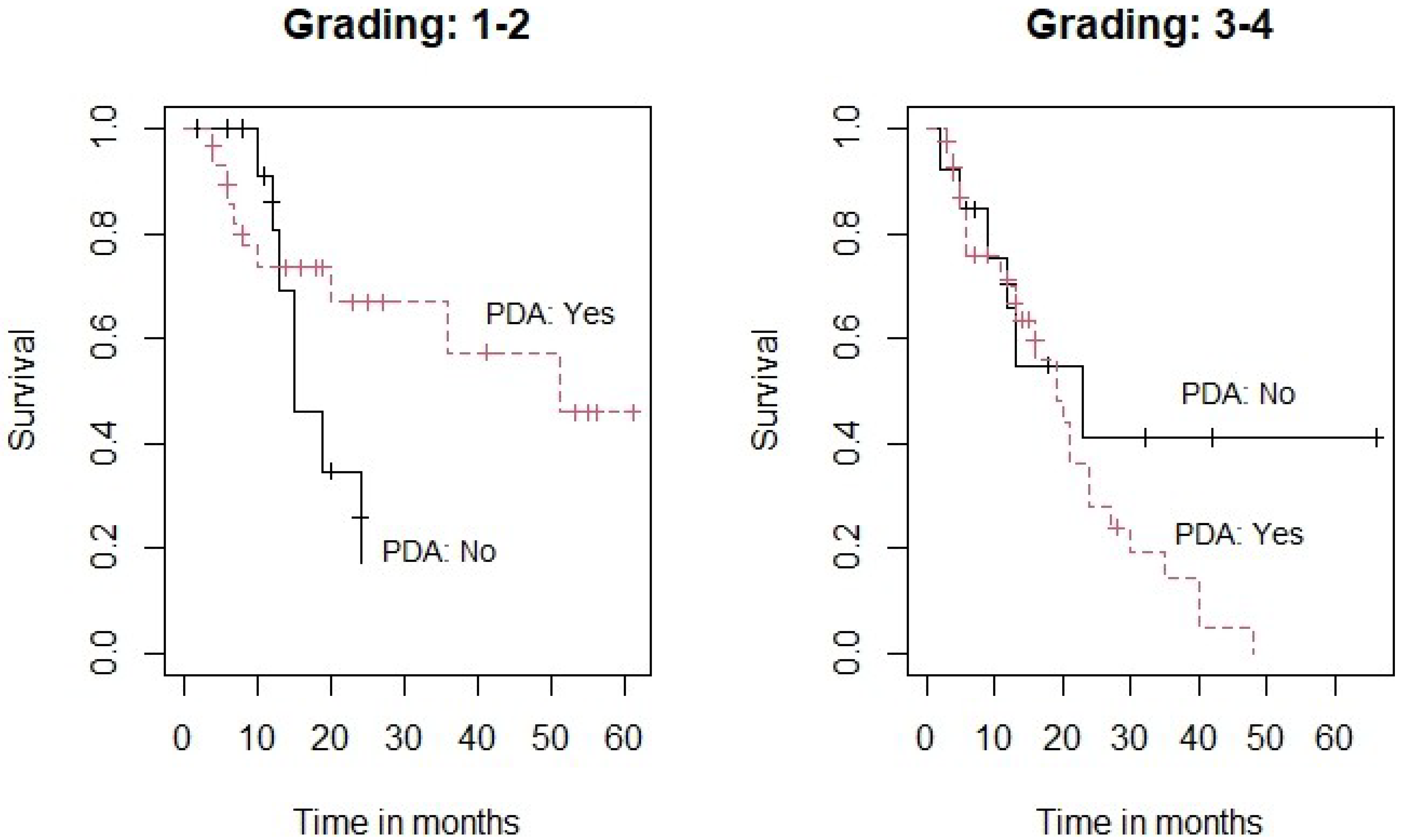
Table 4). However, since the grading showed a certain impact on the results, we decided to perform a subgroup analysis investigating the potential impact of PDA on the grading subgroups G
1–2 vs. G
3–4 (
Figure 1).
Figure 1 demonstrates Kaplan-Meier survival curves for the two subgroups. Subgroup analysis showed that the effect of PDA was not statistically significant for the subgroup of patients with less differentiated tumors (G
3–4) (
p = 0.191) (Right panel). However, there was a clear trend towards an improved survival when PDA was used in the subgroup of patients with a better tumor differentiation (G
1–2) (
p = 0.069) (Left panel).
