Prevalence and Characteristics of Thyroid Abnormalities and Its Association with Anemia in ASIR Region of Saudi Arabia: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Setting
2.2. Precipitant and Data Collection
2.3. Statistical Analysis
2.4. Ethical Approval
3. Results
3.1. General Characteristic of the Study Population
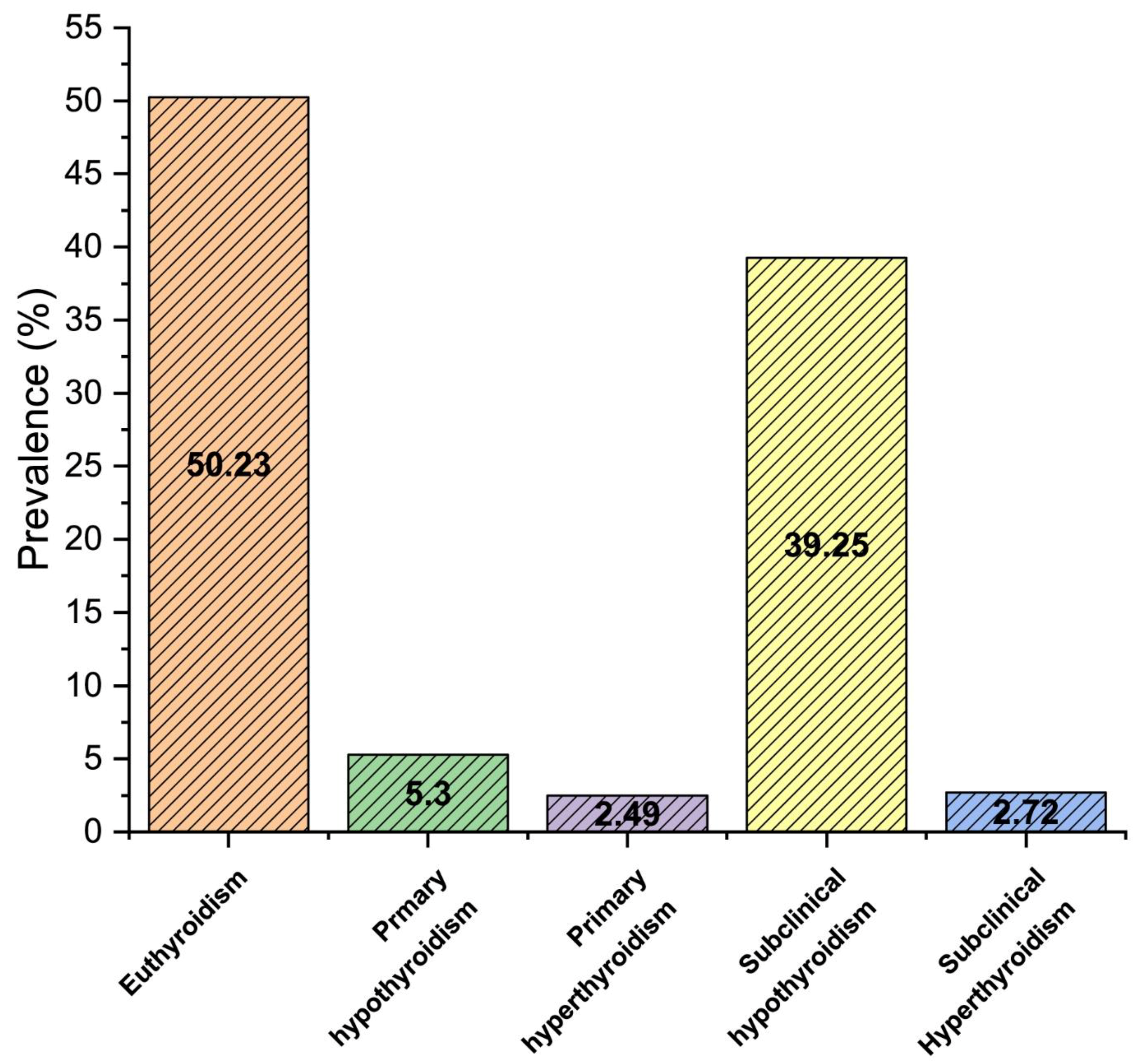
3.2. Thyroid Dysfunction
3.3. Thyroid Profile and Anemia
3.4. Correlation between Thyroid Hormones and Hematological Profiles
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Taylor, P.N.; Albrecht, D.; Scholz, A.; Gutierrez-Buey, G.; Lazarus, J.H.; Dayan, C.M.; Okosieme, O.E. Global epidemiology of hyperthyroidism and hypothyroidism. Nat. Rev. Endocrinol. 2018, 14, 301–316. [Google Scholar] [CrossRef]
- Garmendia Madariaga, A.; Santos Palacios, S.; Guillén-Grima, F.; Galofré, J.C. The incidence and prevalence of thyroid dysfunction in Europe: A meta-analysis. J. Clin. Endocrinol. Metab. 2014, 99, 923–931. [Google Scholar] [CrossRef]
- Ittermann, T.; Khattak, R.M.; Nauck, M.; Cordova, C.M.; Völzke, H. Shift of the TSH reference range with improved iodine supply in Northeast Germany. Eur. J. Endocrinol. 2015, 172, 261–267. [Google Scholar] [CrossRef]
- Meisinger, C.; Ittermann, T.; Wallaschofski, H.; Heier, M.; Below, H.; Kramer, A.; Döring, A.; Nauck, M.; Völzke, H. Geographic variations in the frequency of thyroid disorders and thyroid peroxidase antibodies in persons without former thyroid disease within Germany. Eur. J. Endocrinol. 2012, 167, 363–371. [Google Scholar] [CrossRef]
- Diab, N.; Daya, N.R.; Juraschek, S.P.; Martin, S.S.; McEvoy, J.W.; Schultheiß, U.T.; Köttgen, A.; Selvin, E. Prevalence and Risk Factors of Thyroid Dysfunction in Older Adults in the Community. Sci. Rep. 2019, 9, 13156. [Google Scholar] [CrossRef]
- Scappaticcio, L.; Longo, M.; Maiorino, M.I.; Pernice, V.; Caruso, P.; Esposito, K.; Bellastella, G. Abnormal Liver Blood Tests in Patients with Hyperthyroidism: Systematic Review and Meta-Analysis. Thyroid 2021, 31, 884–894. [Google Scholar] [CrossRef]
- Tan, Z.S.; Beiser, A.; Vasan, R.S.; Au, R.; Auerbach, S.; Kiel, D.P.; Wolf, P.A.; Seshadri, S. Thyroid function and the risk of Alzheimer disease: The Framingham Study. Arch. Intern. Med. 2008, 168, 1514–1520. [Google Scholar] [CrossRef] [PubMed]
