A Review on the Prevalence of Poxvirus Disease in Free-Living and Captive Wild Birds
Abstract
:1. Introduction
2. Host Range
3. Pathology
4. Epidemiology
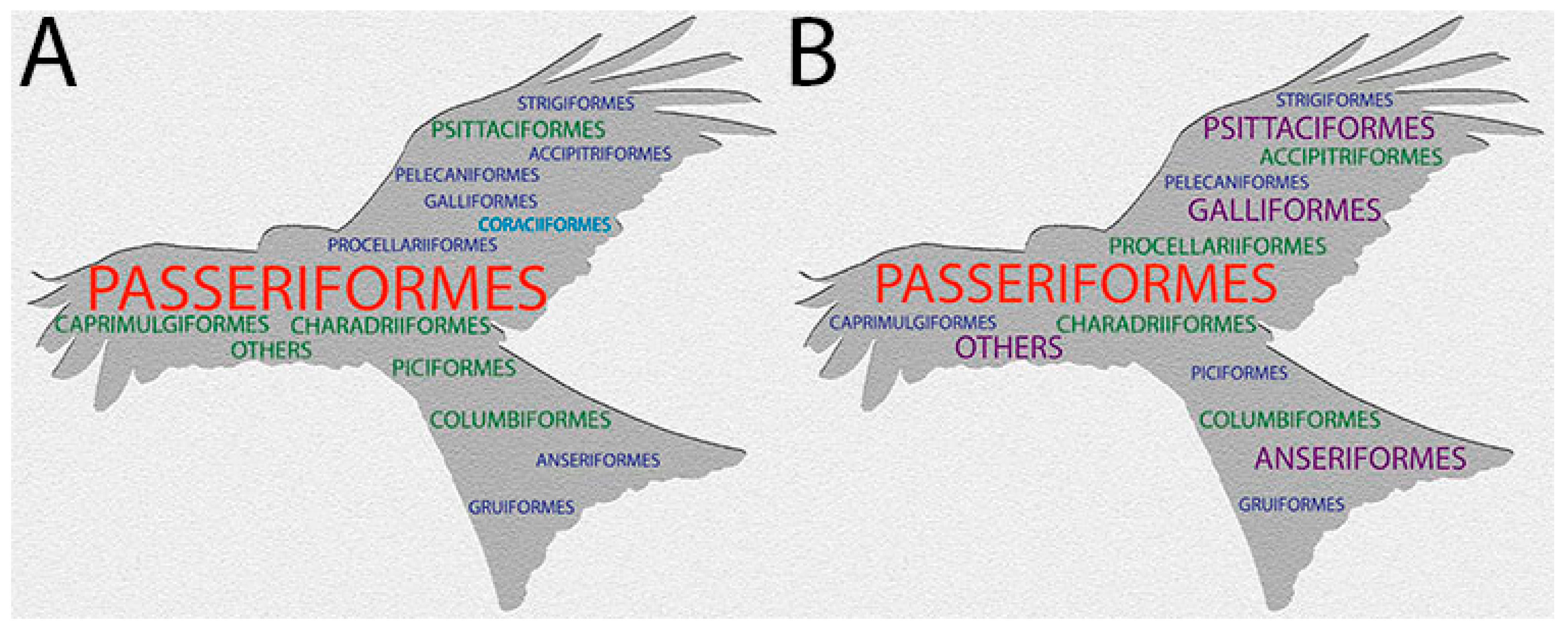
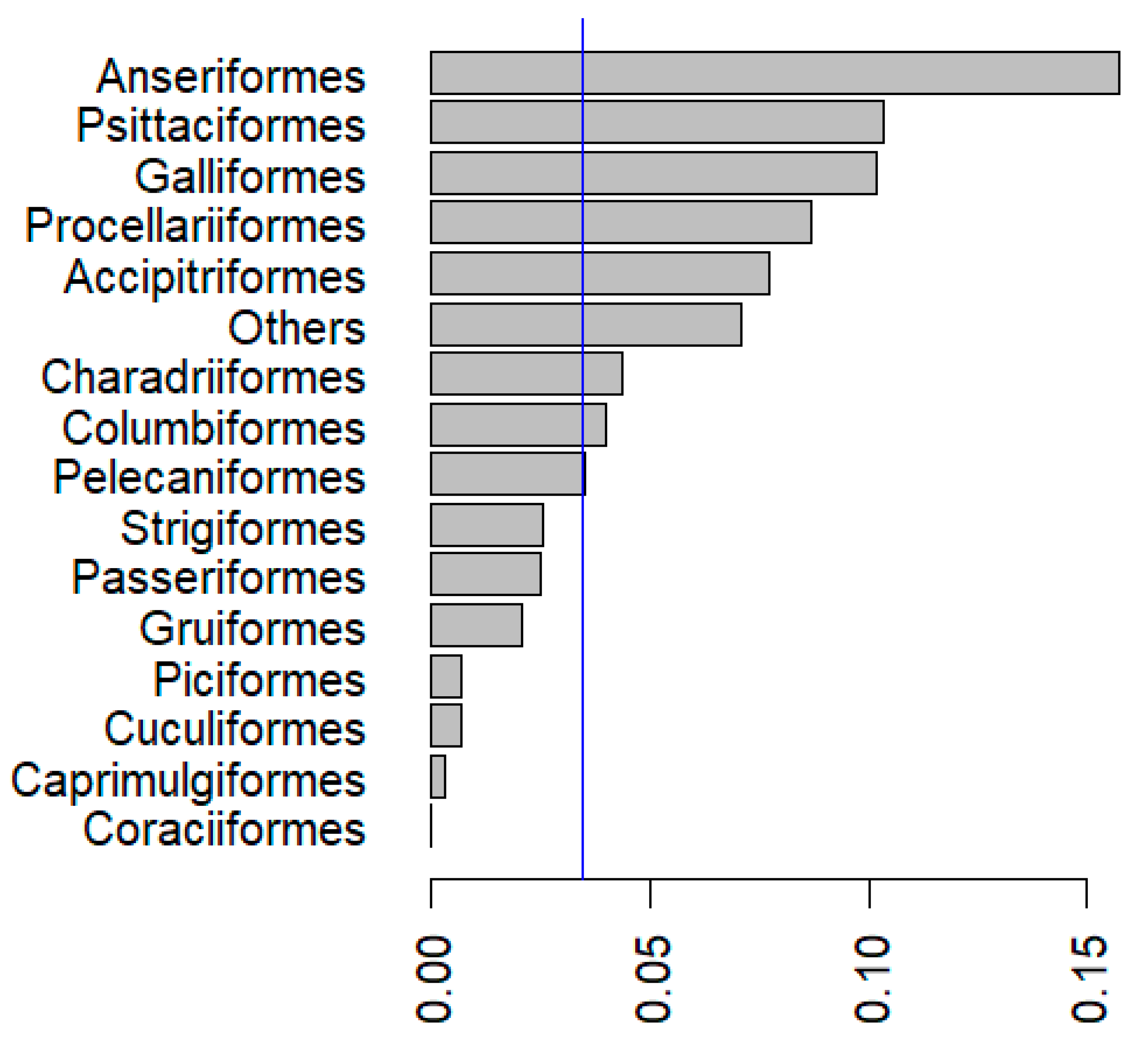
5. Prevalence of Pox Lesions and APV Infections in Wild Birds
| Order | Genera | Species | Common Name | Sample Size | Prevalence (%) | Location (Country) | Period | Reference | Range |
|---|---|---|---|---|---|---|---|---|---|
| Several | Several | 94 | Several | 889 | 0.0 | French Guiana | 2015–2016 | [59] | Continent |
| Several | Several | 3 | Doves and Gambel´s quail | 1792 | 0.0 | Arizona, USA | 1962 | [60] | Continent |
| Passeriformes | Centronyx | 1 | Henslow´s sparrow | 165 | 9.0 | Wisconsin, USA | 2005–2008 | [61] | Continent |
| Spheniscidae | Spheniscus | 1 | Magellanic Penguin | 54,000 | 0.2 | Punta Tombo and Cabo Dos Bahıas, Argentina | 1982–2010 | [31] | Continent |
| Several | Several | 132 | Several | 941 | 0.3 | Ecuador | 2015 | [57] | Continent |
| Passeriformes | Parus | 1 | Great tit | 1819 | 0.82 | Pilis Mountains, Hungary | 2006–2007 | [56] | Continent |
| Several | Several | 37 | Several | 1075 | 1.3 | Czech Republic | 2005 | [55] | Continent |
| Passeriformes | Passer | 1 | House sparrow | 2729 | 3.1 | Central/South, Spain | 2012–2014 | [46] | Continent |
| Passeriformes | Sylvia | 2 | Blackcap | 244 | 3.7 | Czech Republic | 2005 | [55] | Continent |
| Columbiformes | Streptopelia | 1 | Oriental turtle dove | 600 | 4.8 | Republic of Korea | 2011–2013 | [23] | Continent |
| Passeriformes | Bucanetes | 1 | Trumpeter finch | 228 | 0.0 | Canary Islands, Spain | 2002–2003 | [48] | Islands |
| Passeriformes | Passer | 1 | Spanish sparrow | 128 | 0.0 | Canary Islands, Spain | 2002–2003 | [48] | Islands |
| Passeriformes | Sitta | 1 | Nuthatch | 107 | 0.0 | Oxford, UK | 2009–2011 | [39] | Islands |
| Passeriformes | Cyanistes | 1 | Blue tit | 6700 | 0.06 | Oxford, UK | 2009–2011 | [39] | Islands |
| Passeriformes | Periparus | 1 | Coal tit | 1013 | 0.58 | Oxford, UK | 2009–2011 | [39] | Islands |
| Passeriformes | Poecile | 1 | Marsh tit | 579 | 0.85 | Oxford, UK | 2009–2011 | [39] | Islands |
| Passeriformes | Parus | 1 | Great tit | 3661 | 5.01 | Oxford, UK | 2009–2011 | [39] | Islands |
| Passeriformes | Aegithalos | 1 | Long-tailed tit | 42 | 0.0 | Oxford, UK | 2009–2011 | [39] | Islands |
| Phaethontiformes | Phaethon | 1 | White-tailed tropicbird | 81 | 0.3 | Castle Harbour Island Natural Reserve, Bermuda | 1958–1978 | [58] | Islands |
| Several | Several | 4 | Several | 9514 | 1,82 | Trinidad, Trinidad and Tobago | 1964–1971 | [52] | Islands |
| Passeriformes | Several | 11 | Passerines | 3122 | 13.5 | Hawaii, USA | 1977–1980 | [30] | Islands |
| Passeriformes | Hemignathus | 1 | Hawaii Amakihi | 94 | 2 | Hawaii, USA | 2002 | [62] | Islands |
| Procellariiformes | Phoebastria | 1 | Laysan albatross | 70 | 3 | Hawaii, USA | 2003/2007 | [42] | Islands |
| Passeriformes | Several | 9 | Passerines | 484 | 5 | Hawaii, USA | 1995 | [63] | Islands |
| Procellariiformes | Phoebastria | 1 | Waved albatross | 314 | 6 | Galapagos Islands, Ecuador | 2013–2014 | [24] | Islands |
