An Exploratory Investigation of Organic Chemicals Detected in Baby Teeth: Differences in Children with and without Autism
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample and Recruitment
2.2. Laboratory Methods
2.3. Statistical Analysis
2.4. Statistical Power
3. Results
4. Discussion
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Maenner, M.J.; Shaw, K.A.; Baio, J.; Washington, A.; Patrick, M.; DiRienzo, M.; Christensen, D.L.; Wiggins, L.D.; Pettygrove, S.; Andrews, J.G.; et al. Prevalence of Autism Spectrum Disorder among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2016. MMWR Surveill. Summ. 2020, 69, 1–12. [Google Scholar] [CrossRef]
- Li, Q.; Li, Y.; Liu, B.; Chen, Q.; Xing, X.; Xu, G.; Yang, W. Prevalence of Autism Spectrum Disorder Among Children and Adolescents in the United States from 2019 to 2020. JAMA Pediatr. 2022, 176, 943–945. [Google Scholar] [CrossRef]
- Kogan, M.D.; Vladutiu, C.J.; Schieve, L.A.; Ghandour, R.M.; Blumberg, S.J.; Zablotsky, B.; Perrin, J.M.; Shattuck, P.; Kuhlthau, K.A.; Harwood, R.L.; et al. The Prevalence of Parent-Reported Autism Spectrum Disorder Among US Children. Pediatrics 2018, 142, e20174161. [Google Scholar] [CrossRef]
- Modabbernia, A.; Velthorst, E.; Reichenberg, A. Environmental risk factors for autism: An evidence-based review of systematic reviews and meta-analyses. Mol. Autism 2017, 8, 13. [Google Scholar] [CrossRef]
- Masini, E.; Loi, E.; Vega-Benedetti, A.F.; Carta, M.; Doneddu, G.; Fadda, R.; Zavattari, P. An Overview of the Main Genetic, Epigenetic and Environmental Factors Involved in Autism Spectrum Disorder Focusing on Synaptic Activity. Int. J. Mol. Sci. 2020, 21, 8290. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, R.; Paliwal, J.K.; Kuhad, A. Neuropsychopathology of Autism Spectrum Disorder: Complex Interplay of Genetic, Epigenetic, and Environmental Factors. Adv. Neurobiol. 2020, 24, 97–141. [Google Scholar] [CrossRef] [PubMed]
- Carter, C.J.; Blizard, R.A. Autism genes are selectively targeted by environmental pollutants including pesticides, heavy metals, bisphenol A, phthalates and many others in food, cosmetics, or household products. Neurochem. Int. 2016, 101, 83–109. [Google Scholar] [CrossRef] [PubMed]
- Saxena, R.; Babadi, M.; Namvarhaghighi, H.; Roullet, F.I. Role of environmental factors and epigenetics in autism spectrum disorders. Prog. Mol. Biol. Transl. Sci. 2020, 173, 35–60. [Google Scholar] [CrossRef] [PubMed]
- Krimsky, S. The unsteady state and inertia of chemical regulation under the U.S.Toxic Substances Control Act. PLoS Biol. 2017, 15, e2002404. [Google Scholar] [CrossRef]
- Gross, L.; Birnbaum, L. Regulating toxic chemicals for public and environmental health. PLoS Biol. 2017, 15, e2004814. [Google Scholar] [CrossRef]
- Gwinn, M.R.; Axelrad, D.A.; Bahadori, T.; Bussard, D.; Cascio, W.E.; Deener, K.; Dix, D.; Thomas, R.S.; Kavlock, R.J.; Burke, T.A. Chemical risk assessment: Traditional vs public health perspectives. AJPH Am. J. Public Health 2017, 107, 1032–1039. [Google Scholar] [CrossRef] [PubMed]
- Vogel, S.A.; Roberts, J.A. Why the toxic substances control act needs an overhaul, and how to strengthen oversight of chemicals in the interim. Health Aff. 2011, 30, 898–905. [Google Scholar] [CrossRef]
- McPartland, J.; Shaffer, R.M.; Fox, M.A.; Nachman, K.E.; Burke, T.A.; Denison, R.A. Charting a Path Forward: Assessing the Science of Chemical Risk Evaluations under the Toxic Substances Control Act in the Context of Recent National Academies Recommendations. Environ. Health Perspect. 2022, 130, 25003. [Google Scholar] [CrossRef] [PubMed]
- Rayasam, S.D.G.; Koman, P.D.; Axelrad, D.A.; Woodruff, T.J.; Chartres, N. Toxic Substances Control Act (TSCA) Implementation: How the Amended Law Has Failed to Protect Vulnerable Populations from Toxic Chemicals in the United States. Environ. Sci. Technol. 2022, 56, 11969–11982. [Google Scholar] [CrossRef]
- Department of Health and Human Services Centers for Disease Control and Prevention. CDC National Report on Human Exposure to Environmental Chemicals. 2022. Available online: https://www.cdc.gov/exposurereport/index.html (accessed on 9 May 2022).