- Das, K.C.; Mukherjee, M.; Sarkar, T.K.; Dash, R.J.; Rastogi, G.K. Erythropoiesis and erythropoietin in hypo- and hyperthyroidism. J. Clin. Endocrinol. Metab. 1975, 40, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Dorgalaleh, A.; Mahmoodi, M.; Varmaghani, B.; Kiani Node, F.; Saeeidi Kia, O.; Alizadeh, S.; Tabibian, S.; Bamedi, T.; Momeni, M.; Abbasian, S.; et al. Effect of thyroid dysfunctions on blood cell count and red blood cell indice. Iran. J. Ped. Hematol. Oncol. 2013, 3, 73–77. [Google Scholar] [PubMed]
- Golde, D.W.; Bersch, N.; Chopra, I.J.; Cline, M.J. Thyroid hormones stimulate erythropoiesis in vitro. Br. J. Haematol. 1977, 37, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Kawa, M.P.; Grymula, K.; Paczkowska, E.; Baskiewicz-Masiuk, M.; Dabkowska, E.; Koziolek, M.; Tarnowski, M.; Kłos, P.; Dziedziejko, V.; Kucia, M.; et al. Clinical relevance of thyroid dysfunction in human haematopoiesis: Biochemical and molecular studies. Eur. J. Endocrinol. 2010, 162, 295–305. [Google Scholar] [CrossRef]
- Ahmed, S.S.; Mohammed, A.A. Effects of thyroid dysfunction on hematological parameters: Case controlled study. Ann. Med. Surg. (Lond.) 2020, 57, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Al Shanqeeti, S.A.; Alkhudairy, Y.N.; Alabdulwahed, A.A.; Ahmed, A.E.; Al-Adham, M.S.; Mahmood, N.M. Prevalence of subclinical hypothyroidism in pregnancy in Saudi Arabia. Saudi. Med. J. 2018, 39, 254–260. [Google Scholar] [CrossRef]
- Erdogan, M.; Kösenli, A.; Ganidagli, S.; Kulaksizoglu, M. Characteristics of anemia in subclinical and overt hypothyroid patients. Endocr. J. 2012, 59, 213–220. [Google Scholar] [CrossRef]
- Fatima, Q.; Dotasara, P.; Gauri, L.A. Hematological profile in primary hypothyroidism. Int. J. Med. Biomed. Stud. 2020, 4. [Google Scholar] [CrossRef]
- Horton, L.; Coburn, R.J.; England, J.M.; Himsworth, R.L. The haematology of hypothyroidism. Q. J. Med. 1976, 45, 101–123. [Google Scholar]
- Fein, H.G.; Rivlin, R.S. Anemia in thyroid diseases. Med. Clin. N. Am. 1975, 59, 1133–1145. [Google Scholar] [CrossRef]
- Vanderpump, M.P. The epidemiology of thyroid disease. Br. Med. Bull. 2011, 99, 39–51. [Google Scholar] [CrossRef]
- Iddah, M.A.; Macharia, B.N.; Ng’wena, A.G.; Keter, A.; Ofulla, A.V. Thryroid hormones and hematological indices levels in thyroid disorders patients at moi teaching and referral hospital, Western kenya. ISRN Endocrinol. 2013, 2013, 385940. [Google Scholar] [CrossRef] [PubMed]
