Microcystin Concentrations and Detection of the mcyA Gene in Water Collected from Agricultural, Urban, and Recreational Areas in a Karst Aquifer in the Yucatan Peninsula of Mexico
Abstract
1. Introduction
2. Materials and Methods
2.1. Water Collection
2.2. Physicochemical Parameters
2.3. Water Quality Variables
2.4. Microcystin Quantification
2.5. Phytoplankton Collection and DNA Isolation
2.6. PCR Amplification and Cloning of the mcyA Gene
2.7. Sequence Analysis
2.8. Phylogenetic Analysis
2.9. Statistical Analysis
3. Results
3.1. Physicochemical Variables
3.2. Microcystin Concentrations in Water
3.3. Hydrochemistry
3.4. Nutrient Analysis
3.5. Generalized Additive Models (GAMs)
3.6. mcyA Gene Sequence Analysis
3.7. mcyA Phylogenetic Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wurtsbaugh, W.A.; Paerl, H.W.; Dodds, W.K. Nutrients, eutrophication and harmful algal blooms along the freshwater to marine continuum. Wiley Interdisciplinary Reviews. Water 2019, 6, e1373. [Google Scholar] [CrossRef]
- Beversdorf, L.J.; Chaston, S.D.; Miller, T.R.; McMahon, K.D. Microcystin mcyA and mcyE gene abundances are not appropriate indicators of microcystin concentrations in lakes. PLoS ONE 2015, 10, e0125353. [Google Scholar] [CrossRef] [PubMed]
- Kimambo, O.N.; Gumbo, J.R.; Chikoore, H. The occurrence of cyanobacteria blooms in freshwater ecosystems and their link with hydro-meteorological and environmental variations in Tanzania. Heliyon 2019, 5, e01312. [Google Scholar] [CrossRef]
- Sinha, R.; Pearson, L.A.; Davis, T.W.; Burford, M.A.; Orr, P.T.; Neilan, B.A. Increased incidence of Cylindrospermopsis raciborskii in temperate zones—Is climate change responsible? Water Res. 2012, 46, 1408–1419. [Google Scholar] [CrossRef] [PubMed]
- Beretta-Blanco, A.; Carrasco-Letelier, L. Relevant factors in the eutrophication of the Uruguay River and the Río Negro. Sci. Total Environ. 2021, 761, 143–299. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Zhu, G.; Cai, Y.; Xu, H.; Zhu, M.; Gong, Z.; Qin, B. Quantifying the dependence of cyanobacterial growth to nutrient for the eutrophication management of temperate-subtropical shallow lakes. Water Res. 2020, 177, 115806. [Google Scholar] [CrossRef]
- Giannuzzi, L.; Hernando, M. The Eco-Physiological Role of Microcystis aeruginosa in a Changing World. Microorganisms 2022, 10, 685. [Google Scholar] [CrossRef] [PubMed]
- Mowe, M.A.; Porojan, C.; Abbas, F.; Mitrovic, S.M.; Lim, R.P.; Furey, A.; Yeo, D.C. Rising temperatures may increase growth rates and microcystin production in tropical Microcystis species. Harmful Algae 2015, 50, 88–98. [Google Scholar] [CrossRef]
- Paerl, H.W.; Otten, T.G. Harmful cyanobacterial blooms: Causes, consequences, and controls. Microb. Ecol. 2013, 65, 995–1010. [Google Scholar] [CrossRef]
- Sangolkar, L.N.; Maske, S.S.; Muthal, P.L.; Kashyap, S.M.; Chakrabarti, T. Isolation and characterization of microcystin producing Microcystis from a Central Indian water bloom. Harmful Algae 2009, 8, 674–684. [Google Scholar] [CrossRef]
- Álvarez, X.; Valero, E.; Santos, R.M.; Varandas, S.G.P.; Fernandes, L.S.; Pacheco, F.A.L. Anthropogenic nutrients and eutrophication in multiple land use watersheds: Best management practices and policies for the protection of water resources. Land Use Policy 2017, 69, 1–11. [Google Scholar] [CrossRef]
- Paerl, H.W. Mitigating toxic planktonic cyanobacterial blooms in aquatic ecosystems facing increasing anthropogenic and climatic pressures. Toxins 2018, 10, 76. [Google Scholar] [CrossRef] [PubMed]
- Kubickova, B.; Babica, P.; Hilscherová, K.; Šindlerová, L. Effects of cyanobacterial toxins on the human gastrointestinal tract and the mucosal innate immune system. Environ. Sci. Eur. 2019, 31, 1–27. [Google Scholar] [CrossRef]
- Schaefer, A.M.; Yrastorza, L.; Stockley, N.; Harvey, K.; Harris, N.; Grady, R.; Sullivan, J.; McFarland, M.; Reif, J.S. Exposure to microcystin among coastal residents during a cyanobacteria bloom in Florida. Harmful Algae 2020, 92, 101769. [Google Scholar] [CrossRef] [PubMed]
- Francis, G. Poisonous Australian Lake. Nature 1878, 18, 11–12. [Google Scholar] [CrossRef]
- Codd, G.A.; Bell, S.G.; Brooks, W.P. Cyanobacterial toxins in water. Water Sci. Technol. 1989, 21, 1–13. [Google Scholar] [CrossRef]
- Stewart, I.; Seawright, A.A.; Shaw, G.R. Cyanobacterial poisoning in livestock, wild mammals and birds—An overview. State of the Science and Research Needs. Adv. Exp. Med. Biol. 2008, 619, 613–637. [Google Scholar] [CrossRef]
- McLellan, N.L.; Manderville, R.A. Toxic mechanisms of microcystins in mammals. Toxicol. Res. 2017, 6, 391–405. [Google Scholar] [CrossRef]
- Pouria, S.; de Andrade, A.; Barbosa, J.; Cavalcanti, R.L.; Barreto, V.T.S.; Ward, C.J.; Preiser, W.; Poon, G.K.; Neild, G.H.; Codd, G.A. Fatal microcystin intoxication in hemodialysis unit in Caruaru, Brazil. Lancet 1998, 352, 21–26. [Google Scholar] [CrossRef]
- SEGOB. Proyecto de Norma Oficial Mexicana PROY-NOM-127-SSA1-2017, Agua para Uso y Consumo Humano. Límites Permisibles de la Calidad del Agua; SEGOB: Mexico City, Mexico, 2017. [Google Scholar]
- SEGOB Agua Determinación de Acidez Total y Alcalinidad Total, Norma Oficial Mexicana NOM-AA-36-1980.DOF01/08/2001. Available online: https://www.dof.gob.mx/nota_detalle.php?codigo=761733&fecha=01/08/2001#gsc.tab=0 (accessed on 31 March 2023).
