Proliferative Verrucous Leukoplakia Revisited: A Retrospective Clinicopathological Study
Abstract
1. Introduction
2. Materials and Methods
- (1)
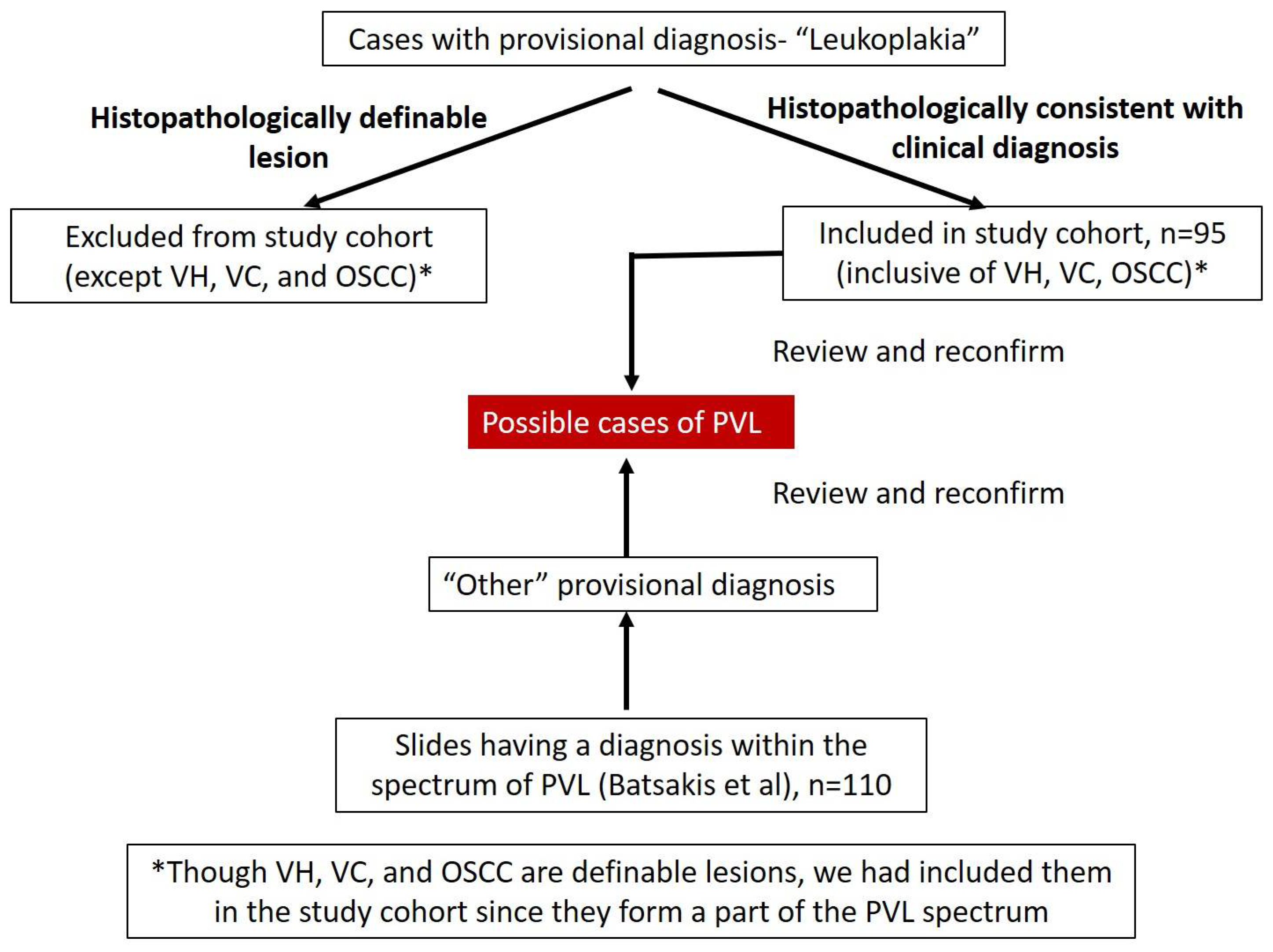
- Patients with a provisional diagnosis of leukoplakia, and their corresponding slides. Cases with any histopathological label with the clinical diagnosis of leukoplakia (n = 95) were included in the study (group 1). Any histopathological diagnosed cases without the available clinical information were excluded from the study.