- Woodruff, T.J.; Zota, A.R.; Schwartz, J.M. Environmental chemicals in pregnant women in the United States: NHANES 2003–2004. Environ. Health Perspect. 2011, 119, 878–885. [Google Scholar] [CrossRef]
- Herbstman, J.B.; Sjodin, A.; Kurzon, M.; Lederman, S.A.; Jones, R.S.; Rauh, V.; Needham, L.L.; Tang, D.; Niedzwiecki, M.; Wang, R.Y.; et al. Prenatal exposure to PBDEs and neurodevelopment. Environ. Health Perspect. 2010, 118, 712–719. [Google Scholar] [CrossRef]
- Wigle, D.T.; Arbuckle, T.E.; Walker, M.; Wade, M.G.; Liu, S.; Krewski, D. Environmental hazards: Evidence for effects on child health. J. Toxicol. Environ. Health B Crit. Rev. 2007, 10, 3–39. [Google Scholar] [CrossRef]
- Talsness, C.E.; Andrade, A.J.M.; Kuriyama, S.N.; Taylor, J.A.; vom Saal, F.S. Components of plastic: Experimental studies in animals and relevance for human health. Philos. Trans. R. Soc. B 2009, 364, 2079–2096. [Google Scholar] [CrossRef]
- Mostafalou, S.; Abdollahi, M. Pesticides: An update of human exposure and toxicity. Arch. Toxicol. 2017, 91, 549–599. [Google Scholar] [CrossRef]
- Rochester, J.R. Bisphenol A, and human health: A review of the literature. Reprod. Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef]
- Fisher, B.E. Most unwanted. Environ. Health Perspect. 1999, 107, A18–A23. [Google Scholar] [CrossRef] [PubMed]
- Alavanja, M.C. Introduction: Pesticides use and exposure extensive worldwide. Rev. Environ. Health 2009, 24, 303–309. [Google Scholar] [CrossRef] [PubMed]
- von Ehrenstein, O.S.; Ling, C.; Cui, X.; Cockburn, M.; Park, A.S.; Yu, F.; Wu, J.; Ritz, B. Prenatal and infant exposure to ambient pesticides and autism spectrum disorder in children: Population-based case-control study. BMJ 2019, 364, l962. [Google Scholar] [CrossRef] [PubMed]
- Hertz-Picciotto, I.; Park, H.Y.; Dostal, M.; Kocan, A. Prenatal exposures to persistent and non-persistent organic compounds and effects on immune system development. Basic Clin. Pharmacol. Toxicol. 2008, 102, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Rosas, L.G.; Eskenazi, B. Pesticides, and child neurodevelopment. Curr. Opin. Pediatr. 2008, 20, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Rosas, L.G.; Marks, A.R.; Bradman, A.; Harley, K.; Holland, N.; Johnson, C.; Fenster, L.; Barr, D.B. Pesticide toxicity and the developing brain. Basic. Clin. Pharmacol. Toxicol. 2008, 102, 228–236. [Google Scholar] [CrossRef]
- Aldridge, J.E.; Seidler, F.J.; Meyer, A.; Thillai, I.; Slotkin, T. Serotonergic systems targeted by developmental exposure to chlorpyrifos: Effects during different critical periods. Environ. Health Perspect. 2003, 111, 1736–1743. [Google Scholar] [CrossRef]
