Testosterone Usage Leading to Pulmonary Embolisms and Deep Vein Thrombosis: A Case Report and Review of the Literature
Abstract
1. Introduction
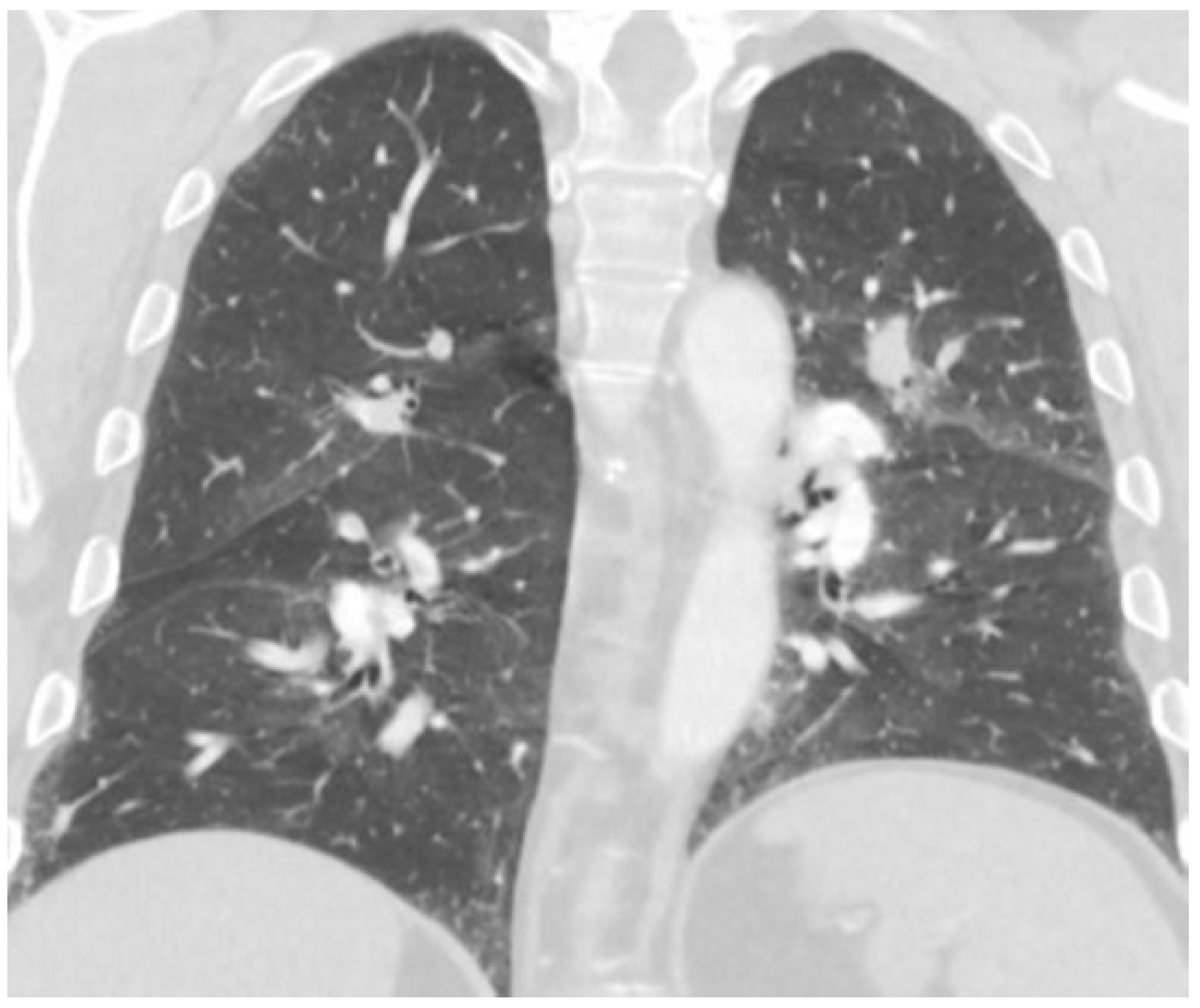
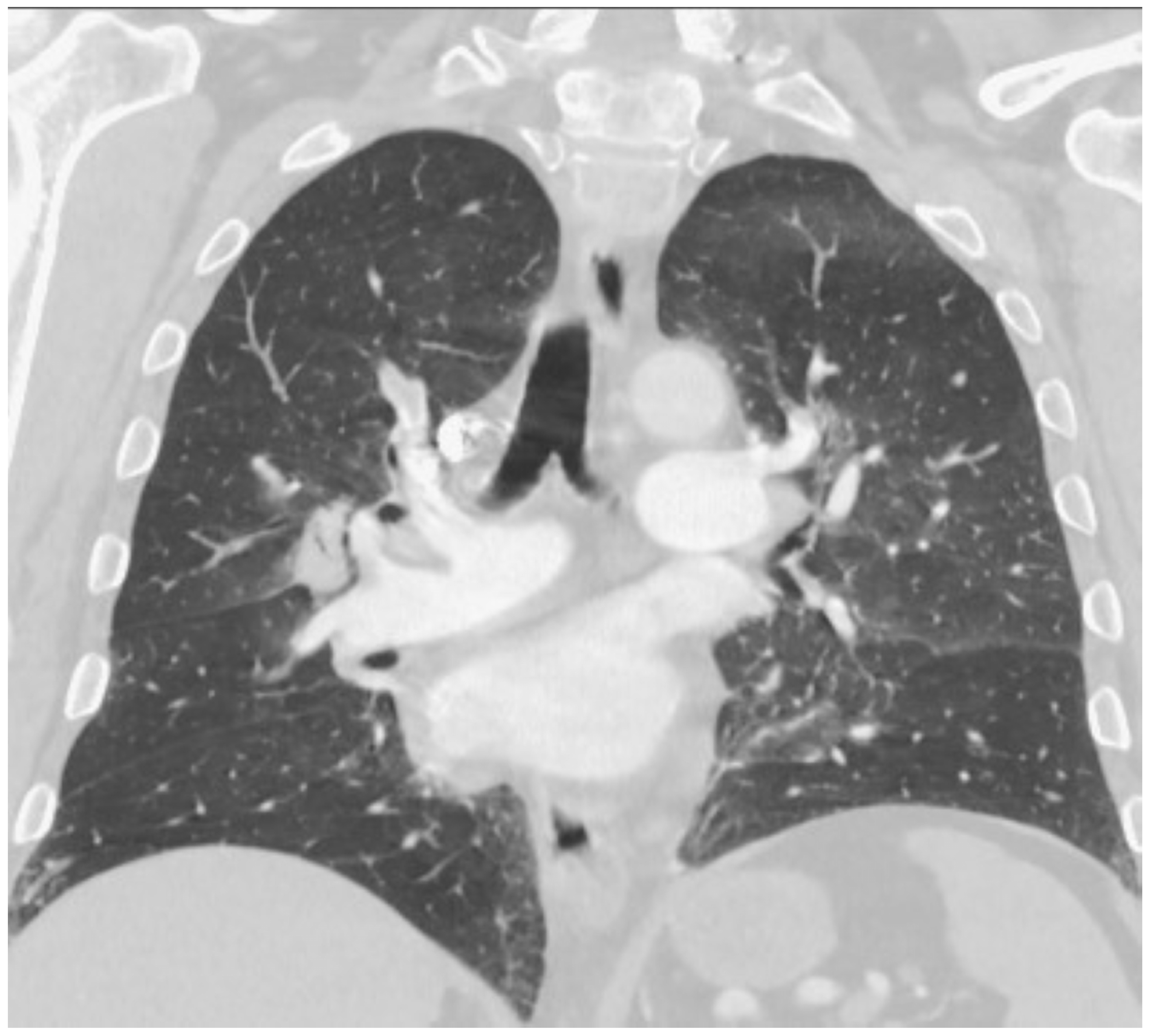
2. Case Report
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sagoe, D.; Molde, H.; Andreassen, C.S.; Torsheim, T.; Pallesen, S. The global epidemiology of anabolic-androgenic steroid use: A meta-analysis and meta-regression analysis. Ann. Epidemiol. 2014, 24, 383–398. [Google Scholar] [CrossRef] [PubMed]
- Pope, H.G., Jr.; Wood, R.I.; Rogol, A.; Nyberg, F.; Bowers, L.; Bhasin, S. Adverse health consequences of performance-enhancing drugs: An Endocrine Society scientific statement. Endocr. Rev. 2014, 35, 341–375. [Google Scholar] [CrossRef] [PubMed]
- Buckman, J.F.; Farris, S.G.; Yusko, D.A. A national study of substance use behaviors among NCAA male athletes who use banned performance enhancing substances. Drug Alcohol Depend. 2013, 131, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Ip, E.J.; Lu, D.H.; Barnett, M.J.; Tenerowicz, M.J.; Vo, J.C.; Perry, P.J. Psychological and physical impact of anabolic-androgenic steroid dependence. Pharmacotherapy 2012, 32, 910–919. [Google Scholar] [CrossRef]