| Passeriformes | Several | 17 (at least) | Darwin’s finch and Galapagos mockingbird | 3607 | 6.3 | Galapagos Islands, Ecuador | 1898–1906 | [54] | Islands |
| Passeriformes | Several | 10 | Darwin´s finch | 898 | 7 | Galapagos Islands, Ecuador | 2000–2002/2004 | [38] | Islands |
| Passeriformes | Anthus | 1 | Berthelot´s pipit | 447 | 8 | Canary Islands, Spain. Madeira and Ilhas Selvagens, Portugal | ND | [64] | Islands |
6. Contribution to Wild Population Declines
7. Outbreaks in Captive Wild Birds
8. Seroprevalence Surveillance
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Beukema, E.L.; Brown, M.P.; Hayball, J.D. The potential role of fowlpox virus in rational vaccine design. Expert Rev. Vaccines 2006, 5, 565–577. [Google Scholar] [CrossRef]
- van Riper, C.; Forrester, D. Avian Pox. In Infectious Diseases of Wild Birds; Thomas, N.J., Hunter, D.B., Eds.; Blackwell Publishing: Ames, IA, USA, 2007; pp. 131–176. [Google Scholar]
- Shearn-Bochsler, V.; Green, D.E.; Converse, K.A.; Docherty, D.E.; Thiel, T.; Geisz, H.N.; Fraser, W.R.; Patterson-Fraser, D.L. Cutaneous and diphtheritic avian poxvirus infection in a nestling Southern Giant Petrel (Macronectes giganteus) from Antarctica. Polar Biol. 2008, 31, 569–573. [Google Scholar] [CrossRef]
- Yeo, G.; Wang, Y.; Chong, S.M.; Humaidi, M.; Lim, X.F.; Mailepessov, D.; Chan, S.; How, C.B.; Lin, Y.N.; Huangfu, T.; et al. Characterization of Fowlpox virus in chickens and bird-biting mosquitoes: A molecular approach to investigating avipoxvirus transmission. J. Gen. Virol. 2019, 100, 838–850. [Google Scholar] [CrossRef] [PubMed]
- Kligler, I.J.; Aschner, M. Demonstration of presence of fowl pox virus in wild caught mosquitoes (Culex Pipiens). Proc. Soc. Exp. Biol. Med. 1931, 28, 463–465. [Google Scholar] [CrossRef]
- Thi, C.; Huong, T.; Murano, T.; Uno, Y.; Usui, T.; Yamaguchi, T. Avian pathology molecular detection of avian pathogens in poultry red mite (Dermanyssus gallinae) collected in chicken farms. J. Vet. Med. Sci. 2014, 14, 0253. [Google Scholar] [CrossRef]
- Walker, P.J.; Siddell, S.G.; Lefkowitz, E.J.; Mushegian, A.R.; Adriaenssens, E.M.; Dempsey, D.M.; Dutilh, B.E.; Harrach, B.; Harrison, R.L.; Hendrickson, R.C.; et al. Changes to virus taxonomy and the Statutes ratified by the International Committee on Taxonomy of Viruses (2020). Arch. Virol. 2020, 165, 2737–2748. [Google Scholar] [CrossRef]
- Adams, M.J.; Lefkowitz, E.J.; King, A.M.Q.; Harrach, B.; Harrison, R.L.; Knowles, N.J.; Kropinski, A.M.; Krupovic, M.; Kuhn, J.H.; Mushegian, A.R.; et al. Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2016). Arch. Virol. 2016, 161, 2921–2949. [Google Scholar] [CrossRef] [Green Version]
- MacDonald, A.M.; Gibson, D.J.; Barta, J.R.; Poulson, R.; Brown, J.D.; Allison, A.B.; Nemeth, N.M. Bayesian phylogenetic analysis of avipoxviruses from North American wild birds demonstrates new insights into host specificity and interspecies transmission. Avian Dis. 2019, 63, 427–432. [Google Scholar] [CrossRef]
- Lüschow, D.; Hoffmann, T.; Hafez, H.M. Differentiation of avian poxvirus strains on the basis of nucleotide sequences of 4b gene fragment. Avian Dis. 2004, 48, 453–462. [Google Scholar] [CrossRef]
