Endoscopic and Histopathological Findings of the Esophagus, Stomach, and Duodenum in Patients with Crohn’s Disease from a Reference Center in Bahia, Brazil
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Subjects
2.2. Esophagogastroduodenoscopy
2.3. Histopathology
2.4. Statistical Analysis
2.5. Ethical Considerations
3. Results
3.1. Demographic and Clinical Characteristics
3.2. Endoscopic Findings
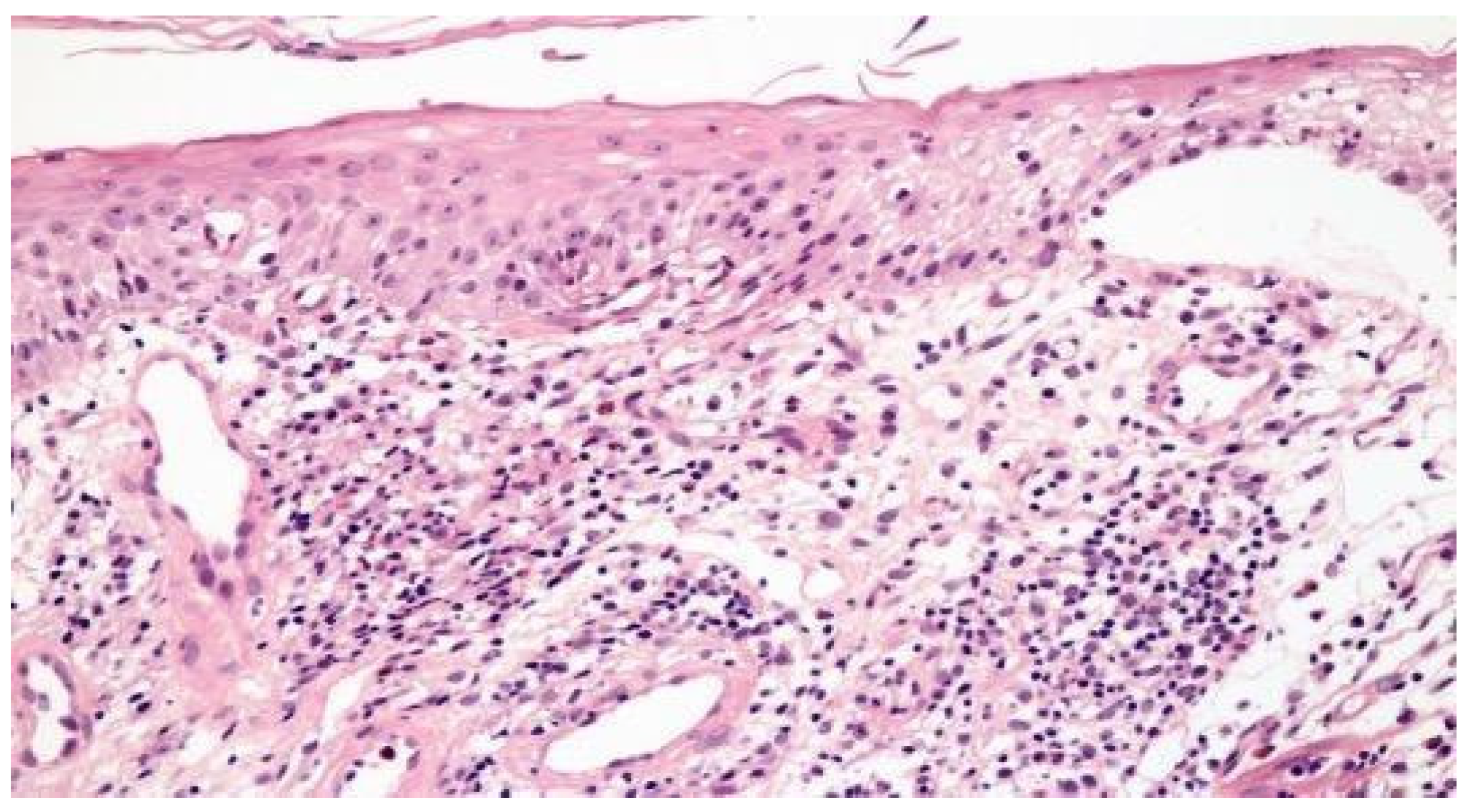
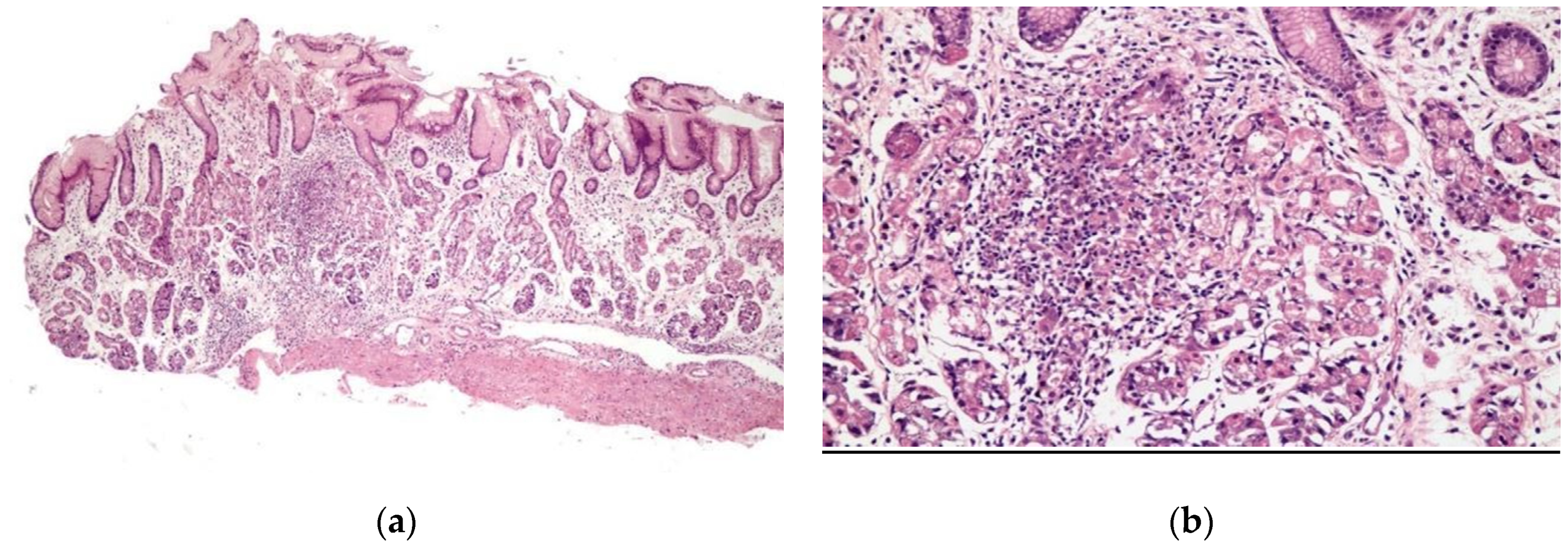
3.3. Histopathological Findings
3.4. Comparison between H. Pylori-Positive and H.-Pylori-Negative Patients
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fielding, J.F.; Toye, D.K.; Beton, D.C.; Cooke, W.T. Crohn’s Disease of the Stomach and Duodenum. Gut 1970, 11, 1001–1006. [Google Scholar] [CrossRef]
- Nugent, F.W.; Richmond, M.; Park, S.K. Crohn’s Disease of the Duodenum. Gut 1977, 18, 115–120. [Google Scholar] [CrossRef]
- Reynolds, H.L.; Stellato, T.A. Crohn’s Disease of the Foregut. Surg. Clin. N. Am. 2001, 81, 117–135. [Google Scholar] [CrossRef]
- Annunziata, M.L.; Caviglia, R.; Papparella, L.G.; Cicala, M. Upper Gastrointestinal Involvement of Crohn’s Disease: A Prospective Study on the Role of Upper Endoscopy in the Diagnostic Work-Up. Dig. Dis. Sci. 2012, 57, 1618–1623. [Google Scholar] [CrossRef]
- Horje, C.S.H.T.; Meijer, J.; Rovers, L.; Van Lochem, E.G.; Groenen, M.J.M.; Wahab, P.J. Prevalence of Upper Gastrointestinal Lesions at Primary Diagnosis in Adults with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2016, 22, 1896–1901. [Google Scholar] [CrossRef]
- Maaser, C.; Sturm, A.; Vavricka, S.R.; Kucharzik, T.; Fiorino, G.; Annese, V.; Calabrese, E.; Baumgart, D.C.; Bettenworth, D.; Borralho Nunes, P.; et al. ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initial Diagnosis, Monitoring of Known IBD, Detection of Complications. J. Crohn’s Colitis 2019, 13, 144–164. [Google Scholar] [CrossRef]
- Escher, J.C. Inflammatory Bowel Disease in Children and Adolescents: Recommendations for Diagnosis—The Porto Criteria. J. Pediatric Gastroenterol. Nutr. 2005, 41, 1–7. [Google Scholar]
- Davis, K.G. Crohn’s Disease of the Foregut. In Surgical Clinics of North America; W.B. Saunders: Philadelphia, PA, USA, 2015; pp. 1183–1193. [Google Scholar]