- Shelton, J.F.; Geraghty, E.M.; Tancredi, D.J.; Delwiche, L.D.; Schmidt, R.J.; Ritz, B.; Hertz-Picciotto, I. Neurodevelopmental disorders, and prenatal residential proximity to agricultural pesticides: The CHARGE Study. Environ. Health Perspect. 2014, 122, A266. [Google Scholar] [CrossRef]
- Sarigiannis, D.A.; Papaioannou, N.; Handakas, E.; Anesti, O.; Polanska, K.; Hanke, W.; Salifoglou, A.; Gabriel, C.; Karakitsios, S. Neurodevelopmental exposome: The effect of in utero co-exposure to heavy metals and phthalates on child neurodevelopment. Environ. Res. 2021, 197, 110949. [Google Scholar] [CrossRef]
- Kardas, F.; Bayram, A.K.; Demirci, E.; Akin, L.; Ozmen, S.; Kendirci, M.; Canpolat, M.; Oztop, D.B.; Narin, F.; Gumus, H.; et al. Increased Serum Phthalates (MEHP, DEHP) and Bisphenol A Concentrations in Children with Autism Spectrum Disorder: The Role of Endocrine Disruptors in Autism Etiopathogenesis. J. Child. Neurol. 2016, 31, 629–635. [Google Scholar] [CrossRef]
- Testa, C.; Nuti, F.; Hayek, J.; De Felice, C.; Chelli, M.; Rovero, P.; Latini, G.; Papini, A.M. Di-(2-ethylhexyl) phthalate and autism spectrum disorders. ASN Neuro 2012, 4, 223–229. [Google Scholar] [CrossRef]
- Rappaport, S.M.; Barupal, D.K.; Wishart, D.; Vineis, P.; Scalbert, A. The blood exposome and its role in discovering causes of disease. Environ. Health Perspect. 2014, 22, 769–774. [Google Scholar] [CrossRef] [PubMed]
- Drexler, H.; Shukla, A. Importance of Exposure Level for Toxicological Risk Assessment. In Regulatory Toxicology; Springer: Cham, Switzerland, 2020; pp. 1–8. [Google Scholar]
- Arora, M.; Austin, C. Teeth as a biomarker of past chemical exposure. Curr. Opin. Pediatr. 2013, 25, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Andra, S.S.; Austin, C.; Arora, M. Tooth matrix analysis for biomonitoring of organic chemical exposure: Current status, challenges, and opportunities. Environ. Res. 2015, 142, 387–406. [Google Scholar] [CrossRef] [PubMed]
- Dumitriu, D.; Baldwin, E.; Coenen, R.J.J.; Hammond, L.A.; Peterka, D.S.; Heilbrun, L.; Frye, R.E.; Palmer, R.; Norrman, H.N.; Fridell, A.; et al. Deciduous tooth biomarkers reveal atypical fetal inflammatory regulation in autism spectrum disorder. iScience 2023, 26, 106247. [Google Scholar] [CrossRef] [PubMed]
- Berkovitz, B.K.B.; Holland, G.R.; Moxham, B.J. A Colour Atlas and Textbook of Oral Anatomy, Histology and Embryology; Wolfe Publishing Ltd.: London, UK, 1992. [Google Scholar]
- Rabinowitz, M.B.; Leviton, A.; Bellinger, D. Relationships between serial blood lead levels and exfoliated tooth dentin lead levels: Models of tooth lead kinetics. Calcif. Tissue Int. 1993, 53, 338–341. [Google Scholar] [CrossRef] [PubMed]
