Oral Iron Supplementation—Gastrointestinal Side Effects and the Impact on the Gut Microbiota
Abstract
1. Introduction
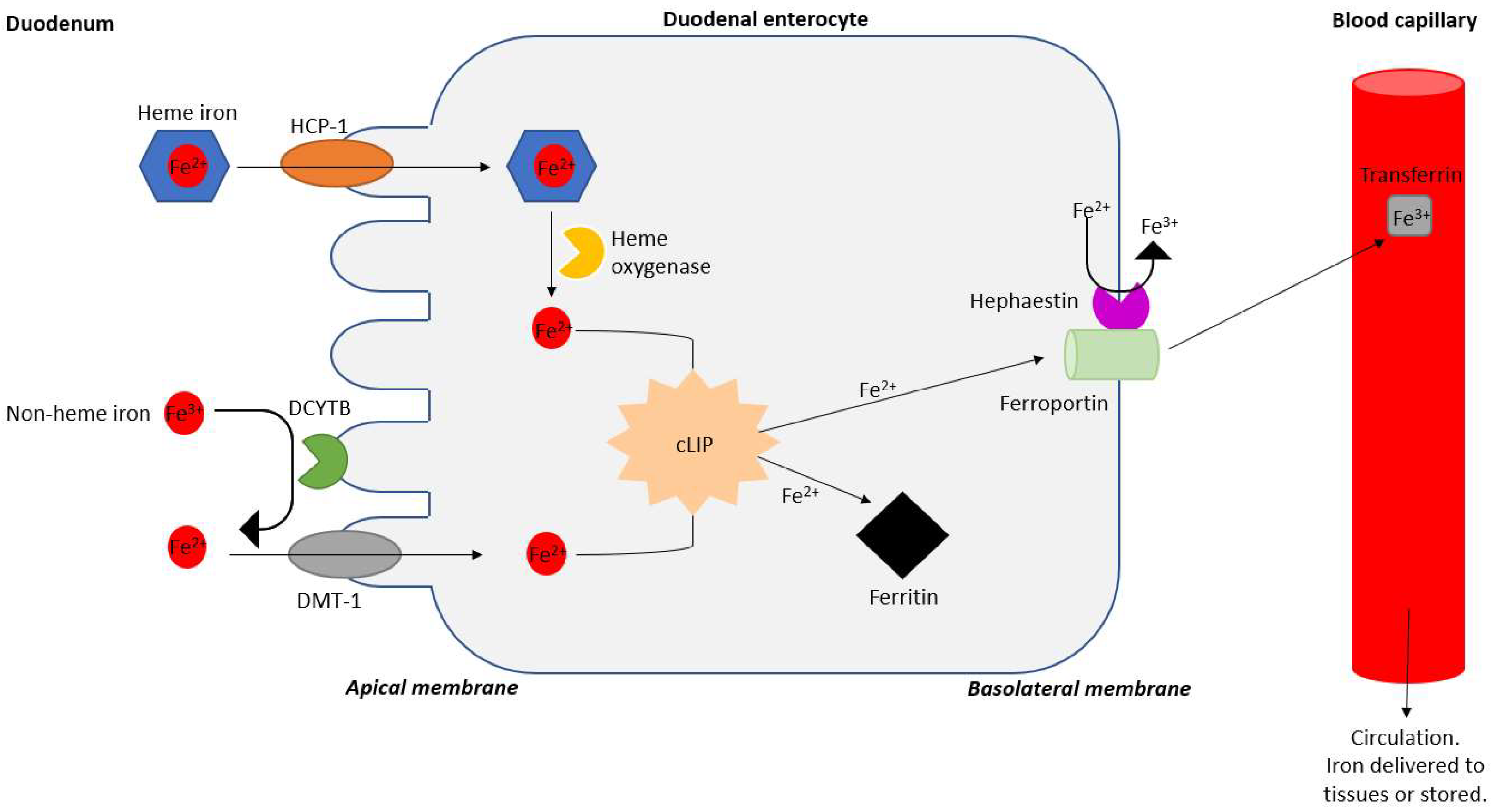
2. Iron Absorption and Dosing of Iron Therapy
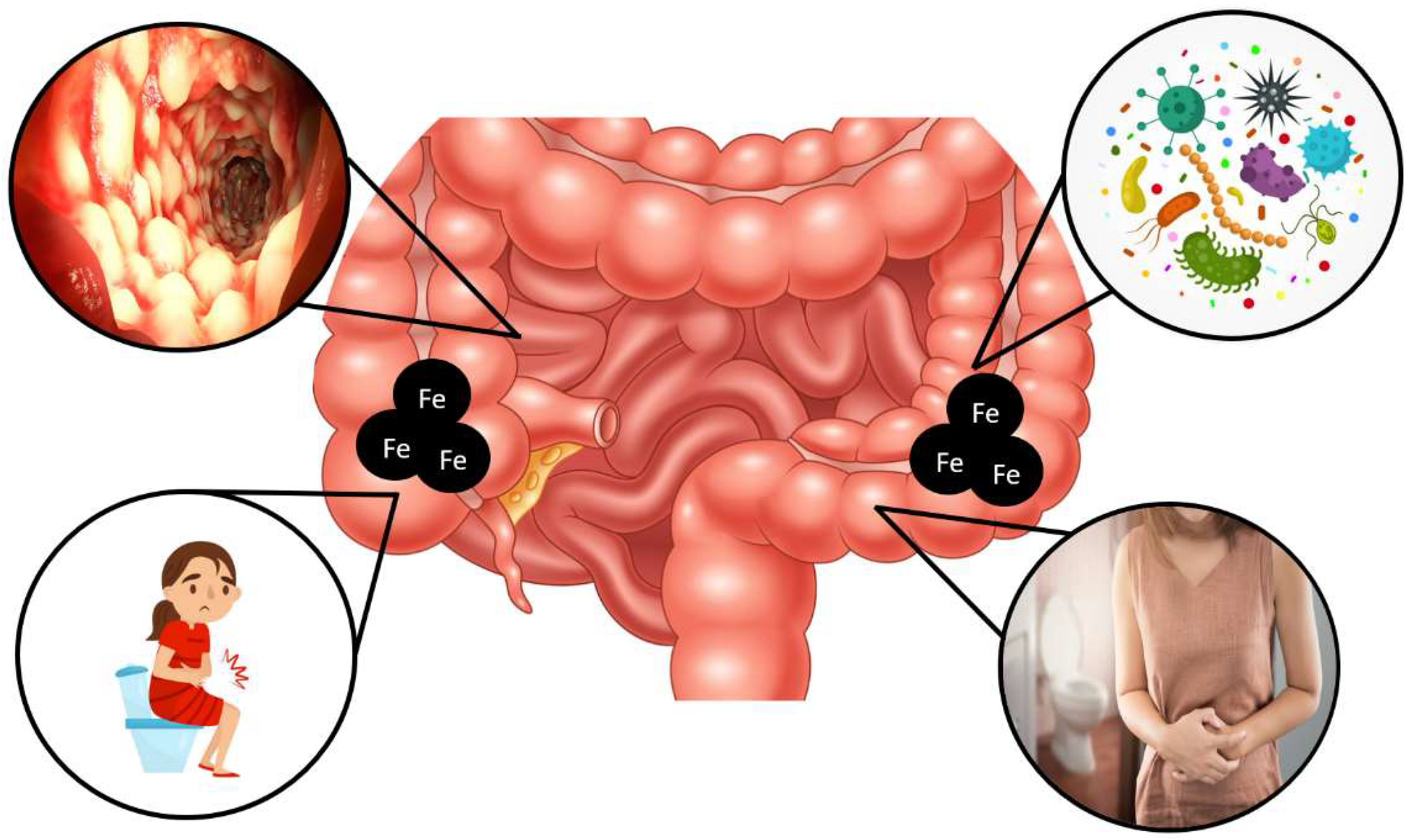
3. Iron and Inflammation
4. Iron and the Gut Microbiota
5. Methanogens, Methanogenesis, and GI Symptoms
6. Methanogenesis and Iron
7. Modulating Methane and Methanogens
8. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Bethell, D.R.; Huang, J. Recombinant Human Lactoferrin Treatment for Global Health Issues: Iron Deficiency and Acute Diarrhea. J. Biometals 2004, 17, 337–342. [Google Scholar] [CrossRef]
- Zimmermann, M.B.; Hurrell, R.F. Nutritional Iron Deficiency. Lancet 2007, 370, 511–520. [Google Scholar] [CrossRef]
- Barton, C.; Cowan, K.; Faulds, J.; Holloway, D.; Johnston, S.; Mason, I.; McMahon, A. Iron Deficiency and Anaemia in Adults: RCN Guidance for Nursing Practice. Available online: https://www.rcn.org.uk/professional-development/publications/pub-007460 (accessed on 4 January 2021).