- Magro, F.; Langner, C.; Driessen, A.; Ensari, A.; Geboes, K.; Mantzaris, G.J.; Villanacci, V.; Becheanu, G.; Nunes, P.B.; Cathomas, G.; et al. European Consensus on the Histopathology of Inflammatory Bowel Disease. J. Crohn’s Colitis 2013, 7, 827–851. [Google Scholar] [CrossRef]
- Diaz, L.; Hernandez-Oquet, R.E.; Deshpande, A.R.; Moshiree, B. Upper Gastrointestinal Involvement in Crohn Disease: Histopathologic and Endoscopic Findings. South. Med. J. 2015, 108, 695–700. [Google Scholar] [CrossRef]
- Luther, J.; Dave, M.; Higgins, P.D.R.; Kao, J.Y. Association between Helicobacter Pylori Infection and Inflammatory Bowel Disease: A Meta-Analysis and Systematic Review of the Literature. Inflamm. Bowel Dis. 2010, 16, 1077–1084. [Google Scholar] [CrossRef]
- Papamichael, K.; Konstantopoulos, P.; Mantzaris, G.J. Helicobacter Pylori Infection and Inflammatory Bowel Disease: Is There a Link? World J. Gastroenterol. 2014, 20, 6374–6385. [Google Scholar] [CrossRef]
- De Magalhães-Costa, M.H.; Dos Reis, B.R.; Chagas, V.L.A.; Nunes, T.; De Souza, H.S.P.; Zaltman, C. Focal Enhanced Gastritis and Macrophage Microaggregates in Thegastric Mucosa: Potential Role in the Differential Diagnosis between Crohn’s Disease and Ulcerative Colitis. Arq. Gastroenterol. 2014, 51, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Gomollón, F.; Dignass, A.; Annese, V.; Tilg, H.; Van Assche, G.; Lindsay, J.O.; Peyrin-Biroulet, L.; Cullen, G.J.; Daperno, M.; Kucharzik, T.; et al. 3rd European Evidence-Based Consensus on the Diagnosis and Management of Crohn’s Disease 2016: Part 1: Diagnosis and Medical Management. J. Crohn’s Colitis 2017, 11, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Satsangi, J.; Silverberg, M.S.; Vermeire, S.; Colombel, J.F. The Montreal Classification of Inflammatory Bowel Disease: Controversies, Consensus, and Implications. Gut 2006, 55, 749–753. [Google Scholar] [CrossRef]
- Harvey, R.F.; Bradshaw, J.M. A Simple Index of Crohn’s-Disease Activity. Lancet 1980, 315, 514. [Google Scholar] [CrossRef]
- Lundell, L.R.; Dent, J.; Bennett, J.R.; Blum, A.L.; Armstrong, D.; Galmiche, J.P.; Johnson, F.; Hongo, M.; Richter, J.E.; Spechler, S.J.; et al. Endoscopic Assessment of Oesophagitis: Clinical and Functional Correlates and Further Validation of the Los Angeles Classification. Gut 1999, 45, 172–180. [Google Scholar] [CrossRef]
- Sipponen, P.; Price, A.B. The Sydney System for Classification of Gastritis 20 Years Ago. J. Gastroenterol. Hepatol. 2011, 26 (Suppl. 1), 31–34. [Google Scholar] [CrossRef]