- Gyuranecz, M.; Foster, J.T.; Dan, A.; Ip, H.S.; Egstad, K.F.; Parker, P.G.; Higashiguchi, J.M.; Skinner, M.A.; Hofle, U.; Kreizinger, Z.; et al. Worldwide phylogenetic relationship of avian poxviruses. J. Virol. 2013, 87, 4938–4951. [Google Scholar] [CrossRef] [Green Version]
- Jarmin, S.; Manvell, R.; Gough, R.E.; Laidlaw, S.M.; Skinner, M.A. Avipoxvirus phylogenetics: Identification of a PCR length polymorphism that discriminates between the two major clades. J. Gen. Virol. 2006. [Google Scholar] [CrossRef] [PubMed]
- Bolte, A.L.; Meurer, J.; Kaleta, E.F. Avian host spectrum of avipoxviruses. Avian Pathol. 1999, 28, 415–432. [Google Scholar] [CrossRef] [Green Version]
- Clements, J.F.; Schulenberg, T.S.; Iliff, M.J.; Billerman, S.M.; Fredericks, T.A.; Sullivan, B.L.; Wood, C.L. The eBird/Clements Checklist of Birds of the World: v2019. Available online: https://www.birds.cornell.edu/clementschecklist/download/ (accessed on 25 March 2021).
- Davidson, W.R.; Kellogg, F.E. An epornitic of avian pox in wild bobwhite quail. J. Wildl. Dis. 1980, 16, 293–298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Docherty, D.E.; Long, R.I.; Flickinger, E.L.; Locke, L.N. Isolation of poxvirus from debilitating cutaneous lesions on four immature grackles (Quiscalus sp.). Avian Dis. 1991, 244–247. [Google Scholar] [CrossRef] [Green Version]
- Lachish, S.; Bonsall, M.B.; Lawson, B.; Cunningham, A.A.; Sheldon, B.C. Individual and population-level impacts of an emerging poxvirus disease in a wild population of great tits. PLoS ONE 2012, 7, e48545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McDonald, S.E.; Lowenstine, L.J.; Ardans, A.A. Avian pox in blue-fronted Amazon parrots. J. Am. Vet. Med. Assoc. 1981, 179, 1218–1222. [Google Scholar]
- Medina, F.M.; Ramírez, G.A.; Hernández, A. Avian pox in white-tailed laurel-pigeons from the Canary Islands. J. Wildl. Dis. 2004, 40, 351–355. [Google Scholar] [CrossRef] [Green Version]
- Fanke, J.; Wibbelt, G.; Krone, O. Mortality factors and diseases in free-ranging Eurasian cranes (Grus grus) in Germany. J. Wildl. Dis. 2011, 47, 627–637. [Google Scholar] [CrossRef]
- Perelman, B.; Gur-Lavie, A.; Samberg, Y. Pox in ostriches. Avian Pathol. 1988, 17, 735–739. [Google Scholar] [CrossRef]
- Donnelly, T.M.; Crane, L.A. An epornitic of avian pox in a research aviary. Avian Dis. 1984, 28, 517–525. [Google Scholar] [CrossRef]
- Lee, H.R.; Koo, B.-S.; Kim, J.-T.; Kim, H.-C.; Kim, M.-S.; Klein, T.A.; Shin, M.-S.; Lee, S.; Jeon, E.-O.; Min, K.-C.; et al. Molecular epidemiology of avian poxvirus in the oriental turtle dove (Streptopelia orientalis) and the biting midge (Culicoides arakawae) in the Republic of Korea. J. Wildl. Dis. 2017, 53, 749–760. [Google Scholar] [CrossRef]
- Tompkins, E.M.; Anderson, D.J.; Pabilonia, K.L.; Huyvaert, K.P. Avian pox discovered in the critically endangered waved albatross (Phoebastria irrorata) from the Galápagos Islands, Ecuador. J. Wildl. Dis. 2017, 53, 891–895. [Google Scholar] [CrossRef]
- Tripathy, D.N.; Reed, W.M. Pox. In Diseases of Poultry; John Wiley & Sons: Halbrook, NJ, USA, 2020; pp. 364–381. [Google Scholar] [CrossRef]
- Weli, S.C.; Tryland, M. Avipoxviruses: Infection biology and their use as vaccine vectors. Virol. J. 2011, 8, 49. [Google Scholar] [CrossRef] [Green Version]
