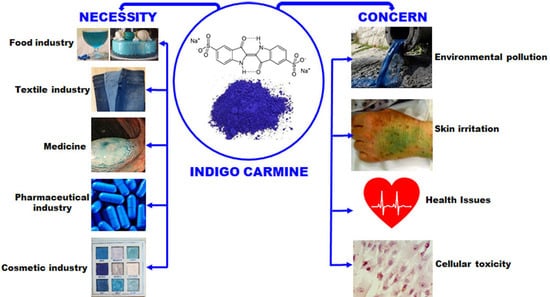
Indigo Carmine: Between Necessity and Concern
Abstract
1. Introduction
2. Materials and Methods
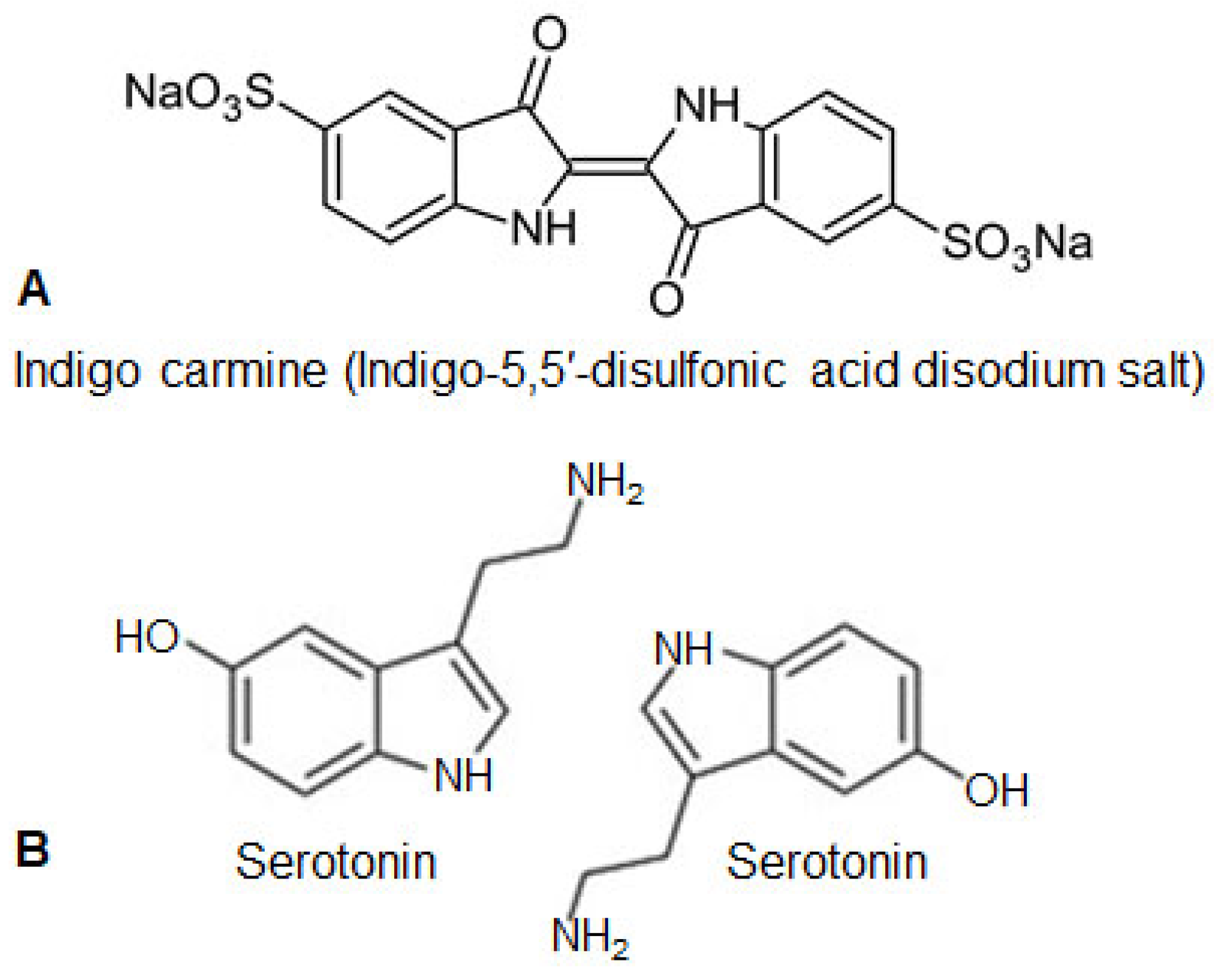
3. The Structure and Properties of Indigo Carmine
4. The Manufacture of Indigo Carmine
5. Indigo Carmine in the Food Industry
Adverse Effects of Indigo Carmine Consumption
6. Indigo Carmine in the Textile Industry
Indigo Carmine and Water Pollution
7. Indigo Carmine in Medicine
Adverse Effects of Using Indigo Carmine in Medicine
8. Other Applications of Indigo Carmine
9. Toxicity Testing of Indigo Carmine
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ngaha, M.C.D.; Njanja, E.; Doungmo, G.; Kamdem, A.T.; Tonle, I.L. Indigo carmine and 2,6 dichlorophenolindophenol removal using cetyltrimethylammonium bromide-modified palm oil fiber: Adsorption isotherms and mass transfer kinetics. Int. J. Biomater. 2019, 2019, 6862825. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Saeed, K.; Zekker, I.; Zhang, B.; Hendi, A.A.; Ahmad, A.; Ahmad, S.; Zada, N.; Ahmad, H.; Shah, L.A.; et al. Review on methylene blue: Its properties, uses, toxicity and photodegradation. Water 2022, 14, 242. [Google Scholar] [CrossRef]
- Benkhaya, S.; Mrabet, S.; Elharfi, A. A review on classifications, recent synthesis and applications of textile dyes. Inorg. Chem. Commun. 2020, 115, 107891. [Google Scholar] [CrossRef]
- Tkaczyk, A.; Mitrowska, K.; Posyniak, A. Synthetic organic dyes as contaminants of the aquatic environment and their implications for ecosystems: A review. Sci. Total Environ. 2020, 717, 137222. [Google Scholar] [CrossRef]
- Ahmad, A.; Mohd-Setapar, S.H.; Chuong, C.S.; Khatoon, A.; Wani, W.A.; Kumar, R.; Rafatullah, M. Recent advances in new generation dye removal technologies: Novel search for approaches to reprocess wastewater. RSC Adv. 2015, 5, 30801–30818. [Google Scholar] [CrossRef]
- Kesraoui, A.; Selmi, T.; Seffen, M.; Brouers, F. Influence of alternating current on the adsorption of indigo carmine. Environ. Sci. Pollut. Res. Int. 2017, 24, 9940–9950. [Google Scholar] [CrossRef]
- Alencar, L.V.T.D.; Passos, L.M.S.; Soares, C.M.F.; Lima, A.S.; Souza, R.L. Efficiency method for methylene blue recovery using aqueous two-phase systems based on cholinium-ionic liquids. J. Fashion Technol. Text. 2020, 6, 13–20. [Google Scholar] [CrossRef]
- Pandey, S.; Do, J.Y.; Kim, J.; Kang, M. Fast and highly efficient removal of dye from aqueous solution using natural locust bean gum based hydrogels as adsorbent. Int. J. Biol. Macromol. 2020, 143, 60–75. [Google Scholar] [CrossRef]
- Ahmad, M.; Rehman, W.; Khan, M.M.; Qureshi, M.T.; Gul, A.; Haq, S.; Ullah, R.; Rab, A.; Menaa, F. Phytogenic fabrication of ZnO and gold decorated ZnO nanoparticles for photocatalytic degradation of Rhodamine B. J. Environ. Chem. Eng. 2020, 9, 104725. [Google Scholar] [CrossRef]
- Khan, S.; Naushad, M.; Govarthanan, M.; Iqbal, J.; Alfadul, S.M. Emerging contaminants of high concern for the environment: Current trends and future. Environ. Res. 2022, 207, 112609. [Google Scholar] [CrossRef]
- Ventura-Camargo, B.C.; Marin-Morales, M.A. Azo dyes: Characterization and toxicity—A review. Text. Light Ind. Sci. Technol. 2013, 2, 85–103. [Google Scholar]
- Kekes, T.; Tzia, C. Adsorption of indigo carmine on functional chitosan and β-cyclodextrin/chitosan beads: Equilibrium, kinetics and mechanism studies. J. Environ. Manag. 2020, 262, 110372. [Google Scholar] [CrossRef] [PubMed]
- Bouras, H.D.; Isik, Z.; Arikan, E.B.; Yeddou, A.; Bouras, N.; Chergui, A.; Favier, L.; Amrane, A.; Dizge, N. Biosorption characteristics of methylene blue by two fungal biomasses. Int. J. Environ. Stud. 2020, 78, 365–381. [Google Scholar] [CrossRef]
- Castillo-Suárez, L.A.; Sierra-Sánchez, A.G.; Linares-Hernádes, I.; Martínez-Miranda, V.; Teutli-Sequeira, E.A. A critical review of textile industry wastewater: Green technologies for the removal of indigo dyes. Int. J. Environ. Sci. Technol. 2023, 20, 10553–10590. [Google Scholar] [CrossRef] [PubMed]
- Fong, W.M.; Affam, A.C.; Chung, W.C. Synthesis of Ag/Fe/CAC for colour and COD removal from methylene blue dye wastewater. Int. J. Environ. Sci. Technol. 2020, 17, 3485–3494. [Google Scholar] [CrossRef]
- Pagnacco, M.; Maksimović, J.P.; Nikolić, N.T.; Bogdanović, D.V.B.; Kragović, M.M.; Stojmenović, M.D.; Blagojević, S.N.; Senćanski, J.V. Indigo carmine in a food dye: Spectroscopic characterization and determining its micro-concentration through the clock reaction. Molecules 2022, 27, 4853. [Google Scholar] [CrossRef]
- Wainwright, M. Dyes for the medical industry. In Handbook of Textile and Industrial Dyeing: Applications of Dyes; Clark, M., Ed.; Woodhead Publishing: Liverpool, UK, 2011; Volume 2, pp. 204–230. [Google Scholar]
- Lakshmi, U.R.; Srivastava, V.C.; Mall, I.D.; Lataye, D.H. Rice husk ash as an effective adsorbent: Evaluation of adsorptive characteristics for indigo carmine dye. J. Environ. Manag. 2009, 90, 710–720. [Google Scholar] [CrossRef]
- Edwin, D.S.S.; Manjunatha, J.G.; Raril, C.; Girish, T.; Ravishankar, D.K.; Arpitha, H.J. Electrochemical analysis of indigo carmine using polyarginine modified carbon paste electrode. J. Electrochem. Sci. Eng. 2021, 11, 87–96. [Google Scholar] [CrossRef]