- Prakash Gyawali, C.; Kahrilas, P.J.; Savarino, E.; Zerbib, F.; Mion, F.; Smout, A.J.P.M.; Vaezi, M.; Sifrim, D.; Fox, M.R.; Vela, M.F.; et al. Modern Diagnosis of GERD: The Lyon Consensus. Gut 2018, 67, 1351–1362. [Google Scholar] [CrossRef]
- Anton, G.; Decker, G.; Loftus, E.V.; Pasha, T.M.; Tremaine, W.J.; Sandborn, W.J. Crohn’s Disease of the Esophagus: Clinical Features and Outcomes. Inflamm. Bowel Dis. 2001, 7, 113–119. [Google Scholar]
- De Felice, K.M.; Katzka, D.A.; Raffals, L.E. Crohn’s Disease of the Esophagus: Clinical Features and Treatment Outcomes in the Biologic Era. Inflamm. Bowel Dis. 2015, 21, 2106–2113. [Google Scholar] [CrossRef]
- D’Haens, G.; Rutgeerts, P.; Geboes, K.; Vantrappen, G. The Natural History of Esophageal Crohn’s Disease: Three Patterns of Evolution. Gastrointest. Endosc. 1994, 40, 296–300. [Google Scholar] [CrossRef]
- Nomura, Y.; Moriichi, K.; Fujiya, M.; Okumura, T. The Endoscopic Findings of the Upper Gastrointestinal Tract in Patients with Crohn’s Disease. Clin. J. Gastroenterol. 2017, 10, 289–296. [Google Scholar] [CrossRef]
- Weinstein, T.; Valderrama, E.; Pettei, M.; Levine, J. Esophageal Crohn’s Disease: Medical Management and Correlation between Clinical, Endoscopic, and Histologic Features. Inflamm. Bowel Dis. 1997, 3, 79–83. [Google Scholar] [CrossRef]
- Sakuraba, A.; Iwao, Y.; Matsuoka, K.; Naganuma, M.; Ogata, H.; Kanai, T.; Hibi, T. Endoscopic and Pathologic Changes of the Upper Gastrointestinal Tract in Crohn’s Disease. Biomed Res. Int. 2014, 2014, 610767. [Google Scholar] [CrossRef]
- So, H.; Ye, B.D.; Park, Y.S.; Kim, J.; Kim, J.S.; Moon, W.; Lee, K.M.; Kim, Y.S.; Keum, B.; Kim, S.E.; et al. Gastric Lesions in Patients with Crohn’s Disease in Korea: A Multicenter Study. Intest. Res. 2016, 14, 60–68. [Google Scholar] [CrossRef][Green Version]
- Gasche, C.; Scholmerich, J.; Brynskov, J.; D’Haens, G.; Hanauer, S.B.; Irvine, E.J.; Jewell, D.P.; Rachmilewitz, D.; Sachar, D.B.; Sandborn, W.J.; et al. A Simple Classification of Crohn’s Disease: Report of the Working Party for the World Congresses of Gastroenterology, Vienna 1998. Inflamm. Bowel Dis. 2000, 6, 8–15. [Google Scholar] [CrossRef]
- Sonnenberg, A.; Melton, S.D.; Genta, R.M. Frequent Occurrence of Gastritis and Duodenitis in Patients with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2011, 17, 39–44. [Google Scholar] [CrossRef]