- Bonovas, S.; Fiorino, G.; Allocca, M.; Lytras, T.; Tsantes, A.; Peyrin-Biroulet, L.; Danese, S. Intravenous Versus Oral Iron for the Treatment of Anemia in Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Medicine 2016, 95, e2308. [Google Scholar] [CrossRef] [PubMed]
- Santiago, P. Ferrous versus Ferric Oral Iron Formulations for the Treatment of Iron Deficiency: A Clinical Overview. Sci. World J. 2012, 2012, 846824. [Google Scholar] [CrossRef]
- Grzywacz, A.; Lubas, A.; Fiedor, P.; Fiedor, M.; Niemczyk, S. Safety and Efficacy of Intravenous Administration of Iron Preparations. Acta Pol. Pharm 2017, 74, 13–24. [Google Scholar] [PubMed]
- Low, M.S.Y.; Speedy, J.; Styles, C.E.; De-Regil, L.M.; Pasricha, S.-R. Daily Iron Supplementation for Improving Anaemia, Iron Status and Health in Menstruating Women. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Wu, T.-W.; Tsai, F.-P. Comparison of the Therapeutic Effects and Side Effects of Oral Iron Supplements in Iron Deficiency Anemia. Drug Res. 2016, 66, 257–261. [Google Scholar] [CrossRef]
- Tolkien, Z.; Stecher, L.; Mander, A.P.; Pereira, D.I.; Powell, J.J. Ferrous Sulfate Supplementation Causes Significant Gastrointestinal Side-Effects in Adults: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0117383. [Google Scholar] [CrossRef]
- Cançado, R.D.; Muñoz, M. Intravenous Iron Therapy: How Far Have We Come? Rev. Bras. Hematol. Hemoter. 2011, 33, 461–469. [Google Scholar] [CrossRef]
- Macdougall, I.C.; Vernon, K. Complement Activation-Related Pseudo-Allergy: A Fresh Look at Hypersensitivity Reactions to Intravenous Iron. Am. J. Nephrol. 2017, 45, 60–62. [Google Scholar] [CrossRef] [PubMed]
- Onken, J.E.; Bregman, D.B.; Harrington, R.A.; Morris, D.; Acs, P.; Akright, B.; Barish, C.; Bhaskar, B.S.; Smith-Nguyen, G.N.; Butcher, A.; et al. A Multicenter, Randomized, Active-Controlled Study to Investigate the Efficacy and Safety of Intravenous Ferric Carboxymaltose in Patients with Iron Deficiency Anemia. Transfusion 2014, 54, 306–315. [Google Scholar] [CrossRef]
- Bhandal, N.; Russell, R. Intravenous versus Oral Iron Therapy for Postpartum Anaemia. BJOG 2006, 113, 1248–1252. [Google Scholar] [CrossRef]
- Stein, J.; Dignass, A.U. Management of Iron Deficiency Anemia in Inflammatory Bowel Disease—A Practical Approach. Ann. Gastroenterol. 2013, 26, 104–113. [Google Scholar] [PubMed]
- Kortman, G.A.; Boleij, A.; Swinkels, D.W.; Tjalsma, H. Iron Availability Increases the Pathogenic Potential of Salmonella Typhimurium and Other Enteric Pathogens at the Intestinal Epithelial Interface. PLoS ONE 2012, 7, e29968. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.W.; Kolber, M.R.; Fedorak, R.N.; van Zanten, S.V. Iron Replacement Therapy in Inflammatory Bowel Disease Patients with Iron Deficiency Anemia: A Systematic Review and Meta-Analysis. J. Crohn’s Colitis 2012, 6, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Aksan, A.; Schoepfer, A.; Juillerat, P.; Vavricka, S.; Bettencourt, M.; Ramirez de Arellano, A.; Gavata, S.; Morin, N.; Valentine, W.; Hunt, B. Iron Formulations for the Treatment of Iron Deficiency Anemia in Patients with Inflammatory Bowel Disease: A Cost-Effectiveness Analysis in Switzerland. Adv. Ther. 2021, 38, 660–677. [Google Scholar] [CrossRef]
- Qi, X.; Zhang, Y.; Guo, H.; Hai, Y.; Luo, Y.; Yue, T. Mechanism and Intervention Measures of Iron Side Effects on the Intestine. Crit. Rev. Food Sci. Nutr. 2019, 60, 2113–2125. [Google Scholar] [CrossRef]
- Przybyszewska, J.; Żekanowska, E. The Role of Hepcidin, Ferroportin, HCP1, and DMT1 Protein in Iron Absorption in the Human Digestive Tract. Prz. Gastroenterol. 2014, 9, 208–213. [Google Scholar] [CrossRef]
- Rishi, G.; Subramaniam, V.N. The Liver in Regulation of Iron Homeostasis. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 313, G157–G165. [Google Scholar] [CrossRef]
- Fuqua, B.K.; Vulpe, C.D.; Anderson, G.J. Intestinal Iron Absorption. J. Trace Elem. Med. Biol. Organ Soc. Miner. Trace Elem. 2012, 26, 115–119. [Google Scholar] [CrossRef]
- Deschemin, J.-C.; Noordine, M.-L.; Remot, A.; Willemetz, A.; Afif, C.; Canonne-Hergaux, F.; Langella, P.; Karim, Z.; Vaulont, S.; Thomas, M.; et al. The Microbiota Shifts the Iron Sensing of Intestinal Cells. FASEB J. 2016, 30, 252–261. [Google Scholar] [CrossRef]
- Schrier, S.L. So You Know How to Treat Iron Deficiency Anemia. Blood 2015, 126, 1971. [Google Scholar] [CrossRef] [PubMed]
- NICE Scenario: Management|Management|Anaemia—Iron Deficienc|CKS|NICE. Available online: https://cks.nice.org.uk/topics/anaemia-iron-deficiency/management/management/ (accessed on 11 January 2021).