- El-Kammah, M.; Elkhatib, E.; Gouveia, S.; Cameselle, C.; Aboukila, E. Enhanced removal of indigo carmine dye from textile effluent using green cost-efficient nanomaterial: Adsorption, kinetics, thermodynamics and mechanisms. Sustain. Chem. Pharm. 2022, 29, 100753. [Google Scholar] [CrossRef]
- Pasdaran, A.; Azarpira, N.; Heidari, R.; Nourinejad, S.; Zare, M.; Hamedi, A. Effects of some cosmetic dyes and pigments on the proliferation of human foreskin fibroblasts and cellular oxidative stress; potential cytotoxicity of chlorophyllin and indigo carmine on fibroblasts. J. Cosmet. Dermatol. 2022, 21, 3979–3985. [Google Scholar] [CrossRef]
- Tabti, S.; Benchettara, A.; Smaili, F.; Benchettara, A.; Berrabah, S.E. Electrodeposition of lead dioxide on Fe electrode application to the degradation of indigo carmine dye. J. Appl. Electrochem. 2022, 52, 1207–1217. [Google Scholar] [CrossRef]
- Kim, I.S.; Kim, K.H.; Shin, S.W.; Kim, T.K.; Kim, J.I. Indigo carmine for the selective endoscopic intervertebral nuclectomy. J. Korean Med. Sci. 2005, 20, 702–703. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, K.H.; Kim, Y.S.; Kuh, S.U.; Park, H.S.; Park, J.Y.; Chin, D.K.; Kim, K.S.; Cho, Y.E. Time and dose dependent cytotoxicities of ioxitalamate and indigocarmine in human nucleus pulposus cells. Spine J. 2013, 13, 564–571. [Google Scholar] [CrossRef] [PubMed]
- EFSA ANS Panel (European Food Safety Authority Panel on Food Additives and Nutrient Sources Added to Food). Scientific opinion on the re-evaluation of indigo carmine (E 132) as a food additive. EFSA J. 2014, 12, 3768. [Google Scholar] [CrossRef]
- Caprarescu, S.; Miron, A.R.; Purcar, V.; Radu, A.L.; Sarbu, A.; Ion-Ebrasu, D.; Atanase, L.I.; Ghiurea, M. Efficient removal of indigo carmine dye by a separation process. Water Sci. Technol. 2016, 74, 2462–2473. [Google Scholar] [CrossRef]
- Pereira, P.C.G.; Reimao, R.V.; Pavesi, T.; Saggioro, E.M.; Moreira, J.C.; Correira, F.V. Lethal and sub-lethal evaluation if indigo carmine dye and byproducts after TiO2 photocatalysis in the immune system of Eisenia andrei earthworms. Ecotoxicol. Environ. Saf. 2017, 143, 275–282. [Google Scholar] [CrossRef]
- Rodriguez-Ferreras, A.; Ruiz-Salazar, J. Indigo carmine related tooth discoloration. Excipients: A pending subject. Farm. Hosp. 2019, 43, 36–38. [Google Scholar]
- Secula, M.S.; Cretescu, I.; Petrescu, S. An experimental study of indigo carmine removal from aqueous solution by electrocoagulation. Desalination 2011, 277, 227–235. [Google Scholar] [CrossRef]
- Vautier, M.; Guillard, C.; Herrmann, J.M. Photocatalytic degradation of dyes in water: Case study of indigo and of indigo carmine. J. Catal. 2001, 201, 46–59. [Google Scholar] [CrossRef]
- JECFA (Joint FAO/WHO Expert Committee on Food Additives). Compendium of Food Additive Specifications—86th Meeting; Joint FAO/WHO Expert Committee on Food Additives: Geneva, Switzerland, 2018. [Google Scholar]
- Rancan, E.A.; Frota, E.I.; de Freitas, T.M.N.; Jordani, M.C.; Évora, P.R.B.; Castro-E-Silva, O. Evaluation of indigo carmine on hepatic ischemia and reperfusion injury. Acta Cir. Bras. 2020, 35, 202000901. [Google Scholar] [CrossRef]
- JECFA (Joint FAO/WHO Expert Committee on Food Additives). Combined Compendium of Food Additive Specifications—All Specifications Monographs from the 1st to the 73rd Meeting (1956–2010); Joint FAO/WHO Expert Committee on Food Additives: Geneva, Switzerland, 2010. [Google Scholar]
- Olas, B.; Bialecki, J.; Urbańska, K.; Brys, M. The effects of natural and synthetic blue dyes on human health: A review of current knowledge and therapeutic perspectives. Adv. Nutr. 2021, 12, 2301–2311. [Google Scholar] [CrossRef] [PubMed]
- König, J. Food colour additives of synthetic origin. In Colour Additives for Foods and Beverages; Scotter, M.J., Ed.; Woodhead Publishing: Viena, Austria, 2015; pp. 35–60. [Google Scholar]
- Arvand, M.; Saberi, M.; Ardaki, M.S.; Mohammadi, A. Mediated electrochemical method for the determination of indigo carmine levels in food products. Talanta 2017, 173, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Nagao, S.; Tsuji, Y.; Sakaguchi, Y.; Takahashi, Y.; Minatsuki, C.; Niimi, K.; Yamashita, H.; Yamamichi, N.; Seto, Y.; Tada, T.; et al. Highly accurate artificial intelligence systems to predict the invasion depth of gastric cancer: Efficacy of conventional white-light imaging, nonmagnifying narrow-band imaging, and indigo carmine dye contrast imaging. Gastrointest. Endosc. 2020, 92, 866–873. [Google Scholar] [CrossRef]
- European Commission. Commission regulation (EU) No 231/2012 of 9 March 2012 Laying Down Specifications for Food Additives Listed in Annexes II and III to Regulation (EC), No 1333/2008 of the European Parliament and of the Council. OJEU. 2012. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32012R0231 (accessed on 11 August 2023).
- Rebelo, S.L.H.; Linhares, M.; Simoes, M.M.Q.; Silva, A.M.S.; Neves, M.G.P.; Cavaleiro, J.A.S.; Freire, C. Indigo dye production by enzymatic mimicking based on an iron (III) porphyrin. J. Catal. 2014, 315, 33–40. [Google Scholar] [CrossRef]
- Steingruber, E. Indigo and indigo colorants. In Ullmann’s Encyclopedia of Industrial Chemistry; Elvers, B., Ed.; Wiley-VCH: Frankenthal, Germany, 2004; pp. 55–63. [Google Scholar]
- Okafor, S.N.; Obonga, W.; Ezeokonkwo, M.A. Assessment of the health implications of synthetic and natural food colourants—A critical review. J. Pharm. Biosci. 2016, 4, 1–11. [Google Scholar] [CrossRef]