- Simeonov, L.; Kochubovski, M.; Simeonova, B.; Draghici, C.; Chirila, E.; Canfield, R. NATO Advanced research workshop on environmental heavy metal pollution and effects on child mental development: Discussion conclusions and recommendations. In Environmental Heavy Metal Pollution and Effects on Child Mental Development; Simeonov, L., Kochubovski, M., Simeonova, B., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 331–342. [Google Scholar]
- Camann, D.E.; Schultz, S.T.; Yau, A.Y.; Heilbrun, L.P.; Zuniga, M.M.; Palmer, R.F.; Miller, C.S. Acetaminophen, pesticide, and diethylhexyl phthalate metabolites, anandamide, and fatty acids in deciduous molars: Potential biomarkers of perinatal exposure. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Palmer, R.F.; Heilbrun, L.; Camann, D.; Yau, A.; Schultz, S.; Elisco, V.; Tapia, B.; Garza, N.; Miller, C.S. Organic compounds detected in deciduous teeth: A replication study from children with autism in two samples. J. Environ. Public Health 2015, 2015, 862414. [Google Scholar] [CrossRef]
- Yu, M.; Tu, P.; Dolios, G.; Dassanayake, P.S.; Volk, H.; Newschaffer, C.; Fallin, M.D.; Croen, L.; Lyall, K.; Schmidt, R.; et al. Tooth biomarkers to characterize the temporal dynamics of the fetal and early-life exposome. Environ. Int. 2021, 157, 106849. [Google Scholar] [CrossRef]
- Lee, H.; Marvin, A.R.; Watson, T.; Piggot, J.; Law, J.K.; Law, P.A. Accuracy of phenotyping of autistic children based on internet implemented parent report. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2010, 153B, 1119–1126. [Google Scholar] [CrossRef]
- Daniels, A.M.; Rosenberg, R.E.; Anderson, C.; Law, J.K.; Marvin, A.R.; Law, P.A. Verification of parent-report of child autism spectrum disorder diagnosis to a web-based autism registry. J. Autism Dev. Disord. 2012, 42, 257–265. [Google Scholar] [CrossRef]
- Favela, K.; Hartnett, M.; Janssen, J.; Vickers, D.; Schaub, A.; Spidle, H.; Pickens, K. Nontargeted Analysis of Face Masks: Comparison of Manual Curation to Automated GCxGC Processing Tools. J. Am. Soc. Mass Spectrom. 2021, 32, 860–871. [Google Scholar] [CrossRef]
- Place, B.; Ulrich, E.; Challis, J.; Chao, A.; Du, B.; Favela, K.; Feng, Y.; Fisher, C.; Gardinali, P.; Hood, A.; et al. An Introduction to the Benchmarking and Publications for Non-Targeted Analysis Working Group. Anal. Chem. 2021, 93, 16289–16296. [Google Scholar] [CrossRef]
- Kim, S.; Chen, J.; Cheng, T.; Gindulyte, A.; He, J.; He, S.; Li, Q.; Shoemaker, B.A.; Thiessen, P.A.; Yu, B.; et al. PubChem 2023 update. Nucleic Acids Res. 2023, 51, D1373–D1380. [Google Scholar] [CrossRef]
- Birkett, M.A.; Day, S.J. Internal pilot studies for estimating sample size. Stat. Med. 1994, 13, 2455–2463. [Google Scholar] [CrossRef]
- Browne, R.H. On the use of a pilot study for sample size determination. Stat. Med. 1995, 14, 1933–1940. [Google Scholar] [CrossRef]
- Lee, E.; Whitehead, A.L.; Jacques, R.M.; Julious, S.A. The statistical interpretation of pilot trials: Should significance thresholds be reconsidered? BMC Med. Res. Methodol. 2014, 14, 41. [Google Scholar] [CrossRef]