- Cook, J.D. Diagnosis and Management of Iron-Deficiency Anaemia. Best Pract. Res. Clin. Haematol. 2005, 18, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Zariwala, M.G.; Somavarapu, S.; Farnaud, S.; Renshaw, D. Comparison Study of Oral Iron Preparations Using a Human Intestinal Model. Sci. Pharm. 2013, 81, 1123–1139. [Google Scholar] [CrossRef]
- Zijp, I.M.; Korver, O.; Tijburg, L.B.M. Effect of Tea and Other Dietary Factors on Iron Absorption. Crit. Rev. Food Sci. Nutr. 2000, 40, 371–398. [Google Scholar] [CrossRef] [PubMed]
- Tempel, M.; Chawla, A.; Messina, C.; Çeliker, M.Y. Effects of Omeprazole on Iron Absorption: Preliminary Study. Turk. J. Hematol. 2013, 30, 307. [Google Scholar] [CrossRef]
- Halksworth, G.; Moseley, L.; Carter, K.; Worwood, M. Iron Absorption from Spatone (a Natural Mineral Water) for Prevention of Iron Deficiency in Pregnancy. Clin. Lab. Haematol. 2003, 25, 227–231. [Google Scholar] [CrossRef] [PubMed][Green Version]
- McKenna, D.; Spence, D.; Haggan, S.E.; McCrum, E.; Dornan, J.C.; Lappin, T.R. A Randomized Trial Investigating an Iron-Rich Natural Mineral Water as a Prophylaxis against Iron Deficiency in Pregnancy. Clin. Lab. Haematol. 2003, 25, 99–103. [Google Scholar] [CrossRef]
- Worwood, M.; Evans, W.D.; Villis, R.J.; Burnett, A.K. Iron Absorption from a Natural Mineral Water (Spatone Iron-Plus). Available online: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1365-2257.1996.tb00732.x (accessed on 10 July 2019).
- Gómez-Ramírez, S.; Brilli, E.; Tarantino, G.; Muñoz, M. Sucrosomial® Iron: A New Generation Iron for Improving Oral Supplementation. Pharmaceuticals 2018, 11, 97. [Google Scholar] [CrossRef]
- Moretti, D.; Goede, J.S.; Zeder, C.; Jiskra, M.; Chatzinakou, V.; Tjalsma, H.; Melse-Boonstra, A.; Brittenham, G.; Swinkels, D.W.; Zimmermann, M.B. Oral Iron Supplements Increase Hepcidin and Decrease Iron Absorption from Daily or Twice-Daily Doses in Iron-Depleted Young Women. Blood 2015, 126, 1981–1989. [Google Scholar] [CrossRef]
- Stoffel, N.U.; Zeder, C.; Brittenham, G.M.; Moretti, D.; Zimmermann, M.B. Iron Absorption from Supplements Is Greater with Alternate Day than with Consecutive Day Dosing in Iron-Deficient Anemic Women. Haematologica 2019, 105, 1232. [Google Scholar] [CrossRef] [PubMed]
- Hawamdeh, H.M.; Rawashdeh, M.; Aughsteen, A.A. Comparison between Once Weekly, Twice Weekly, and Daily Oral Iron Therapy in Jordanian Children Suffering from Iron Deficiency Anemia. Matern. Child Health J. 2013, 17, 368–373. [Google Scholar] [CrossRef]
- Stoffel, N.U.; Cercamondi, C.I.; Brittenham, G.; Zeder, C.; Geurts-Moespot, A.J.; Swinkels, D.W.; Moretti, D.; Zimmermann, M.B. Iron Absorption from Oral Iron Supplements given on Consecutive versus Alternate Days and as Single Morning Doses versus Twice-Daily Split Dosing in Iron-Depleted Women: Two Open-Label, Randomised Controlled Trials. Lancet Haematol. 2017, 4, e524–e533. [Google Scholar] [CrossRef]
- Junqueira Franco, M.M.V.; Nicoletti, C.F.; Nonino, C.B.; Vannucchi, H.; Marchini, J.S. Oxidative Stress after Iron Supplementation in Crohn’s Disease. J. Clin. Case Rep. 2016, 6, 2. [Google Scholar] [CrossRef]
- Rajendran, S.; Bobby, Z.; Habeebullah, S.; Jacob, S.E. Differences in the Response to Iron Supplementation on Oxidative Stress, Inflammation, and Hematological Parameters in Nonanemic and Anemic Pregnant Women. J. Matern. Fetal Neonatal Med. 2020, 1–7. [Google Scholar] [CrossRef]
- Ding, H.; Yu, X.; Chen, L.; Han, J.; Zhao, Y.; Feng, J. Tolerable Upper Intake Level of Iron Damages the Intestine and Alters the Intestinal Flora in Weaned Piglets. Metallomics 2020, 12, 1356–1369. [Google Scholar] [CrossRef]