- Martynov, V.O.; Brygadyrenko, V.V. The influence of the synthetic food colourings tartrazine, allura red and indigo carmine on the body weight of Tenebrio molitor (Coleoptera, Tenebrionidae) larvae. Regul. Mech. 2018, 9, 479–484. [Google Scholar] [CrossRef]
- Amchova, P.; Kotolova, H.; Ruda-Kucerova, J. Health safety issues of synthetic food colorants. Regul. Toxicol. Pharmacol. 2015, 73, 914–922. [Google Scholar] [CrossRef]
- Neves, M.I.L.; Silva, E.K.; Meireles, M.A. Natural blue food colorants: Consumer acceptance, current alternatives, trends, challenges, and future strategies. Trends Food Sci. Technol. 2021, 112, 163–173. [Google Scholar] [CrossRef]
- Merinas-Amo, R.; Martínez-Jurado, M.; Jurado-Güeto, S.; Alonso-Moraga, A.; Merinas-Amo, T. Biological effects of food coloring in in vivo and in vitro model systems. Foods 2019, 8, 176. [Google Scholar] [CrossRef]
- Ahmed, M.A.; Al-Khalifa, A.S.; Al-Nouri, D.M.; El-din, M.F.S. Dietary intake of artificial food color additives containing food products by school-going children. Saudi J. Biol. Sci. 2021, 28, 27–34. [Google Scholar] [CrossRef]
- GSFA (General Standard Food Additives). Food Additive Details: GSFA Provisions for Indigotine (Indigo Carmine). Available online: https://www.fao.org/gsfaonline/additives/details.html?id=96&d-3586470-s=2&d-3586470-o=2&print=true (accessed on 11 August 2023).
- JECFA (Joint FAO/WHO Expert Committee on Food Additives). Safety Evaluation of Certain Food Additives: Prepared by the Eighty-Sixth Meeting of the JECFA; Joint FAO/WHO Expert Committee on Food Additives: Geneva, Switzerland, 2020. [Google Scholar]
- Kiseleva, M.G.; Pimenova, V.V.; Eller, K.I. Optimization of conditions for the HPLC determination of synthetic dyes in food. J. Anal. Chem. 2003, 58, 685–690. [Google Scholar] [CrossRef]
- Minioti, K.S.; Sakellariou, C.F.; Thomaidis, N.S. Determination of 13 synthetic food colorants in water-soluble foods by reversed-phase high-performance liquid chromatography coupled with diode-array detector. Anal. Chim. Acta 2007, 583, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Chao, J.; Feng, F.; Chen, Z.; Chu, X. Highly sensitive determination of 10 dyes in food with complex matrices using SPE followed by UPLC-DAD-tandem mass spectrometry. J. Liq. Chromatogr. Relat. 2011, 34, 93–105. [Google Scholar]
- Feng, F.; Zhao, Y.; Yong, W.; Jiang, G.; Chu, X. Highly sensitive and accurate screening of 40 dyes in soft drinks by liquid chromatography-electrospray tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2011, 879, 1813–1818. [Google Scholar] [CrossRef]
- Huang, H.; Shih, Y.C.; Chen, Y.C. Determining eight colorants in milk beverages by capillary electrophoresis. J. Chromatogr. A. 2002, 959, 317–325. [Google Scholar] [CrossRef]
- Prado, M.A.; Boas, L.F.V.; Bronze, M.R.; Godoy, H.T. Validation of methodology for simultaneous determination of synthetic dyes in alcoholic beverages by capillary electrophoresis. J. Chromatogr. A 2006, 1136, 231–236. [Google Scholar] [CrossRef]
- European Commission. Analysis of needs in post-market monitoring of food additives and preparatory work for future projects in this field. EFSA Support. Publ. 2013, 10, 419E. [Google Scholar]
- Harp, B.P.; Miranda-Bermudez, E.; Barrows, J.N. Determination of seven certified color additives in food products using liquid chromatography. J. Agric. Food Chem. 2013, 61, 3726–3736. [Google Scholar] [CrossRef]
- EFSA ANS Panel (European Food Safety Authority Panel on Food Additives and Nutrient Sources Added to Food). Follow-up of the re-evaluation of indigo carmine (E 132) as a food additive. EFSA J. 2023, 21, 8108. [Google Scholar]
- Zanoni, T.B.; Cardoso, A.A.; Zanoni, M.V.B.; Ferreira, A.A.P. Exploratory study on sequestration of some essential metals by indigo carmine food dye. Braz. J. Pharm. Sci. 2010, 46, 723–730. [Google Scholar] [CrossRef]
- Chowdhury, M.F.; Khandaker, S.; Sarker, F.; Islam, A.; Rahman, M.T.; Awual, M.R. Current treatment technologies and mechanisms for removal of indigo carmine dyes from wastewater: A review. J. Mol. Liq. 2020, 318, 114061. [Google Scholar] [CrossRef]
- de Keijzer, M.; van Bommel, M.R.; Hofmann-de-Keijzer, R.; Knaller, R.; Oberhumer, E. Indigo carmine: Understanding a problematic blue dye. Stud. Conserv. 2013, 57, 87–95. [Google Scholar] [CrossRef]
- Sari, M.M. Removal of acidic indigo carmine textile dye from aqueous solutions using radiation induced cationic hydrogels. Water Sci. Technol. 2010, 61, 2097–2104. [Google Scholar] [CrossRef] [PubMed]
- Fabara, A.N.; Fraaije, M.W. Production of indigo through the use of a dual-function substrate and a bifunctional fusion enzyme. Enzyme Microb. Technol. 2020, 142, 109692. [Google Scholar] [CrossRef] [PubMed]
- Othman, I.; Mohamed, R.M.; Ibrahim, I.A.; Mohamed, M.M. Synthesis and modification of ZSM-5 with manganese and lanthanum and their effects on decolorization of indigo carmine dye. Appl. Catal. 2006, 299, 95–102. [Google Scholar] [CrossRef]
- Pattanaik, L.; Duraivadivel, P.; Hariprasad, P.; Naik, S.N. Utilization and re-use of solid and liquid waste generated from the natural indigo dye production process-a zero waste approach. Bioresour. Technol. 2020, 301, 122721. [Google Scholar] [CrossRef]
- dos Anjos, F.S.; Vieira, E.F.; Cestari, A.R. Interaction of indigo carmine dye with chitosan evaluated by adsorption and thermochemical data. J. Colloid Interface Sci. 2002, 253, 243–246. [Google Scholar] [CrossRef]