- Scappaticcio, L.; Maiorino, M.I.; Maio, A.; Esposito, K.; Bellastella, G. Neutropenia in patients with hyperthyroidism: Systematic review and meta-analysis. Clin. Endocrinol. (Oxf.) 2021, 94, 473–483. [Google Scholar] [CrossRef]
- Lima, C.S.; Zantut Wittmann, D.E.; Castro, V.; Tambascia, M.A.; Lorand-Metze, I.; Saad, S.T.; Costa, F.F. Pancytopenia in untreated patients with Graves’ disease. Thyroid 2006, 16, 403–409. [Google Scholar] [CrossRef]
- Canaris, G.J.; Manowitz, N.R.; Mayor, G.; Ridgway, E.C. The Colorado thyroid disease prevalence study. Arch. Intern. Med. 2000, 160, 526–534. [Google Scholar] [CrossRef]
- Wilson, G.R.; Curry, R.W.J. Subclinical thyroid disease. Am. Fam. Phys. 2005, 72, 1517–1524. [Google Scholar]
- Bekkering, G.E.; Agoritsas, T.; Lytvyn, L.; Heen, A.F.; Feller, M.; Moutzouri, E.; Abdulazeem, H.; Aertgeerts, B.; Beecher, D.; Brito, J.P.; et al. Thyroid hormones treatment for subclinical hypothyroidism: A clinical practice guideline. BMJ 2019, 365, l2006. [Google Scholar] [CrossRef]
- Refaat, B. Prevalence and characteristics of anemia associated with thyroid disorders in non-pregnant Saudi women during the childbearing age: A cross-sectional study. Biomed. J. 2015, 38, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Thyroidfriends. ATA Professional Guidelines. Available online: https://www.thyroid.org/professionals/ata-professional-guidelines/ (accessed on 28 July 2021).
- Ali, A.A.G.; Altahir, S.A. Prevalence of thyroids dysfunction among Saudi adult males and females from (June–September 2016). Endocrinol. Diab. 2016, 3, 1–3. [Google Scholar]
- Aljabri, K.S.J.; Facharatz, A.I.; Bokhari, S.A. The frequency of hypothyroidism in saudi community-based hospital: A retrospective single centre study. Trends. Diabetes. Metab. 2019, 2, 1–4. [Google Scholar] [CrossRef][Green Version]
- Alqahtiani, N.M.; Alramadhan, Z.T.; bin Obaid, M.R.; Kurdi, A.N.; Alhelal, A.A.; Aljaafar, F.A.; AlDairam, W.H.; Alyaqoub, Z.A.Y.; Sharufana, A.S.; Abdalla, A.A. Hypothyroidism in Saudi Arabia; Prevalence, risk factors, and its relation with Diabetes Mellitus. Arch. Pharm. Pract. 2020, 11, 56–63. [Google Scholar]
- Iodine Deficiency. Available online: https://www.thyroid.org/iodine-deficiency/ (accessed on 28 July 2021).