- Paulo, S.; Catroxo, M.M. Identification of poxvirus under transmission electron microscopy during outbreak period in wild birds, in São Paulo, Brazil. Int. J. Morphol 2009, 27, 577–585. [Google Scholar]
- Reza, K.; Nasrin, A.; Mahmoud, S. Clinical and pathological findings of concurrent poxvirus lesions and aspergillosis infection in canaries. Asian Pac. J. Trop. Biomed. 2013, 3, 182–185. [Google Scholar] [CrossRef] [Green Version]
- Kirmse, P. Host specificity and pathogenicity of pox viruses from wild birds. Bull. Wildl. Dis. Assoc. 1969, 5, 376–386. [Google Scholar] [CrossRef]
- van Riper, C., III; van Riper, S.G.; Hansen, W.R. Epizootiology and effect of avian pox on Hawaiian forest birds. Auk 2002, 119, 929–942. [Google Scholar] [CrossRef]
- Kane, O.J.; Uhart, M.M.; Rago, V.; Pereda, A.J.; Smith, J.R.; Van Buren, A.; Clark, J.A.; Boersma, P.D. Avian pox in Magellanic Penguins (Spheniscus magellanicus). J. Wildl. Dis. 2012, 48, 790–794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilcoxen, T.E.; Horn, D.J.; Hogan, B.M.; Hubble, C.N.; Huber, S.J.; Flamm, J.; Knott, M.; Lundstrom, L.; Salik, F.; Wassenhove, S.J.; et al. Effects of bird-feeding activities on the health of wild birds. Conserv. Physiol. 2015, 3, cov058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giraudeau, M.; Mousel, M.; Earl, S.; McGraw, K. Parasites in the city: Degree of urbanization predicts poxvirus and coccidian infections in house finches (Haemorhous mexicanus). PLoS ONE 2014, 9, e86747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akey, B.L.; Nayar, J.K.; Forrester, D.J. Avian pox in Florida wild turkeys: Culex nigripalpus and Wyeomyia vanduzeei as experimental vectors. J. Wildl. Dis. 1981, 17, 597–599. [Google Scholar] [CrossRef]
- Forrester, D.J. The ecology and epizootiology of avian pox and malaria in wild turkeys. Bull. Soc. Vector Ecol. 1991, 16, 127–148. [Google Scholar]
- Samuel, M.D.; Woodworth, B.L.; Atkinson, C.T.; Hart, P.J.; LaPointe, D.A. The epidemiology of avian pox and interaction with avian malaria in Hawaiian forest birds. Ecol. Monogr. 2018, 88, 621–637. [Google Scholar] [CrossRef]
- VanderWerf, E.A. Distribution and potential impacts of avian poxlike lesions in ‘elepaio at Hakalau Forest National Wildlife Refuge. Stud. Avian Biol. 2001, 22, 247–253. [Google Scholar]
- Kleindorfer, S.; Dudaniec, R.Y. Increasing prevalence of avian poxvirus in Darwin’s finches and its effect on male pairing success. J. Avian Biol. 2006, 37, 69–76. [Google Scholar] [CrossRef]
- Lachish, S.; Lawson, B.; Cunningham, A.A.; Sheldon, B.C. Epidemiology of the emergent disease Paridae pox in an intensively studied wild bird population. PLoS ONE 2012, 7, e38316. [Google Scholar] [CrossRef] [Green Version]
- Buenestado, F.; Gortázar, C.; Millán, J.; Höfle, U.; Villafuerte, R. Descriptive study of an avian pox outbreak in wild red-legged partridges (Alectoris rufa) in Spain. Epidemiol. Infect. 2004, 132, 369–374. [Google Scholar] [CrossRef] [Green Version]
- Le Loc’h, G.; Paul, M.C.; Camus-Bouclainville, C.; Bertagnoli, S. Outbreaks of pox disease due to canarypox-like and fowlpox-like viruses in large-scale houbara bustard captive-breeding programmes, in Morocco and the United Arab Emirates. Transbound. Emerg. Dis. 2016, 63, e187–e196. [Google Scholar] [CrossRef] [PubMed]