- NIDA. How Are Anabolic Steroids Used? 22 November 2021. Available online: https://nida.nih.gov/publications/research-reports/steroids-other-appearance-performance-enhancing-drugs-apeds/how-are-anabolic-steroids-used (accessed on 7 December 2022).
- de Ronde, W.; Smit, D.L. Anabolic androgenic steroid abuse in young males. Endocr. Connect. 2020, 9, R102–R111. [Google Scholar] [CrossRef] [PubMed]
- Turetz, M.; Sideris, A.T.; Friedman, O.A.; Triphathi, N.; Horowitz, J.M. Epidemiology, Pathophysiology, and Natural History of Pulmonary Embolism. Semin. Interv. Radiol. 2018, 35, 92–98. [Google Scholar] [CrossRef]
- Horlander, K.T.; Mannino, D.M.; Leeper, K.V. Pulmonary Embolism Mortality in the United States, 1979–1998: An analysis using multiple-cause mortality data. Arch. Intern. Med. 2003, 163, 1711–1717. [Google Scholar] [CrossRef]
- Stein, P.D.; Beemath, A.; Matta, F.; Weg, J.G.; Yusen, R.D.; Hales, C.A.; Hull, R.D.; Leeper, K.V.; Sostman, H.D.; Tapson, V.F.; et al. Clinical Characteristics of Patients with Acute Pulmonary Embolism: Data from PIOPED II. Am. J. Med. 2007, 120, 871–879. [Google Scholar] [CrossRef]