- Arzate-Cárdenas, M.A.; Olvera-Ramírez, R.; Martínez-Jerónimo, F. Microcystis toxigenic strains in urban lakes: A case of study in Mexico City. Ecotoxicology 2010, 19, 1157–1165. [Google Scholar] [CrossRef]
- Pineda-Mendoza, R.M.; Briones-Roblero, C.I.; Gonzalez-Escobedo, R.; Rivera-Orduña, F.N.; Martínez-Jerónimo, F.; Zúñiga, G. Seasonal changes in the bacterial community structure of three eutrophicated urban lakes in Mexico City, with emphasis on Microcystis spp. Toxicon 2020, 179, 8–20. [Google Scholar] [CrossRef]
- Montero, E.; Vázquez, G.; Caballero, M.; Favila, M.E.; Martínez-Jerónimo, F. Seasonal variation of Microcystis aeruginosa and factors related to blooms in a deep warm monomictic lake in Mexico. J. Limnol. 2021, 80, 1–20. [Google Scholar] [CrossRef]
- Cagle, S.E.; Roelke, D.L.; Hernández-Zepeda, C.; Rosiles-González, G.; Carrillo-Jovel, V.H.; Ortega-Camacho, D.; Cejudo, E. Cyanobacteria and nitrates in karstic systems of Yucatan (Mexico) and Texas (USA). Aquat. Sci. 2021, 83, 74. [Google Scholar] [CrossRef]
- Guardiola, J.; González-Gómez, F.; Lendechy-Grajales, Á. The Influence of Water Access in Subjective Well-Being: Some Evidence in Yucatan, Mexico. Soc. Indic. Res. 2013, 110, 207–218. [Google Scholar] [CrossRef]
- Baxa, D.V.; Kurobe, T.; Ger, K.A.; Lehman, P.W.; Teh, S.J. Estimating the abundance of toxic Microcystis in the San Francisco Estuary using quantitative real-time PCR. Harmful Algae 2010, 9, 342–349. [Google Scholar] [CrossRef]
- Feist, S.M.; Lance, R.F. Genetic detection of freshwater harmful algal blooms: A review focused on the use of environmental DNA (eDNA) in Microcystis aeruginosa and Prymnesium parvum. Harmful Algae 2021, 110, 102124. [Google Scholar] [CrossRef]
- Hisbergues, M.; Christiansen, G.; Rouhiainen, L.; Sivonen, K.; Börner, T. PCR-based identification of microcystin-producing genotypes of different cyanobacterial genera. Arch. Microbiol. 2003, 180, 402–410. [Google Scholar] [CrossRef]
- Li, D.; Gu, A.Z.; He, M. Quantification and genetic diversity of total and microcystin-producing Microcystis during blooming season in Tai and Yang-cheng lakes, China. J. Appl. Microbiol. 2014, 116, 1482–1494. [Google Scholar] [CrossRef]
- Sidelev, S.I. A Novel Multiplex PCR-based Technique for Detection of Toxigenic Cyanobacteria. Microbiology 2019, 88, 375–377. [Google Scholar] [CrossRef]
- Tillett, D.; Dittmann, E.; Erhard, M.; von Döhren, H.; Börner, T.; Neilan, B.A. Structural organization of microcystin biosynthesis in Microcystis aeruginosa PCC7806: An integrated peptide-polyketide synthetase system. Chem. Biol. 2000, 7, 753–764. [Google Scholar] [CrossRef]
- Li, L.; Jia, R.; Liu, Y.; Zhang, H. Detection of microcystin-producing cyanobacteria in a reservoir by whole cell quantitative PCR. Procedia Environ. Sci. 2011, 10, 2272–2279. [Google Scholar] [CrossRef][Green Version]
- Schmitter-Soto, J.J.; Comín, F.A.; Escobar-Briones, E.; Herrera-Silveira, J.; Alcocer, J.; Suárez-Morales, E.; Steinich, B. Hydrogeochemical and biological characteristics of cenotes in the Yucatan Peninsula (SE Mexico). Hydrobiologia 2002, 467, 215–228. [Google Scholar] [CrossRef]
- Tavera, R.; Novelo, E.; López, S. Cyanoprokaryota (Cyanobacteria) in karst environments in Yucatan, Mexico. Bot. Sci. 2013, 91, 27–52. [Google Scholar] [CrossRef][Green Version]
- Valadez, F.; Rosiles-González, G.; Almazán-Becerril, A.; Merino-Ibarra, M. Las cianobacterias planctonicas del lago tropical carstico Lagartos de la Peninsula de Yucatan, Mexico. Rev. Biol. Trop. 2013, 61, 971–979. [Google Scholar] [CrossRef]
- Strickland, J.D.; Parsons, T.R. A Practical Handbook of Seawater Analysis; Fisheries Research Board of Canada: Ottawa, ON, Canada, 1972. [Google Scholar] [CrossRef]
- Bower, C.E.; Holm-Hansen, T. A salicylate–hypochlorite method for determining ammonia in seawater. Can. J. Fish. Aquat. Sci. 1980, 37, 794–798. [Google Scholar] [CrossRef]
- Lorenzen, C.J. Determination of Chlorophyll and Pheo-Pigments: Spectrophotometric equations. Limnol. Oceanogr. 1967, 12, 343–346. [Google Scholar] [CrossRef]
- Fernandez-Figueroa, E.G.; Buley, R.P.; Barros, M.U.; Gladfelter, M.F.; McClimans, W.D.; Wilson, A.E. Carlson’s Trophic State Index is a poor predictor of cyanobacterial dominance in drinking water reservoirs. AWWA Water Sci. 2021, 3, e1219. [Google Scholar] [CrossRef]