- (2)
- A diagnosis of PVL is usually made retrospectively, since it represents a disease that progresses in a continuum over time. There are chances that the patient may have PVL in any one of the transitional stages of the continuum. Hence, we also extracted archival slides of other oral lesions (n = 110), with a histological diagnosis within the spectrum of PVL (group 2), described by Batsakis et al. [2]; i.e., VH, VC, and OSCC (Table 2).
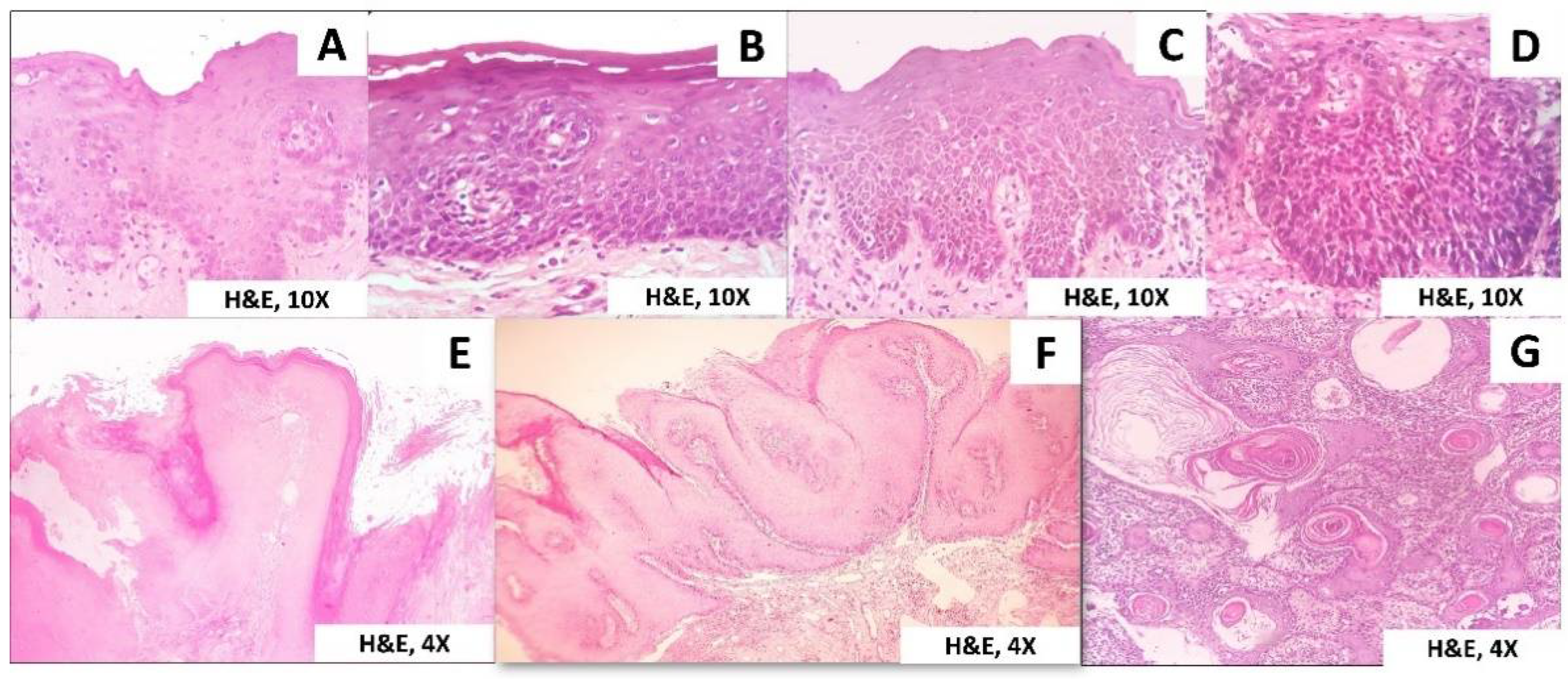
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hansen, L.S.; Olson, J.A.; Silverman, S. Proliferative verrucous leukoplakia: A long-term study of thirty patients. Oral Surg. Oral Med. Oral Pathol. 1985, 60, 285–298. [Google Scholar] [CrossRef]
- Batsakis, J.G.; Suarez, P.; El-Naggar, A.K. Proliferative verrucous leukoplakia and its related lesions. Oral Oncol. 1999, 35, 354–359. [Google Scholar] [CrossRef]
- Zakrzewska, J.M.; Lopesm, V.; Speightm, P.; Hopper, C. Proliferative verrucous leukoplakia: A report of ten cases. Oral Surg. Oral Med. Oral Pathol. 1996, 82, 396–401. [Google Scholar]
- Silverman, S.; Gorsky, M. Proliferative verrucous leukoplakia: A follow-up study of 54 cases. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1997, 84, 154–157. [Google Scholar] [CrossRef]
- Ghazali, N.; Bakri, M.M.; Zain, R.B. Aggressive, multifocal oral verrucous leukoplakia: Proliferative verrucous leukoplakia or not? J. Oral Pathol. Med. 2003, 32, 383–392. [Google Scholar] [CrossRef]
- Bagán, J.V.; Murillo, J.; Poveda, R.; Gavaldá, C.; Jiménez, Y.; Scully, C. Proliferative verrucous leukoplakia: Unusual locations of oral squamous cell carcinomas, and field cancerization as shown by the appearance of multiple OSCCs. Oral Oncol. 2004, 40, 440–443. [Google Scholar] [CrossRef]
- Cabay, R.J.; Morton, T.H.; Epstein, J.B. Proliferative verrucous leukoplakia and its progression to oral carcinoma: A review of the literature. J. Oral Pathol. Med. 2007, 36, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Bagan, J.; Scully, C.; Jimenez, Y.; Martorell, M. Proliferative verrucous leukoplakia: A concise update. Oral Dis. 2010, 16, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Wu, Y.; Wu, L.; Zhang, L.; Xie, B.; Zeng, X.; Lin, M.; Zhou, H. Case report of rapidly progressive proliferative verrucous leukoplakia and a proposal for aetiology in mainland China. World J. Surg. Oncol. 2011, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Issrani, R.; Prabhu, N.; Keluskar, V. Oral proliferative verrucous leukoplakia: A case report with an update. Contemp. Clin. Dent. 2013, 4, 258. [Google Scholar] [CrossRef] [PubMed]
