A Rare Cause of Drug-Induced Pancytopenia: Trimethoprim–Sulfamethoxazole-Induced Pancytopenia
Abstract
1. Introduction
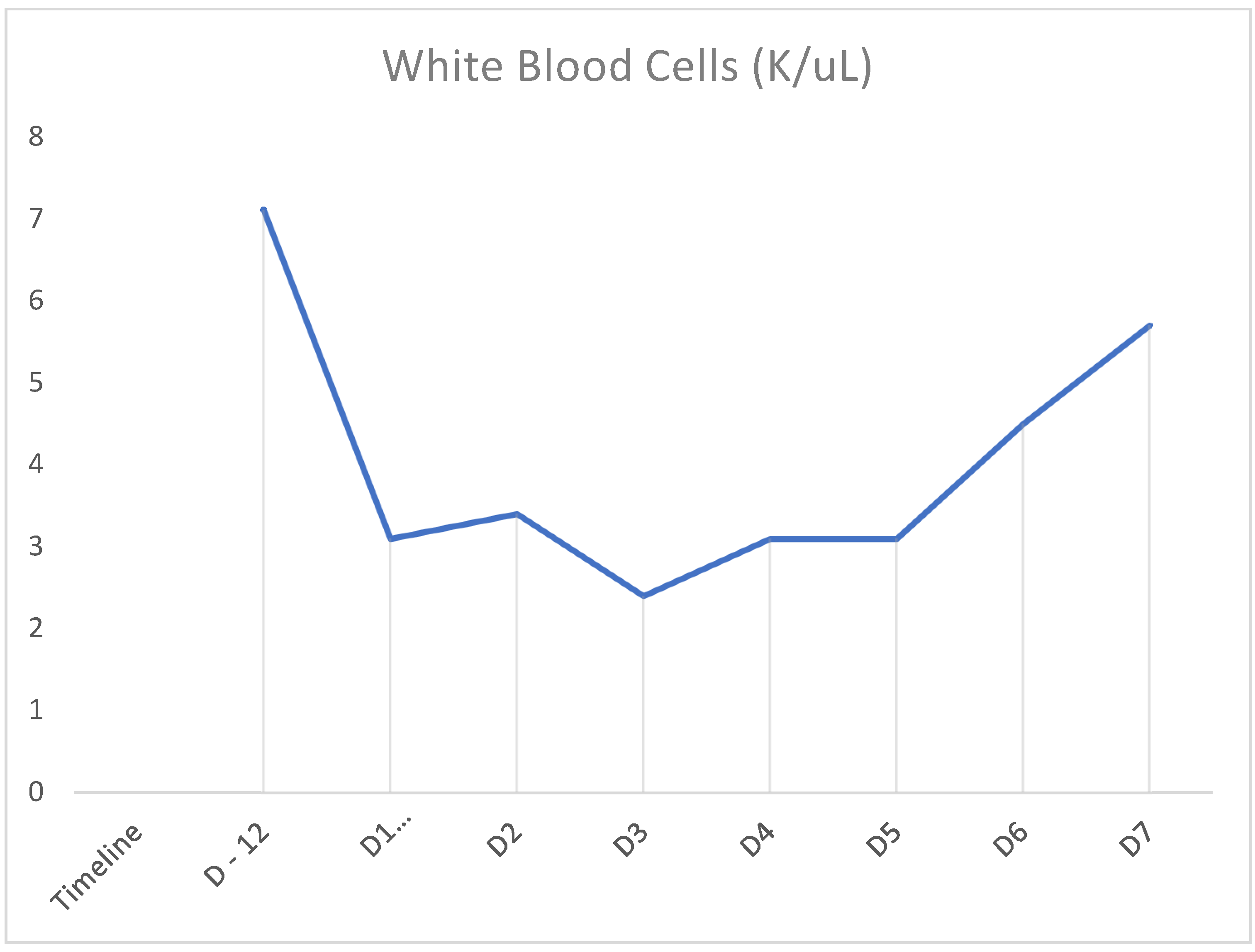
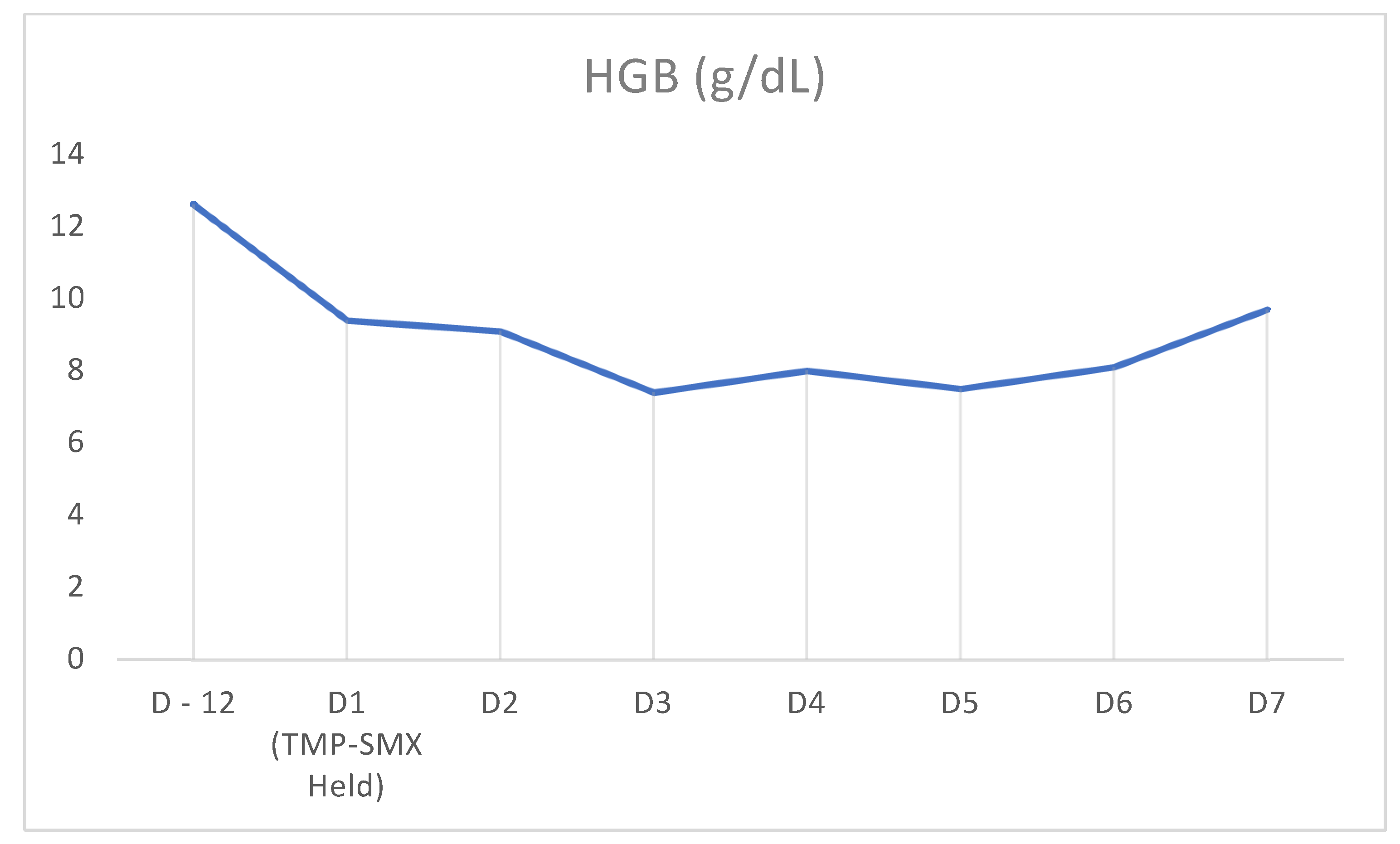
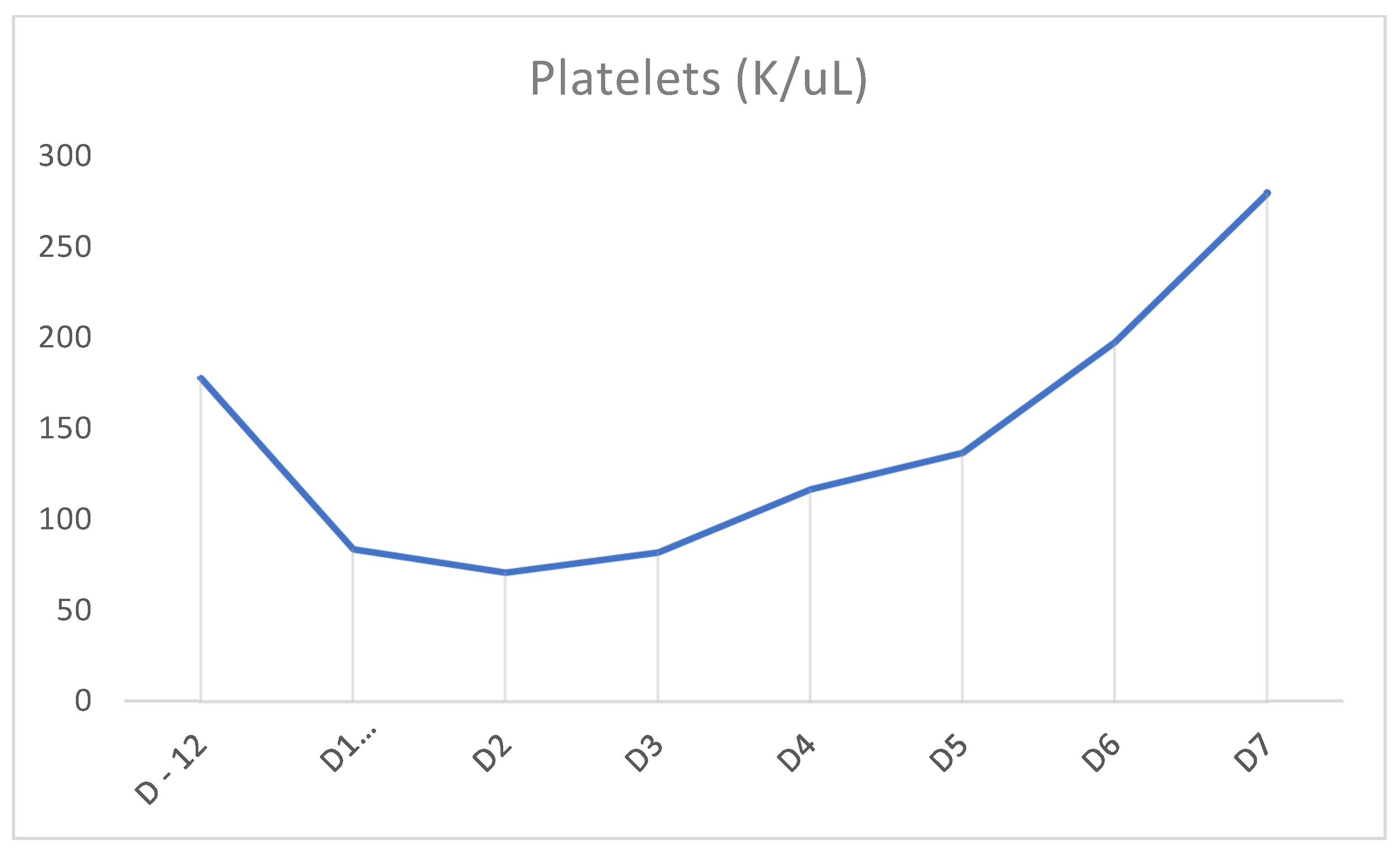
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Valent, P. Low blood counts: Immune mediated, idiopathic, or myelodysplasia. Hematol. Am. Soc. Hematol. Educ. Program. 2012, 2012, 485–491. [Google Scholar] [CrossRef]
- Young, N.S.; Abkowitz, J.L.; Luzzatto, L. New Insights into the Pathophysiology of Acquired Cytopenias. Hematol. Am. Soc. Hematol. Educ. Program. 2000, 1, 18–38. [Google Scholar] [CrossRef]
- Pascutti, M.F.; Erkelens, M.N.; Nolte, M.A. Impact of Viral Infections on Hematopoiesis: From Beneficial to Detrimental Effects on Bone Marrow Output. Front. Immunol. 2016, 7, 364. [Google Scholar] [CrossRef] [PubMed]