- Young, L.C.; VanderWerf, E.A. Prevalence of avian pox virus and effect on the fledging success of Laysan Albatross. J. Field Ornithol. 2008, 79, 93–98. [Google Scholar] [CrossRef]
- VanderWerf, E.A.; Burt, M.D.; Rohrer, J.L.; Mosher, S.M. Distribution and prevalence of mosquito-borne diseases in O’ahu’elepaio. Condor 2006, 108, 770–777. [Google Scholar] [CrossRef]
- Gortázar, C.; Millán, J.; Höfle, U.; Buenestado, F.J.; Villafuerte, R.; Kaleta, E.F. Pathology of avian pox in wild red-legged partridges (Alectoris rufa) in Spain. Ann. N. Y. Acad. Sci. 2002, 969, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.A. Differential prevalence of avian pox in adult and immature California quail. J. Wildl. Dis. 1986, 22, 564–566. [Google Scholar] [CrossRef] [Green Version]
- Ruiz-Martínez, J.; Ferraguti, M.; Figuerola, J.; Martínez-De La Puente, J.; Williams, R.A.J.; Herrera-Dueñas, A.; Aguirre, J.I.; Soriguer, R.; Escudero, C.; Moens, M.A.J.; et al. Prevalence and genetic diversity of avipoxvirus in house sparrows in Spain. PLoS ONE 2016, 11, e0168690. [Google Scholar] [CrossRef] [Green Version]
- Fukui, D.; Nakamura, M.; Yamaguchi, T.; Takenaka, M.; Murakami, M.; Yanai, T.; Fukushi, H.; Yanagida, K.; Bando, G.; Matsuno, K.; et al. An epizootic of emerging novel avian pox in carrion crows (Corvus corone) and large-billed crows (Corvus macrorhynchos) in Japan. J. Wildl. Dis. 2016, 52, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Smits, J.E.; Tella, J.L.; Carrete, M.; Serrano, D.; López, G. An epizootic of avian pox in endemic short-toed larks (Calandrella rufescens) and Berthelot’s pipits (Anthus berthelotti) in the Canary Islands, Spain. Vet. Pathol. 2005, 42, 59–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Curry, R.L.; Grant, P.R. Demography of the cooperatively breeding Galapagos mockingbird, Nesomimus parvulus, in a climatically variable environment. J. Anim. Ecol. 1989, 58, 441–463. [Google Scholar] [CrossRef]
- Zylberberg, M.; Lee, K.A.; Klasing, K.C.; Wikelski, M. Increasing avian pox prevalence varies by species, and with immune function, in Galápagos finches. Biol. Conserv. 2012, 153, 72–79. [Google Scholar] [CrossRef]
- McClure, H.E. Epizootic lesions of house finches in Ventura county, California. J. Field Ornithol. 1989, 60, 421–430. [Google Scholar]
- Tikasingh, E.S.; Worth, C.B.; Spence, L.; Aitken, T.H. Avian pox in birds from Trinidad. J. Wildl. Dis. 1982, 18, 133–139. [Google Scholar] [CrossRef] [Green Version]
- Tarello, W. Prevalence and clinical signs of avipoxvirus infection in falcons from the Middle East. Vet. Dermatol. 2008, 19, 101–104. [Google Scholar] [CrossRef]
- Parker, P.G.; Buckles, E.L.; Farrington, H.; Petren, K.; Whiteman, N.K.; Ricklefs, R.E.; Bollmer, J.L.; Jiménez-Uzcá, G. 110 years of avipoxvirus in the Galapagos Islands. PLoS ONE 2011, 6, e15989. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kulich, P.; Roubalová, E.; Dubská, L.; Sychra, O.; Šmíd, B.; Literák, I. Avipoxvirus in blackcaps (Sylvia atricapilla). Avian Pathol. 2008, 37, 101–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palade, E.; Biró, N.; Dobos-Kovács, M.; Demeter, Z.; Mándoki, M.; Rusvai, M. Poxvirus infection in Hungarian great tits (Parus major): Case report. Acta Vet. Hung. 2008, 56, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Moens, M.A.J.; Pérez-Tris, J.; Milá, B.; Benítez, L. The biological background of a recurrently emerging infectious disease: Prevalence, diversity and host specificity of Avipoxvirus in wild Neotropical birds. J. Avian Biol. 2017, 48, 1041–1046. [Google Scholar] [CrossRef] [Green Version]