- Babu, A.N.; Reddy, D.S.; Sharma, P.; Kumar, G.S.; Ravindhranath, K.; Mohan, G.V.K. Removal of hazardous indigo carmine dye from wastet water using treated red mud. Mater. Today Proc. 2019, 17, 198–208. [Google Scholar] [CrossRef]
- Achieng, G.O.; Kowenje, C.O.; Lalah, J.O.; Ojwach, S.O. Synthesis and characterization of FSB@Fe3O4 composites and application in removal of indigo carmine dye from industrial wastewaters. Environ. Sci. Pollut. Res. Int. 2021, 28, 54876–54890. [Google Scholar] [CrossRef]
- Tanyol, M.; Yildirim, N.C.; Alparslan, D. Electrocoagulation induced treatment of indigo carmine textile dye in an aqueous medium: The effect of process variables on efficiency evaluated using biochemical response of Gammarus pulex. Environ. Sci. Pollut. Res. 2021, 28, 55315–55329. [Google Scholar] [CrossRef]
- Hessel, C.; Allegre, C.; Maisseu, M.; Charbit, F.; Moulin, P. Guidelines and legislation for dye house effluents. J. Environ. Manag. 2007, 83, 171–180. [Google Scholar] [CrossRef]
- Berradi, M.; Hsissou, R.; Khudhair, M.; Assouag, M.; Cherkaoui, O.; El Bachiri, A.; El Harfi, A. Textile finishing dyes and their impact on aquatic environs. Heliyon 2019, 5, 2711. [Google Scholar] [CrossRef]
- Ortiz, E.; Gόmez-Chávez, V.; Cortés-Romero, C.M.; Solís, H.; Ruiz-Ramos, R.; Loera-Serna, S. Degradation of indigo carmine using advanced oxidation processes: Synergy effects and toxicological study. J. Environ. Prot. Sci. 2016, 7, 1693–1706. [Google Scholar] [CrossRef]
- Gupta, T.; Ansari, K.; Lataye, D.; Kadu, M.; Khan, M.A.; Mubarak, N.M.; Garg, R.; Karri, R.R. Adsorption of indigo carmine dye by Acacia nilotica sawdust activated carbon in fixed bed column. Sci. Rep. 2022, 12, 15522. [Google Scholar] [CrossRef]
- Ahmed, M.A.; Ahmed, M.A.; Mohamed, A.A. Synthesis, characterization and application of chitosan/grapheme oxide/copper ferrite nanocomposite for the adsorptive removal of anionic and cationic dyes from wastewater. RSC Adv. 2023, 13, 5337–5352. [Google Scholar] [CrossRef]
- Forgacs, E.; Cserháti, T.; Oros, G. Removal of synthetic dyes from wastewaters: A review. Environ. Int. 2004, 30, 953–971. [Google Scholar] [CrossRef]
- Karthik, V.; Saravanan, K.; Bharathi, P.; Dharanya, V.; Meiaraj, C. An overview of treatments for the removal of textile dyes. J. Chem. Pharm. Sci. 2014, 7, 301–307. [Google Scholar]
- Abid, M.F.; Zablouk, M.A.; Abid-Alameer, A.M. Experimental study of dye removal from industrial wastewater by membrane technologies of reverse osmosis and nanofiltration. Iran J. Environ. Health Sci. Eng. 2012, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.Z.; Sun, S.P.; Li, F.Y.; Ong, Y.K.; Chung, T.S. Treatment of highly concentrated wastewater containing multiple synthetic dyes by a combined process of coagulation/flocculation and nanofiltration. J. Membr. Sci. 2014, 469, 306–315. [Google Scholar] [CrossRef]
- Răducan, A.; Puiu, M.; Oancea, P.; Colbea, C.; Velea, A.; Dinu, B.; Mihăilescu, A.M.; Galaon, T. Fast decolourization of indigo carmine and crystal violet in aqueous environments through micellar catalysis. Sep. Purif. Technol. 2019, 210, 698–709. [Google Scholar] [CrossRef]
- Huy, B.T.; Paeng, D.S.; Thao, C.T.B.; Phuong, N.T.K.; Lee, Y. ZnO-Bi2O3/graphitic carbon nitride photocatalytic system with H2O2-assisted enhanced degradation of indigo carmine under visible light. Arab. J. Chem. 2020, 13, 3790–3800. [Google Scholar] [CrossRef]
- Zukawa, T.; Sasaki, Y.; Kurosawa, T.; Kamiko, N. Photolysis of indigo carmine solution by planar vacuum-ultraviolet (147 nm) light source. Chemosphere 2019, 214, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Li, H.X.; Xu, B.; Tang, L.; Zhang, J.H.M.; Mao, Z.G. Reductive decolorization of indigo carmine dye with Bacillus sp. MZS10. Int. Biodeterior. Biodegrad. 2015, 103, 30–37. [Google Scholar] [CrossRef]
- Wu, L. Analysis of food additives. In Innovative Food Analysis; Galanakis, C.M., Ed.; Acadamic Press: Cambridge, UK, 2021; pp. 157–180. [Google Scholar]
- Gutiérrez-Segura, E.; Solache-Ríos, M.; Colín-Cruz, A. Sorption of indigo carmine by a Fe-zeolitic tuff and carbonaceous material from pyrolyzed sewage sludge. J. Hazard. Mater. 2009, 170, 1227–1235. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, P.; Zhang, S.; Zhou, Q. Comparative study on the adsorption of tartrazine and indigo carmine onto maize cob carbon. Sep. Sci. Technol. 2014, 49, 877–886. [Google Scholar] [CrossRef]
- Zhou, Y.; Lu, J.; Zhou, Y.; Liu, Y. Recent advances for dyes removal using novel adsorbents: A review. Environ. Pollut. 2019, 252, 352–365. [Google Scholar] [CrossRef]
- Siyal, A.A.; Shamsuddin, M.R.; Khan, M.I.; Rabat, N.E.; Zulfiqar, M.; Man, Z.; Siame, J.; Azizli, K.A. A review on geopolymers as emerging materials for the adsorption of heavy metals and dyes. J. Environ. Manag. 2018, 224, 327–339. [Google Scholar] [CrossRef]
- Cestari, A.R.; Vieira, E.F.S.; Tavares, A.M.G.; Bruns, R.E. The removal of the indigo carmine dye from aqueous solutions using cross-linked chitosan—Evaluation of adsorption thermodynamics using a full factorial design. J. Hazard. Mater. 2007, 153, 566–574. [Google Scholar] [CrossRef]
- Pan, J.; Zhou, L.; Chen, H.; Liu, X.; Hong, C.; Chen, D.; Pan, B. Mechanistically understanding adsorption of methyl orange, indigo carmine, and methylene blue onto ionic/nonionic polystyrene adsorbents. J. Hazard. Mater. 2021, 418, 126300. [Google Scholar] [CrossRef]