- Di Nisio, M.; van Es, N.; Büller, H.R. Deep vein thrombosis and pulmonary embolism. Lancet 2016, 388, 3060–3073. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Toraby, S.; Cotterman, R.; Oriowo, B.; Fish, J. A Tumultuous Course of Exogenous Testosterone by a Bodybuilder Causing a Catastrophic Hypercoagulable State in the Surgical Intensive Care Unit. Case Rep. Vasc. Med. 2019, 2019, 3097865. [Google Scholar] [CrossRef] [PubMed]
- Liljeqvist, S.; Helldén, A.; Bergman, U.; Söderberg, M. Pulmonary embolism associated with the use of anabolic steroids. Eur. J. Intern. Med. 2008, 19, 214–215. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, S.M.; Ko, N.K.; Sattar, A.S.; Ipek, E.G.; Ali, S. Pulmonary Embolism Secondary to Testosterone-Enhancing Herbal Supplement Use. Cureus 2017, 9, e1545. [Google Scholar] [CrossRef] [PubMed]
- Handelsman, D.J. Androgen Misuse and Abuse. Endocr. Rev. 2021, 42, 457–501. [Google Scholar] [CrossRef]
- Montisci, M.; El Mazloum, R.; Cecchetto, G.; Terranova, C.; Ferrara, S.D.; Thiene, G.; Basso, C. Anabolic androgenic steroids abuse and cardiac death in athletes: Morphological and toxicological findings in four fatal cases. Forensic Sci. Int. 2012, 217, e13–e18. [Google Scholar] [CrossRef] [PubMed]
- Pillai, A.A.; Fazal, S.; Babiker, H.M. Polycythemia. [Updated 10 July 2022]. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK526081 (accessed on 7 December 2022).