- Al-Nuaim, A.R.; Al-Mazrou, Y.; Kamel, M.; Al-Attas, O.; Al-Daghari, N.; Sulimani, R. Iodine deficiency in Saudi Arabia. Ann. Saudi. Med. 1997, 17, 293–297. [Google Scholar] [CrossRef]
- Abbag, F.I.; Abu-Eshy, S.A.; Mahfouz, A.A.; Al-Fifi, S.A.; El-Wadie, H.; Abdallah, S.M.; Musa, M.G.; Devansan, C.S.; Patel, A. Iodine-deficiency disorders in the Aseer region, south-western Saudi Arabia: 20 years after the national survey and universal salt iodization. Public Health Nutr. 2015, 18, 2523–2529. [Google Scholar] [CrossRef][Green Version]
- Abuye, C.; Berhane, Y.; Ersumo, T. The role of changing diet and altitude on goitre prevalence in five regional states in Ethiopia. East Afr. J. Public Health 2008, 5, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Abu-Eshy, S.A.; Abolfotouh, M.A.; Al-Naggar, Y.M. Endemic goitre in schoolchildren in high and low altitude areas of Asir region, Saudi Arabia. Saudi. Med. J. 2001, 22, 146–149. [Google Scholar]
- Moussa, S.; Alshammari, A.; Gada Alshammari, G.; Alshammar, K.; Alanzi, M.; Alanzi, N.; Alshammari, Z. Pattern of Thyroid Disease in Hail Region, Saudi Arabia. Int. J. Sci. Res. 2013, 5, 276–282. [Google Scholar] [CrossRef]
- Aljarbou, A.; Morgan, A.; Alshaalan, F.; Alshehri, D.; Alshathri, M.; Alsayyali, K. The Patterns of Surgically Treated Thyroid Disease in Central Rural Region of Saudi Arabia. Egypt. J. Hosp. Med. 2018, 70, 1066–1071. [Google Scholar] [CrossRef]
- Rana, H.; Mirah, J.A.; Al-Shahrani, N.; Nouf, A.; Afrah, A.; Basma, O.; Sumbul, F. Incidence of thyroid diseases in female Saudi adults visiting a tertiary care hospital in Riyadh. Epidemiol. Open Access 2017, 7, 286. [Google Scholar]
- Alrowaili, A.M.Z.; Alanazi, M.A.M.; Alenezi, M.H.M.; Alrwuaili, A.M.; Ahmed, A.A.B.; Alruwaili, A.E.M.; Alshalan, M.H.F.; Alfuhigi, Z.D.M.; Abdulhamid, M.A.A.; Alanzi, N.D.Y. Prevalence of Hypothyroidism and Its Associated Risk Factors in Arar City, Saudi Arabia. Egypt. J. Hosp. Med. 2018, 71, 2765–2769. [Google Scholar] [CrossRef]
- Bashir, H.; Bhat, M.H.; Farooq, R.; Majid, S.; Shoib, S.; Hamid, R.; Mattoo, A.A.; Rashid, T.; Bhat, A.A.; Wani, H.A.; et al. Comparison of hematological parameters in untreated and treated subclinical hypothyroidism and primary hypothyroidism patients. Med. J. Islam. Repub. Iran 2012, 26, 172–178. [Google Scholar]
- Bremner, A.P.; Feddema, P.; Joske, D.J.; Leedman, P.J.; O’Leary, P.C.; Olynyk, J.K.; Walsh, J.P. Significant association between thyroid hormones and erythrocyte indices in euthyroid subjects. Clin. Endocrinol. (Oxf.) 2012, 76, 304–311. [Google Scholar] [CrossRef]