4. Discussion
The use of peridural anesthesia and its association with improved survival after cancer surgery has been described for different tumor entities in multiple retrospective studies [
33,
34,
35]. Initial studies on the role of PDA on cancer outcome have stated a reduction in tumor recurrence and metastases of breast and prostatic cancer when surgery was performed under general anesthesia combined with regional analgesia. This prognostic impact was attributed to a lesser impairment of immune surveillance, caused by a decrease of the neuroendocrine stress response to surgical trauma [
21,
22].
Both prostate cancer as well as breast cancer are frequently dependent on hormone receptor status and are characterized by a rather prolonged disease course. However, to date there is less evidence on the prognostic impact of peridural analgesia in malignant entities with a more aggressive tumor biology. Its effect for pancreatic cancer remains mostly unexplored. To the best of our knowledge, this is the first study investigating the role of peridural anesthesia on the outcome of patients with pancreatic adenocarcinoma, which is known for its very aggressive biological potential. Reported 5-year survival rates after surgery range between 4–25% [
36,
37,
38]. In our collective the 5-year survival time was calculated at 17%, which is in line with other reports, considering the large proportion of patients with locally advanced disease and lymph node involvement.
Collectively, our study revealed no significant prognostic benefit when PDA was administered, yet a subgroup analysis of patients with better tumor differentiation (G1–2) showed that PDA in these patients was correlated with an improved overall survival. In contrast, no prognostic impact was observed in patients with poorly differentiated tumors.
Interestingly, further studies have reported positive prognostic effects associated with PDA in certain subgroups. In a study analyzing the effect of PDA in patients with colorectal cancer, a prognostic benefit for a subgroup of patients with rectal cancer was found [
39]. Furthermore, an improvement in survival in patients receiving peridural anesthesia for colorectal cancer surgery was shown [
40], but solely for metastasis-free patients for 1.5 years. Likewise for colorectal cancer, a potential benefit for patients > 64 years was observed [
41], and prolonged survival, yet no impact on cancer recurrence [
24] was described. This corresponds to our findings. The effect may depend on the specific type, location or aggressiveness of the tumor.
Generally, the data reported on this subject is characterized by rather controversial results. In addition to prostatic and breast cancer, several analyses could show a significant association of epidural anesthesia and improved overall survival for patients with gastric cancer [
25,
42,
43,
44,
45]. A study including patients with bladder cancer revealed an increased five-year survival in patients who received regional anesthesia, although this association was not significant [
46]. Investigating the effect of perioperative dexamethasone on survival after resection of pancreatic adenocarcinoma, Call et al. found increased survival when PDA was used [
47].
In contrast, no benefit of PDA on overall survival or cancer recurrence was found in patients with colorectal [
48], prostatic [
49] or bladder cancer [
8,
50]. Evaluating patients with major abdominal surgery for cancer, further studies [
8,
26,
27,
45] could not observe any significant influence of anaesthetic technique on patients’ outcome, either. Yet, in these collectives, the patients had various surgical procedures including gastrectomy, pancreatectomy, colectomy, hepatectomy, cystectomy, nephrectomy or prostatectomy. Thus, a potential effect of PDA regarding cancer recurrence or overall survival might be multifactorial and therefore inhomogenous.
In summary, our findings, as well as several of the above-mentioned results of previous studies, suggest that PDA might have a favourable effect on long-term prognosis of patients with solid malignant tumors. However, it remains unclear if this effect is also present in patients with more aggressive and poorly differentiated primary tumors. Presumably, these patients could benefit from a reduced preoperative stress response and opioid requirements.
The limitation of our study includes the retrospective observational nature of the analysis. Moreover, ropivacaine and sufentanil doses were selected at the discretion of the attending anesthesiologist and were not administered according to a standardised protocol. The received amount of PDA may therefore differ between patients.
5. Conclusions
Overall, we could not show any significant prognostic benefit. Consistent with previously published data, which show prognostic impacts of PDA within subgroups, our findings suggest an improved survival for the subgroup of patients with better tumor differentiation (G1–2). Therefore, patients with better tumor differentiation of pancreatic adenocarcinoma may benefit with respect to overall survival.
The assumption that regional anesthesia may have an effect on outcome after oncological surgery is supported by numerous retrospective clinical studies.
To date, a variety of observational clinical data have been released—the overall conflicting findings indicate the need for further evidence from large prospective, randomized-controlled trials.
Author Contributions
Study conception, design and overall analysis and interpretation of data, drafting, revising the manuscript (A.A., N.L.-T., A.R., W.T.K., P.K.), Data analysis and interpretation (A.A., N.L.-T., P.E.V., S.R.), Manuscript preparation (A.A., N.L.-T., A.R., N.P.), Conceptual contributions and manuscript revision (A.A., N.L.-T., A.R., C.F.E., W.T.K., P.K.), the doctoral thesis of S.R. was based on the data of this study. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
All collected data were adhered to the guidelines established by the Declaration of Helsinki and have been approved by the ethics committee of the Medical Faculty, Heinrich-Heine-University, Düsseldorf, Germany (2020-848).