- Marks, P.W. Hematologic manifestations of liver disease. Semin. Hematol. 2013, 50, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Risitano, A.M.; Maciejewski, J.P.; Selleri, C.; Rotoli, B. Function and malfunction of hematopoietic stem cells in primary bone marrow failure syndromes. Curr. Stem. Cell. Res. Ther. 2007, 2, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Kalkut, G. Sulfonamides and trimethoprim. Cancer Investig. 1998, 16, 612–615. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.L.; Hossler, P.A.; Calhoun, D.H.; Meshnick, S.R. Inhibition of recombinant Pneumocystis carinii dihydropteroate synthetase by sulfa drugs. Antimicrob. Agents Chemother. 1995, 39, 1756–1763. [Google Scholar] [CrossRef] [PubMed]
- Pattishall, K.H.; Acar, J.; Burchall, J.J.; Goldstein, F.W.; Harvey, R.J. Two distinct types of trimethoprim-resistant dihydrofolate reductase specified by R-plasmids of different compatibility groups. J. Biol. Chem. 1977, 252, 2319–2323. [Google Scholar] [CrossRef]
- George, J.N.; Aster, R.H. Drug-induced thrombocytopenia: Pathogenesis, evaluation, and management. Hematology 2009, 1, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, G.C.; Hughes, D.T.; Hall, P.C. A