- Karkoutly, S.; Hammoud, T.; Al-Quobaili, F. Effect of hypothyroidism on haematological parameters: A gender-based comparison. N. Z. J. Med. Lab. Sci. 2020, 74, 98. [Google Scholar]
- Omar, S.; Hadj Taeib, S.; Kanoun, F.; Hammami, M.B.; Kamoun, S.; Ben Romdhane, N.; Feki, M.; Slimane, H.; Kaabachi, N. Erythrocyte abnormalities in thyroid dysfunction. Tunis. Med. 2010, 88, 783–788. [Google Scholar] [PubMed]
- Jafarzadeh, A.; Poorgholami, M.; Izadi, N.; Nemati, M.; Rezayati, M. Immunological and hematological changes in patients with hyperthyroidism or hypothyroidism. Clin. Investig. Med. 2010, 33, E271–E279. [Google Scholar] [CrossRef] [PubMed]
- Wopereis, D.M.; Du Puy, R.S.; van Heemst, D.; Walsh, J.P.; Bremner, A.; Bakker, S.J.L.; Bauer, D.C.; Cappola, A.R.; Ceresini, G.; Degryse, J.; et al. The Relation Between Thyroid Function and Anemia: A Pooled Analysis of Individual Participant Data. J. Clin. Endocrinol. Metab. 2018, 103, 3658–3667. [Google Scholar] [CrossRef] [PubMed]
| Age Range (Years) | Gender | |
|---|---|---|
| FEMALE% (n = 6146) | MALE% (n = 3831) | |
| Below 20 | 4.7% (290) | 6.0% (229) |
| 20–30 | 14.5% (895) | 9.8% (376) |
| 31–40 | 31.6% (1949) | 26.9% (1030) |
| 41–50 | 20.4% (1259) | 22.4% (861) |
| Above 50 | 28.7% (1767) | 34.9% (1336) |
| Parameters | Female | Male | p * | ||
|---|---|---|---|---|---|
| Mean ± SD | Median ± IQR | Mean ± SD | Median ± IQR | ||
| TSH (μIU/mL) | 4.32 ± 6.7 | 2.86 ± 4.1 | 4.22 ± 5.8 | 2.90 ± 4.1 | 0.668 |
| FT4 (ng/dL) | 0.99 ± 0.17 | 0.98 ± 0.2 | 1.01 ± 1.3 | 0.99 ± 0.2 | 0.066 |
| Hb (g/dL) | 12.84 ± 2.1 | 13.90 ± 2.3 | 14.75 ± 2.1 | 15.0 ± 2.6 | 0.039 |
| HCT (%) | 40.73 ± 5.1 | 40.80 ± 6.0 | 42.75 ± 5.7 | 43.40 ± 6.8 | 0.000 |
| MCV (fL) | 79.7 ± 9.7 | 81.60 ± 7.2 | 80.27 ± 8.4 | 81.35 ± 5.9 | 0.000 |
| MCH (Pg) | 26.92 ± 4.0 | 27.80 ± 3.6 | 27.42 ± 3.6 | 28.10 ± 3.1 | 0.000 |
| MCHC (g/dL) | 33.95 ± 1.6 | 34.10 ± 1.9 | 34.33 ± 1.5 | 34.50 ± 1.9 | 0.000 |
| RDW (%) | 14.38 ± 2.1 | 13.80 ± 1.9 | 14.09 ± 1.8 | 13.60 ± 1.6 | 0.000 |
| RBC (1012/L) | 4.82 ± 1.3 | 5.01 ± 0.8 | 5.17 ± 1.1 | 5.34 ± 0.8 | 0.000 |
| Thyroid Dysfunction | Prevalence in Different Age Sub-Group (Years) | ||||||
|---|---|---|---|---|---|---|---|
| Below 20 | 20–30 | 31–40 | 41–50 | >50 | Total | χ2 | |
| Female | 102.18 a (p = 0.000) a 105.32 b (p = 0.000) b | ||||||
| Euthyroidism | 53.1% (154) | 51.6% (462) | 52.6% (1025) | 50.6% (637) | 49.8% (880) | 31.6% (3158) | |
| Primary Hypothyroidism | 1.7% (5) | 3% (27) | 3% (59) | 6.5% (81) | 6.5% (115) | 2.9% (287) | |
| Primary Hyperthyroidism | 0.3% (1) | 1.7% (15) | 1.6% (32) | 1.8% (22) | 3.7% (65) | 1.4% (135) | |
