Maggot Therapy as a Part of a Holistic Approach in the Treatment of Multimorbid Patients with Chronic Ulcer
Abstract
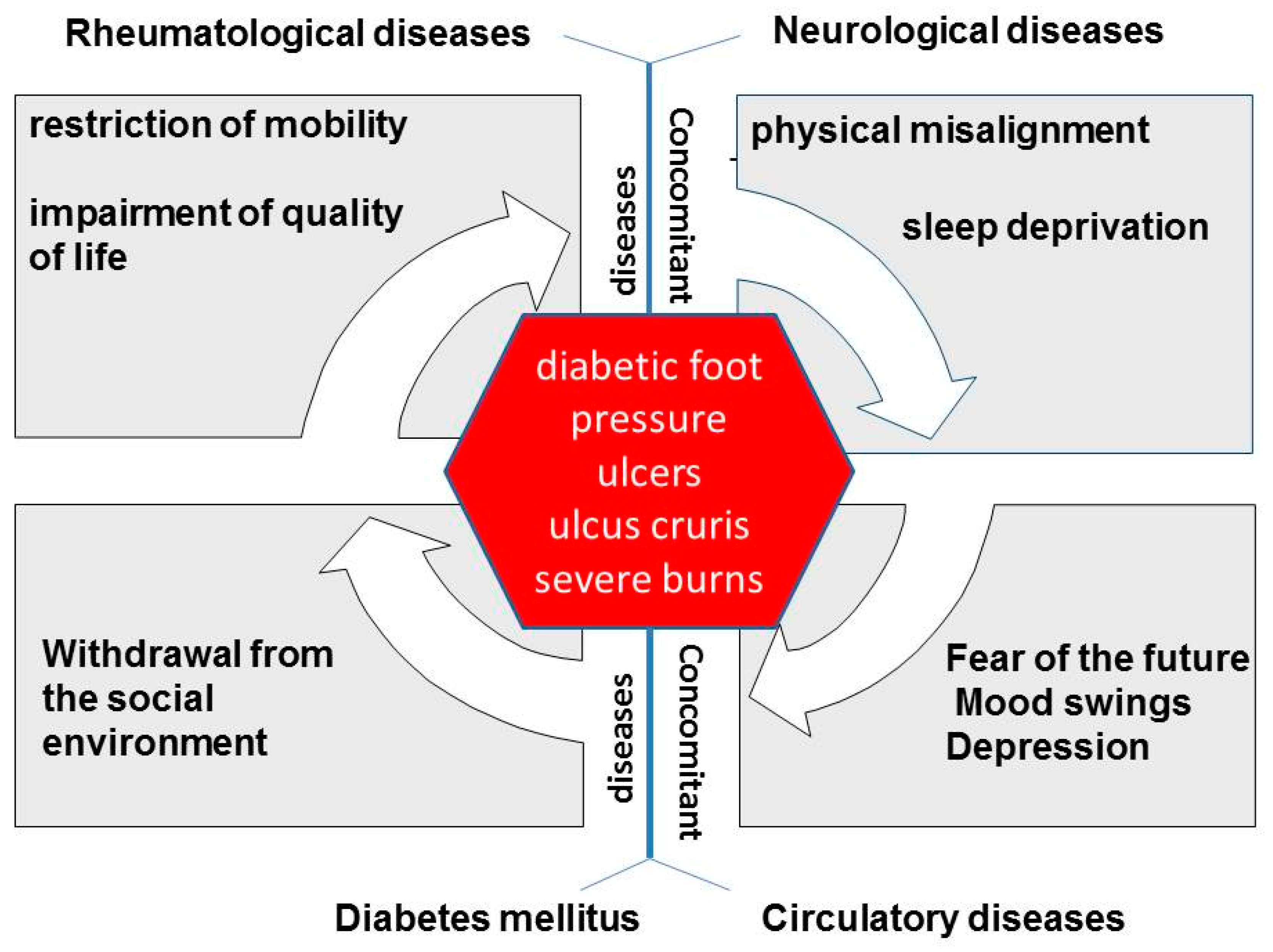
1. Introduction
2. Case Presentation
2.1. Diagnosis
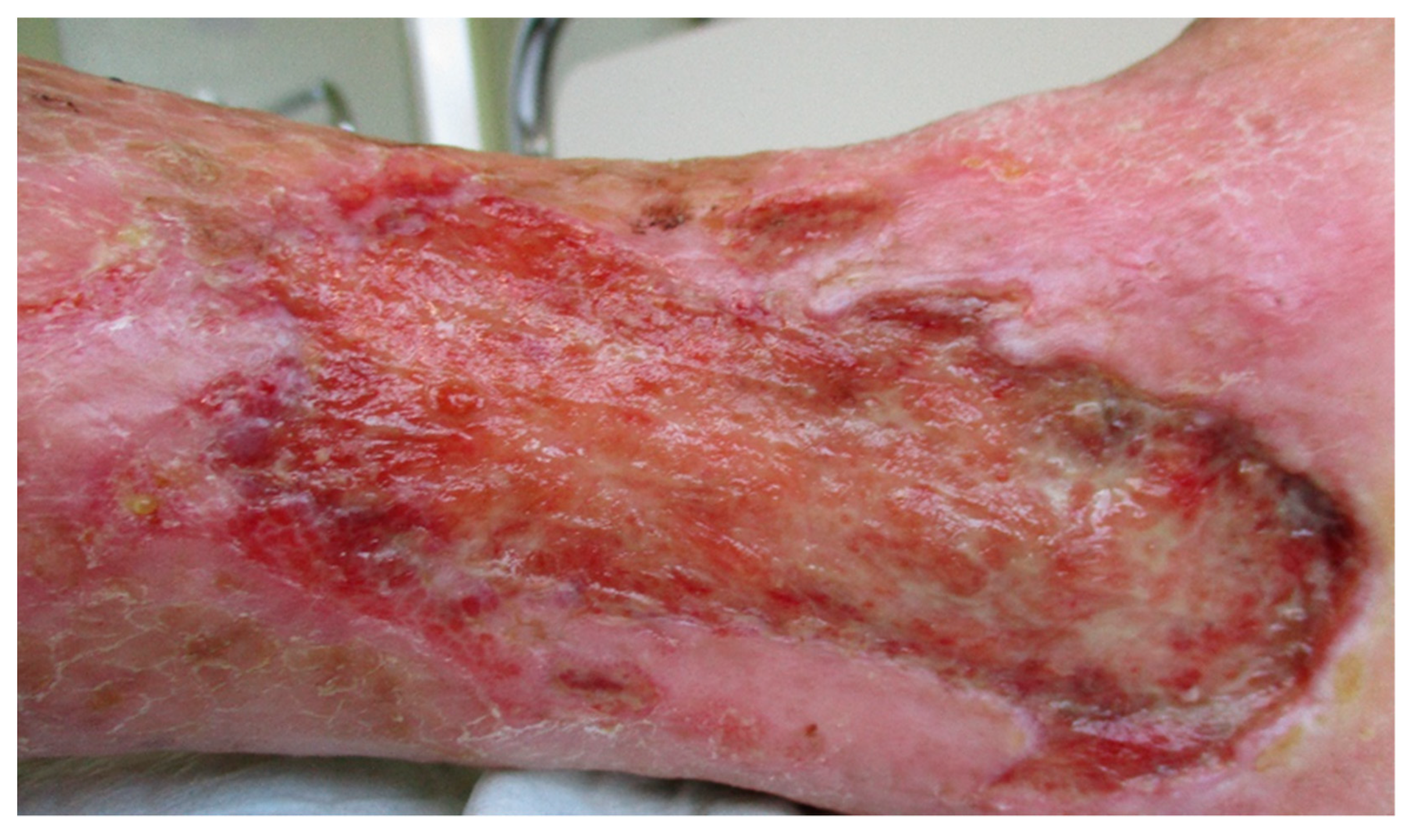
- Ulcus cruris varicosum, left lower leg, with chronic venous insufficiency with signs of inflammation and therapy-resistant course, first diagnosis, 03/1994.Locations of the lesions:
- -
- Lower leg left medial, distal third (12 cm × 4.6 cm), in existence since 1994;
- -
- Lower leg left medial, proximal from 1. (2 cm × 1 cm), time of origin unknown;
- -
- Lower leg left medial, anterior from 1. (2.5 cm × 1.5 cm), time of origin unknown;
- -
- Lower leg left lateral, malleolus lateralis (5 cm × 3 cm), since the end of 05/2020;
with sharp, tense pain. - Diabetic foot ulcer, left midfoot, no bacterial infection, since 1992;
- Pressure point with hematoma and fresh lesion 0.5 cm in diameter, right dorsum pedis between MCT 3 and 4, distal third;
- Lymphedema lower third of lower leg and left foot;
- Benign paroxismal vertigo;
- Normochromic normocytic anemia in chronic inflammation;
- Status after thrombosis of the popliteal vein and all 3 lower leg veins on the left, first diagnosis 04/2019;
- -
- History of anticoagulation with Clexane.