- Kharma, M.Y.; Tarakji, B. Current Evidence in Diagnosis and Treatment of Proliferative Verrucous Leukoplakia. Ann. Saudi Med. 2012, 32, 412–414. [Google Scholar] [CrossRef]
- van der Waal, I.; Reichart, P.A. Oral proliferative verrucous leukoplakia revisited. Oral Oncol. 2008, 44, 719–721. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.S.; Patil, S.; Sanketh, D. Oral proliferative verrucous leukoplakia- A case report. Int. J. Clin. Dent. Sci. 2014, 5, 20–24. [Google Scholar]
- El-Naggar, A.K.; Chan, J.K.C.; Grandis, J.R.; Takata, T.; Slootweg, P.J. Tumours of the oral cavity and mobile tongue. In WHO Classification of Tumours of the Head and Neck, 4th ed.; IARC Press: Lyon, France, 2017. [Google Scholar]
- Cerero-Lapiedra, R.; Baladé-Martínez, D.; Moreno-López, L.-A.; Esparza-Gómez, G.; Bagán, J.V. Proliferative verrucous leukoplakia: A proposal for diagnostic criteria. Med. Oral Patol. Oral Cir. Bucal. 2010, 15, 839–845. [Google Scholar] [CrossRef]
- Carrard, V.; Brouns, E.; van der Waal, I. Proliferative verrucous leukoplakia; a critical appraisal of the diagnostic criteria. Med. Oral Patol. Oral Cir. Bucal. 2013, 18, 411–413. [Google Scholar] [CrossRef]
- Garcia-Chias, B.; Casado-De La Cruz, L.; Esparza-Gomez, G.; Cerero-Lapiedra, R. Diagnostic criteria in proliferative verrucous leukoplakia: Evaluation. Med. Oral Patol. Oral y Cir. Bucal. 2014, 19, 335–339. [Google Scholar] [CrossRef]
- Villa, A.; Menon, R.S.; Kerr, A.R.; De Abreu Alves, F.; Guollo, A.; Ojeda, D.; Woo, S.B. Proliferative leukoplakia: Proposed new clinical diagnostic criteria. Oral Dis. 2018, 24, 749–760. [Google Scholar] [CrossRef]
- Gillenwater, A.M.; Vigneswaran, N.; Fatani, H.; Saintigny, P.; El-Naggar, A.K. Proliferative verrucous leukoplakia: Recognition and differentiation from conventional leukoplakia and mimics. Head Neck 2014, 36, 1662–1668. [Google Scholar] [CrossRef] [PubMed]
- Ha, P.K.; Califano, J.A. The molecular biology of mucosal field cancerization of the head and neck. Crit. Rev. Oral Biol. Med. 2003, 14, 363–369. [Google Scholar] [CrossRef]
- Braakhuis, B.J.M.; Tabor, M.P.; Kummer, J.A.; Leemans, C.R.; Brakenhoff, R.H. A genetic explanation of Slaughter’s concept of field cancerization: Evidence and clinical implications. Cancer Res. 2003, 63, 1727–1730. Available online: http://www.ncbi.nlm.nih.gov/pubmed/12702551 (accessed on 2 August 2020).