Informed Consent Statement
Not applicable.
Data Availability Statement
Supplementary data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ASA | American Society of Anesthesiologists |
| GA | general anaesthesia |
| NK cells | natural killer cells |
| PDA | peridural anesthesia |
| POD | postoperative day |
| T cells | thymus cells |
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [Green Version]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [Green Version]
- Mancuso, A.; Calabrò, F.; Sternberg, C.N. Current therapies and advances in the treatment of pancreatic cancer. Crit. Rev. Oncol. 2006, 58, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Staley, C.A.; Lee, J.E.; Cleary, K.R.; Abbruzzese, J.L.; Fenoglio, C.J.; Rich, T.A.; Evans, U.B. Preoperative chemoradiation, pancreaticoduodenectomy, and intraoperative radiation therapy for adenocarcinoma of the pancreatic head. Am. J. Surg. 1996, 171, 118–125. [Google Scholar] [CrossRef]
- Bundscherer, A.; Malsy, M.; Gebhardt, K.; Metterlein, T.; Plank, C.; Wiese, C.; Gruber, M.; Graf, B. Effects of ropivacaine, bupivacaine and sufentanil in colon and pancreatic cancer cells in vitro. Pharmacol. Res. 2015, 95–96, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Buggy, D.; Smith, G. Epidural anaesthesia and analgesia: Better outcome after major surgery? BMJ 1999, 319, 530–531. [Google Scholar] [CrossRef] [PubMed]
- Ben-Eliyahu, S. Epidural anaesthesia and analgesia: Better outcome after major surgery? Growing evidence suggests so. Lancet Oncol. 2002, 3, 578–579. [Google Scholar]
- Chipollini, J.; Alford, B.; Boulware, D.C.; Forget, P.; Gilbert, S.M.; Lockhart, J.L.; Pow-Sang, J.M.; Sexton, W.J.; Spiess, P.E.; Poch, M.A.; et al. Epidural anesthesia and cancer outcomes in bladder cancer patients: Is it the technique or the medication? A matched-cohort analysis from a tertiary referral center. BMC Anesthesiol. 2018, 18, 157. [Google Scholar] [CrossRef] [PubMed]
- Dubowitz, J.A.; Sloan, E.; Riedel, B.J. Implicating anaesthesia and the perioperative period in cancer recurrence and metastasis. Clin. Exp. Metastasis 2017, 35, 347–358. [Google Scholar] [CrossRef]
- Wall, T.; Sherwin, A.; Ma, D.; Buggy, D. Influence of perioperative anaesthetic and analgesic interventions on oncological outcomes: A narrative review. Br. J. Anaesth. 2019, 123, 135–150. [Google Scholar] [CrossRef]
- Cata, J.P.; Hernandez, M.; Lewis, V.O.; Kurz, A. Can Regional Anesthesia and Analgesia Prolong Cancer Survival After Orthopaedic Oncologic Surgery? Clin. Orthop. Relat. Res. 2014, 472, 1434–1441. [Google Scholar] [CrossRef] [Green Version]
- Sessler, D.I. Does regional analgesia reduce the risk of cancer recurrence? A hypothesis. Eur. J. Cancer Prev. 2008, 17, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Cata, J. Impact of anesthesia on cancer recurrence. Rev. Española Anestesiol. Y Reanim. 2015, 62, 570–575. [Google Scholar] [CrossRef]
- Kim, R. Anesthetic technique and cancer recurrence in oncologic surgery: Unraveling the puzzle. Cancer Metastasis Rev. 2017, 36, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Kim, R. Effects of surgery and anesthetic choice on immunosuppression and cancer recurrence. J. Transl. Med. 2018, 16, 8. [Google Scholar] [CrossRef]