- Ramesh, T.N.; Sreenivasa, V.P. Removal of indigo carmine dye from aqueous solution using magnesium hydroxide as an adsorbent. J. Mater. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Robinson, T.; Chandran, B.; Nigam, P. Removal of dyes from a synthetic textile dye effluent by biosorption on apple pomace and wheat straw. Water Res. 2002, 36, 2824–2830. [Google Scholar] [CrossRef] [PubMed]
- Ansari, R.; Seyghali, B.; Mohammad-khah, A.; Zanjanchi, M.A. Application of nano surfactant modified biosorbent as an efficient adsorbent for dye removal. Sep. Sci. Technol. 2012, 47, 1802–1812. [Google Scholar] [CrossRef]
- Shikuku, V.O.; Atina, G.O.A.; Kowenje, C. Removal of dyes from wastewater by adsorption onto low-cost adsorbents. In Impact of Textile Dyes on Public Health and the Environment; Wani, K.A., Jangid, N.K., Eds.; IGI Global Publishers: Hershey, PA, USA, 2019; pp. 239–257. [Google Scholar]
- Ahmad, M.B.; Soomro, U.; Muqeet, M.; Ahmed, Z. Adsorption of indigo carmine dye onto the surface-modified adsorbent prepared from municipal waste and simulation using deep neural network. J. Hazard. Mater. 2020, 408, 124433. [Google Scholar] [CrossRef] [PubMed]
- Lekshmi, K.P.V.; Yesodharan, S.; Yesodharan, E.P. MnO2 efficiently removes indigo carmine dyes from polluted water. Heliyon 2018, 4, 897. [Google Scholar] [CrossRef]
- Pavel, O.D.; Stamate, A.E.; Zavoianu, R.; Cruceanu, A.; Tirsoaga, A.; Birjega, R.; Brezestean, I.A.; Ciorita, A.; Culita, D.A.; Dias, A.P.S. Mo-LDH-GO hybrid catalysts for indigo carmine advanced oxidation. Materials 2023, 16, 3025. [Google Scholar] [CrossRef]
- Zaidan, F.; Freitas, P.A.M. Flow injection analysis of indigo carmine using green coconut (Cocos Nucifera L.) fiber as a bioadsorbent. Int. J. Eng. Res. 2015, 1, 39–46. [Google Scholar]
- Erickson, J.C.; Widmer, B.A. The vasopressor effect of indigo carmine. Anesthesiology 1968, 29, 188–189. [Google Scholar] [CrossRef]
- Naitoh, J.; Fox, B.M. Severe hypotension, bronchospasm, and urticaria from intravenous indigo carmine. Urology 1994, 44, 271–272. [Google Scholar] [CrossRef]
- Park, H.J.; Lee, S.M.; Choi, J.A.; Park, N.H.; Kim, H.S.; Park, S.I. Preoperative localization of cystic lesions in the knee using ultrasound-guided injection of indigo carmine. J. Clin. Ultrasound 2010, 38, 305–308. [Google Scholar] [CrossRef]
- Jeon, H.J.; Yoon, J.S.; Cho, S.S.; Kang, K.O. Indigo carmine-induced hypotension in patients undergoing general anaesthesia. Singap. Med. J. 2012, 53, 57–59. [Google Scholar]
- Kennedy, W.F.; Wirjoatmadja, K.; Akamatsu, T.J.; Bonica, J.J. Cardiovascular and respiratory effects of indigo carmine. J. Urol. 1968, 100, 775–778. [Google Scholar] [CrossRef] [PubMed]
- Jeffords, D.L.; Lance, P.H.; DeWolf, W.C. Severe hypertensive reaction to indigo carmine. Urology 1977, 9, 180–181. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.N.; Lazar, J.; Badger, J. A postoperative blue rash: Indigo carmine dye extravasation. Int. J. Dermatol. 2015, 54, 371–372. [Google Scholar] [CrossRef] [PubMed]
- Isosu, T.; Satoh, T.; Oishi, R.; Imaizumi, T.; Hakozaki, T.; Obara, S.; Ikegami, Y.; Kurosawa, S.; Murakawa, M. Effects of indigo carmine intravenous injection on noninvasive and continuous total hemoglobin measurement. J. Clin. Monit. Comput. 2016, 30, 313–316. [Google Scholar] [CrossRef]
- Zippelius, T.; Hoburg, A.; Preininger, B.; Vörös, P.; Perka, C.; Matziolis, G.; Röhner, E. Effect of indigo carmine on human chondrocytes in vitro. Open Orthop. J. 2013, 7, 8–11. [Google Scholar] [CrossRef]
- Choulis, N.H. Miscellaneous drugs, materials, medical devices and techniques. In Side Effects of Drugs Annual; Ray, S.D., Ed.; Elsevier: Manchester, UK, 2014; pp. 725–746. [Google Scholar]
- Tsou, M.P.M. Posterolateral percutane endoscopic lumbar discectomy. Oper. Technol. Orthop. 2003, 13, 207–213. [Google Scholar] [CrossRef]
- Yao, J.; Trindade, M.C.D. Color-aided visualization of dorsal wrist ganglion stalks aids in complete arthroscopic excision. Arthroscopy 2011, 27, 425–429. [Google Scholar] [CrossRef]
- Sheynkin, Y.R.; Starr, C.; Li, P.S.; Goldstein, M. Effect of methylene blue, indigo carmine, and Renografin on human sperm motility. Urology 1999, 53, 214–217. [Google Scholar] [CrossRef]
- Yang, J.; Monk, T.G.; White, P.F. Acute hemodynamic effects of indigo carmine in the presence of compromised cardiac function. J. Clin. Anesth. 1991, 3, 320–323. [Google Scholar] [CrossRef]
- Yanagidate, F.; Hamaya, Y.; Dohi, S. Vaginal indigo carmine-induced severe hypotension. Anesth. Analg. 2001, 92, 556–557. [Google Scholar] [CrossRef]
- Luketic, L.; Murji, A. Options to evaluate ureter patency at cystoscopy in a world without indigo carmine. J. Minim. Invasive Gynecol. 2016, 23, 878–885. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan, M.M.; Weitzman, G.A.; Hogan, S.; Breckinridge, S.; Miller, M.M. Methylene blue but not indigo carmine is toxic to human luteal cells in vitro. Reprod. Toxicol. 1993, 7, 631–633. [Google Scholar] [CrossRef] [PubMed]
- Imai, K.; Chikazawa, K.; Yonemori, E.; Kuwata, T. Visualizing the dissectable layer for vaginal hysterectomy with indigo carmine. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022, 278, 195–196. [Google Scholar] [CrossRef] [PubMed]