- Tendency to fall;
- Urge incontinence;
- Condition after hysterectomy and ovariectomy approx. 5 years ago;
- Condition after appendectomy at the age of 9–10 years;
- Condition after febrile bronchitis, 06/2019;
2.2. Cases Characteristics
3. Diagnostic
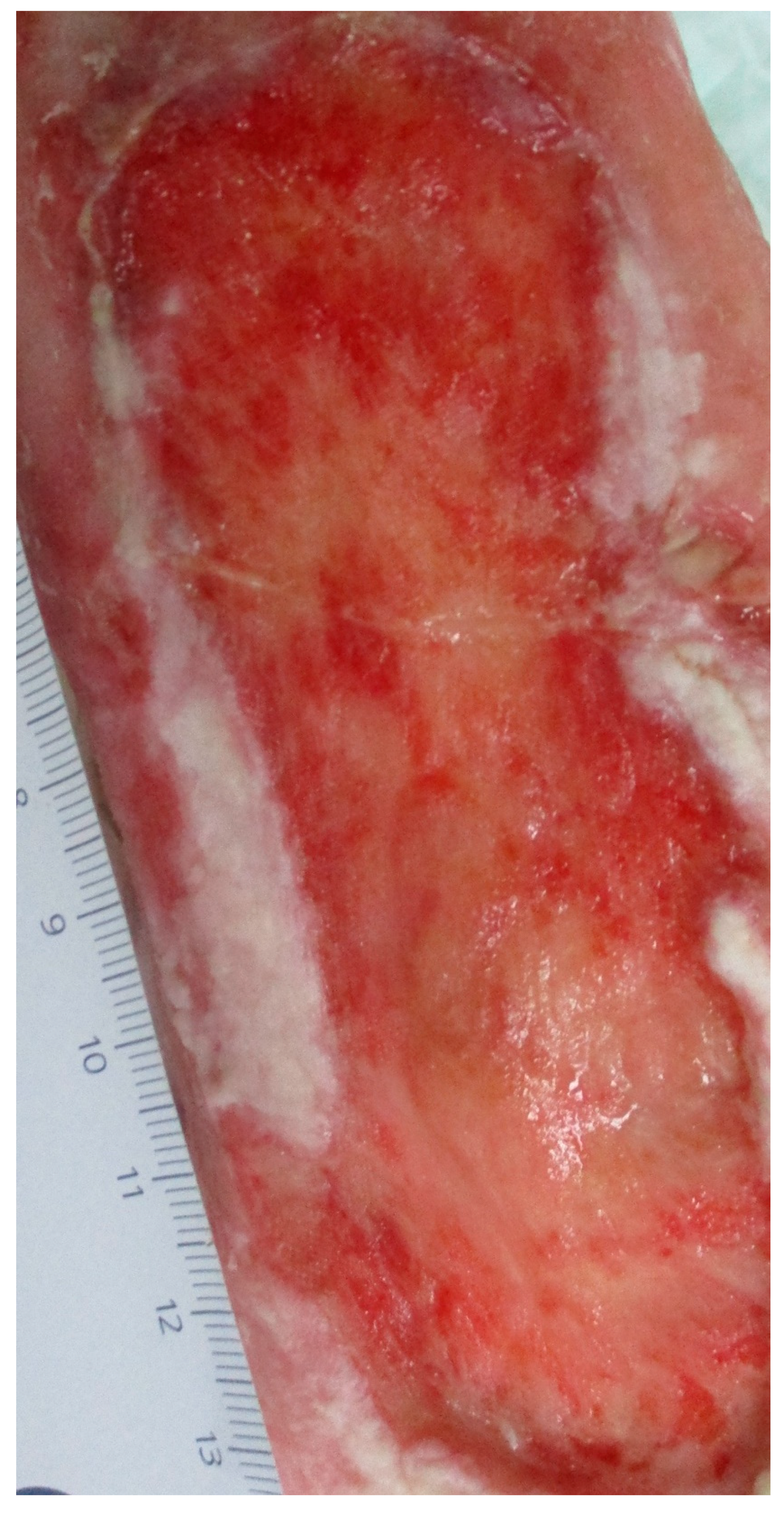
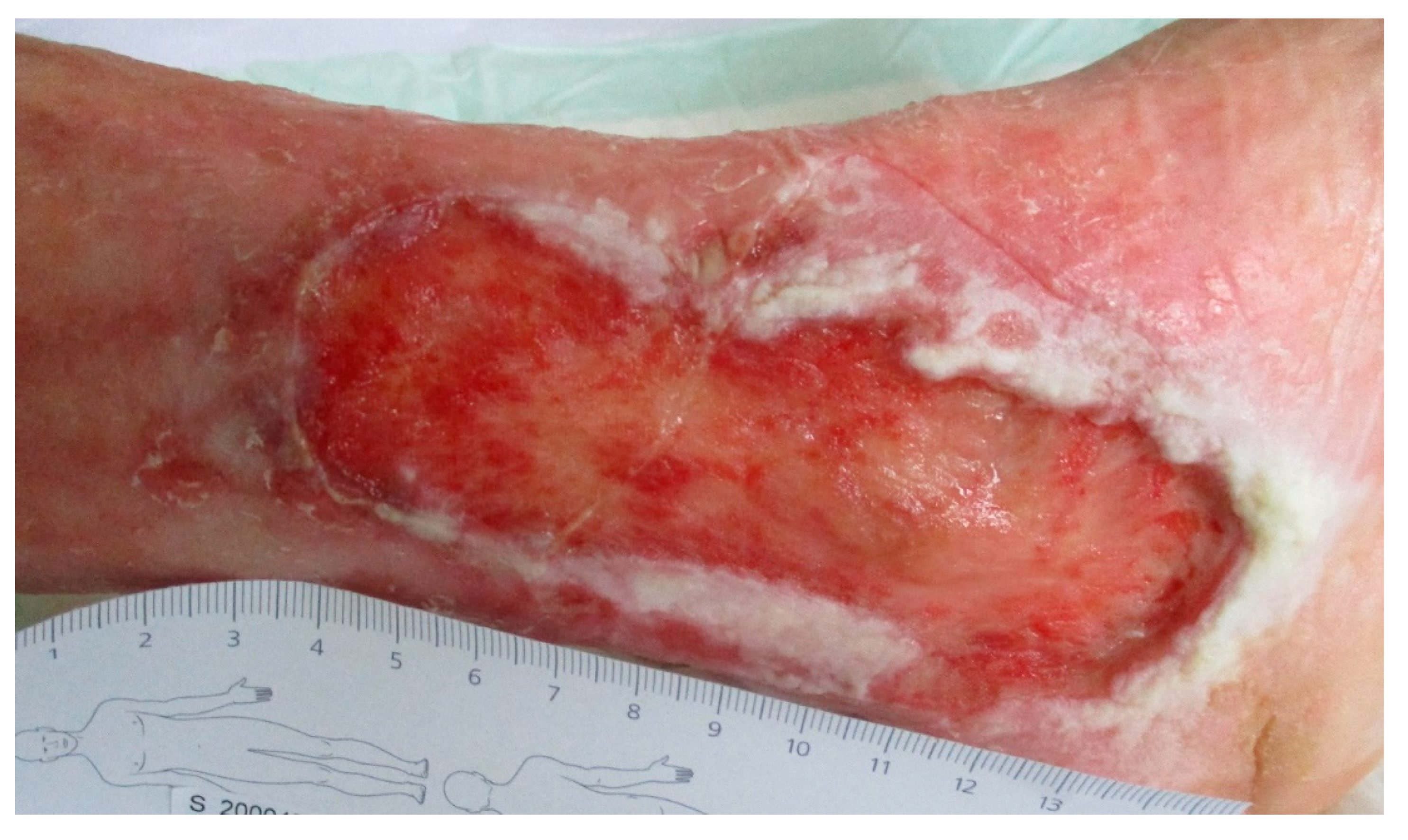
4. Treatment and Progress
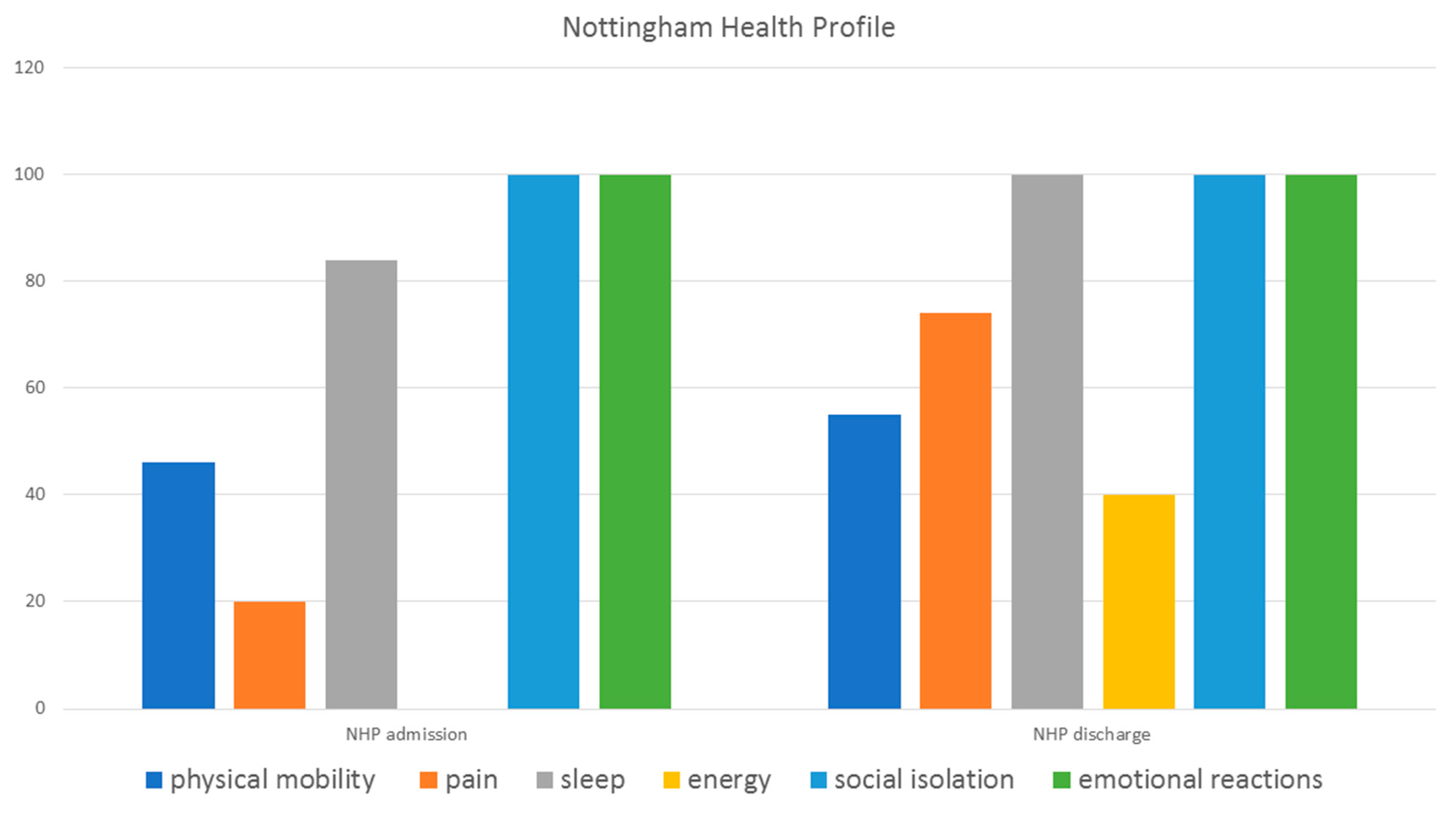
5. Discussion
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Müller-Bühl, U.; Leutgeb, R.; Bungartz, J.; Szecsenyi, J.; Laux, G. Expenditure of chronic venous leg ulcer management in German primary care: Results from a population–based study. Int. Wound J. 2013, 10, 52–56. [Google Scholar] [CrossRef]
- Newbern, S. Identifying Pain and Effects on Quality of Life from Chronic Wounds Secondary to Lower-Extremity Vascular Disease: An Integrative Review. Adv. Skin Wound Care 2018, 31, 102–108. [Google Scholar] [CrossRef]
- Cole-King, A.; Harding, K.G. Psychological factors and delayed healing in chronic wounds. Psychosom. Med. 2001, 63, 216–220. [Google Scholar] [CrossRef]
- Protz, K.; Heyer, K.; Dörler, M.; Stücker, M.; Hampel-Kalthoff, C.; Augustin, M. Kompressionstherapie—Kenntnisse und Anwendungspraxis. J. Dtsch. Dermatol. Ges. 2014, 12, 794–802. [Google Scholar] [CrossRef]
- Augustin, M.; Brocatti, L.K.; Rustenbach, S.J.; Schäfer, I.; Herberger, K. Cost-of-illness of leg ulcers in the community. Int. Wound J. 2014, 11, 283–292. [Google Scholar] [CrossRef]
- Nussbaum, S.R.; Carter, M.J.; Fife, C.E.; DaVanzo, J.; Haught, R.; Nusgart, M.; Cartwright, D. An economic evaluation of the impact, cost, and medicare policy implications of chronic non-healing wounds. Value Health 2018, 21, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Guest, J.F.; Ayoub, N.; McIlwraith, T.; Uchegbu, I.; Gerrish, A.; Weidlich, D. Health economic burden that