- Sambrook, J.; Russell, D.W. Molecular Cloning—A Laboratory Manual, 3rd ed.; Cold Spring Harbor Laboratory Press: Long Island, NY, USA, 2001. [Google Scholar]
- Sherwood, A.R.; Presting, G.G. Universal primers amplify a 23S rDNA plastid marker in eukaryotic algae and cyanobacteria. J. Phycol. 2007, 43, 605–608. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef]
- Anderson, M.J.; Walsh, D.C. PERMANOVA, ANOSIM, and the Mantel test in the face of heterogeneous dispersions: What null hypothesis are you testing? Ecol. Monogr. 2013, 83, 557–574. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v6: User Manual/Tutorial, 190; PRIMER-E: Plymouth, UK, 2006. [Google Scholar]
- Le Hen, G.; Balzani, P.; Haase, P.; Kouba, A.; Liu, C.; Nagelkerke, L.A.J.; Theissen, N.; Renault, D.; Soto, I.; Haubrock, P.J. Alien species and climate change drive shifts in a riverine fish community and trait compositions over 35 years. Sci. Total Environ. 2023, 867, 161486. [Google Scholar] [CrossRef] [PubMed]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org (accessed on 21 April 2023).
- Pacheco, J.; Marín, L.; Cabrera, A.; Steinich, B.; Escolero, O. Nitrate Temporal and Spatial Patterns in 12 Water-Supply Wells, Yucatan, Mexico. Environ. Geol. 2001, 40, 708–715. [Google Scholar] [CrossRef]
- Moujabber, M.; Samra, B.; Darwish, T.; Atallah, T. Comparison of Different Indicators for Groundwater Contamination by Seawater Intrusion on the Lebanese Coast. Water Resour. Manag. 2006, 20, 161–180. [Google Scholar] [CrossRef]
- Asare, A.; Appiah-Adjei, E.K.; Ali, B.; Owusu-Nimo, F. Assessment of Seawater Intrusion Using Ionic Ratios: The Case of Coastal Communities along the Central Region of Ghana. Environ. Earth Sci. 2021, 80, 1–14. [Google Scholar] [CrossRef]
- Perry, E.; Velázquez-Oliman, G.; Marín, L. The Hydrogeochemistry of the Karst Aquifer System of the Northern Yucatan Peninsula, Mexico. Int. Geol. Rev. 2002, 44, 191–221. [Google Scholar] [CrossRef]
- Ye, X.; Zhou, Y.; Lu, Y.; Du, X. Hydrochemical Evolution and Quality Assessment of Groundwater in the Sanjiang Plain, China. Water 2022, 14, 1265. [Google Scholar] [CrossRef]
- Sarker, M.; Rahman, M.; Van Camp, M.; Hossain, D.; Islam, M.; Ahmed, N.; Masud Karim, M.; Quaiyum Bhuiyan, M.A.; Walraevens, K. Groundwater Salinization and Freshening Processes in Coastal Aquifers from Southwest Bangladesh. Sci. Total Environ. 2021, 779, 146339. [Google Scholar] [CrossRef] [PubMed]
- Ahamad, A.; Madhav, S.; Singh, P.; Pandey, J.; Khan, A.H. Assessment of Groundwater Quality with Special Emphasis on Nitrate Contamination in Parts of Varanasi City, Uttar Pradesh, India. Appl. Water Sci. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Song, L.; Sano, T.; Li, R.; Watanabe, M.M.; Liu, Y.; Kaya, K. Microcystin production of Microcystis viridis (cyanobacteria) under different culture conditions. Phycol. Res. 1998, 46, 19–23. [Google Scholar] [CrossRef]
- Yang, J.; Tang, H.; Zhang, X.; Zhu, Z.; Huang, Y.; Yang, Z. High temperature and pH favor Microcystis aeruginosa to outcompete Scenedesmus obliquus. Environ. Sci. Pollut. Res. 2018, 25, 4794–4802. [Google Scholar] [CrossRef] [PubMed]
- Dai, R.; Wang, P.; Jia, P.; Zhang, Y.; Chu, X.; Wang, Y. A review on factors affecting microcystins production by algae in aquatic environments. World J. Microbiol. Biotechnol. 2016, 32, 51. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, J.; Huang, W.; Kong, F.; Li, Y.; Xi, M.; Zheng, Z. Comparative bioavailability of ammonium, nitrate, nitrite and urea to typically harmful cyanobacterium Microcystis aeruginosa. Mar. Pollut. Bull. 2016, 110, 93–98. [Google Scholar] [CrossRef]
- Smith, D.N.; Ortega-Camacho, D.; Acosta-González, G.; Leal-Bautista, R.M.; Fox III, W.E.; Cejudo, E. A multi-approach assessment of land use effects on groundwater quality in a karstic aquifer. Heliyon 2020, 6, e03970. [Google Scholar] [CrossRef]
- Herrera-Silveira, J.A.; Martín, B.M.; Díaz-Arce, V. Variaciones del fitoplancton en cuatro lagunas costeras del Estado de Yucatán, México. Rev. Biol. Trop. 2016, 47, 47–56. Available online: https://revistas.ucr.ac.cr/index.php/rbt/article/view/26140 (accessed on 31 January 2023).