- Sarode, S.; Anand, R.; Sarode, G.; Patil, S. Somatic Mutation Theory/Tissue Organization Field Theory: Has the Premise been Wrong All along? World J. Dent. 2016, 7, 167–168. [Google Scholar]
- Murrah, V.A.; Batsakis, J.G. Proliferative Verrucous Leukoplakia and Verrucous Hyperplasia. Ann. Otol. Rhinol. Laryngol. 1994, 103, 660–663. [Google Scholar] [CrossRef] [PubMed]
- Favia, G.; Capodiferro, S.; Limongelli, L.; Tempesta, A.; Maiorano, E. Malignant transformation of oral proliferative verrucous leukoplakia: A series of 48 patients with suggestions for management. Int. J. Oral Maxillofac. Surg. 2021, 50, 14–20. [Google Scholar] [CrossRef]
- Upadhyaya, J.D.; Fitzpatrick, S.G.; Islam, M.N.; Bhattacharyya, I. Cohen DM. A retrospective 20-year analysis of proliferative verrucous leukoplakia and its progression to malignancy and association with high-risk human papillomavirus. Head Neck Pathol. 2018, 12, 500–510. [Google Scholar] [CrossRef] [PubMed]
| Major Criteria (MC) | Minor Criteria (mc) |
|---|---|
| A leukoplakia lesion with more than two different oral sites (A) | Leukoplakia lesion occupies at least 3 cm when adding all the affected areas (a) |
| Existence of a verrucous area (B) | Patient is female (b) |
| Lesions have spread or engrossed during the course of the disease (C) | Patient (male or female) is a non-smoker (c) |
| Recurrence in a previously treated area (D) | Disease evolution higher than 5 years (d) |
| Histopathology diagnosis (E *) |
| Provisional Diagnosis | Final Diagnosis | |||||||
|---|---|---|---|---|---|---|---|---|
| Hyperkeratosis | Dysplasia | VH | VC | OSCC | Total | |||
| M | Mo | S | ||||||
| Leukoplakia | 18 | 40 | 20 | 02 | 05 | 02 | 08 | 95 |
| Others * | 7 | 6 | 5 | 10 | 6 | 7 | 69 | 110 |
| Total | 25 | 46 | 25 | 12 | 11 | 9 | 77 | 205 |
| Lesions | Hyperkeratosis | Epithelial Dysplasia | VC | VH | OSCC | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| n = 25 | n = 83 | n = 9 | n = 11 | n = 77 | ||||||
| M n = 46 | Mo n = 25 | S n = 12 | WD n = 52 | MD n = 22 | PD n = 3 | |||||
| Age | Mean | 47.4 | 45 | 49.2 | 56 | 64.7 | 54.45 | 56.44 | 53.8 | 47.66 |
| SD | 16.317 | 14.995 | 14.957 | 10.807 | 9.022 | 17.403 | 12.475 | 13.757 | 24.00 | |
| Range | 20–71 | 19–75 | 28–79 | 41–72 | 46–78 | 28–85 | 28–83 | 29–75 | 18–65 | |
| Gender | Male | 17 | 40 | 23 | 8 | 6 | 9 | 23 | 13 | 2 |
| Female | 8 | 6 | 2 | 4 | 3 | 2 | 29 | 9 | 1 | |