- Wingate, D.B.; Barker, I.K. Poxvirus infection of the white-tailed tropicbird (Phaethon lepturus) in Bermuda. J. Wildl. Dis. 1980, 16, 619–622. [Google Scholar] [CrossRef] [Green Version]
- Moens, M.A.J.; Pérez-Tris, J.; Cortey, M.; Benítez, L. Identification of two novel CRESS DNA viruses associated with an Avipoxvirus lesion of a blue-and-gray Tanager (Thraupis episcopus). Infect. Genet. Evol. 2018, 60, 89–96. [Google Scholar] [CrossRef]
- Blankenship, L.H.; Reed, R.E.; Irby, H.D. Pox in mourning doves and Gambel’s quail in Southern Arizona. J. Wildl. Manag. 1966, 30, 253–257. [Google Scholar] [CrossRef]
- Ellison, K.S.; Hofmeister, E.K.; Ribic, C.A.; Sample, D.W. Relatively high prevalence of pox-like lesions in Henslow’s sparrow among nine species of migratory grassland passerines in Wisconsin, USA. J. Wildl. Dis. 2014, 50, 810–816. [Google Scholar] [CrossRef]
- Aruch, S.; Atkinson, C.T.; Savage, A.F.; LaPointe, D.A. Prevalence and distribution of pox-like lesions, avian malaria, and mosquito vectors in Kīpahulu valley, Haleakalā National Park, Hawai’i, USA. J. Wildl. Dis. 2007, 43, 567–575. [Google Scholar] [CrossRef]
- Atkinson, C.T.; Lease, J.K.; Dusek, R.J.; Samuel, M.D. Prevalence of pox-like lesions and malaria in forest bird communities on leeward Mauna Loa Volcano, Hawaii. Condor 2005, 107, 537–546. [Google Scholar] [CrossRef]
- Illera, J.C.; Emerson, B.C.; Richardson, D.S. Genetic characterization, distribution and prevalence of avian pox and avian malaria in the Berthelot’s pipit (Anthus berthelotii) in Macaronesia. Parasitol. Res. 2008, 103, 1435–1443. [Google Scholar] [CrossRef]
- Thiel, T.; Whiteman, N.K.; Tirapé, A.; Baquero, M.I.; Cedeño, V.; Walsh, T.; Uzcátegui, G.J.; Parker, P.G. Characterization of canarypox-like viruses infecting endemic birds in the Galápagos Islands. J. Wildl. Dis. 2005, 41, 342–353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wikelski, M.; Foufopoulos, J.; Vargas, H.; Snell, H. Galápagos birds and diseases: Invasive pathogens as threats for island species. Ecol. Soc. 2004, 9, 5. [Google Scholar] [CrossRef]
- Crawford, J.A.; Oates, R.M.; Helfer, D.H. Avian pox in California quail from Oregon. J. Wildl. Dis. 1979, 15, 447–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crawford, J.A.; Kilbride, K.M. Prevalence of Poxvirus in a Population of California Quail from Oregon, 1975-1987. J. Wildl. Dis. 1988, 24, 718–719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vargas, H. Frequency and effect of pox-like lesions in Galapagos mockingbirds. J. Field Ornithol. 1987, 58, 101–102. [Google Scholar]