- Higashimori, A.; Takahara, M.; Utsunomiya, M.; Fukunaga, M.; Kawasaki, D.; Mori, S.; Takimura, H.; Hirano, K.; Tsubakimoto, Y.; Nakama, T.; et al. Utility of indigo carmine angiography in patients with critical limb ischemia: Prospective multi-center intervention study (DIESEL-study). Catheter. Cardiovasc. Interv. 2019, 93, 108–112. [Google Scholar] [CrossRef]
- Monson, F.C.; Wein, A.J.; McKenna, B.A.; Whitmore, K.; Levin, R.M. Indigo carmine as a quantitative indicator of urothelial integrity. J. Urol. 1991, 145, 842–845. [Google Scholar] [CrossRef]
- Mimura, M.; Alameddine, R.M.; Korn, B.S.; Kikkawa, D.O.; Oku, H.; Sato, B.; Ikeda, T. Endoscopic evaluation of lacrimal mucosa with indigo carmine stain. Ophthalm. Plast. Reconstr. Surg. 2020, 36, 49–54. [Google Scholar] [CrossRef]
- Kubo, K.; Kimura, N.; Kato, M. Texture and color enhancement imaging with indigo carmine dye accentuates slightly depressed early gastric cancer lesions. Clin. Gastroenterol. Hepatol. 2022, 20, 23–24. [Google Scholar] [CrossRef]
- Yasuda, T.; Yagi, N.; Omatsu, T.; Hayashi, S.; Nakahata, Y.; Yasuda, Y.; Obora, A.; Kojima, T.; Naito, Y.; Itoh, Y. Benefits of linked color imaging for recognition of early differentiated-type gastric cancer: In comparison with indigo carmine contrast method and blue laser imaging. Surg. Endosc. 2021, 35, 2750–2758. [Google Scholar] [CrossRef]
- Resindra, A.; Monno, Y.; Imahori, K.; Okutomi, M.; Suzuki, S.; Gotoda, T.; Miki, K. 3D reconstruction of whole stomach from endoscope video using structure-from-motion. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2019, 2019, 3900–3904. [Google Scholar]
- González-Bernardo, O.S.; Riestra, S.; Vivas, S.; de Francisco, R.; Pérez-Martínez, I.; Castoño-García, A.; Jiménez-Beltrán, V.; Rollé, V.; Suárez, P.; Suárez, A. Chromoendoscopy with indigo carmine vs virtual chromoendoscopy (iSCAN1) for neoplasia screening in patients with inflammatory bowel disease: A prospective randomized study. Inflamm. Bowel Dis. 2021, 27, 1256–1262. [Google Scholar] [CrossRef]
- Lord, R.; Burr, N.E.; Mohammed, N.; Subramanian, V. Colonic lesion characterization in inflammatory bowel disease: A systematic review and meta-analysis. World J. Gastroenterol. 2018, 24, 1167–1180. [Google Scholar] [CrossRef] [PubMed]
- Fu, K.; Sano, Y.; Kato, S.; Fujii, T.; Nagashima, F.; Yoshino, T.; Okuna, T.; Yoshida, S.; Fujimori, T. Chromoendoscopy using indigo carmine dye spraying with magnifying observation is the most reliable method for differential diagnosis between non-neoplastic and neoplastic colorectal lesions: A prospective study. Endoscopy 2004, 36, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, T.; Itoh, M.; Yoshimi, N. Pressure dye-spray: A simple and reliable method for differentiating adenomas from hyperplastic polyps in the colon. Gastrointest. Endosc. 2002, 55, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Hurlstone, D.P.; Brown, S.; Cross, S.S. The role of flat and depressed colorectal lesions in colorectal carcinogenesis: New insights from clinicopathological findings in high-magnification chromoscopic colonoscopy. Histopathology 2003, 43, 413–426. [Google Scholar] [CrossRef]
- Kiesslich, R.; Jung, M.; DiSario, J.A.; Galle, P.R.; Neurath, M.F. Perspectives of chromo and magnifying endoscopy: How, how much, when, and whom should we stain? J. Clin. Gastroenterol. 2004, 38, 7–13. [Google Scholar] [CrossRef]
- Kudo, S.; Tamura, S.; Nakajima, T.; Yamano, H.; Kusaka, H.; Watanabe, H. Diagnosis of colorectal timorous lesions by magnifying endoscopy. Gastrointest. Endosc. 1996, 44, 8–14. [Google Scholar] [CrossRef]
- Buchner, A.M. The role of chromoendoscopy in evaluating colorectal dysplasia. Gastroenterol. Hepatol. 2017, 13, 336–347. [Google Scholar]
- Pham, N.B.; Vu, K.T.; Nguyen, N.H.; Doan, H.T.; Tran, T.T. Magnifying chromoendoscopy with flexible spectral imaging color enhancement, indigo carmine, and crystal violet in predicting the histopathology of colorectal polyps: Diagnostic value in a scare-setting resource. Gastroenterol. Res. Pract. 2022, 2022, 6402904. [Google Scholar] [CrossRef]
- Ng, T.Y.; Datta, T.D.; Kirimli, B.I. Reaction to indigo carmine. J. Urol. 1976, 116, 132–133. [Google Scholar] [CrossRef]
- Kawaguchi, Y.; Hashimoto, H.; Kitayama, M.; Hirota, K. Intravenous indigo carmine might cause cerebral ischemia. Acta Anaesthesiol. Scand. 2007, 51, 776–777. [Google Scholar] [CrossRef]
- Craik, J.D.; Khan, D.; Afifi, R. The safety of intravenous indigo carmine to assess ureteric patency during transvaginal uterosacral suspension of the vaginal vault. J. Pelvic Med. Surg. 2009, 15, 11–15. [Google Scholar] [CrossRef]
- Sutton, E.; Pietrzak, A. Indigo carmine-induced hypotension in a parturient with idiopathic pulmonary artery hypertension, hypertrophic cardiomyopathy, and LAD myocardial bridging. J. Cardiothorac. Vasc. Anesth. 2016, 30, 158–161. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, J.F.; Connors, D.F.; Sprung, J.; Ballard, L.A. Upper extremity discoloration caused by subcutaneous indigo carmine injection. Anesth. Analg. 1996, 83, 1126–1128. [Google Scholar] [CrossRef]
- Choi, J.W.; Lee, J.J.; Kim, G.H.; Hong, S.H. Extensive skin color change caused by extravasation of indigo carmine. Korean J. Anesthesiol. 2012, 62, 499–500. [Google Scholar] [CrossRef] [PubMed]