| Site of lesion | Buccal mucosa | 20 | 37 | 17 | 8 | 4 | 7 | 28 | 5 | 1 |
| Gingiva | 4 | 0 | 2 | 1 | 1 | 1 | 2 | 0 | 1 | |
| Labial mucosa | 2 | 2 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | |
| Gingival buccal Sulcus | 0 | 0 | 0 | 0 | 2 | 3 | 5 | 1 | 0 | |
| Retro molar Trigone | 4 | 6 | 5 | 0 | 2 | 1 | 2 | 3 | 0 | |
| Lower Lip | 2 | 3 | 1 | 0 | 0 | 2 | 3 | 0 | 0 | |
| Tongue | 1 | 1 | 2 | 2 | 0 | 2 | 4 | 7 | 0 | |
| Alveolar ridge | 2 | 4 | 2 | 2 | 2 | 1 | 10 | 7 | 1 | |
| Palate | 0 | 1 | 0 | 0 | 0 | 1 | 3 | 1 | 1 | |
| Description | Patch | 22 | 35 | 20 | 6 | 1 | 4 | 6 | 1 | 0 |
| Exophytic (Plaque/Verruco- Papillary | 3 | 9 | 1 | 1 | 5 | 2 | 5 | 2 | 2 | |
| Ulcer | 0 | 2 | 2 | 2 | 0 | 0 | 19 | 6 | 1 | |
| Ulcero-proliferative | 0 | 0 | 2 | 3 | 3 | 5 | 22 | 13 | 0 | |
| Colour | White | 20 | 40 | 15 | 8 | 3 | 7 | 18 | 4 | 0 |
| Red/pink | 0 | 0 | 1 | 3 | 0 | 1 | 7 | 5 | 1 | |
| Mixed (Red-White) | 5 | 6 | 9 | 1 | 6 | 3 | 27 | 13 | 2 | |
| Size(cm) | <1 | 2 | 3 | 1 | 2 | 0 | 1 | 5 | 1 | 0 |
| 1–2 | 20 | 18 | 13 | 3 | 1 | 4 | 25 | 9 | 3 | |
| 2–4 | 0 | 19 | 10 | 6 | 7 | 0 | 18 | 12 | 0 | |
| >4 | 3 | 6 | 1 | 1 | 1 | 6 | 4 | 0 | 0 | |
| Habits | Smokeless tobacco | 10 | 16 | 10 | 9 | 7 | 5 | 41 | 13 | 2 |
| Smoking | 6 | 22 | 17 | 6 | 2 | 4 | 20 | 11 | 2 | |
| Betel quid | 2 | 3 | 0 | 2 | 0 | 1 | 2 | 0 | 0 | |
| Alcohol | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | |
| No Habits | 10 | 7 | 1 | 1 | 2 | 2 | 3 | 1 | 1 | |
| Major Criteria (mc) | No. of Cases with Positive Criteria n (%) | Minor Criteria (mc) | No. of Cases with Positive Criteria n (%) |
|---|---|---|---|
| A leukoplakia lesion with more than two different oral sites (A) | 15 (7.3%) | Leukoplakia lesion occupies at least 3 cm when adding all the affected areas (a) | 47 (22.9%) |
| Existence of a verrucous area (B) | 30 (14%) | The patient is female (b) | 64 (31.2%) |
| Lesions have spread or engrossed during the course of the disease (C) | 2 (0.9%) | Patient (male or female) is a non-smoker (c) | 28 (13.6%) |
| Recurrence in a previously treated area (D) | 2 (0.9%) | Disease evolution higher than 5 years (d) | 0 |
| Histopathology diagnosis (E) | 205 (100%) | ||
| Total number of patients satisfying criteria for PVL–11 | |||
| Case | Age | Sex | Site * | Habit ** | Provisional Diagnosis | Microscopic Diagnosis | Disease Evolution (No. of Years) | MC/mc Combination | |
|---|---|---|---|---|---|---|---|---|---|
| Group 1 | 1 | 56 | F | B (L and R), P | ST | Leukoplakia | Well Diff SCC | NA | AE/ab |