- Tan, X.; Gu, H.; Ruan, Y.; Zhong, J.; Parajuli, K.; Hu, J. Effects of nitrogen on interspecific competition between two cell-size cyanobacteria: Microcystis aeruginosa and Synechococcus sp. Harmful Algae 2019, 89, 101661. [Google Scholar] [CrossRef] [PubMed]
- Böhlke, J.K.; Smith, R.L.; Miller, D.N. Ammonium transport and reaction in contaminated groundwater: Application of isotope tracers and isotope fractionation studies. Water Resour. Res. 2006, 42, 4349. [Google Scholar] [CrossRef]
- González-Herrera, R.; Martinez-Santibañez, E.; Pacheco-Avila, J.; Cabrera-Sansores, A. Leaching and dilution of fertilizers in the Yucatan karstic aquifer. Environ. Earth Sci. 2014, 72, 2879–2886. [Google Scholar] [CrossRef]
- Kim, H.; Jo, B.Y.; Kim, H.S. Effect of different concentrations and ratios of ammonium, nitrate, and phosphate on growth of the blue-green alga (cyanobacterium) Microcystis aeruginosa isolated from the Nakdong River, Korea. Algae 2017, 32, 275–284. [Google Scholar] [CrossRef]
- Melaram, R.; Lopez-Dueñas, B. Detection and Occurrence of Microcystins and Nodularins in Lake Manatee and Lake Washington-Two Floridian Drinking Water Systems. Front. Water 2022, 4, 899572. [Google Scholar] [CrossRef]
- Kramer, B.J.; Davis, T.W.; Meyer, K.A.; Rosen, B.H.; Goleski, J.A.; Dick, G.J.; Gobler, C.J. Nitrogen limitation, toxin synthesis potential, and toxicity of cyanobacterial populations in Lake Okeechobee and the St. Lucie River Estuary, Florida, during the 2016 state of emergency event. PLoS ONE 2018, 13, e0196278. [Google Scholar] [CrossRef]