| Subclinical Hypothyroidism | 44.8% (130) | 41.2% (369) | 40.6% (789) | 37.6% (473) | 36.0% (637) | 24.0% (2398) | |
| Subclinical Hyperthyroidism | 0.0% (0) | 2.5% (22) | 2.3% (44) | 3.7% (46) | 4.0% (70) | 1.8% (182) | |
| Male | 59.16 a (p = 0.000) a 69.67 b (p = 0.000) b | ||||||
| Euthyroidism | 51.1% (117) | 51.1% (192) | 49% (505) | 49.5% (427) | 46.4% (620) | 18.6% (1861) | |
| Primary Hypothyroidism | 0% (0.0) | 4.5% (17) | 4.8% (49) | 7% (60) | 8.8% (117) | 2.4% (243) | |
| Primary Hyperthyroidism | 0.4% (1) | 2.7% (10) | 2.2% (23) | 3.6% (31) | 3.7% (49) | 1.1% (114) | |
| Subclinical Hypothyroidism | 48% (110) | 40.4% (152) | 41.6% (428) | 37.3% (321) | 38.4% (513) | 15.3% (1524) | |
| Subclinical Hyperthyroidism | 0.4% (1) | 1.3% (5) | 2.4% (25) | 2.6% (22) | 2.8% (37) | 0.9% (90) | |
| Anemia | Euthyroidism (n = 5019) | Primary Hypothyroidism (n = 530) | Primary Hyperthyroidism (n = 249) | Subclinical Hypothyroidism (n = 3922) | Subclinical Hyperthyroidism (n = 272) | χ2 | |
|---|---|---|---|---|---|---|---|
| Non-anemic (n = 6491) | Female (n = 3645) | 30.6% (1987) | 2.1% (137) | 1.0% (66) | 20.9% (1354) | 1.6% (101) | 86.82 a (p = 0.000) a 86.39 a (p = 0.000) b |
| Male (n = 2846) | 20.6% (1340) | 3.4 (221) | 1.6% (106) | 17.0% (1101) | 1.2% (78) | ||
| Microcytic hypochromic (n = 2189) | Female (n = 1574) | 32.4% (709) | 4.2% (92) | 1.6% (34) | 31.4% (687) | 2.4% (52) | 28.38 a (p = 0.000) a 30.81 b (p = 0.000) b |
| Male (n = 615) | 14.6% (319) | 0.5% (12) | 0.2% (5) | 12.3% (270) | 0.4% (9) | ||
| Normocytic normochromic (n = 1312) | Female (n = 941) | 35.2% (462) | 4.4% (58) | 2.7% (35) | 27.2% (357) | 2.2% (29) | 21.84 a (p = 0.000) 23.87 b (p = 0.000) b |
| Male (n = 371) | 15.4% (202) | 0.8% (10) | 0.2% (3) | 11.7% (153) | 0.2% (3) |
| Thyroid Dysfunction | Hb (g/dL) | MCV (fL) | MCH (pg/cell) | HCT (%) | RBC (×106) | MCHC (g/dL) | RDW (%) |
|---|---|---|---|---|---|---|---|
| Female | |||||||
| Euthyroidism | 14.02 ± 2.01 | 81.94 ± 9.5 | 27.06 ± 4.01 | 41.05 ± 5.11 | 4.82 ± 1.34 | 34.08 ± 1.51 | 14.28 ± 1.97 |
| Primary Hypothyroidism | 11.22 ± 1.93 | 79.48 ± 11.2 | 26.67 ± 4.15 | 39.86 ± 5.60 | 4.73 ± 0.94 | 33.07 ± 1.73 | 14.61 ± 2.22 |
| Primary Hyperthyroidism | 13.48 ± 1.32 | 80.66 ± 6.86 | 27.36 ± 2.43 | 40.30 ± 3.52 | 4.96 ± 0.64 | 33.43 ± 1.32 | 14.22 ± 1.76 |
| Subclinical Hypothyroidism | 11.83 ± 2.14 | 79.17 ± 9.92 | 26.76 ± 4.11 | 39.48 ± 5.02 | 4.78 ± 1.34 | 33.96 ± 1.62 | 14.48 ± 2.03 |
| Subclinical Hyperthyroidism | 13.55 ± 1.76 | 79.74 ± 7.21 | 26.93 ± 2.92 | 40.17 ± 4.24 | 5.03 ± 0.52 | 33.65 ± 1.56 | 14.49 ± 1.87 |
| p value | 0.000 | 0.002 | 0.035 | 0.000 | 0.080 | 0.000 | 0.001 |