different wound types impose on the UK’s National Health Service. Int. Wound J. 2017, 14, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, I.S.; Twine, C.; Whitaker, M.J.; Welck, M.; Brown, C.S.; Shandall, A. Larval therapy from antiquity to the present day: Mechanisms of action, clinical applications and future potential. Postgrad. Med. J. 2007, 83, 409–413. [Google Scholar] [CrossRef]
- Pechter, E.A.; Sherman, R.A. Maggot therapy: The surgical metamorphosis. Plast. Reconstr. Surg. 1983, 72, 567–570. [Google Scholar] [CrossRef]
- Sherman, R.A.; Hall, M.J.; Thomas, S. Medicinal maggots: An ancient remedy for some contemporary afflictions. Annu. Rev. Entomol. 2000, 45, 55–81. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S. Surgical Dressings and Wound Management; Medetec Publications: Cardiff, South Wales, UK, 2010; ISBN-10: 1-84426-834-9; ISBN-13: 978-1-84426-834-4. [Google Scholar]
- Gottrup, F.; Jørgensen, B. Maggot debridement: An alternative method for debridement. Eplasty 2011, 11, e33. [Google Scholar]
- Pöppel, A.K.; Koch, A.; Kogel, K.H.; Vogel, H.; Kollewe, C.; Wiesner, J. Lucimycin, an antifungal peptide from the therapeutic maggot of the common green bottle fly Lucilia sericata. Biol. Chem. 2014, 395, 649–656. [Google Scholar] [CrossRef]
- Stegeman, S.A.; Steenvoorde, P. Maggot debridement therapy. Proc. Neth. Entomol. Soc. Meet. 2011, 22, 61–66. [Google Scholar]
- Sherman, R.A. Maggot versus conservative debridement therapy for the treatment of pressure ulcers. Wound Repair Regen. 2002, 10, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Mudge, E.; Price, P.; Walkley, N.; Harding, K.G. A randomized controlled trial of larval therapy for the debridement of leg ulcers: Results of a multicenter, randomized, controlled, open, observer blind, parallel group study. Wound Repair Regen. 2014, 22, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Dumville, J.C.; Worthy, G.; Soares, M.O.; Bland, J.M.; Cullum, N.; Dowson, C.; Torgerson, D.J. VenUS II: A randomised controlled trial of larval therapy in the management of leg ulcers. Health Technol. Assess. 2009, 13, 1–182. [Google Scholar] [CrossRef]