- Wright, C.L.; Riddell, R.H. Histology of the Stomach and Duodenum in Crohn’s Disease. Am. J. Surg. Pathol. 1998, 22, 383–390. [Google Scholar] [CrossRef]
- Oberhuber, G.; Püspök, A.; Dejaco, C.; Hammer, J.; Zauner, C.; Pfeffel, F.; Fiedler, R.; Pötzi, R.; Allerberger, F.; Wrba, F. Minimal Chronic Inactive Gastritis: Indicator of Pre-Existing Helicobacter Pylori Gastritis? Pathol. Res. Pract. 1996, 192, 1016–1021. [Google Scholar] [CrossRef]
- Nasser, S.C.; Slim, M.; Nassif, J.G.; Nasser, S.M. Influence of Proton Pump Inhibitors on Gastritis Diagnosis and Pathologic Gastric Changes. World J. Gastroenterol. 2015, 21, 4599–4606. [Google Scholar] [CrossRef] [PubMed]
- Parente, F.; Cucino, C.; Bollani, S.; Imbesi, V.; Maconi, G.; Bonetto, S.; Vago, L.; Bianchi Porro, G. Focal Gastric Inflammatory Infiltrates in Inflammatory Bowel Diseases: Prevalence, Immunohistochemical Characteristics, and Diagnostic Role. Am. J. Gastroenterol. 2000, 95, 705–711. [Google Scholar] [CrossRef]
- Hooi, J.K.Y.; Lai, W.Y.; Ng, W.K.; Suen, M.M.Y.; Underwood, F.E.; Tanyingoh, D.; Malfertheiner, P.; Graham, D.Y.; Wong, V.W.S.; Wu, J.C.Y.; et al. Global Prevalence of Helicobacter Pylori Infection: Systematic Review and Meta-Analysis. Gastroenterology 2017, 153, 420–429. [Google Scholar] [CrossRef]
- Wu, X.W.; Ji, H.Z.; Yang, M.F.; Wu, L.; Wang, F.Y. Helicobacter Pylori Infection and Inflammatory Bowel Disease in Asians: A Meta-Analysis. World J. Gastroenterol. 2015, 21, 4750–4756. [Google Scholar] [CrossRef] [PubMed]
- Schmitz-Moormann, P.; Malchow, H.; Pittner, P. Endoscopic and Bioptic Study of the Upper Gastrointestinal Tract in Crohn’s Disease Patients. Pathol. Res. Pract. 1985, 179, 377–387. [Google Scholar] [CrossRef]
- Johnson, C.M.; Hartman, D.J.; Ramos-Rivers, C.; Rao, B.B.; Bhattacharya, A.; Regueiro, M.; Schwartz, M.; Swoger, J.; Al Hashash, J.; Barrie, A.; et al. Epithelioid Granulomas Associate with Increased Severity and Progression of Crohn’s Disease, Based on 6-Year Follow-Up. Clin. Gastroenterol. Hepatol. 2018, 16, 900–907. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | (n = 58) |
|---|---|
| Female, n (%) | 30 (51.7) |
| Age (y), mean ± SD | 42.1 ± 12.79 |
| Age at diagnosis (y), mean ± SD | 36.6 ± 12.26 |
| Duration of CD (m), median (range) | 48 (1-312) |
| Montreal disease age at diagnosis, n (%) | – |
| A1 (16 y or younger) | – |