- Porter, J.F.; McKay, G.; Choy, K.H. The prediction of sorption from a binary mixture of acidic dyes using single and mixed isotherm variants of the ideal adsorbed solute theory. Chem. Eng. Sci. 1999, 54, 5863–5885. [Google Scholar] [CrossRef]
- Kettle, A.J.; Clark, B.M.; Winterbourn, C.C. Superoxide converts indigo carmine to isatin sulfonic acid: Implications for the hypothesis that neutrophils produce ozone. J. Biol. Chem. 2004, 279, 18521–18525. [Google Scholar] [CrossRef] [PubMed]
- Savel, J.; Kosin, P.; Broz, A. Indigo carmine degradation in the presence of maltose and ethanol. J. Inst. Brew. 2015, 121, 548–552. [Google Scholar] [CrossRef]
- Benedict, C.S. Dissolved gases. In Brewing Materials and Processes. A Practical Approach to Beer Excellence; Bamforth, C.W., Ed.; Academic Press: Davis, CA, USA, 2016; pp. 157–174. [Google Scholar]
- Gaunt, I.F.; Kiss, I.S.; Grasso, P.; Gangolli, S.D. Short-term toxicity study on indigo carmine in the pig. Food Cosmet. Toxicol. 1969, 7, 17–24. [Google Scholar] [CrossRef]
- Hooson, J.; Gaunt, I.F.; Kiss, I.S.; Grasso, P.; Butterworth, K.R. Long-term in toxicity carmine in mice. Food Cosmet. Toxicol. 1975, 13, 167–176. [Google Scholar] [CrossRef]
- Hansen, W.H.; Fitzhugh, O.G.; Nelson, A.A.; Davis, K.J. Chronic toxicity of two food colors, Brilliant Blue FCF and Indigotine. Toxicol. Appl. Pharmacol. 1966, 8, 29–36. [Google Scholar] [CrossRef]
- Singh, S.; Das, M.; Khanna, S.K. Bio-metabolism of green S and Indigo Carmine through caecal microflora of rats. Biochem. Biophys. Res. Commun. 1993, 195, 490–496. [Google Scholar] [CrossRef]
- Ozaki, A.; Kitano, M.; Itoh, N.; Kuroda, K.; Furusawa, N.; Masuda, T.; Yamaguchi, H. Mutagenicity and DNA-damaging activity of decomposed products of food colours under UV radiation. Food Chem. Toxicol. 1998, 36, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Dixit, A.; Goyal, R.P. Evaluation of reproductive toxicity caused by indigo carmine on male swiss albino mice. Pharmacologyonline 2013, 1, 218–224. [Google Scholar]
- Peng, Y.J.; Chen, C.M.; Li, Y.F.; Guo, Y.T.; Chen, Y.T.; Chao, K.H.; Yang, J.J. Patent blue versus methylene blue and indigo carmine as a better dye for chromodiscography: In vitro staining efficacy and cytotoxicity study using bovine coccygeal intervertebral discs. Spine J. 2023, 23, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Yoo, W.H.; Kim, S.R.; Kim, S.H.; Lee, J.; Mok, J.; Shin, D.H.; Ahn, H.Y.; Eom, J.S. Stability and safety of transbronchial dye mixture for preoperative localization in a porcine model. Thorac. Cancer 2023, 14, 834–839. [Google Scholar] [CrossRef] [PubMed]


| Properties of Indigo Carmine Dye | Authors | |
|---|---|---|
| Molecular formula | C16H8N2Na2O8S2 | [26,27,35] |
| IUPAC name | 3,3′-dioxo-2,2′-bis indolyden-5,5-disulfonic acid disodium salt | |
| EINECS | 212-728-8 | [25] |
| Consistency | powder | [26,27,35,36] |
| Color | dark blue | |
| Color index number | 73,015 | [25] |
| Molecular weight | 466.367 g/mol | [23,26,37] |
| Melting point | >300 °C | [26] |
| Maximum absorption | 608–612 nm | |
| Composition | dye content ~ 85%; ˂18% disodium 3,3′-dioxo-2,2′-bi-indolylidene-5,7′-disulphonate | |
| Reactive group | amines, phosphines, pyridines, salts basic, ketones, hydrocarbons, aliphatic unsaturated | |
| Purity | water-insoluble matter ˂ 2%; auxiliary coloring substances ˂ 1%; organic compounds (isatin-5-sulfonic acid, 5-sulfoanthranilic acid, anthranilic acid) ˂ 1%; unsulfonated primary aromatic amines ˂ 0.01%; ether extractable matter ˂ 0.2%; arsenic ˂ 3 mg/kg; lead ˂ 2 mg/kg; cadmium ˂ 1 mg/kg; mercury ˂ 1 mg/kg | [25,38] |
| Solubility | 10 g/L | [27] |
| Density | 0.4–0.6 | |
| Food Category No. | Food Category | Restriction/Exceptions | EFSA MPL mg/kg | GSFA MPL mg/kg |
|---|---|---|---|---|
| 01.1.2 | Dairy-based drinks, flavored and/or fermented | - | 300 | |
| 01.4 | Flavored fermented milk products including heat-treated products | 150 | - | |
| 01.6.1 | Unripened cheese | For use in surface treatment only | 150 | 200 |
| 01.6.2.2 | Rind of ripened cheese | Refers to the rind only of the cheese | - | 100 |
| 01.6.3 | Other creams | 150 | - | |
| 01.6.4.2 | Flavored processed cheese, including containing fruit, vegetables, meat, etc. | - | 100 | |
| 01.6.5 | Cheese analogues | - | 200 | |
| 01.7 | Dairy-based desserts-pudding, fruit, or flavored yoghurt | - | 150 | |
| 01.7.3 | Edible cheese rind | quantum satis | - | |
| 01.7.6 | Cheese products | Only flavored unripened products | 100 | - |
| 02.1.3 | Lard, tallow, fish oil and other animal fats | - | 300 | |
| 02.3 | Fat emulsions mainly of type oil-in-water, including mixed and/or flavored products based on fat emulsions | - | 300 | |
| 02.4 | Fat-based desserts excluding dairy-based dessert | - | 150 | |
| 03.0 | Edible ices, including sherbet and sorbet | 150 | 150 | |
| 04.1.2.11 | Fruit fillings for pastries | - | 150 | |
| 04.1.2.5 | Jams, jellies, marmalades | - | 300 | |
| 04.1.2.6 | Fruit-based spreads | - | 300 | |
| 04.1.2.7 | Candied fruit | - | 200 | |
| 04.1.2.8 | Fruit preparations, including pulp, purees, fruit toppings and coconut milk | - | 150 | |
| 04.1.2.9 | Fruit-based desserts, including fruit-flavored water-based desserts | - | 150 | |
| 04.2.2.3 | Vegetables (mushrooms and fungi, roots and tubers, pulses and legumes, and aloe vera), and seaweeds in vinegar, oil, brine, or soybean sauce | - | 150 | |