- Opletalová, K.; Blaizot, X.; Mourgeon, B.; Chêne, Y.; Creveuil, C.; Combemale, P.; Laplaud, A.-L.; Sohyer-Lebreuilly, I.; Dompmartin, A. Maggot therapy for wound debridement: A randomized multicentertrial. Arch. Derm. 2012, 148, 432–438. [Google Scholar]
- Sherman, R.A.; Pechter, E.A. Maggot therapy: A review of the therapeutic applications of fly larvae in human medicine, especially for treating osteomyelitis. Med. Vet. Entomol. 1988, 2, 225–230. [Google Scholar] [CrossRef]
- Sherman, R.A.; Shimoda, K.J. Presurgical maggot debridement of soft tissue wounds is associated with decreased rates of postoperative infection. Clin. Infect. Dis. 2004, 39, 1067–1070. [Google Scholar] [CrossRef]
- Nasoori, A.; Hoomand, R. Maggot debridement therapy for an electrical burn injury with instructions for the use of Lucilia sericata larvae. J. Wound Care 2017, 26, 734–741. [Google Scholar] [CrossRef]
- Hwang, J.H.; Modi, H.N.; Suh, S.W.; Hong, J.Y.; Yang, J.H.; Park, J.H. Maggot debridement therapy for postsurgical wound infection in scoliosis: A case series in five patients. Spine 2011, 36, 313–319. [Google Scholar] [CrossRef]
- Romeyke, T.; Stummer, H. Evidence-based complementary and alternative medicine in inpatient care: Take a look at Europe. J. Evid. Based Complementary Altern. Med. 2015, 20, 87–93. [Google Scholar] [CrossRef]
- Pereira, R.; Santos, E.; Queiroz, M.; Junior, M.; Carvalho, M.; Salomé, G. Depression and wellness in elderly patients with venous ulcers. Rev. Bras. Cir. Plástica 2001, 29, 567–574. [Google Scholar] [CrossRef]