- Oh, C.-K.; Moon, Y. Dietary and Sentinel Factors Leading to Hemochromatosis. Nutrients 2019, 11, 1047. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.B.; Chassard, C.; Rohner, F.; N’goran, E.K.; Nindjin, C.; Dostal, A.; Utzinger, J.; Ghattas, H.; Lacroix, C.; Hurrell, R.F. The Effects of Iron Fortification on the Gut Microbiota in African Children: A Randomized Controlled Trial in Cote d’Ivoire. Am. J. Clin. Nutr. 2010, 92, 1406–1415. [Google Scholar] [CrossRef] [PubMed]
- Jaeggi, T.; Kortman, G.A.M.; Moretti, D.; Chassard, C.; Holding, P.; Dostal, A.; Boekhorst, J.; Timmerman, H.M.; Swinkels, D.W.; Tjalsma, H.; et al. Iron Fortification Adversely Affects the Gut Microbiome, Increases Pathogen Abundance and Induces Intestinal Inflammation in Kenyan Infants. Gut 2015, 64, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, B.; Li, H. Gut Microbiota and Iron: The Crucial Actors in Health and Disease. Pharmaceuticals 2018, 11, 98. [Google Scholar] [CrossRef] [PubMed]
- Poole, R.K. Advances in Bacterial Pathogen Biology; Elsevier Science & Technology: Kent, UK, 2014; ISBN 978-0-12-800305-3. [Google Scholar]
- Cherayil, B.J. The Role of Iron in the Immune Response to Bacterial Infection. Immunol. Res. 2011, 50, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Iatsenko, I.; Marra, A.; Boquete, J.-P.; Peña, J.; Lemaitre, B. Iron Sequestration by Transferrin 1 Mediates Nutritional Immunity in Drosophila Melanogaster. Proc. Natl. Acad. Sci. USA 2020, 117, 7317–7325. [Google Scholar] [CrossRef] [PubMed]
- Mikulic, N.; Uyoga, M.A.; Mwasi, E.; Stoffel, N.U.; Zeder, C.; Karanja, S.; Zimmermann, M.B. Iron Absorption Is Greater from Apo-Lactoferrin and Is Similar Between Holo-Lactoferrin and Ferrous Sulfate: Stable Iron Isotope Studies in Kenyan Infants. J. Nutr. 2020, 150, 3200–3207. [Google Scholar] [CrossRef]
- Kell, D.B.; Heyden, E.L.; Pretorius, E. The Biology of Lactoferrin, an Iron-Binding Protein That Can Help Defend Against Viruses and Bacteria. Front. Immunol. 2020, 11, 1221. [Google Scholar] [CrossRef]
- Drago-Serrano, M.E.; Campos-Rodríguez, R.; Carrero, J.C.; de la Garza, M. Lactoferrin: Balancing Ups and Downs of Inflammation Due to Microbial Infections. Int. J. Mol. Sci. 2017, 18, 501. [Google Scholar] [CrossRef]
- González, A.; Gálvez, N.; Martín, J.; Reyes, F.; Pérez-Victoria, I.; Dominguez-Vera, J.M. Identification of the Key Excreted Molecule by Lactobacillus Fermentum Related to Host Iron Absorption. Food Chem. 2017, 228, 374–380. [Google Scholar] [CrossRef]
- Paganini, D.; Uyoga, M.A.; Kortman, G.A.M.; Cercamondi, C.I.; Moretti, D.; Barth-Jaeggi, T.; Schwab, C.; Boekhorst, J.; Timmerman, H.M.; Lacroix, C.; et al. Prebiotic Galacto-Oligosaccharides Mitigate the Adverse Effects of Iron Fortification on the Gut Microbiome: A Randomised Controlled Study in Kenyan Infants. Gut 2017, 66, 1956–1967. [Google Scholar] [CrossRef] [PubMed]
- Rusu, I.G.; Suharoschi, R.; Vodnar, D.C.; Pop, C.R.; Socaci, S.A.; Vulturar, R.; Istrati, M.; Moroșan, I.; Fărcaș, A.C.; Kerezsi, A.D.; et al. Iron Supplementation Influence on the Gut Microbiota and Probiotic Intake Effect in Iron Deficiency—A Literature-Based Review. Nutrients 2020, 12, 1993. [Google Scholar] [CrossRef]
- Zimmermann, M.B. Global Look at Nutritional and Functional Iron Deficiency in Infancy. Hematol. Am. Soc. Hematol. Educ. Program 2020, 2020, 471–477. [Google Scholar] [CrossRef]
- Attaluri, A.; Jackson, M.; Valestin, J.; Rao, S.S.C. Methanogenic Flora Is Associated with Altered Colonic Transit but Not Stool Characteristics in Constipation without IBS. Am. J. Gastroenterol. 2010, 105, 1407–1411. [Google Scholar] [CrossRef]
- Stephen, A.M.; Wiggins, H.S.; Englyst, H.N.; Cole, T.J.; Wayman, B.J.; Cummings, J.H. The Effect of Age, Sex and Level of Intake of Dietary Fibre from Wheat on Large-Bowel Function in Thirty Healthy Subjects. Br. J. Nutr. 1986, 56, 349–361. [Google Scholar] [CrossRef]