- Vasconcelos, V.; Martins, A.; Vale, M.; Antunes, A.; Azevedo, J.; Welker, M.; Lopez, O.; Montejano, G. First report on the occurrence of microcystins in planktonic cyanobacteria from Central Mexico. Toxicon 2010, 56, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Alillo-Sánchez, J.L.; Gaytán-Herrera, M.L.; Martínez-Almeida, V.M.; Ramírez-García, P. Microcystin-LR equivalents and their correlation with Anabaena spp. in the main reservoir of a hydraulic system of Central Mexico. Inland Waters 2014, 4, 327–336. [Google Scholar] [CrossRef]
- Zamora-Barrios, C.A.; Nandini, S.; Sarma, S.S.S. Effect of crude extracts from cyanobacterial blooms in Lake Texcoco (Mexico) on the population growth of Brachionus calyciflorus (Rotifera). Toxicon 2017, 139, 45–53. [Google Scholar] [CrossRef]
- Tooming-Klunderud, A.; Mikalsen, B.; Kristensen, T.; Jakobsen, K.S. The mosaic structure of the mcyABC operon in Microcystis. Microbiology 2008, 154, 1886–1899. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Choi, J.; Fatka, M.; Swanner, E.; Ikuma, K.; Liang, X.; Howe, A. Improved detection of mcyA genes and their phylogenetic origins in harmful algal blooms. Water Res. 2020, 176, 115730. [Google Scholar] [CrossRef]
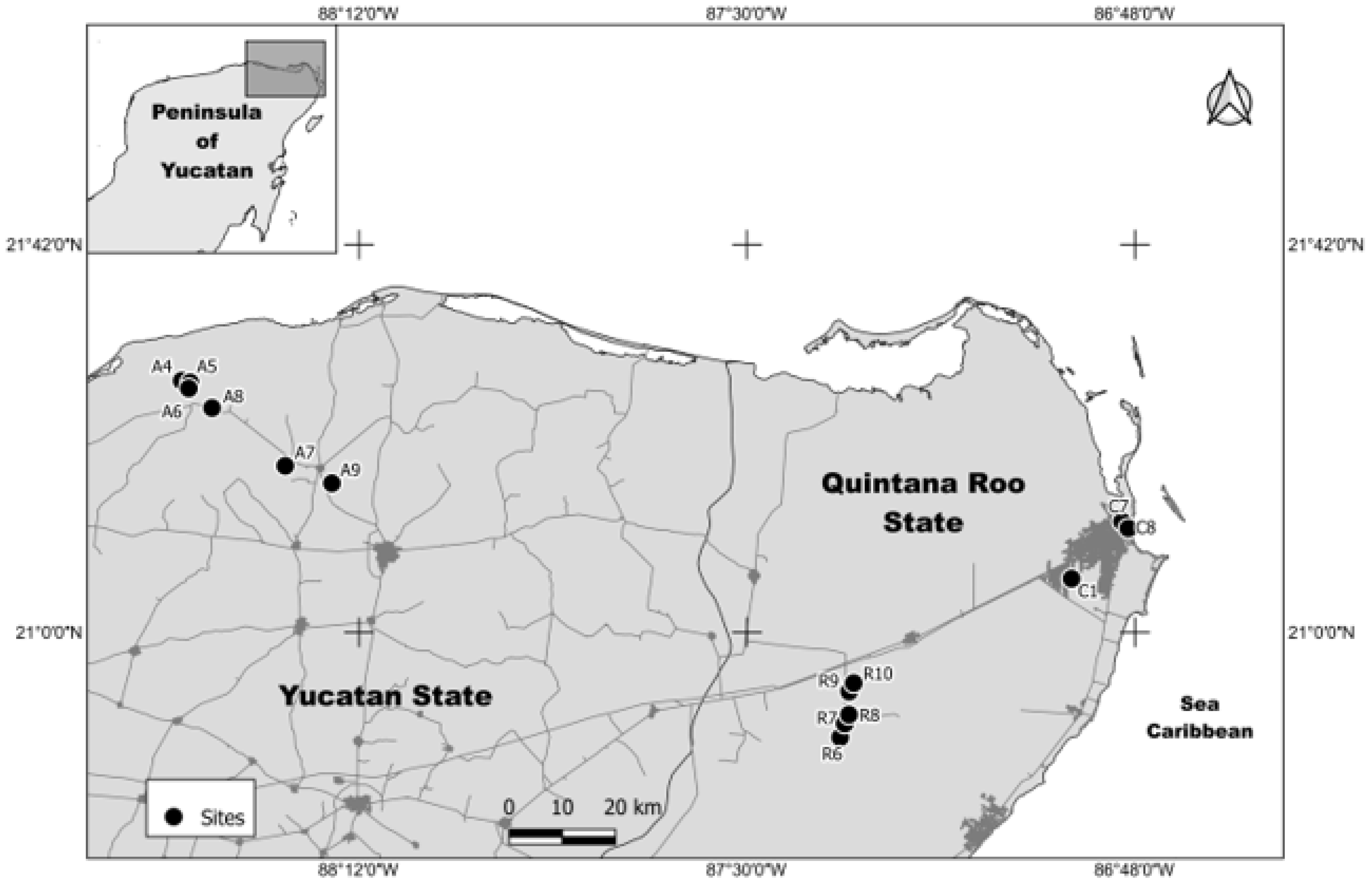
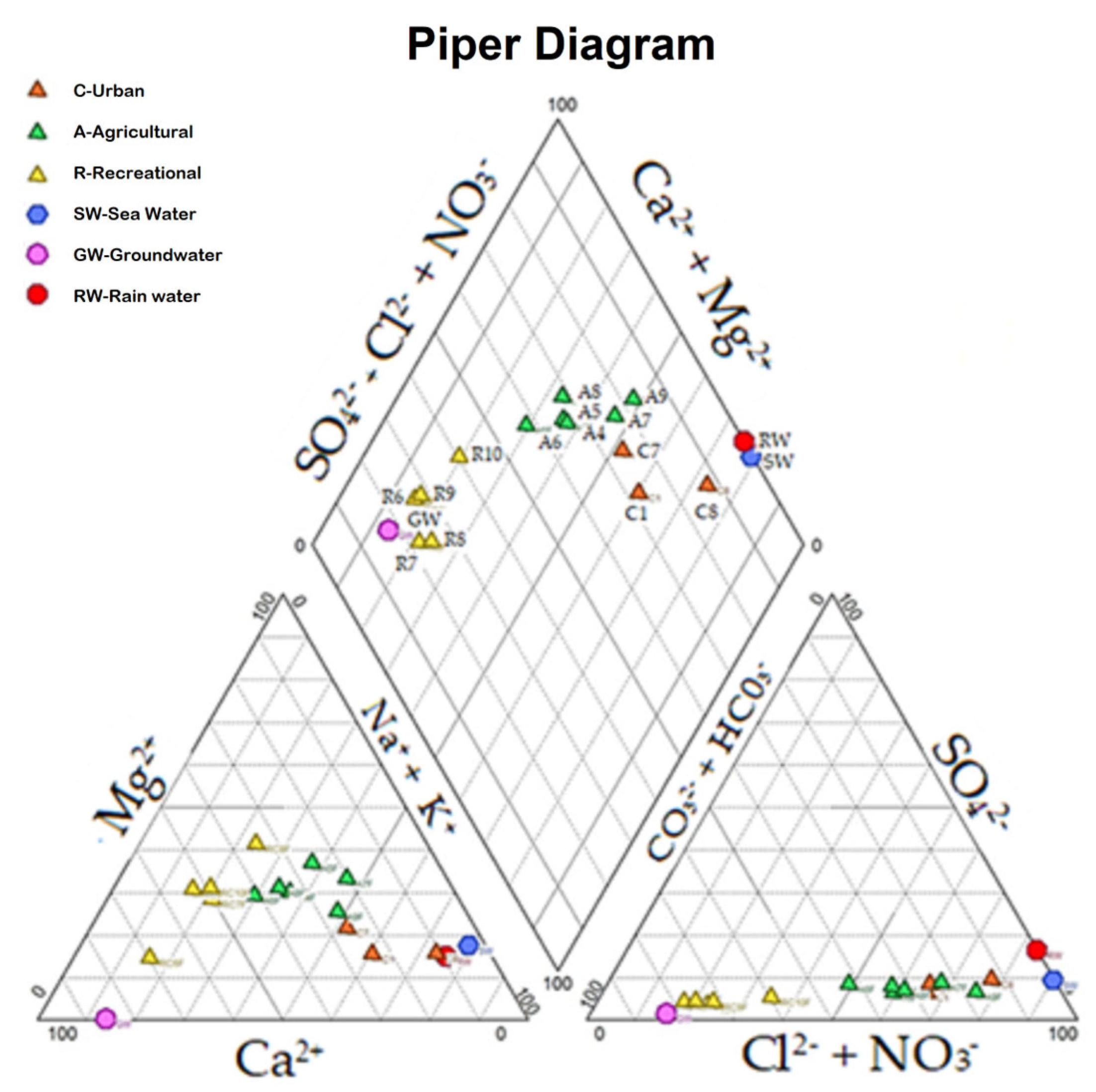
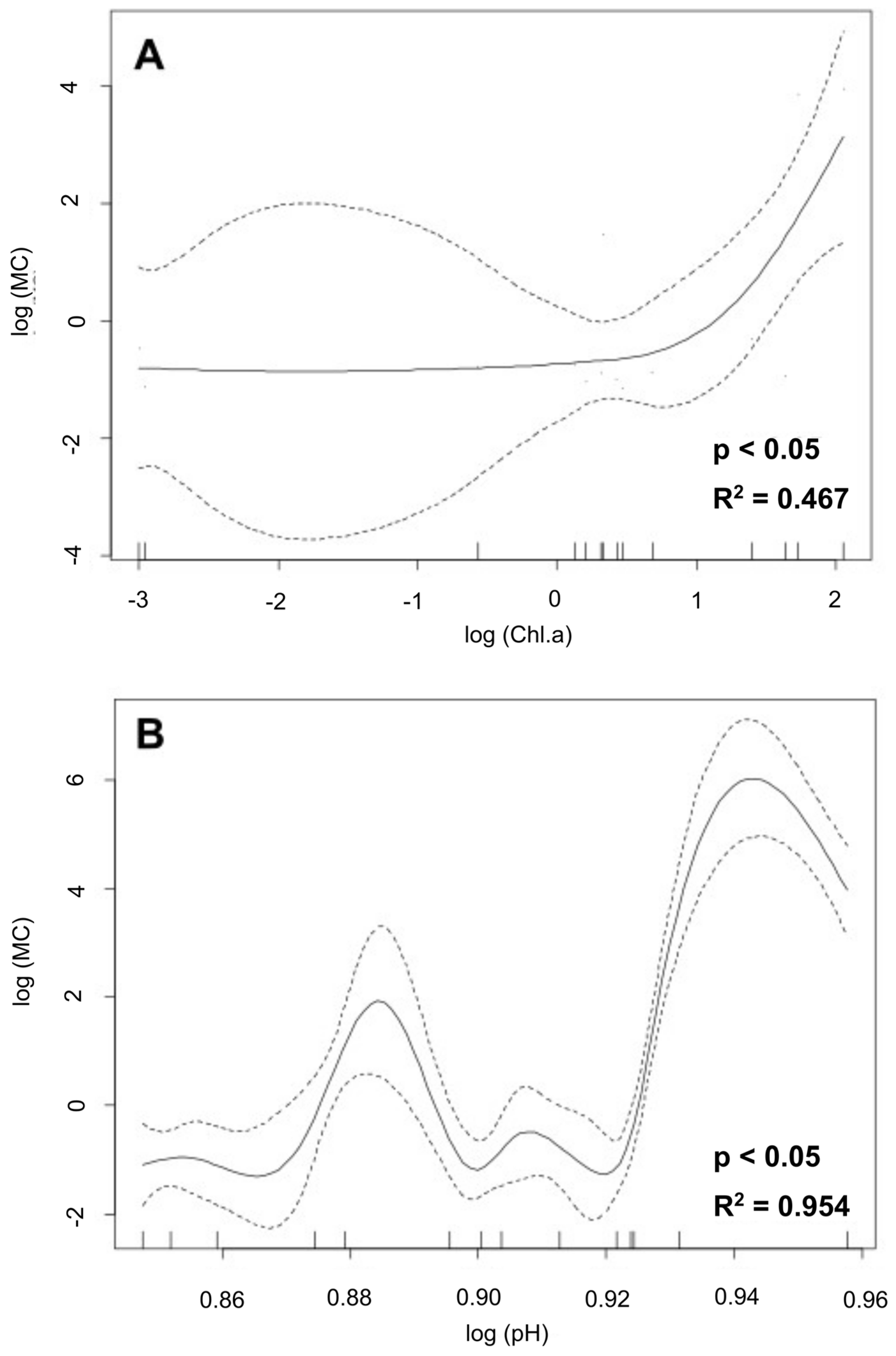
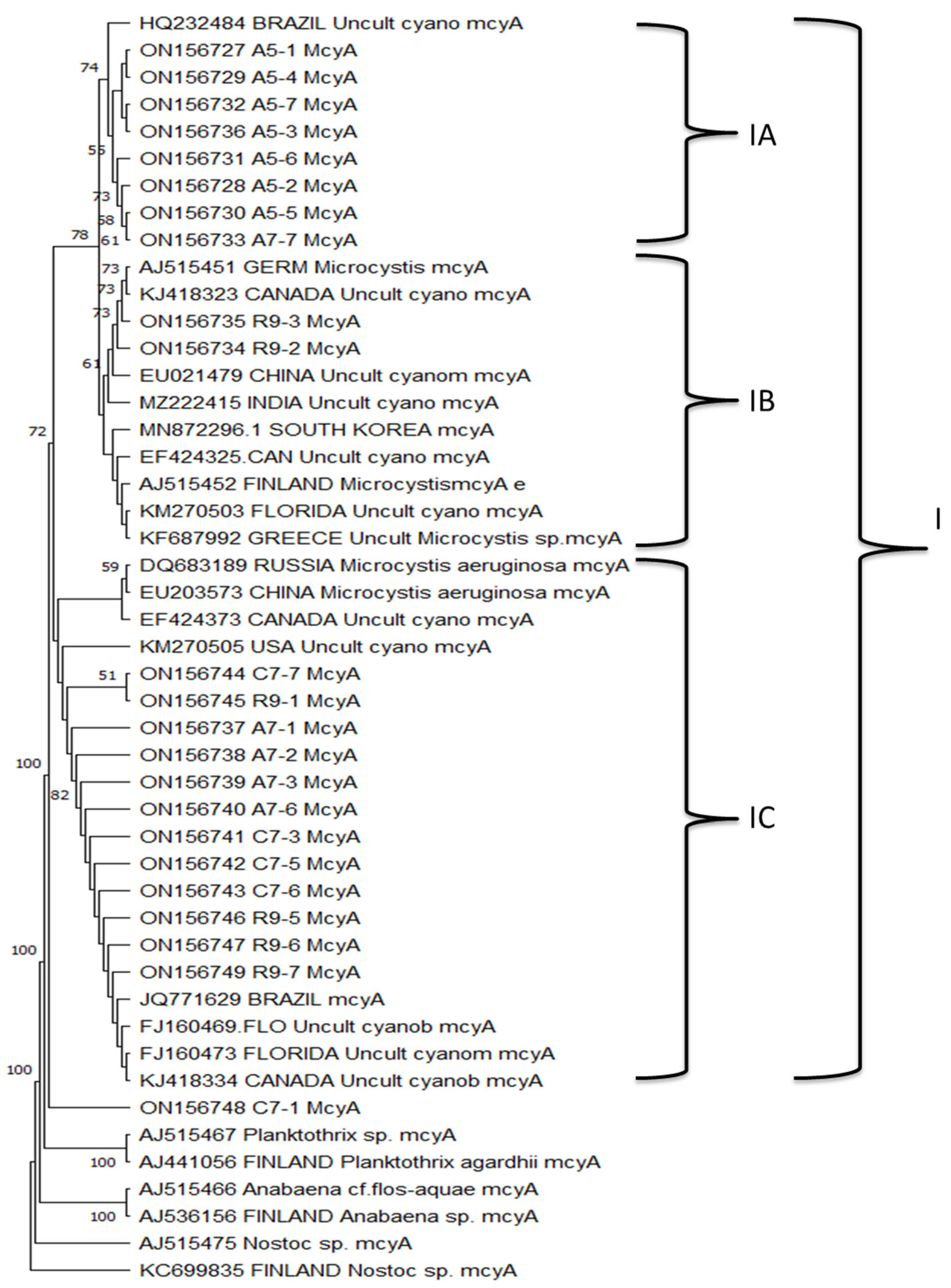
| Site | pH | T °C | EC µS/cm | DO mg/L | Chl-a mg/m3 | MC µg/L |
|---|---|---|---|---|---|---|
| C1 | 8.18 | 30.40 | 710.00 | 6.90 | 0.27 | 0.51 |
| C7 | 8.54 | 27.60 | 1205.00 | 7.90 | 53.46 | >5.00 |
| C8 | 7.10 | 27.40 | 5050.00 | 1.10 | 4.81 | 0.41 |
| Average of each variable in urban sites | 7.94 | 28.47 | 2321.67 | 5.30 | 19.51 | 0.46 |
| A4 | 7.23 | 25.90 | 1231.00 | 1.80 | n.d. | 0.15 |
| A5 | 9.07 | 25.20 | 824.00 | 5.00 | 112.86 | >5.00 |
| A6 | 7.49 | 26.90 | 1010.00 | 1.50 | n.d. | 0.81 |
| A7 | 7.57 | 26.90 | 1372.00 | 9.10 | 2.14 | 2.77 |
| A8 | 8.40 | 28.80 | 1058.00 | 15.70 | 25.06 | 0.98 |
| A9 | 7.04 | 27.90 | 1700.00 | 1.10 | 2.94 | 0.14 |