- Herber, O.R.; Schnepp, W.; Rieger, M.A. A systematic review on the impact of leg ulceration on patients’ quality of life. Health Qual. Life Outcomes 2007, 5, 44. [Google Scholar] [CrossRef]
- Yan, L.; Chu, J.; Li, M.; Wang, X.; Zong, J.; Zhang, X.; Song, M.; Wang, S. Pharmacological Properties of the Medical Maggot: A Novel Therapy Overview. Evid. Based Complement. Alternat. Med. 2018, 3, 4934890. [Google Scholar] [CrossRef] [PubMed]
- Bugaj, M.; Struzyna, J.; Madry, R.; Korzeniowski, T.; Antonov, S. The use of Lucilia sericata larvae in the treatment of burns. Chir. Plast. Oparz. 2014, 2, 91–96. [Google Scholar]
- Edwards, J.; Stapley, S. Debridement of diabetic foot ulcers. Cochrane Database Syst. Rev. 2010, 20, CD003556. [Google Scholar] [CrossRef]
- Dunville, J.C.; Worthy, G.; Bland, M.; Cullum, N.; Dowson, C.; Iglesias, C.; Torgerson, D.J. Larval therapy for leg ulcers (VenUS II): Randomized controlled trial. BMJ 2009, 338, b773. [Google Scholar] [CrossRef] [PubMed]
- Romeyke, T.; Scheuer, H.C.; Stummer, H. Physiotherapy as one column of an integrative and interdisciplinary medical approach—Impact of the introduction of diagnosis related groups in Germany. Complementary Ther. Med. 2013, 21, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Sherman, R.A.; Mendez, S.; McMillan, C. Using maggots in wound care: Part 1: Learn about this simple, effective, low-risk, low-cost wound debridement technique. Wound Care Advis. 2014, 3, 12. [Google Scholar]
- Sherman, R.A. Maggot therapy for treating diabetic foot ulcers unresponsive to conventional therapy. Diabetes Care 2003, 26, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Wayman, J.; Nirojogi, V.; Walker, A.; Sowinski, A.; Walker, M.A. The cost effectiveness of larval therapy in venous ulcers. J. Tissue Viability 2000, 10, 91–94. [Google Scholar] [CrossRef]

| Indicator | Value | Further Information/Associations with Multiple Comorbidities |