- Pimentel, M.; Lin, H.C.; Enayati, P.; van den Burg, B.; Lee, H.R.; Chen, J.H.; Park, S.; Kong, Y.; Conklin, J. Methane, a Gas Produced by Enteric Bacteria, Slows Intestinal Transit and Augments Small Intestinal Contractile Activity. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 290, G1089–G1095. [Google Scholar] [CrossRef]
- Triantafyllou, K.; Chang, C.; Pimentel, M. Methanogens, Methane and Gastrointestinal Motility. J. Neurogastroenterol. Motil. 2014, 20, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Ojetti, V.; Petruzziello, C.; Migneco, A.; Gnarra, M.; Gasbarrini, A.; Franceschi, F.L. Reuteri in Methane Producer Constipated Patients. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 1702–1708. [Google Scholar]
- Gaci, N.; Borrel, G.; Tottey, W.; O’Toole, P.W.; Brugère, J.F. Archaea and the Human Gut: New Beginning of an Old Story. World J. Gastroenterol. 2014, 20, 16062–16078. [Google Scholar] [CrossRef] [PubMed]
- Nkamga, V.D.; Henrissat, B.; Drancourt, M. Archaea: Essential Inhabitants of the Human Digestive Microbiota. Hum. Microbiome J. 2017, 3, 1–8. [Google Scholar] [CrossRef]
- Bray, M.S.; Wu, J.; Reed, B.C.; Kretz, C.B.; Belli, K.M.; Simister, R.L.; Henny, C.; Stewart, F.J.; DiChristina, T.J.; Brandes, J.A.; et al. Shifting Microbial Communities Sustain Multiyear Iron Reduction and Methanogenesis in Ferruginous Sediment Incubations. Geobiology 2017, 15, 678–689. [Google Scholar] [CrossRef]
- Geng, S.; Song, K.; Li, L.; Xie, F. Improved Algal Sludge Methane Production and Dewaterability by Zerovalent Iron-Assisted Fermentation. Acs Omega 2020, 5, 6146–6152. [Google Scholar] [CrossRef]
- Ruaud, A.; Esquivel-Elizondo, S.; de la Cuesta-Zuluaga, J.; Waters, J.L.; Angenent, L.T.; Youngblut, N.D.; Ley, R.E. Syntrophy via Interspecies H2 Transfer between Christensenella and Methanobrevibacter Underlies Their Global Cooccurrence in the Human Gut. mBio 2020, 11, e03235-19. [Google Scholar] [CrossRef] [PubMed]
- Suri, J.; Kataria, R.; Malik, Z.A.; Parkman, H.P.; Schey, R. Elevated Methane Levels in Small Intestinal Bacterial Overgrowth Suggests Delayed Small Bowel and Colonic Transit. Gastroenterology 2017, 152, S525. [Google Scholar] [CrossRef]
- Pimentel, M.; Kong, Y.; Park, S. IBS Subjects with Methane on Lactulose Breath Test Have Lower Postprandial Serotonin Levels than Subjects with Hydrogen. Dig. Dis. Sci. 2004, 49, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, A.W.; Laughton, S.N.; Wiesner, M.R. Enhanced Biogas Production from Nanoscale Zero Valent Iron-Amended Anaerobic Bioreactors. Environ. Eng. Sci. 2015, 32, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Ünal, B.; Perry, V.R.; Sheth, M.; Gomez-Alvarez, V.; Chin, K.-J.; Nüsslein, K. Trace Elements Affect Methanogenic Activity and Diversity in Enrichments from Subsurface Coal Bed Produced Water. Front. Microbiol. 2012, 3, 175. [Google Scholar] [CrossRef]
- Deere, T.M.; Lessner, F.H.; Duin, E.C.; Lessner, D.J. Building an ancient cofactor: Iron-sulfur cluster biogenesis in methanogenic archaea. 1. In Proceedings of the Diverse Life and its Detection on Different Worlds, Mesa, AZ, USA, 24–28 April 2017; p. 3507. [Google Scholar]
- Deere, T.M.; Prakash, D.; Lessner, F.H.; Duin, E.C.; Lessner, D.J. Methanosarcina Acetivorans Contains a Functional ISC System for Iron-Sulfur Cluster Biogenesis. BMC Microbiol. 2020, 20, 323. [Google Scholar] [CrossRef]
- Daniels, L.; Belay, N.; Rajagopal, B.S.; Weimer, P.J. Bacterial Methanogenesis and Growth from CO2 with Elemental Iron as the Sole Source of Electrons. Science 1987, 237, 509–511. [Google Scholar] [CrossRef] [PubMed]
- Weiss, D.S.; Thauer, R.K. Methanogenesis and the Unity of Biochemistry. Cell 1993, 72, 819–822. [Google Scholar] [CrossRef]