| Average of each variable in agricultural sites | 7.80 | 26.93 | 1199.17 | 5.70 | 23.83 | 0.97 |
| R6 | 8.01 | 26.30 | 460.00 | 6.90 | 43.44 | 0.34 |
| R7 | 7.86 | 26.40 | 648.00 | 13.00 | 2.67 | 0.28 |
| R8 | 7.95 | 27.20 | 561.00 | 9.10 | 1.34 | 0.54 |
| R9 | 8.35 | 28.40 | 531.00 | 9.00 | 2.06 | 0.39 |
| R10 | 8.39 | 27.40 | 459.00 | 8.40 | 1.60 | 0.25 |
| Average of each variable in recreational sites | 8.11 | 27.14 | 531.80 | 9.28 | 10.22 | 0.36 |
| RDA1 | PC1 | |
|---|---|---|
| Eigenvalue | 2.4337 | 0.7521 |
| Proportion Explained | 0.7639 | 0.2361 |
| Cumulative Proportion | 0.7639 | 1 |
| Site | N-NH4+ mg/L | S.D. | N-NO2− mg/L | S.D. | N-NO3− mg/L | S.D. | P-PO43− mg/L | S.D. |
|---|---|---|---|---|---|---|---|---|
| C1 | 0.028 | 0.001 | 0.024 | 0.001 | 0.025 * | - | 0.025 * | - |
| C7 | 0.14 | 0.002 | 0.0025 * | - | 0.025 * | - | 0.025 * | - |
| C8 | 0.731 | 0.001 | 0.0025 * | - | 0.025 * | - | 0.025 * | - |
| Average of each variable in urban sites | 0.30 | 0.01 | - | 0.03 | - | 0.03 | - | |
| S.E. | 0.18 | 0.01 | 0.00 | 0.00 | ||||
| A4 | 0.0035 * | - | 1.587 | 0.040 | 4.12 | 0.220 | 0.025 * | - |
| A5 | 0.103 | 0.001 | 0.0025 * | - | 0.902 | 0.096 | 0.025 * | - |
| A6 | 1.072 | 0.017 | 0.0025 * | - | 0.776 | 0.027 | 2.5 | 0.053 |
| A7 | 0.0035 * | - | 0.019 | 0.000 | 18.3 | 2.290 | 1.04 | 0.010 |
| A8 | 0.0035 * | - | 0.0025 * | - | 0.824 | 0.096 | 1.09 | 0.005 |
| A9 | 0.35 | 0.033 | 0.185 | 0.02 | 6.41 | 0.137 | 0.025 * | - |
| Average of each variable in agricultural sites | 0.26 | 0.30 | 5.22 | 0.78 | ||||
| S.E. | 0.16 | 0.24 | 2.54 | 0.37 | ||||
| R6 | 0.11 | 0.008 | 0.0025 * | - | 0.669 | 0.041 | 0.025 * | - |
| R7 | 0.0035 * | - | 0.03 | 0.000 | 1.78 | 0.096 | 0.38 | 0.001 |
| R8 | 0.0035 * | - | 0.0025 * | - | 0.377 | 0.013 | 0.62 | 0.016 |