- Leon, A.; Davis, L.; Kraemer, H. The role and interpretation of pilot studies in clinical research. J. Psychiatr. Res. 2011, 45, 626–629. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, D. Statistical considerations for pilot studies. Int. J. Radiat. Oncol. Biol. Phys. 1980, 6, 371–374. [Google Scholar] [CrossRef]
- SAS Institute Inc. SAS® 9.4 Statements: Reference; SAS Institute Inc.: Cary, NC, USA, 2013. [Google Scholar]
- Frye, R.E.; Rossignol, D.A. Metabolic disorders and abnormalities associated with autism spectrum disorder. J. Pediatr. Biochem. 2012, 2, 181–191. [Google Scholar]
- Nisar, S.; Haris, M. Neuroimaging genetics approaches to identify new biomarkers for the early diagnosis of autism spectrum disorder. Mol. Psychiatry 2023. [Google Scholar] [CrossRef]
- Biosca-Brull, J.; Pérez-Fernández, C.; Mora, S.; Carrillo, B.; Pinos, H.; Conejo, N.M.; Collado, P.; Arias, J.L.; Martín-Sánchez, F.; Sánchez-Santed, F.; et al. Relationship between Autism Spectrum Disorder and Pesticides: A Systematic Review of Human and Preclinical Models. Int. J. Environ. Res. Public. Health 2021, 18, 5190. [Google Scholar] [CrossRef] [PubMed]
- Maleki, M.; Noorimotlagh, Z.; Mirzaee, S.A.; Jaafarzadeh, N.; Martinez, S.S.; Rahim, F.; Kaffashian, M. An updated systematic review on the maternal exposure to environmental pesticides and involved mechanisms of autism spectrum disorder (ASD) progression risk in children. Rev. Environ. Health 2022. ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Yang, X.; Chen, D.; Xu, Y.; Lan, L.; Zhao, S.; Liu, Q.; Snijders, A.M.; Xia, Y. Maternal exposure to pesticides and autism or attention-deficit/hyperactivity disorders in offspring: A meta-analysis. Chemosphere 2023, 313, 137459. [Google Scholar] [CrossRef] [PubMed]
- Eales, J.; Bethel, A.; Galloway, T.; Hopkinson, P.; Morrissey, K.; Short, R.E.; Garside, R. Human health impacts of exposure to phthalate plasticizers: An overview of reviews. Environ. Int. 2022, 158, 106903. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, T.; Morisaki, N.; Honda, Y.; Sampei, M.; Tani, Y. Chemicals, Nutrition, and autism spectrum disorder: A Mini-Review. Front. Neurosci. 2016, 10, 174. [Google Scholar] [CrossRef]
- Volk, H.E.; Ames, J.L.; Chen, A.; Fallin, M.D.; Hertz-Picciotto, I.; Halladay, A.; Hirtz, D.; Lavin, A.; Ritz, B.; Zoeller, T.; et al. Considering toxic chemicals in the etiology of autism. Pediatrics 2022, 149, e2021053012. [Google Scholar] [CrossRef]
- Keil-Stietz, K.; Lein, P.J. Gene× environment interactions in autism spectrum disorders. Curr. Top. Dev. Biol. 2023, 152, 221–284. [Google Scholar]