- Zylberberg, M.; Lee, K.A.; Klasing, K.C.; Wikelski, M. Variation with land use of immune function and prevalence of avian pox in Galapagos finches. Conserv. Biol. 2013, 27, 103–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenkins, C.D.; Temple, S.A.; van Riper, C., III; Hansen, W.R. Disease-related aspects of conserving the endangered Hawaiian crow. In Disease and Management of Threatened Bird Populations; Cooper, J.E., Ed.; ICBP Technical Publication Series No. 10; International Council for Bird Preservation: Cambridge, UK, 1989; pp. 77–87. [Google Scholar]
- Fuller, T.; Bensch, S.; Müller, I.; Novembre, J.; Pérez-Tris, J.; Ricklefs, R.E.; Smith, T.B.; Waldenström, J. The ecology of emerging infectious diseases in migratory birds: An assessment of the role of climate change and priorities for future research. Ecohealth 2012, 9, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Lawson, B.; Lachish, S.; Colvile, K.M.; Durrant, C.; Peck, K.M.; Toms, M.P.; Sheldon, B.C.; Cunningham, A.A. Emergence of a novel avian pox disease in British tit species. PLoS ONE 2012, 7, e40176. [Google Scholar] [CrossRef] [Green Version]
- Becker, D.J.; Streicker, D.G.; Altizer, S. Linking anthropogenic resources to wildlife-pathogen dynamics: A review and meta-analysis. Ecol. Lett. 2015, 18, 483–495. [Google Scholar] [CrossRef]
- Warner, R.E. The role of introduced diseases in the extinction of the endemic Hawaiian avifauna. Condor 1968, 70, 101–120. [Google Scholar] [CrossRef]
- Tripathy, D.N.; Schnitzlein, W.M.; Morris, P.J.; Janssen, D.L.; Zuba, J.K.; Massey, G.; Atkinson, C.T. Characterization of poxviruses from forest birds in Hawaii. J. Wildl. Dis. 2000, 36, 225–230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munro, G. Outbreak of Avian Pox Virus in Gentoo Penguins in the Falklands; CABI: Oxon, UK, 2006. [Google Scholar]
- Sileo, L.; Sievert, P.R.; Samuel, M.D. Causes of mortality of albatross chicks at Midway Atoll. J. Wildl. Dis. 1990, 26, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Giotis, E.S.; Skinner, M.A. Spotlight on avian pathology: Fowlpox virus. Avian Pathol. 2019, 48, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Al Falluji, M.M.; Tantawi, H.H.; Al-bana, A.; Sheikhly, S. Pox infection among captive peacocks. J. Wildl. Dis. 1979, 15, 597–600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hess, C.; Maegdefrau-Pollan, B.; Bilic, I.; Liebhart, D.; Richter, S.; Mitsch, P.; Hess, M. Outbreak of cutaneous form of poxvirus on a commercial turkey farm caused by the species fowlpox. Avian Dis. 2011, 55, 714–718. [Google Scholar] [CrossRef] [PubMed]
- Samour, J.H.; Kaaden, O.R.; Wernery, U.; Bailey, T.A. An epornitic of avian pox in Houbara Bustards (Chlamydotis undulata macqueenii). J. Vet. Med. Ser. B 1996, 43, 287–292. [Google Scholar] [CrossRef]
- González-Hein, G.; González, C.; Hidalgo, H. Case report: An avian pox outbreak in captive psittacine birds in Chile. J. Exot. Pet Med. 2008, 17, 210–215. [Google Scholar] [CrossRef]
- Esteves, F.C.; Marín, S.Y.; Resende, M.; Silva, A.S.; Coelho, H.L.; Barbosa, M.B.; D’Aparecida, N.S.; de Resende, J.S.; Torres, A.C.D.; Martins, N.R. Avian pox in native captive psittacines, Brazil, 2015. Emerg. Infect. Dis. 2017, 23, 154–156. [Google Scholar] [CrossRef] [Green Version]
- Johnson, B.J.; Castro, A.E. Canary pox causing high mortality in an aviary. J. Am. Vet. Med. Assoc. 1986, 189, 1345–1347. [Google Scholar]