- McMullin, M.F.; Bareford, D.; Campbell, P.; Green, A.R.; Harrison, C.; Hunt, B.; Oscier, D.; Polkey, M.I.; Reilly, J.T.; Rosenthal, E.; et al. Guidelines for the diagnosis, investigation and management of polycythaemia/erythrocytosis. Br. J. Haematol. 2005, 130, 174–195. [Google Scholar] [CrossRef]
- Wells, R.E.; Merrill, E.W. Influence of Flow Properties of Blood Upon Viscosity-Hematocrit Relationships. J. Clin. Investig. 1962, 41, 1591–1598. [Google Scholar] [CrossRef]
- Maggio, M.; Snyder, P.J.; Ceda, G.P.; Milaneschi, Y.; Luci, M.; Cattabiani, C.; Masoni, S.; Vignali, A.; Volpi, R.; Lauretani, F.; et al. Is the haematopoietic effect of testosterone mediated by erythropoietin? The results of a clinical trial in older men. Andrology 2013, 1, 24–28. [Google Scholar] [CrossRef]
- Bachman, E.; Feng, R.; Travison, T.; Li, M.; Olbina, G.; Ostland, V.; Ulloor, J.; Zhang, A.; Basaria, S.; Ganz, T.; et al. Testosterone suppresses hepcidin in men: A potential mechanism for testosterone-induced erythrocytosis. J. Clin. Endocrinol. Metab. 2010, 95, 4743–4747. [Google Scholar] [CrossRef] [PubMed]
- Bachman, E.; Travison, T.G.; Basaria, S.; Davda, M.N.; Guo, W.; Li, M.; Westfall, J.C.; Bae, H.; Gordeuk, V.; Bhasin, S. Testosterone Induces Erythrocytosis via Increased Erythropoietin and Suppressed Hepcidin: Evidence for a New Erythropoietin/Hemoglobin Set Point. J. Gerontol. Ser. A Biomed. Sci. Med. Sci. 2014, 69, 725–735. [Google Scholar] [CrossRef]
- Calado, R.T.; Yewdell, W.T.; Wilkerson, K.L.; Regal, J.A.; Kajigaya, S.; Stratakis, C.A.; Young, N.S. Sex hormones, acting on the TERT gene, increase telomerase activity in human primary hematopoietic cells. Blood 2009, 114, 2236–2243. [Google Scholar] [CrossRef]
- Jones, S.D., Jr.; Dukovac, T.; Sangkum, P.; Yafi, F.A.; Hellstrom, W.J. Erythrocytosis and Polycythemia Secondary to Testosterone Replacement Therapy in the Aging Male. Sex Med. Rev. 2015, 3, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Ory, J.; Nackeeran, S.; Balaji, N.C.; Hare, J.M.; Ramasamy, R. Secondary Polycythemia in Men Receiving Testosterone Therapy Increases Risk of Major Adverse Cardiovascular Events and Venous Thromboembolism in the First Year of Therapy. J. Urol. 2022, 207, 1295–1301. [Google Scholar] [CrossRef]
- Ohlander, S.J.; Varghese, B.; Pastuszak, A.W. Erythrocytosis Following Testosterone Therapy. Sex. Med. Rev. 2018, 6, 77–85. [Google Scholar] [CrossRef]
- Madsen, M.C.; van Dijk, D.; Wiepjes, C.M.; Conemans, E.B.; Thijs, A.; Heijer, M.D. Erythrocytosis in a Large Cohort of Trans Men Using Testosterone: A Long-Term Follow-Up Study on Prevalence, Determinants, and Exposure Years. J. Clin. Endocrinol. Metab. 2021, 106, 1710–1717. [Google Scholar] [CrossRef]
- Martinez, C.; Suissa, S.; Rietbrock, S.; Katholing, A.; Freedman, B.; Cohen, A.T.; Handelsman, D.J. Testosterone treatment and risk of venous thromboembolism: Population based case-control study. BMJ 2016, 355, i5968. [Google Scholar] [CrossRef] [PubMed]