| A2 (17–40 y) | 40 (69) |
| A3 (over 40 y) | 18 (31) |
| Montreal disease location, n (%) | – |
| L1 (terminal ileum) | 9 (15.5) |
| L2 (colonic) | 23 (39.7) |
| L3 (ileocolonic) | 26 (44.8) |
| L4 (upper gastrointestinal tract) | 3 (5.2) |
| Montreal disease behavior, n (%) | – |
| B1 (nonstricturing, nonpenetrating) | 36 (62.1) |
| B2 (stricturing) | 12 (20.7) |
| B3 (penetrating) | 10 (17.2) |
| Perianal | 23 (39.7) |
| Perianal fistula, n (%) | 17 (29.3) |
| HBI, n (%) | – |
| Remission | 35 (60.3) |
| Activity | 23 (39.7) |
| UGI symptoms, n (%) | 39 (67.2) |
| CRP elevated, n (%) | 16 (27.6) |
| Anemia, n (%) | 15 (25.9) |
| Smoking status, n (%) | – |
| Active | 6 (10.3) |
| Absent | 52 (89.7) |
| Alcohol status, n (%) | – |
| Active | 8 (13.8) |
| Absent | 50 (86.2) |
| Treatment, n (%) | 55 (94.8) |
| Sulfasalazine | 8 (13.8) |
| Topic mesalamine | 8 (13.8) |
| Oral mesalamine | 6 (10.3) |
| Azathioprine | 40 (69) |
| Methotrexate | 1 (1.7) |
| Adalimumab | 6 (10.3) |
| Infliximab | 12 (20.7) |
| Steroid therapy | 6 (10.3) |
| Surgery, n (%) | 13 (22.4) |
| Endoscopic Findings | Present Upper GI Symptoms | Absent Upper GI Symptoms | p Value |
|---|---|---|---|
| (n = 39) | (n = 19) | ||
| Normal endoscopy | 5 (12.8) | 2 (10.5) | 1.00 b |
| Altered endoscopy | 34 (87.2) | 17 (89.5) | – |
| Esophagus | – | – | – |
| Erosive esophagitis (Los Angeles) | 17 (43.6) | 8 (42.1) | 0.92 a |
| A | 14 (35.9) | 7 (36.8) | – |
| B | 2 (5.1) | 1 (5.3) | – |
| C | 1 (2.6) | – | – |
| Normal | 18 (46.2) | 10 (52.6) | 0.64 a |
| Stomach | – | – | – |
| Gastritis | 22 (56.4) | 10 (52.6) | 0.14 b |
| Erythematous | 11 (50) | 2 (20) | – |
| Erosive gastritis | 11 (50) | 8 (80) | – |
| Gastritis location | – | – | – |
| Body | 4 (18.2) | 1 (10) | 0.27 b |
| Antrum | 8 (36.4) | 7 (70) | 0.22 b |
| Pangastritis | 10 (45.4) | 2 (20) | – |
| Gastritis intensity | – | – | 0.14 b |
| Mild | 16 (72.7) | 10 (100) | – |
| Moderate to severe | 6 (27.3) | – | – |
| Nodularity | 7 (17.9) | 3 (15.8) | 1.00 b |
| Ulcers (Sakita classification) | – | – | – |
| A2 | 1 (100) | – | – |
| Normal | 13 (33.3) | 5 (26.3) | 0.59 a |
| Duodenum | – | – | – |
| Duodenitis | – | – | 1.00 b |
| Erythematous | 4 (10.3) | 1 (5.3) | – |
| Erosive | 5 (12.8) | 3 (15.8) | – |
| Ulcer (Sakita classification) | – | – | 0.43 a |
| A2 | 3 (7.7) | – | – |
| S2 | 2 (5.1) | 2 (10.5) | – |
| Normal | 23 (59) | 13 (68.4) | 0.47 a |