- Sivan, O.; Shusta, S.S.; Valentine, D.L. Methanogens Rapidly Transition from Methane Production to Iron Reduction. Geobiology 2016, 14, 190–203. [Google Scholar] [CrossRef]
- Chou, H.-H.; Huang, J.-S.; Chen, W.-G.; Ohara, R. Competitive Reaction Kinetics of Sulfate-Reducing Bacteria and Methanogenic Bacteria in Anaerobic Filters. Bioresour. Technol. 2008, 99, 8061–8067. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Zhang, L.; Batstone, D.J.; Keller, J.; Yuan, Z. Impact of Iron Salt Dosage to Sewers on Downstream Anaerobic Sludge Digesters: Sulfide Control and Methane Production. J. Environ. Eng. 2013, 139, 594–601. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.; Ni, B.-J. Zero Valent Iron Simultaneously Enhances Methane Production and Sulfate Reduction in Anaerobic Granular Sludge Reactors. Water Res. 2015, 75, 292–300. [Google Scholar] [CrossRef]
- Pimentel, M.; Chang, C.; Chua, K.S.; Mirocha, J.; DiBaise, J.; Rao, S.; Amichai, M. Antibiotic Treatment of Constipation-Predominant Irritable Bowel Syndrome. Dig. Dis. Sci. 2014, 59, 1278–1285. [Google Scholar] [CrossRef]
- Pimentel, M.; Lembo, A.; Chey, W.D.; Zakko, S.; Ringel, Y.; Yu, J.; Mareya, S.M.; Shaw, A.L.; Bortey, E.; Forbes, W.P.; et al. Rifaximin Therapy for Patients with Irritable Bowel Syndrome without Constipation. N. Engl. J. Med. 2011, 364, 22–32. [Google Scholar] [CrossRef]
- Pimentel, M.; Chatterjee, S.; Chow, E.J.; Park, S.; Kong, Y. Neomycin Improves Constipation-Predominant Irritable Bowel Syndrome in a Fashion That Is Dependent on the Presence of Methane Gas: Subanalysis of a Double-Blind Randomized Controlled Study. Dig. Dis. Sci. 2006, 51, 1297–1301. [Google Scholar] [CrossRef]
- Kresser, C.; Pimentel, M. A New Understanding of SIBO and IBS, with Mark Pimentel. Available online: https://chriskresser.com/a-new-understanding-of-sibo-and-ibs-with-mark-pimentel/ (accessed on 20 October 2020).
- Gottlieb, K.; Le, C.; Wacher, V.; Sliman, J.; Cruz, C.; Porter, T.; Carter, S. Selection of a Cut-off for High- and Low-Methane Producers Using a Spot-Methane Breath Test: Results from a Large North American Dataset of Hydrogen, Methane and Carbon Dioxide Measurements in Breath. Gastroenterol. Rep. 2017, 5, 193–199. [Google Scholar] [CrossRef]
- Hubert, S.; Chadwick, A.; Wacher, V.; Coughlin, O.; Kokai-Kun, J.; Bristol, A. Development of a Modified-Release Formulation of Lovastatin Targeted to Intestinal Methanogens Implicated in Irritable Bowel Syndrome With Constipation. J. Pharm. Sci. 2018, 107, 662–671. [Google Scholar] [CrossRef]
- Candyrine, S.C.L.; Mahadzir, M.F.; Garba, S.; Jahromi, M.F.; Ebrahimi, M.; Goh, Y.M.; Samsudin, A.A.; Sazili, A.Q.; Chen, W.L.; Ganesh, S.; et al. Effects of Naturally-Produced Lovastatin on Feed Digestibility, Rumen Fermentation, Microbiota and Methane Emissions in Goats over a 12-Week Treatment Period. PLoS ONE 2018, 13, e0199840. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, M.; Dubey, C.; Tulasi, V.; Kislai, P.; Manohar, N. Investigation of Lovastatin, the Anti-Hypercholesterolemia Drug Molecule from Three Oyster Mushroom Species. Int. J. Biomed. Clin. Sci. 2017, 2, 26–31. [Google Scholar]
- Brown, K.; Scott-Hoy, B.; Jennings, L.W. Response of Irritable Bowel Syndrome with Constipation Patients Administered a Combined Quebracho/Conker Tree/M. Balsamea Willd Extract. World J Gastrointest Pharm. 2016, 7, 463–468. [Google Scholar] [CrossRef]
- Thompson, L.R.; Rowntree, J.E. Invited Review: Methane Sources, Quantification, and Mitigation in Grazing Beef Systems. Appl. Anim. Sci. 2020, 36, 556–573. [Google Scholar] [CrossRef]
- Kirby, E.J. The Seaweed-Eating Sheep that Belch in a Good Way. Available online: https://www.bbc.com/news/stories-50856275 (accessed on 30 March 2020).