| R9 | 0.0035 * | - | 0.0025 * | - | 0.64 | 0.027 | 1.31 | 0.022 |
| R10 | 0.0035 * | - | 0.0025 * | - | 0.474 | 0.013 | 0.025 * | - |
| Average of each variable in recreational sites | 0.02 | 0.01 | 0.79 | 0.47 | ||||
| S.E. | 0.02 | 0.005 | 0.23 | 0.21 |
| Sequence ID Accession Num | GenBank Description | Query Coverage | Percentage of Nucleotide Identity | Accession Number and Location from Where the Sequence was Isolated |
|---|---|---|---|---|
| A7-1 ON156737 | Microcystis aeruginosa NPLJ-4 peptide synthetase (mcyA) gene, partial cds | 97% | 97% | JQ771629.1 BRAZIL |
| A7-2 ON156738 | 97% | 99% | ||
| A7-3 ON156739 | ||||
| A7-6 ON156740 | ||||
| C7-3 ON156741 | ||||
| |C7-5 ON156742 | ||||
| C7-6 ON156743 | ||||
| R9-5 ON156746 | ||||
| R9-6 ON156747 | ||||
| C7-7 ON156744 | 99% | 97% | ||
| A5-1 ON156727 | Microcystis aeruginosa NRERC-214 (mcyA) gene, partial cds | 95% | 95% | MN872296.1 South Korea |
| A5-2 ON156728 | Microcystis sp. 199 partial mcyA gene | 95% | 99% | AJ515452.1 Finland |
| A7-7 ON156733 | ||||
| A5-5 ON156730 | 98% | |||
| A5-4 ON156729 | 98% | 98% | ||
| R9-2 ON156734 | Microcystis sp. HUB 5.2.4 partial mcyA gene | 95% | 99.70% | AJ515451.1 Germany |
| R9-3 ON156735 | ||||
| A5-6 ON156731 | Uncultured cyanobacterium clone 01LO-IB7 (mcyA) gene, partial cds | 95% | 98.60% | EF424325.1 Canada |
| R9-1 ON156745 | Uncultured cyanobacterium clone 1 (mcyA) gene, partial cds | 95% | 97.30% | FJ160469 USA Florida |
| R9-7 ON156749 | 89% | 100% | ||
| C7-1 ON156748 | Uncultured cyanobacterium clone 4 (mcyA) gene, partial cds | 95% | 87% | FJ160473 USA Florida |
| A5-3 ON156736 | Uncultured cyanobacterium clone 6 protein (mcyA) gene, partial cds | 98% | 93.31 | KM270503.1 Canada |