|---|---|---|
| Neutrophil, Tsd/µL | 5.84 | Norm: 1.9–6.1 Infections with bacteria, viruses, fungi, or parasites can increase the value. |
| Basophil, Tsd/µL | 0.02 | Norm: ≤0.08. Diseases with higher concentrations of lipids in the blood (Diabetes mellitus, Nephropathien, Myxedema) can be associated with higher levels of basophiles. |
| Creatine, mg/dL | 0.55 | Norm: ≤1.10 (for individuals over 60). In case the norm is surpassed, the reasons are either acute kidney failure, chronic kidney disease, or desiccosis (lack of water, dehydration). |
| Lymphocyte absolute Tsd/µL | 1.93 | |
| C-reactive protein (CPR), mg/L | 37.1 | Norm: ≤5. Elevated CRP-levels are associated with bacterial and viral infections, rheumatic diseases, coronary diseases, heart attacks, etc. |
| Triglyceride, mg/dL | 61 | Norm: <150. Elevated values indicate metabolism disorders. Patients with diabetes, kidney diseases, or overweight often have higher levels. |
| Urea, mg/dL | 26 | Norm:10–50. Higher values in the blood serum indicate a reduced kidney function. |
| Uric acid, mg/dL | 3.8 | Norm: ≤7. Higher levels indicate chronic kidney diseases, diabetes, lipid metabolism disorders. |
| Lactate dehydrogenase, U/L | 159 | Norm: ≤250. Higher values indicate coronary heart diseases, myocarditis, cardiac arrhythmias, skeletal muscle diseases. |
| TSH basal µIU/mL | 0.73 | Norm: 0.27–4.2. |
| Protein electrophoresis | Albumin, Alpha-1-Globulin, Alpha-2-Globulin, Beta-Globulin, Gamma globulin. Indicates chronic liver diseases, acute and chronic inflammations, lack of protein, monoclonal gammopathies. | |
| Albumin % | 46.8% | Norm: 54.7–66.0 |
| Alpha-1-Globulin % | 7.2% | Norm: 3.1–5.6 |
| Alpha-2-Globulin % | 12.7% | Norm: 6.8–13.7 |
| Beta-Globulin % | 14.7% | Norm: 9.0–13.7 |
| Gamma globulin % | 18.6% | Norm: 10.6–19.8 |