- Santos, J.X.; Rasga, C.; Marques, A.R.; Martiniano, H.; Asif, M.; Vilela, J.; Oliveira, G.; Sousa, L.; Nunes, A.; Vicente, A.M. A Role for Gene-Environment Interactions in Autism Spectrum Disorder Is Supported by Variants in Genes Regulating the Effects of Exposure to Xenobiotics. Front. Neurosci. 2022, 16, 862315. [Google Scholar] [CrossRef]
- Thomas, T.R.; Koomar, T.; Casten, L.G.; Tener, A.J.; Bahl, E.; Michaelson, J.J. Clinical autism subscales have common genetic liabilities that are heritable, pleiotropic, and generalizable to the general population. Transl. Psychiatry 2022, 12, 247. [Google Scholar] [CrossRef] [PubMed]
- Warrier, V.; Zhang, X.; Reed, P.; Havdahl, A.; Moore, T.M.; Cliquet, F.; Leblond, C.S.; Rolland, T.; Rosengren, A.; EU-AIMS LEAP; et al. Genetic correlates of phenotypic heterogeneity in autism. Nat. Genet. 2022, 54, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Pintus, R.; Dessi, A.; Bosco, A.; Fanos, V. Metabolomics in Pediatric Neuropsychiatry. BRAIN Broad Res. Artif. Intell. Neurosci. 2021, 12, 335–341. [Google Scholar] [CrossRef]
- Vidal, M.; Cusick, M.E.; Barabási, A.L. Interactome networks and human disease. Cell 2011, 144, 986–998. [Google Scholar] [CrossRef] [PubMed]
- Stanton, J.E.; Malijauskaite, S.; McGourty, K.; Grabrucker, A.M. The Metallome as a Link Between the “Omes” in autism spectrum disorders. Front. Mol. Neurosci. 2021, 14, 695873. [Google Scholar] [CrossRef] [PubMed]
- Rechnitzer, T. Unifying ‘the’ Precautionary Principle? Justification and Reflective Equilibrium. Philosophia 2022, 50, 2645–2661. [Google Scholar] [CrossRef] [PubMed]
- Elkin, L. The Precautionary Principle and Expert Disagreement. Erkenntnis 2023, 88, 2717–2726. [Google Scholar] [CrossRef]
- Fowler, S.P.; Gimeno Ruiz de Porras, D.; Swartz, M.D.; Stigler Granados, P.; Heilbrun, L.P.; Palmer, R.F. Daily Early-Life Exposures to Diet Soda and Aspartame Are Associated with Autism in Males: A Case-Control Study. Nutrients 2023, 15, 3772. [Google Scholar] [CrossRef]
- Heilbrun, L.P.; Palmer, R.F.; Jaen, C.R.; Svoboda, M.D.; Perkins, J.; Miller, C.S. Maternal Chemical and Drug Intolerances: Potential Risk Factors for Autism and Attention Deficit Hyperactivity Disorder (ADHD). J. Am. Board. Fam. Med. 2015, 28, 461–470. [Google Scholar] [CrossRef]
- Steinemann, A. Fragranced consumer products: Exposures and effects from emissions. Air Qual. Atmos. Health 2016, 9, 861–866. [Google Scholar] [CrossRef]
| Cases (n = 22) | Controls (n = 20) | p | |
|---|---|---|---|
| Mean (SD) or % | Mean (SD) or % | ||
| Mothers Age | 40.4 (6.4) | 41.1 (5.3) | 0.77 |
| Child Gender (% male) | 80.1% | 70.1% | 0.67 |
| Family Income | |||
| Less than USD 15,000 | 6.7% | 18.5% | |
| USD 15,000–USD 24,999 | 86.7% | 44.4% | |
| USD 25,000–USD 34,999 | 6.7% | 11.1% | |
| Not reported | 0% | 25.9% | 0.61 |
| Ethnicity | |||
| Hispanic | 6.7% | 22.2% | 0.09 |