- Byrne, K.; Levins, K.J.; Buggy, D.J. Can anesthetic-analgesic technique during primary cancer surgery affect recurrence or metastasis? Can. J. Anesth./J. Can. D’anesthésie 2015, 63, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Beattie, W.S.; Badner, N.H.; Choi, P. Epidural Analgesia Reduces Postoperative Myocardial Infarction: A Meta-Analysis. Anesthesia Analg. 2001, 93, 853–858. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, S.; Yan, M. Combined General/Epidural Anesthesia (Ropivacaine 0.375%) Versus General Anesthesia for Upper Abdominal Surgery. Anesth. Analg. 2008, 106, 1562–1565. [Google Scholar] [CrossRef]
- Rodgers, A.; Walker, N.; Schug, S.; McKee, A.; Kehlet, H.; van Zundert, A.; Sage, D.; Futter, M.; Saville, G.; Clark, T.; et al. Reduction of postoperative mortality and morbidity with epidural or spinal anaesthesia: Results from overview of randomised trials. BMJ 2000, 321, 1493. [Google Scholar] [CrossRef] [Green Version]
- Wijeysundera, D.N.; Beattie, W.S.; Austin, P.; E Hux, J.; Laupacis, A. Epidural anaesthesia and survival after intermediate-to-high risk non-cardiac surgery: A population-based cohort study. Lancet 2008, 372, 562–569. [Google Scholar] [CrossRef]
- Biki, B.; Mascha, E.; Moriarty, D.C.; Fitzpatrick, J.M.; Sessler, D.I.; Buggy, D.J. Anesthetic technique for radical prostatectomy surgery affects cancer recurrence: A retrospective analysis. Anesthesiology 2008, 109, 180–187. [Google Scholar] [CrossRef] [Green Version]
- Exadaktylos, A.K.; Buggy, D.J.; Moriarty, D.C.; Mascha, E.; Sessler, D.I. Can anesthetic technique for primary breast cancer surgery affect recurrence or metastasis? Anesthesiology 2006, 105, 660–664. [Google Scholar] [CrossRef] [Green Version]
- Lusty, A.J.; Hosier, G.W.; Koti, M.; Chenard, S.; Mizubuti, G.B.; Jaeger, M.; Siemens, D.R. Anesthetic technique and oncological outcomes in urology: A clinical practice review. Urol. Oncol. Semin. Orig. Investig. 2019, 37, 845–852. [Google Scholar] [CrossRef]
- Cummings, K.C., 3rd; Xu, F.; Cummings, L.C.; Cooper, G.S. A comparison of epidural analgesia and traditional pain management effects on survival and cancer recurrence after colectomy: A population-based study. Anesthesiology 2012, 116, 797–806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Wang, L.; Chen, H.; Xu, Y.; Zheng, X.; Wang, G. The effects of intra- and post-operative anaesthesia and analgesia choice on outcome after gastric cancer resection: A retrospective study. Oncotarget 2017, 8, 62658–62665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Binczak, M.; Tournay, E.; Billard, V.; Rey, A.; Jayr, C. Major abdominal surgery for cancer: Does epidural analgesia have a long-term effect on recurrence-free and overall survival? Ann. Françaises D’anesthésie Réanimation 2013, 32, e81–e88. [Google Scholar] [CrossRef] [PubMed]
- Myles, P.S.; Peyton, P.; Silbert, B.; Hunt, J.; Rigg, J.R.A.; Sessler, D.; The ANZCA Trials Group. Investigators Perioperative epidural analgesia for major abdominal surgery for cancer and recurrence-free survival: Randomised trial. BMJ 2011, 342, d1491. [Google Scholar] [CrossRef] [Green Version]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2013. [Google Scholar]
- Efron, B. Estimation and Accuracy After Model Selection. J. Am. Stat. Assoc. 2014, 109, 991–1007. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 3 August 2021).
- Xie, Y. Dynamic Documents with R and Knitr; Informa UK Limited: London, UK, 2016. [Google Scholar]
- Therneau, T. A Package for Survival Analysis in R. R Package Version 3.2-11. 2021. Available online: https://CRAN.R-project.org/package=survival (accessed on 3 August 2021).