haematological study of patients receiving long-term treatment with trimethoprim and sulphonamide. J. Clin. Pathol. 1970, 23, 392–396. [Google Scholar] [CrossRef] [PubMed]
| Pancytopenia Initial Laboratory Workup | Values | Reference Range |
|---|---|---|
| Reticulocyte Count | 3.15% | 0.6–2.3% |
| Neutrophil % | 85% | 40–70% |
| White Blood Cell | 3.1 K/μL | 4.5–11 |
| Hemoglobin | 9.4 g/dL | 13.5–18 |
| Platelet | 84 K/μL | 150–450 |
| Fibrinogen | 438 md/dL | 207–442 mg/dL |
| D-dimer | 2.22 ug/mlFEU | 0–0.48 ug/mlFEU |
| Lactic Acid | 0.9 mmol/L | 0.7–2 mmol/L |
| Uric Acid | 4.8 mg/dL | 2.5–6.2 mg/dL |
| Lactate Dehydrogenase | 576 U/L | 313–618 U/L |
| Vitamin B12 | 169 pg/mL | 230–931 pg/mL |
| Folate | 7.7 ng/mL | 2.70–19 ng/mL |
| Procalcitonin | 1.22 ng/mL | <0.5 ng/mL |
| Lithium | 0.6 mmol/L | 0.6–1.2 mmol/L |
| Tick Panel | Negative | |
| Hepatitis Panel | Negative | |
| COVID-19 | Negative | |
| Influenzae | Negative | |
| Blood Cultures | Negative | |
