An Uncommon Case of Secondary Organizing Pneumonia Due to Influenza Type B
Abstract
1. Introduction
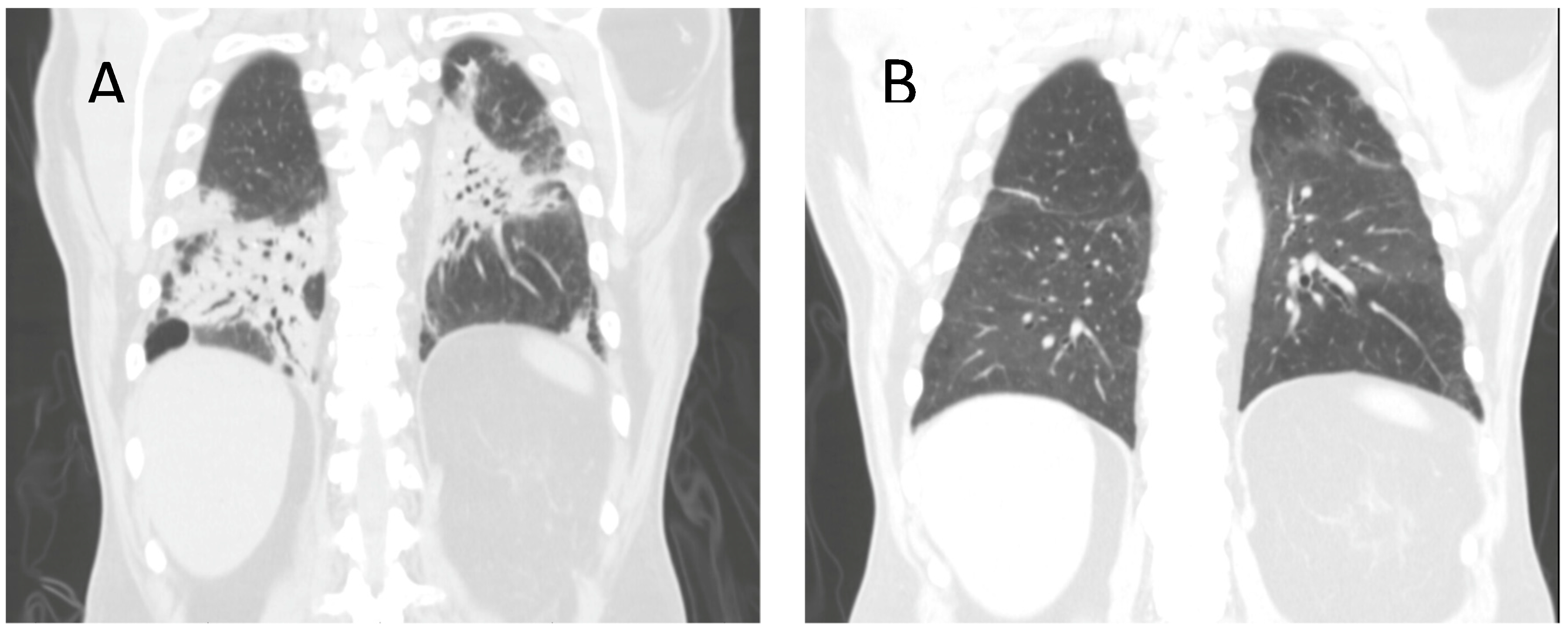
2. Case Discussion
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zare Mehrjardi, M.; Kahkouee, S.; Pourabdollah, M. Radio-pathological correlation of organising pneumonia (OP): A pictorial review. Br. J. Radiol. 2017, 90, 20160723. [Google Scholar] [CrossRef] [PubMed]
- Drakopanagiotakis, F.; Polychronopoulos, V.; Judson, M.A. Organizing pneumonia. Am. J. Med. Sci. 2008, 335, 34–39. [Google Scholar] [CrossRef] [PubMed]
- King, T.E. Organizing pneumonia. People’s Med Publ. House 2011, 5, 981. [Google Scholar]
- Baque-Juston, M.; Pellegrin, A.; Leroy, S.; Marquette, C.H.; Padovani, B. Organizing pneumonia: What is it? A conceptual approach and pictorial review. Diagn. Interv. Imaging 2014, 95, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Bradley, B.; Branley, H.M.; Egan, J.J.; Greaves, M.S.; Hansell, D.M.; Harrison, N.K.; Hirani, N.; Hubbard, R.; Lake, F.; Millar, A.B.; et al. Interstitial lung disease guideline: The british thoracic society in collaboration with the thoracic society of australia and new zealand and the irish thoracic society. Thorax 2008, 63, v1–v58. [Google Scholar] [PubMed]
- Cornejo, R.; Llanos, O.; Fernández, C.; Díaz, J.C.; Cardemil, G.; Salguero, J.; Luengo, C.; Tobar, E.; Romero, C.; Gálvez, L.R. Organizing pneumonia in patients with severe respiratory failure due to novel A (H1N1) influenza. BMJ Case Rep. 2010, 2010, bcr0220102708. [Google Scholar] [CrossRef] [PubMed]
- Asai, N.; Yokoi, T.; Nishiyama, N.; Koizumi, Y.; Sakanashi, D.; Kato, H.; Hagihara, M.; Suematsu, H.; Yamagishi, Y.; Mikamo, H. Secondary organizing pneumonia following viral pneumonia caused by severe influenza B: A case report and literature reviews. BMC Infect. Dis. 2017, 17, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.; Park, S.J.; Koo, S.M.; Kim, Y.K.; Kim, K.U.; Uh, S.-T.; Kim, T.H.; Park, S. Incidence and Risk Factors of Pneumonia in Hospitalized Patients with Seasonal Influenza A or B. Tuberc. Respir. Dis. 2017, 80, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Torrego, A.; Pajares, V.; Mola, A.; Lerma, E.; Franquet, T. Influenza A (H1N1) organising pneumonia. BMJ Case Rep. 2010, 2010, bcr1220092531. [Google Scholar] [CrossRef] [PubMed]
- Kwok, W.C.; Lam, S.H.Y.; Wong, M.P.; Ip, M.S.M.; Lam, D.C.L. Influenza B/streptococcal co-infection complicated by organizing pneumonia. Respirol. Case Rep. 2016, 4, e00170. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shah, P.; Sobash, P.T.; Vedala, K.; Kakkera, K.; Kamoga, G.-R. An Uncommon Case of Secondary Organizing Pneumonia Due to Influenza Type B. Clin. Pract. 2021, 11, 174-177. https://doi.org/10.3390/clinpract11010024
Shah P, Sobash PT, Vedala K, Kakkera K, Kamoga G-R. An Uncommon Case of Secondary Organizing Pneumonia Due to Influenza Type B. Clinics and Practice. 2021; 11(1):174-177. https://doi.org/10.3390/clinpract11010024
Chicago/Turabian StyleShah, Parth, Philip T. Sobash, Krishna Vedala, Krishna Kakkera, and Gilbert-Roy Kamoga. 2021. "An Uncommon Case of Secondary Organizing Pneumonia Due to Influenza Type B" Clinics and Practice 11, no. 1: 174-177. https://doi.org/10.3390/clinpract11010024
APA StyleShah, P., Sobash, P. T., Vedala, K., Kakkera, K., & Kamoga, G.-R. (2021). An Uncommon Case of Secondary Organizing Pneumonia Due to Influenza Type B. Clinics and Practice, 11(1), 174-177. https://doi.org/10.3390/clinpract11010024