| Endoscopic Findings | Positive H. Pylori (n = 19) | Negative H. Pylori (n = 39) | p Value |
|---|---|---|---|
| Normal endoscopy | 1 (5.3) | 6 (15.4) | 0.41 b |
| Altered endoscopy | 18 (94.7) | 33 (84.6) | – |
| Esophagus | – | – | – |
| Erosive esophagitis | 8 (42.1) | 17 (43.6) | 0.91 a |
| Normal | 9 (47.7) | 19 (48.7) | 0.92 a |
| Stomach | – | – | – |
| Gastritis | 11 (57.9) | 21 (53.8) | 0.77 a |
| Erythematous | 5 (45.5) | 8 (38.1) | 0.72 b |
| Erosive | 6 (54.5) | 13 (61.9) | – |
| Gastritis location | – | – | 0.13 b |
| Body | 2 (18.2) | 3 (14.3) | – |
| Antrum | 3 (27.3) | 12 (57.1) | – |
| Pangastritis | 6 (54.5) | 6 (28.6) | – |
| Gastritis intensity | – | – | 0.15 b |
| Mild | 7 (63.6) | 19 (90.5) | – |
| Moderate to severe | 4 (36.4) | 2 (9.5) | – |
| Nodularity | 5 (26.3) | 5 (12.8) | 0.27 b |
| Ulcers (Sakita classification) | – | – | – |
| A2 | 1 (100) | – | – |
| Normal | 4 (21.1) | 14 (35.9) | 0.25 a |
| Duodenum | – | – | – |
| Duodenitis | – | – | – |
| Erythematous | 2 (10.5) | 3 (7.7) | 1.00 b |
| Erosive | 2 (10.5) | 6 (15.4) | |
| Ulcers (Sakita classification) | – | – | 1.00 b |
| A2 | 2 (10.5) | 1 (2.6) | – |
| S2 | 2 (10.5) | 2 (5.1) | – |
| Pseudopolyp | – | 1 | – |
| Flat lesions | 1 (5.3) | 2 (5.1) | – |
| Normal | 12 (63.2) | 24 (61.5) | 0.91 a |
| Histopathological Findings | Positive H. pylori (n = 19) | Negative H. pylori (n = 39) | p Value |
|---|---|---|---|
| Normal | 0 | 3 (7.7) | 0.54 b |
| Altered | 19 (100) | 36 (92.3) | – |
| Stomach | – | – | – |
| Antrum chronic gastritis | 19 (100) | 32 (82.1%) | 0.08 b |
| Activity | – | – | <0.001 a |
| Present | 18 (94.7) | 6 (15.4) | – |
| Absent | 1 (5.3) | 33 (84.6) | – |
| Body chronic gastritis | 19 (100) | 33 (84.6) | 0.16 b |
| Activity | – | – | <0.001 a |
| Present | 18 (94.7) | 4 (10.3) | – |
| Absent | 1 (5.3) | 35 (89.7) | – |
| Inflammatory cells | – | – | – |
| Mononuclear | 2 (10.5) | 35 (89.7) | <0.001 a |
| Mononuclear and granulocytes | 17 (89.5) | – | – |
| Absent | 0 | 4 (10.3) | – |
| Lamina propria infiltrate | – | – | 0.29 b |
| Present | 19 (100) | 35 (89.8) | – |
| Absent | – | 4 (10.3) | – |
| Permeation of glands by inflammatory cells | 14 (73.7) | – | <0.001 a |
| Foveolar hyperplasia | – | 3 (7.7) | – |
| Intestinal metaplasia | 1 (5.3) | – | – |
| Atrophy | 1 (5.3) | – | – |
| Focally enhanced gastritis | – | 4 (10.3) | – |