| A5-7 ON156732 | 95% | 94.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ávila-Torres, G.; Rosiles-González, G.; Carrillo-Jovel, V.H.; Acosta-González, G.; Cejudo-Espinosa, E.; Ortega-Camacho, D.; Hernández-Zepeda, C.; Valenzuela, O.A.M. Microcystin Concentrations and Detection of the mcyA Gene in Water Collected from Agricultural, Urban, and Recreational Areas in a Karst Aquifer in the Yucatan Peninsula of Mexico. Microbiol. Res. 2023, 14, 1168-1184. https://doi.org/10.3390/microbiolres14030078
Ávila-Torres G, Rosiles-González G, Carrillo-Jovel VH, Acosta-González G, Cejudo-Espinosa E, Ortega-Camacho D, Hernández-Zepeda C, Valenzuela OAM. Microcystin Concentrations and Detection of the mcyA Gene in Water Collected from Agricultural, Urban, and Recreational Areas in a Karst Aquifer in the Yucatan Peninsula of Mexico. Microbiology Research. 2023; 14(3):1168-1184. https://doi.org/10.3390/microbiolres14030078
Chicago/Turabian StyleÁvila-Torres, Gerardo, Gabriela Rosiles-González, Victor Hugo Carrillo-Jovel, Gilberto Acosta-González, Eduardo Cejudo-Espinosa, Daniela Ortega-Camacho, Cecilia Hernández-Zepeda, and Oscar Alberto Moreno Valenzuela. 2023. "Microcystin Concentrations and Detection of the mcyA Gene in Water Collected from Agricultural, Urban, and Recreational Areas in a Karst Aquifer in the Yucatan Peninsula of Mexico" Microbiology Research 14, no. 3: 1168-1184. https://doi.org/10.3390/microbiolres14030078
APA StyleÁvila-Torres, G., Rosiles-González, G., Carrillo-Jovel, V. H., Acosta-González, G., Cejudo-Espinosa, E., Ortega-Camacho, D., Hernández-Zepeda, C., & Valenzuela, O. A. M. (2023). Microcystin Concentrations and Detection of the mcyA Gene in Water Collected from Agricultural, Urban, and Recreational Areas in a Karst Aquifer in the Yucatan Peninsula of Mexico. Microbiology Research, 14(3), 1168-1184. https://doi.org/10.3390/microbiolres14030078