| 04.2.2.7 | Fermented vegetable (mushrooms and fungi, roots and tubers, pulses and legumes, and aloe vera) and seaweed products | - | 300 | |
| 04.2.4.1 | Fruit and vegetable preparations excluding compote | Only mostarda di frutta | 200 | - |
| 05.2 | Confectionery including hard and soft candy, nougats | 200–300 | 300 | |
| 05.3 | Chewing gum | 300 | 300 | |
| 05.4 | Decorations for fine bakery wares, toppings (non-fruit) and sweet sauces | Only decorations, coatings, and sauces, except fillings | 300–500 | 300 |
| 05.1.4 | Cocoa and chocolate products | For use in surface decoration only | - | 450 |
| 05.1.5 | Imitation chocolate, chocolate substitute products | - | 300 | |
| 06.5 | Cereal- and starch-based desserts (rice pudding, tapioca pudding) | - | 150 | |
| 06.6 | Batters | Only for coating | 500 | - |
| 07.2 | Fine bakery wares (sweet, salty, savory) and mixes | 200 | 200 | |
| 08.2.3 | Casings and coatings and decorations for meat | Only decorations and coatings except edible external coating of pasturmas | 500 | - |
| 09.1.1 | Fresh fish | For use in decoration, stamping, or branding the product only | - | 300 |
| 09.2 | Processed fish and fishery products including mollusks and crustaceans | Only surimi and similar products and salmon substitutes | 500 | - |
| 09.2.1 | Frozen fish, fish fillets, and fish products, including mollusks, crustaceans, and echinoderms | For use in surimi and fish roe products only | - | 300 |
| 09.2.4.1 | Cooked fish and fish products | For use in surimi and fish roe products only | - | 300 |
| 09.2.4.2 | Cooked mollusks, crustaceans, and echinoderms | For use in glaze, coatings, or decorations | - | 250 |
| 09.3 | Fish roe | Except sturgeon’s eggs (caviar) | 300 | - |
| 09.3.3 | Salmon substitutes, caviar and other fish roe products | - | 300 | |
| 09.3.4 | Semi-preserved fish and fish products, including mollusks, crustaceans, and echinoderms, excluding products of food categories 09.3.1–09.3.3 | - | 300 | |
| 09.4 | Fully preserved, including canned or fermented fish and fish products, including mollusks, crustaceans, and echinoderms | Fish roe, sardines, surimi | - | 300 |
| 10.1 | Fresh eggs | For use in decoration, stamping, or branding the product only | - | 300 |
| 10.4 | Egg-based desserts (e.g., custard) | - | 300 | |
| 11.4 | Other sugars and syrups (e.g., xylose, maple syrup, sugar toppings) | - | 300 | |
| 12.2.2 | Seasonings and condiments | Only seasonings, for example, curry powder, tandoori | 500 | 300 |
| 12.4 | Mustards | 300 | 300 | |
| 12.5 | Soups and broths | 300 | 50 | |
| 12.6 | Sauces and similar products | Including pickles, relishes, chutney, and piccalilli; excluding tomato-based sauces | 500 | 300 |
| 12.9 | Protein products | Only meat and fish analogues based on vegetable proteins | 100 | - |
| 13.2 | Dietary foods for special medical purposes defined in Directive 1999/21/EC | 50 | - | |
| 13.3 | Dietetic foods intended for special medical purposes (excluding products of food category 13.2) | 50 | 50 | |
| 13.4 | Dietetic formulas for slimming purposes and weight reduction | - | 50 | |
| 13.5 | Dietetic foods (e.g., supplementary foods for dietary use) excluding products of food categories 13.1–13.4 and 13.6 | - | 300 | |
| 13.6 | Food supplements | - | 300 | |
| 14.1.4 | Water-based flavored drinks, including sport, energy, or electrolyte drinks and particulated drinks | 100 | 100 | |
| 14.2.2 | Cider and perry | Excluding cidre bouché | 200 | 200 |
| 14.2.4 | Wines (other than grape) | 200 | 200 | |
| 14.2.6 | Distilled spirituous beverages containing more than 15% alcohol | 200 | 300 | |
| 14.2.7 | Aromatized alcoholic beverages (e.g., beer, wine and spirituous cooler-type beverages, low-alcoholic refreshers) | 200 | 200 | |
| 14.2.7.1 | Aromatized wines | Except americano, bitter vino | 200 | - |
| 14.2.7.2 | Aromatized wine-based drinks | 200 | - | |
| 14.2.7.3 | Aromatized wine-product cocktails | 200 | - | |
| 14.2.8 | Other alcoholic drinks including | 200 | - | |
| 15.1 | Snacks—potato-, cereal-, flour-, or starch-based (from roots and tubers, pulses, and legumes) | 100 | 200 | |
| 15.2 | Processed nuts, including coated nuts and nut mixtures (with e.g., dried fruit) | Only savory-coated nuts | 100 | 100 |
| 16 | Desserts excluding products covered in categories 01, 03, and 04 | 150 | - | |
| 17.1 | Food supplements supplied in a solid form (capsules and tablets and similar forms, excluding chewable forms) | 300 | - | |
| 17.2 | Food supplements supplied in a liquid form | 100 | - | |
| 17.3 | Food supplements supplied in a syrup-type or chewable form | 100–300 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ristea, M.-E.; Zarnescu, O. Indigo Carmine: Between Necessity and Concern. J. Xenobiot. 2023, 13, 509-528. https://doi.org/10.3390/jox13030033
Ristea M-E, Zarnescu O. Indigo Carmine: Between Necessity and Concern. Journal of Xenobiotics. 2023; 13(3):509-528. https://doi.org/10.3390/jox13030033
Chicago/Turabian StyleRistea, Madalina-Elena, and Otilia Zarnescu. 2023. "Indigo Carmine: Between Necessity and Concern" Journal of Xenobiotics 13, no. 3: 509-528. https://doi.org/10.3390/jox13030033
APA StyleRistea, M.-E., & Zarnescu, O. (2023). Indigo Carmine: Between Necessity and Concern. Journal of Xenobiotics, 13(3), 509-528. https://doi.org/10.3390/jox13030033