- Ogawa, K.; Hirai, M.; Katsube, T.; Murayama, M.; Hamaguchi, K.; Shimakawa, T.; Naritake, Y.; Hosokawa, T.; Kajiwara, T. Suppression of cellular immunity by surgical stress. Surgery 2000, 127, 329–336. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, X.-R.; Yang, H. Effects of combined epidural and general anesthesia on intraoperative hemodynamic responses, postoperative cellular immunity, and prognosis in patients with gallbladder cancer: A randomized controlled trial. Medicine 2017, 96, e6137. [Google Scholar] [CrossRef]
- Stewart, T.J.I.; Abrams, S. How tumours escape mass destruction. Oncogene 2008, 27, 5894–5903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haller, G.; Myles, P. Regional Block and Cancer Recurrence: Too Early to Tell. Anesthesiology 2007, 107, 354. [Google Scholar] [CrossRef] [Green Version]
- Schnelldorfer, T.; Ware, A.L.; Sarr, M.G.; Smyrk, T.C.; Zhang, L.; Qin, R.; Gullerud, R.E.; Donohue, J.H.; Nagorney, D.M.; Farnell, M.B. Long-term survival after pancreatoduodenectomy for pancreatic adenocarcinoma: Is cure possible? Ann Surg. 2008, 247, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.K.; Southekal, S.; Guda, C. Survival Analysis of Multi-Omics Data Identifies Potential Prognostic Markers of Pancreatic Ductal Adenocarcinoma. Front. Genet. 2019, 10, 624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wuethrich, P.Y.; Hsu Schmitz, S.F.; Kessler, T.M.; Thalmann, G.N.; Studer, U.E.; Stueber, F.; Burkhard, F.C. Potential influence of the anesthetic technique used during open radical prostatectomy on prostate cancer-related outcome: A retrospective study. Anesthesiology 2010, 113, 570–576. [Google Scholar] [CrossRef]
- Christopherson, R.; James, K.E.; Tableman, M.; Marshall, P.; Johnson, F.E. Long-Term Survival After Colon Cancer Surgery: A Variation Associated with Choice of Anesthesia. Anesth. Analg. 2008, 107, 325–332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gottschalk, A.; Ford, J.G.; Regelin, C.C.; You, J.; Mascha, E.J.; Sessler, D.I.; Durieux, M.; Nemergut, E.C. Association between Epidural Analgesia and Cancer Recurrence after Colorectal Cancer Surgery. Anesthesiology 2010, 113, 27–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, B.M.; Ghotra, V.S.; Karam, J.A.; Hernandez, M.; Pratt, G.; Cata, J.P. Regional anesthesia/analgesia and the risk of cancer recurrence and mortality after prostatectomy: A meta-analysis. Pain Manag. 2015, 5, 387–395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cummings, K.C., 3rd; Patel, M.; Htoo, P.T.; Bakaki, P.M.; Cummings, L.C.; Koroukian, S. A comparison of the effects of epidural analgesia versus traditional pain management on outcomes after gastric cancer resection: A population-based study. Reg. Anesth. Pain Med. 2014, 39, 200–207. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Guo, W.; Wu, Q.; Zhang, R.; Fang, J. Impact of Combination Epidural and General Anesthesia on the Long-Term Survival of Gastric Cancer Patients: A Retrospective Study. Med. Sci. Monit. 2016, 22, 2379–2385. [Google Scholar] [CrossRef] [Green Version]
- Pei, L.; Tan, G.; Wang, L.; Guo, W.; Xiao, B.; Gao, X.; Li, H.; Xu, Z.; Zhang, X.; Zhao, J.; et al. Comparison of Combined General-Epidural Anesthesia with General Anesthesia Effects on Survival and Cancer Recurrence: A Meta-Analysis of Retrospective and Prospective Studies. PLoS ONE 2014, 9, e114667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, D.; Lim, C.S.; Shin, Y.S.; Ko, Y.K.; Park, S.I.; Song, S.H.; Kim, B.J. A comparison of regional and general anesthesia effects on 5 year survival and cancer recurrence after transurethral resection of the bladder tumor: A retrospective analysis. BMC Anesthesiol. 2015, 16, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Call, T.R.; Pace, N.L.; Thorup, D.B.; Maxfield, D.; Chortkoff, B.; Christensen, J.P.; Lin, J. Factors associated with improved survival after resection of pancreatic adenocarcinoma: A multivariable model. Anesthesiology 2015, 122, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Day, A.; Smith, R.; Jourdan, I.; Fawcett, W.; Scott, M.; Rockall, T. Retrospective analysis of the effect of postoperative analgesia on survival in patients after laparoscopic resection of colorectal cancer. Br. J. Anaesth. 2012, 109, 185–190. [Google Scholar] [CrossRef] [Green Version]
- Tsui, B.C.H.; Rashiq, S.; Schopflocher, D.; Murtha, A.; Broemling, S.; Pillay, J.; Finucane, B.T. Epidural anesthesia and cancer recurrence rates after radical prostatectomy. Can. J. Anesth./J. Can. D’anesthésie 2009, 57, 107–112. [Google Scholar] [CrossRef] [Green Version]
- Weingarten, T.N.; Taccolini, A.M.; Ahle, S.T.; Dietz, K.R.; Dowd, S.S.; Frank, I.; Boorjian, S.A.; Thapa, P.; Hanson, A.C.; Schroeder, D.R.; et al. Perioperative management and oncological outcomes following radical cystectomy for bladder cancer: A matched retrospective cohort study. Can. J. Anesth./J. Can. D’anesthésie 2016, 63, 584–595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).