| CMV | Negative | |
| EBV | Negative |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sawalha, K.; Sobash, P.T.; Kamoga, G.-R. A Rare Cause of Drug-Induced Pancytopenia: Trimethoprim–Sulfamethoxazole-Induced Pancytopenia. Clin. Pract. 2021, 11, 358-362. https://doi.org/10.3390/clinpract11020050
Sawalha K, Sobash PT, Kamoga G-R. A Rare Cause of Drug-Induced Pancytopenia: Trimethoprim–Sulfamethoxazole-Induced Pancytopenia. Clinics and Practice. 2021; 11(2):358-362. https://doi.org/10.3390/clinpract11020050
Chicago/Turabian StyleSawalha, Khalid, Philip T. Sobash, and Gilbert-Roy Kamoga. 2021. "A Rare Cause of Drug-Induced Pancytopenia: Trimethoprim–Sulfamethoxazole-Induced Pancytopenia" Clinics and Practice 11, no. 2: 358-362. https://doi.org/10.3390/clinpract11020050
APA StyleSawalha, K., Sobash, P. T., & Kamoga, G.-R. (2021). A Rare Cause of Drug-Induced Pancytopenia: Trimethoprim–Sulfamethoxazole-Induced Pancytopenia. Clinics and Practice, 11(2), 358-362. https://doi.org/10.3390/clinpract11020050