| Non-Hispanic White | 60.0% | 44.4% | |
| Other | 26.7% | 7.4% | |
| Not Reported | 6.6% | 25.9% |
| Autism | Typically Developing Controls | p | |||
|---|---|---|---|---|---|
| Mean (SD) | Min–Max | Mean (SD) | Min–Max | ||
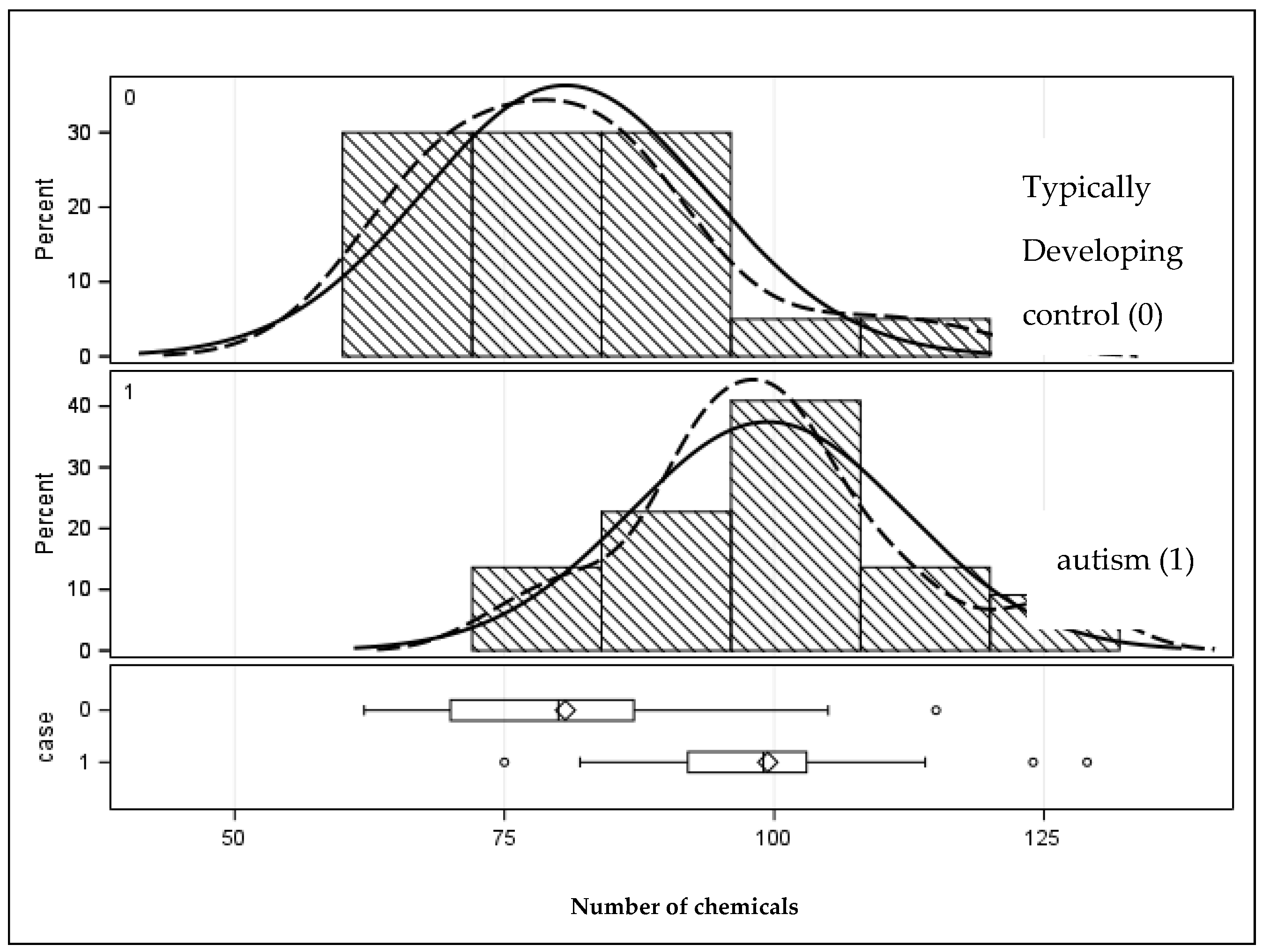
| Total number of chemicals | 99.41 (12.78) | 75–129 | 80.65 (13.18) | 62–115 | 0.0001 |
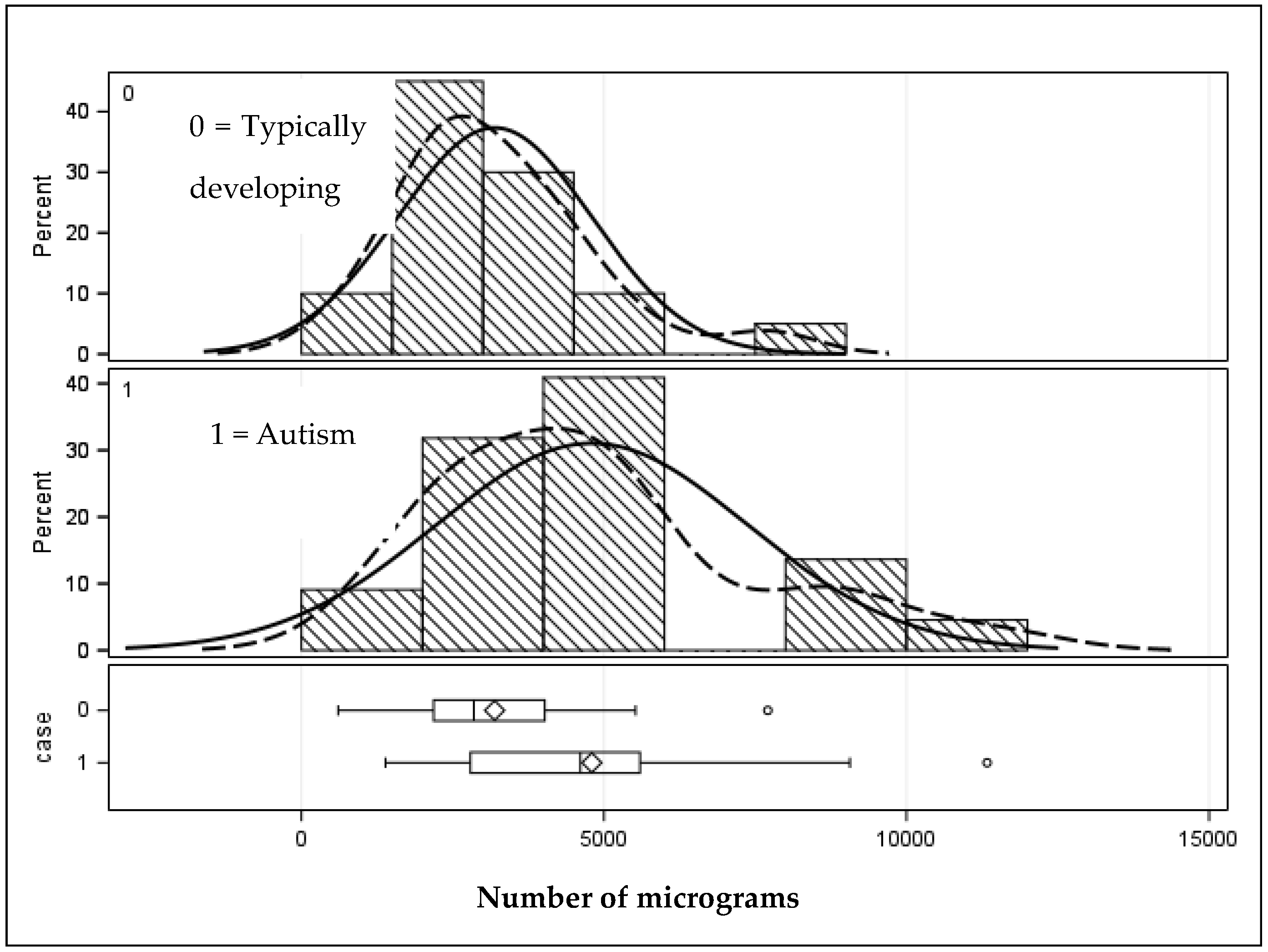
| Total grams | 4799.44 (2570.36) | 1388.16–11,332.95 | 3192.90 (1603.39) | 606.25–7707.95 | 0.01 |
| Chemical | Autism Cases | Typically Developing Child Cases | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | (Std Dev) | Minimum | Maximum | Mean | (Std Dev) | Minimum | Maximum | |
| Chem1 | 0.10 | (0.17) | 0.00 | 0.49 | 0.01 | (0.03) | 0.00 | 0.15 |
| Chem2 | 0.32 | (0.26) | 0.00 | 0.99 | 0.11 | (0.18) | 0.00 | 0.77 |
| Chem3 | 0.51 | (0.60) | 0.00 | 1.84 | 0.16 | (0.32) | 0.00 | 1.05 |
| Chem4 | 1.97 | (1.69) | 0.00 | 7.43 | 0.77 | (1.31) | 0.00 | 5.62 |
| Chem5 | 0.22 | (0.32) | 0.00 | 0.99 | 0.07 | (0.15) | 0.00 | 0.43 |
| Chem6 | 1.08 | (1.23) | 0.00 | 5.13 | 0.30 | (0.52) | 0.00 | 2.00 |
| Chem7 | 0.23 | (0.27) | 0.00 | 0.80 | 0.03 | (0.09) | 0.00 | 0.33 |
| Chem8 | 1.49 | (1.67) | 0.17 | 7.72 | 0.25 | (0.42) | 0.00 | 1.47 |
| Chem9 | 0.15 | (0.24) | 0.00 | 0.78 | 0.04 | (0.16) | 0.00 | 0.68 |
| Chem10 | 15.23 | (22.83) | 0.00 | 49.71 | 2.36 | (10.56) | 0.00 | 47.24 |
| Chem11 | 6.75 | (6.12) | 0.00 | 19.38 | 2.30 | (3.36) | 0.00 | 9.76 |
| Chem12 | 87.56 | (109.22) | 0.00 | 538.23 | 27.17 | (27.88) | 0.00 | 116.99 |
| Chem13 | 1.04 | (0.87) | 0.29 | 3.41 | 0.56 | (0.49) | 0.00 | 2.20 |
| Chem14 | 0.40 | (0.53) | 0.00 | 2.36 | 0.16 | (0.37) | 0.00 | 1.44 |
| Chem15 | 3.38 | (2.88) | 0.94 | 11.76 | 5.29 | (3.18) | 0.00 | 13.88 |
| Chem16 | 0.24 | (0.49) | 0.00 | 1.87 | 1.41 | (1.76) | 0.00 | 6.18 |
| Chem17 | 3.08 | (2.12) | 0.98 | 10.03 | 2.01 | (1.63) | 0.00 | 6.60 |
| Chem18 | 45.60 | (29.33) | 0.00 | 111.20 | 12.20 | (15.62) | 0.00 | 61.78 |
| Chem19 | 1.37 | (2.45) | 0.00 | 10.89 | 0.21 | (0.33) | 0.00 | 1.00 |
| Chem20 | 1.40 | (1.31) | 0.19 | 5.83 | 0.59 | (0.53) | 0.00 | 1.65 |
| Chem21 | 3.32 | (3.31) | 0.58 | 15.02 | 1.86 | (2.03) | 0.00 | 8.58 |
| Chem22 | 0.03 | (0.09) | 0.00 | 0.31 | 0.39 | (0.77) | 0.00 | 2.77 |
| Chem23 | 1.17 | (0.60) | 0.33 | 2.90 | 3.45 | (5.01) | 0.41 | 19.98 |
| Chem24 | 0.90 | (0.97) | 0.00 | 3.36 | 0.25 | (0.95) | 0.00 | 4.21 |
| Chem25 | 1.22 | (0.62) | 0.60 | 2.97 | 0.83 | (0.51) | 0.00 | 2.11 |
| Chem26 | 4.03 | (5.63) | 0.00 | 18.09 | 0.85 | (1.57) | 0.00 | 5.98 |
| Chem27 | 1.19 | (0.75) | 0.00 | 2.80 | 0.15 | (0.37) | 0.00 | 1.40 |
| Chem28 | 0.24 | (0.29) | 0.00 | 1.18 | 0.06 | (0.15) | 0.00 | 0.61 |
| Chem29 | 0.10 | (0.26) | 0.00 | 1.09 | 0.00 | (0.00) | 0.00 | 0.00 |