| 2 | 71 | M | B (L and R), RMT | S, ST | Leukoplakia | Hyperkeratosis without dysplasia | NA | ABE/a | |
| 3 | 60 | F | B, AR, RMT | ST | Leukoplakia | Well Diff SCC | NA | AE/ab | |
| 4 | 65 | F | L, TL | ST | Leukoplakia | Mod. Diff SCC | NA | AE/ab | |
| 5 | 42 | F | B (R and L), RMT | NO HABITS | Leukoplakia | Mod. Epithelial Dysplasia | NA | ABE/abc | |
| Group 2 | 6 | 40 | F | B (R & L) | NO HABITS | Verrucous Leukoplakia | Early Invasive SCC | NA | BE/bc |
| 7 | 45 | F | B, AR | NO HABITS | Carcinoma | Well Diff SCC | NA | BE/bc | |
| 8 | 59 | M | AR, TL, TD | S | Carcinoma | Mod. Diff SCC | NA | BDE/a | |
| 9 | 46 | M | RMT, B, G | ST | Verrucous Leukoplakia | Microinvasive SCC | NA | ABE/a | |
| 10 | 66 | F | L | NO HABITS | Carcinoma | Well Diff SCC | NA | DE/bc | |
| 11 | 70 | M | B (R and L), L | S, ST | Verrucous Leukoplakia | Verrucous Hyperplasia | NA | ABE/a | |
| PVL | Total | Age (Mean) | Sex | Habits n (%) | |||||
| Male n (%) | Female n (%) | ||||||||
| 11 | 56.36 | 7 (63.6 %) | 4 (36.4 %) | 7 (63.63%) | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghosh, S.; Rao, R.S.; Upadhyay, M.K.; Kumari, K.; Sanketh, D.S.; Raj, A.T.; Parveen, S.; Alhazmi, Y.A.; Jethlia, A.; Mushtaq, S.; et al. Proliferative Verrucous Leukoplakia Revisited: A Retrospective Clinicopathological Study. Clin. Pract. 2021, 11, 337-346. https://doi.org/10.3390/clinpract11020048
Ghosh S, Rao RS, Upadhyay MK, Kumari K, Sanketh DS, Raj AT, Parveen S, Alhazmi YA, Jethlia A, Mushtaq S, et al. Proliferative Verrucous Leukoplakia Revisited: A Retrospective Clinicopathological Study. Clinics and Practice. 2021; 11(2):337-346. https://doi.org/10.3390/clinpract11020048
Chicago/Turabian StyleGhosh, Snehashish, Roopa S. Rao, Manoj K. Upadhyay, Karuna Kumari, D. Sharathkumar Sanketh, A. Thirumal Raj, Sameena Parveen, Yaser Ali Alhazmi, Ankur Jethlia, Shazia Mushtaq, and et al. 2021. "Proliferative Verrucous Leukoplakia Revisited: A Retrospective Clinicopathological Study" Clinics and Practice 11, no. 2: 337-346. https://doi.org/10.3390/clinpract11020048
APA StyleGhosh, S., Rao, R. S., Upadhyay, M. K., Kumari, K., Sanketh, D. S., Raj, A. T., Parveen, S., Alhazmi, Y. A., Jethlia, A., Mushtaq, S., Sarode, S., Reda, R., Patil, S., & Testarelli, L. (2021). Proliferative Verrucous Leukoplakia Revisited: A Retrospective Clinicopathological Study. Clinics and Practice, 11(2), 337-346. https://doi.org/10.3390/clinpract11020048