- Ajayi, A.A.L.; Mathur, R.; Halushka, P.V. Testosterone Increases Human Platelet Thromboxane A 2 Receptor Density and Aggregation Responses. Circulation 1995, 91, 2742–2747. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Mazumder, S.; Bhattacharya, S.; Sinha, A.K. The sex specific effects of extraneous testosterone on ADP induced platelet aggregation in platelet-rich plasma from male and female subjects. Int. J. Lab. Hematol. 2014, 36, e74–e77. [Google Scholar] [CrossRef]
- Roşca, A.E.; Vlădăreanu, A.M.; Mititelu, A.; Popescu, B.O.; Badiu, C.; Căruntu, C.; Voiculescu, S.E.; Onisâi, M.; Gologan, Ş.; Mirica, R.; et al. Effects of Exogenous Androgens on Platelet Activity and Their Thrombogenic Potential in Supraphysiological Administration: A Literature Review. J. Clin. Med. 2021, 10, 147. [Google Scholar] [CrossRef]
- Coviello, A.D.; Kaplan, B.; Lakshman, K.M.; Chen, T.; Singh, A.B.; Bhasin, S. Effects of Graded Doses of Testosterone on Erythropoiesis in Healthy Young and Older Men. J. Clin. Endocrinol. Metab. 2008, 93, 914–919. [Google Scholar] [CrossRef]
- Cervi, A.; Balitsky, A.K. Testosterone use causing erythrocytosis. Can. Med. Assoc. J. 2017, 189, E1286–E1288. [Google Scholar] [CrossRef]
- Basaria, S.; Coviello, A.D.; Travison, T.G.; Storer, T.W.; Farwell, W.R.; Jette, A.M.; Eder, R.; Tennstedt, S.; Ulloor, J.; Zhang, A.; et al. Adverse Events Associated with Testosterone Administration. N. Engl. J. Med. 2010, 363, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Finkle, W.D.; Greenland, S.; Ridgeway, G.K.; Adams, J.L.; Frasco, M.A.; Cook, M.B.; Fraumeni, J.F., Jr.; Hoover, R.N. Increased Risk of Non-Fatal Myocardial Infarction Following Testosterone Therapy Prescription in Men. PLoS ONE 2014, 9, e85805. [Google Scholar] [CrossRef] [PubMed]
- Vigen, R. Association of Testosterone Therapy with Mortality, Myocardial Infarction, and Stroke in Men with Low Testosterone Levels. JAMA 2013, 310, 1829–1836. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menakuru, S.R.; Atta, M.; Dhillon, V.S.; Salih, A. Testosterone Usage Leading to Pulmonary Embolisms and Deep Vein Thrombosis: A Case Report and Review of the Literature. Hematol. Rep. 2023, 15, 290-297. https://doi.org/10.3390/hematolrep15020029
Menakuru SR, Atta M, Dhillon VS, Salih A. Testosterone Usage Leading to Pulmonary Embolisms and Deep Vein Thrombosis: A Case Report and Review of the Literature. Hematology Reports. 2023; 15(2):290-297. https://doi.org/10.3390/hematolrep15020029
Chicago/Turabian StyleMenakuru, Sasmith R., Mona Atta, Vijaypal S. Dhillon, and Ahmed Salih. 2023. "Testosterone Usage Leading to Pulmonary Embolisms and Deep Vein Thrombosis: A Case Report and Review of the Literature" Hematology Reports 15, no. 2: 290-297. https://doi.org/10.3390/hematolrep15020029
APA StyleMenakuru, S. R., Atta, M., Dhillon, V. S., & Salih, A. (2023). Testosterone Usage Leading to Pulmonary Embolisms and Deep Vein Thrombosis: A Case Report and Review of the Literature. Hematology Reports, 15(2), 290-297. https://doi.org/10.3390/hematolrep15020029