| Chem30 | 0.37 | (0.45) | 0.00 | 1.87 | 0.08 | (0.16) | 0.00 | 0.62 |
| Chem31 | 8.26 | (11.79) | 0.00 | 30.80 | 0.26 | (0.84) | 0.00 | 3.27 |
| Chem32 | 0.20 | (0.22) | 0.00 | 0.76 | 0.07 | (0.16) | 0.00 | 0.69 |
| Chem33 | 7.90 | (6.86) | 0.00 | 30.65 | 1.53 | (2.04) | 0.00 | 6.28 |
| Chem34 | 0.40 | (0.45) | 0.00 | 1.61 | 0.07 | (0.17) | 0.00 | 0.60 |
| Chem35 | 13.55 | (14.14) | 0.43 | 55.87 | 3.81 | (9.86) | 0.00 | 44.85 |
| Chem36 | 0.26 | (0.24) | 0.00 | 1.04 | 0.14 | (0.19) | 0.00 | 0.59 |
| Chem37 | 104.19 | (84.68) | 40.70 | 364.51 | 34.96 | (39.04) | 0.00 | 149.21 |
| Chem38 | 5.08 | (12.02) | 0.00 | 44.51 | 23.79 | (27.90) | 0.00 | 119.15 |
| Chem39 | 7.55 | (8.19) | 1.94 | 40.46 | 3.77 | (2.19) | 0.46 | 9.13 |
| Chem40 | 3.65 | (3.16) | 0.00 | 12.24 | 1.63 | (1.92) | 0.00 | 7.02 |
| Chem41 | 3.49 | (2.51) | 0.00 | 11.00 | 0.92 | (1.94) | 0.00 | 8.27 |
| Chem42 | 2.96 | (7.63) | 0.00 | 22.41 | 0.00 | (0.00) | 0.00 | 0.00 |
| Chem43 | 6.90 | (11.73) | 0.67 | 57.12 | 2.16 | (2.55) | 0.00 | 11.11 |
| Chem44 | 139.17 | (103.90) | 0.00 | 339.60 | 75.98 | (117.41) | 0.00 | 394.04 |
| Chem45 | 5.73 | (7.64) | 0.86 | 35.25 | 1.46 | (1.64) | 0.00 | 6.26 |
| Chem46 | 340.09 | (485.55) | 30.49 | 1966.46 | 59.84 | (58.48) | 4.53 | 244.78 |
| Chem47 | 35.42 | (28.77) | 5.63 | 122.78 | 17.25 | (29.99) | 0.48 | 136.16 |
| Chem48 | 3.16 | (3.20) | 0.38 | 12.29 | 1.43 | (2.03) | 0.00 | 8.99 |
| Chem49 | 3756.79 | (2500.01) | 895.83 | 10,276.18 | 2643.87 | (1612.12) | 376.14 | 6898.29 |
| Chem50 | 2.40 | (2.86) | 0.00 | 10.10 | 0.34 | (0.67) | 0.00 | 1.99 |
| Chem51 | 6.31 | (9.62) | 0.32 | 44.91 | 0.99 | (2.23) | 0.00 | 9.99 |
| Chem52 | 1.64 | (4.51) | 0.00 | 17.88 | 0.00 | (0.00) | 0.00 | 0.00 |
| Chem53 | 1.59 | (1.92) | 0.00 | 9.14 | 0.41 | (0.65) | 0.00 | 1.90 |
| Chem54 | 75.20 | (51.88) | 0.00 | 189.40 | 32.07 | (39.70) | 0.00 | 126.47 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palmer, R.F. An Exploratory Investigation of Organic Chemicals Detected in Baby Teeth: Differences in Children with and without Autism. J. Xenobiot. 2024, 14, 404-415. https://doi.org/10.3390/jox14010025
Palmer RF. An Exploratory Investigation of Organic Chemicals Detected in Baby Teeth: Differences in Children with and without Autism. Journal of Xenobiotics. 2024; 14(1):404-415. https://doi.org/10.3390/jox14010025
Chicago/Turabian StylePalmer, Raymond F. 2024. "An Exploratory Investigation of Organic Chemicals Detected in Baby Teeth: Differences in Children with and without Autism" Journal of Xenobiotics 14, no. 1: 404-415. https://doi.org/10.3390/jox14010025
APA StylePalmer, R. F. (2024). An Exploratory Investigation of Organic Chemicals Detected in Baby Teeth: Differences in Children with and without Autism. Journal of Xenobiotics, 14(1), 404-415. https://doi.org/10.3390/jox14010025