| Bilirubin mg/dL | 0.21 | Norm: 0–1.2 Higher values indicate damage to liver cells from drugs or infections with salmonella. |
| Erythrocytes Mio/µL | 3.76 | Norm: 3.9–5.2 If erythrocytes <3.9 that indicates anemia. |
| Iron µg/dl | 22 | Norm: 40–160 An iron deficiency leads to fatigue general feeling of weakness combined with fatigue and loss of performance, dizziness, and shortness of breath. |
| Hemoglobin g/dL | 10.4 | Norm: 12–16 A low hemoglobin level indicates that a person has too little iron in their blood. The transport of oxygen and the formation of new red blood cells (erythrocytes) are then at risk. |
| Hematocrit % | 33 | Norm: 36–46 A hematocrit value below the normal range can be the result of anemia or blood loss, but it can also be caused by excessive fluid consumption. |
| Red Blood Cell Distribution Width % | 16.7 | Norm: <15 Higher values indicate anemias (e.g., hemolytic anemia, iron deficiency anemia, pernicious anemia, or spheroidal cell anemia) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romeyke, T. Maggot Therapy as a Part of a Holistic Approach in the Treatment of Multimorbid Patients with Chronic Ulcer. Clin. Pract. 2021, 11, 347-357. https://doi.org/10.3390/clinpract11020049
Romeyke T. Maggot Therapy as a Part of a Holistic Approach in the Treatment of Multimorbid Patients with Chronic Ulcer. Clinics and Practice. 2021; 11(2):347-357. https://doi.org/10.3390/clinpract11020049
Chicago/Turabian StyleRomeyke, Tobias. 2021. "Maggot Therapy as a Part of a Holistic Approach in the Treatment of Multimorbid Patients with Chronic Ulcer" Clinics and Practice 11, no. 2: 347-357. https://doi.org/10.3390/clinpract11020049
APA StyleRomeyke, T. (2021). Maggot Therapy as a Part of a Holistic Approach in the Treatment of Multimorbid Patients with Chronic Ulcer. Clinics and Practice, 11(2), 347-357. https://doi.org/10.3390/clinpract11020049