- University of Adelaide Study Shows Potential for Reduced Methane from Cows. Available online: https://phys.org/news/2019-07-potential-methane-cows.html (accessed on 30 March 2020).
- Roque, B.M.; Venegas, M.; Kinley, R.D.; de Nys, R.; Duarte, T.L.; Yang, X.; Kebreab, E. Red Seaweed (Asparagopsis Taxiformis) Supplementation Reduces Enteric Methane by over 80 Percent in Beef Steers. PLoS ONE 2021, 16, e0247820. [Google Scholar] [CrossRef] [PubMed]
- Smith, J. Seaweed as a Tool for Methane Reduction in Livestock|Coral Reef Ecology. Available online: https://scripps.ucsd.edu/labs/coralreefecology/research/seaweed-as-a-tool-for-methane-reduction-in-livestock/ (accessed on 30 March 2020).
- Roque, B.M.; Brooke, C.G.; Ladau, J.; Polley, T.; Marsh, L.J.; Najafi, N.; Pandey, P.; Singh, L.; Kinley, R.; Salwen, J.K.; et al. Effect of the Macroalgae Asparagopsis Taxiformis on Methane Production and Rumen Microbiome Assemblage. Anim. Microbiome 2019, 1, 3. [Google Scholar] [CrossRef] [PubMed]
- Aroniadis, O.C.; Brandt, L.J. Intestinal Microbiota and the Efficacy of Fecal Microbiota Transplantation in Gastrointestinal Disease. Gastroenterol. Hepatol. 2014, 10, 230–237. [Google Scholar]
- Coccorullo, P.; Strisciuglio, C.; Martinelli, M.; Miele, E.; Greco, L.; Staiano, A. Lactobacillus Reuteri (DSM 17938) in Infants with Functional Chronic Constipation: A Double-Blind, Randomized, Placebo-Controlled Study. J. Pediatrics 2010, 157, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Jiang, J.; Tian, F.; Zhao, J.; Zhang, H.; Zhai, Q.; Chen, W. Meta-Analysis of Randomized Controlled Trials of the Effects of Probiotics on Functional Constipation in Adults. Clin. Nutr. 2020, 39, 2960–2969. [Google Scholar] [CrossRef]
- Axling, U.; Önning, G.; Martinsson Niskanen, T.; Larsson, N.; Hansson, S.R.; Hulthén, L. The Effect of Lactiplantibacillus Plantarum 299v Together with a Low Dose of Iron on Iron Status in Healthy Pregnant Women: A Randomized Clinical Trial. Acta Obstet. Gynecol. Scand. 2021. [Google Scholar] [CrossRef] [PubMed]
| Oral Iron Supplement | Dose (mg) | Elemental Iron Dose (mg) |
|---|---|---|
| Ferrous sulphate | 200 | 65 |
| Ferrous fumarate | 200 | 65 |
| Ferrous gluconate | 300 | 35 |
| Increase Absorption | Decrease Absorption |
|---|---|
| Ascorbic acid (Vitamin C) | Antacid medications (e.g., proton pump inhibitors) |
| Meat | Phytates and polyphenols |
| Fish | Calcium |
| Type of Methanogenesis | Substrates | Mechanism |
|---|---|---|
| Hydrogenotrophic | Hydrogen or formate | The substrates can reduce carbon dioxide to produce methane |
| Methylotrophic | Methanol, methyl-sulphides, methylamines | Methyl group of the substrate is converted to methane using specific methyltransferases |
| Acetotrophic | Acetate | Fermentation of acetate or decarboxylation to carbon dioxide followed by reduction |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bloor, S.R.; Schutte, R.; Hobson, A.R. Oral Iron Supplementation—Gastrointestinal Side Effects and the Impact on the Gut Microbiota. Microbiol. Res. 2021, 12, 491-502. https://doi.org/10.3390/microbiolres12020033
Bloor SR, Schutte R, Hobson AR. Oral Iron Supplementation—Gastrointestinal Side Effects and the Impact on the Gut Microbiota. Microbiology Research. 2021; 12(2):491-502. https://doi.org/10.3390/microbiolres12020033
Chicago/Turabian StyleBloor, Sarah R., Rudolph Schutte, and Anthony R. Hobson. 2021. "Oral Iron Supplementation—Gastrointestinal Side Effects and the Impact on the Gut Microbiota" Microbiology Research 12, no. 2: 491-502. https://doi.org/10.3390/microbiolres12020033
APA StyleBloor, S. R., Schutte, R., & Hobson, A. R. (2021). Oral Iron Supplementation—Gastrointestinal Side Effects and the Impact on the Gut Microbiota. Microbiology Research, 12(2), 491-502. https://doi.org/10.3390/microbiolres12020033