| Normal | – | 5 (12.8) | 0.16 b |
| Duodenum | – | – | – |
| Intraepithelial lymphocytosis | – | 1 (2.6) | – |
| Gastric metaplasia | 1 (5.3) | – | – |
| Duodenitis | 1 (5.3) | 3 (7.7) | – |
| Duodenitis (activity) | – | – | 1.00 b |
| Present | 1 (5.3) | 3 (7.8) | – |
| Absent | 18 (94.7) | 36 (92.3) | – |
| Duodenitis (inflammatory cells) | – | – | – |
| Mononuclear | 1 (5.3) | 2 (5.1) | – |
| Mononuclear and granulocytes | – | 1 (2.6) | – |
| Absent | 18 (94.7) | 36 (92.3) | – |
| Normal | 16 (84.2) | 31 (79.5) | 1.00 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pimentel, A.M.; Freitas, L.A.R.d.; Cruz, R.d.C.R.; Silva, I.N.d.N.; Andrade, L.D.; Marques, P.N.; Braga, J.C.; Fortes, F.M.L.; Brito, K.R.M.; Fontes, J.A.M.; et al. Endoscopic and Histopathological Findings of the Esophagus, Stomach, and Duodenum in Patients with Crohn’s Disease from a Reference Center in Bahia, Brazil. Clin. Pract. 2021, 11, 374-385. https://doi.org/10.3390/clinpract11020052
Pimentel AM, Freitas LARd, Cruz RdCR, Silva INdN, Andrade LD, Marques PN, Braga JC, Fortes FML, Brito KRM, Fontes JAM, et al. Endoscopic and Histopathological Findings of the Esophagus, Stomach, and Duodenum in Patients with Crohn’s Disease from a Reference Center in Bahia, Brazil. Clinics and Practice. 2021; 11(2):374-385. https://doi.org/10.3390/clinpract11020052
Chicago/Turabian StylePimentel, Andrea Maia, Luiz Antônio Rodrigues de Freitas, Rita de Cássia Reis Cruz, Isaac Neri de Novais Silva, Laíla Damasceno Andrade, Paola Nascimento Marques, Júlia Cordeiro Braga, Flora Maria Lorenzo Fortes, Katia Rejane Marques Brito, Jaciane Araújo Mota Fontes, and et al. 2021. "Endoscopic and Histopathological Findings of the Esophagus, Stomach, and Duodenum in Patients with Crohn’s Disease from a Reference Center in Bahia, Brazil" Clinics and Practice 11, no. 2: 374-385. https://doi.org/10.3390/clinpract11020052
APA StylePimentel, A. M., Freitas, L. A. R. d., Cruz, R. d. C. R., Silva, I. N. d. N., Andrade, L. D., Marques, P. N., Braga, J. C., Fortes, F. M. L., Brito, K. R. M., Fontes, J. A. M., Almeida, N. P., Surlo, V. C., Rocha, R., Lyra, A. C., & Santana, G. O. (2021). Endoscopic and Histopathological Findings of the Esophagus, Stomach, and Duodenum in Patients with Crohn’s Disease from a Reference Center in Bahia, Brazil. Clinics and Practice, 11(2), 374-385. https://doi.org/10.3390/clinpract11020052