| Male | |||||||
| Euthyroidism | 14.65 ± 2.12 | 82.10 ± 9.10 | 27.35 ± 3.8 | 42.40 ± 5.82 | 5.14 ± 1.12 | 34.34 ± 1.51 | 14.08 ± 1.79 |
| Primary Hypothyroidism | 12.72 ± 1.59 | 79.10 ± 7.50 | 28.33 ± 2.9 | 40.90 ± 6.62 | 4.91 ± 0.92 | 34.41 ± 1.56 | 13.82 ± 1.54 |
| Primary Hyperthyroidism | 15.60 ± 1.34 | 80.10 ± 5.10 | 27.90 ± 2.1 | 44.90 ± 7.09 | 5.51 ± 0.80 | 33.98 ± 1.44 | 14.24 ± 2.09 |
| Subclinical Hypothyroidism | 12.62 ± 2.14 | 79.10 ± 7.90 | 27.35 ± 3.6 | 40.62 ± 5.11 | 4.81 ± 1.15 | 34.34 ± 1.50 | 14.13 ± 1.75 |
| Subclinical Hyperthyroidism | 15.23 ± 1.68 | 80.01 ± 5.91 | 27.40 ± 2.31 | 43.25 ± 7.82 | 5.48 ± 0.79 | 34.22 ± 1.35 | 14.21 ± 1.85 |
| p value | 0.000 | 0.002 | 0.001 | 0.000 | 0.000 | 0.107 | 0.111 |
| Key Variables | Female | Male | ||||
|---|---|---|---|---|---|---|
| TSH | FT4 | Age | TSH | FT4 | Age | |
| Age | 0.40 ** | −0.15 * | - | 0.12 * | −0.76 ** | - |
| TSH | - | −0.161 ** | 0.40 ** | - | −0.165 ** | 0.12 |
| FT4 | −0.161 ** | - | −0.15 | −0.165 ** | - | −0.76 ** |
| MCV | −0.017 | −0.045 ** | 0.085 ** | −0.018 | −0.048 ** | 0.090 ** |
| MCH | −0.022 | 0.012 | 0.071 ** | −0.012 | −0.016 | 0.077 ** |
| Hb | −0.048 ** | 0.120 ** | 0.004 | −0.075 ** | 0.098 ** | 0.056 ** |
| HCT | −0.050 ** | 0.099 ** | 0.019 | −0.067 ** | 0.082 ** | 0.056 ** |
| MCHC | −0.016 | 0.111 ** | −0.035 ** | −0.017 | 0.076 ** | −0.017 |
| RDW | 0.041 ** | −0.058 ** | −0.001 | 0.045 ** | −0.041 * | −0.001 |
| RBC | −0.053 ** | 0.130 ** | −0.042 ** | −0.074 ** | 0.139 ** | 0.014 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alqahtani, S.A.M. Prevalence and Characteristics of Thyroid Abnormalities and Its Association with Anemia in ASIR Region of Saudi Arabia: A Cross-Sectional Study. Clin. Pract. 2021, 11, 494-504. https://doi.org/10.3390/clinpract11030065
Alqahtani SAM. Prevalence and Characteristics of Thyroid Abnormalities and Its Association with Anemia in ASIR Region of Saudi Arabia: A Cross-Sectional Study. Clinics and Practice. 2021; 11(3):494-504. https://doi.org/10.3390/clinpract11030065
Chicago/Turabian StyleAlqahtani, Saif Aboud M. 2021. "Prevalence and Characteristics of Thyroid Abnormalities and Its Association with Anemia in ASIR Region of Saudi Arabia: A Cross-Sectional Study" Clinics and Practice 11, no. 3: 494-504. https://doi.org/10.3390/clinpract11030065
APA StyleAlqahtani, S. A. M. (2021). Prevalence and Characteristics of Thyroid Abnormalities and Its Association with Anemia in ASIR Region of Saudi Arabia: A Cross-Sectional Study. Clinics and Practice, 11(3), 494-504. https://doi.org/10.3390/clinpract11030065