- Shivaprasad, H.L.; Kim, T.; Tripathy, D.; Woolcock, P.R.; Uzal, F. Unusual pathology of canary poxvirus infection associated with high mortality in young and adult breeder canaries (Serinus canaria). Avian Pathol. 2009, 38, 311–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Catania, S.; Carnaccini, S.; Mainenti, M.; Moronato, M.L.; Gobbo, F.; Calogero, T. Isolation of avipoxvirus from tongue of canaries (Serinus canaria) show severe localized proliferative glossitis. Avian Dis. 2017, 61, 531–535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landolt, M.; Kocan, R.M. Transmission of avian pox from starlings to Rothchild’s mynahs. J. Wildl. Dis. 1976, 12, 353–356. [Google Scholar] [CrossRef]
- Hukkanen, R.R.; Richardson, M.; Wingfield, J.C.; Treuting, P.; Brabb, T. Avipox sp. in a colony of gray-crowned rosy finches (Leucosticte tephrocotis). Comp. Med. 2003, 53, 548–552. [Google Scholar] [PubMed]
- Giddens, W.E.; Swango, L.J.; Henderson, J.D.; Lewis, R.A.; Farner, D.S.; Carlos, A.; Dolowy, W.C. Canary pox in sparrows and canaries (Fringillidae) and in weavers (Ploceidae); pathology and host specificity of the virus. Vet. Pathol. 1971, 8, 260–280. [Google Scholar] [CrossRef] [Green Version]
- Tsai, S.S.; Chang, T.C.; Yang, S.F.; Chi, Y.C.; Cher, R.S.; Chien, M.S.; Itakura, C. Unusual lesions associated with avian poxvirus infection in rosy-faced lovebirds (Agapornis roseicollis). Avian Pathol. 1997, 26, 75–82. [Google Scholar] [CrossRef]
- Lierz, M.; Bergmann, V.; Isa, G.; Czerny, C.P.; Lueschow, D.; Mwanzia, J.; Prusas, C.; Hafez, H.H. Avipoxvirus infection in a collection of captive stone curlews (Burhinus oedicnemus). J. Avian Med. Surg. 2007, 21, 50–56. [Google Scholar] [CrossRef]
- Krone, O.; Essbauer, S.; Wibbelt, G.; Isa, G.; Rudloph, M.; Gough, R.E. Avipoxvirus infection in peregrine falcons (Falco peregrinus) from a reintroduction programme in Germany. Vet. Rec. 2004, 154, 110–113. [Google Scholar] [CrossRef]
- Alley, M.R.; Ha, H.J.; Howe, L.; Hale, K.A.; Cash, W. Concurrent avian malaria and avipox virus infection in translocated South Island saddlebacks (Philesturnus carunculatus carunculatus). N. Z. Vet. J. 2010, 58, 218–223. [Google Scholar] [CrossRef]
- Le Loc’h, G.; Souley, M.-N.A.; Bertagnoli, S.; Paul, M.C. Low impact of avian pox on captive-bred houbara bustard breeding performance. Front. Vet. Sci. 2017, 4, 12. [Google Scholar] [CrossRef] [Green Version]
- Tadese, T.; Potter, E.A.; Reed, W.M. Development of a mixed antigen agar gel enzyme assay (AGEA) for the detection of antibodies to poxvirus in chicken and turkey sera. J. Vet. Med. Sci. 2003, 65, 255–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ha, H.J.; Banda, M.; Alley, M.R.; Howe, L.; Gartrell, B.D. The seroprevalence of avipoxvirus and its association with avian malaria (Plasmodium spp.) infection in introduced passerine birds in the southern regions of the North Island of New Zealand. Avian Dis. 2013, 57, 109–115. [Google Scholar] [CrossRef] [PubMed]
| Order | Species | Common Name | Sample Size | Prevalence (%) | Location, Country | Period | Reference | Range |
|---|---|---|---|---|---|---|---|---|
| Galliformes | Alectoris rufa | Red-legged partridge | 70 | 41.4 | Cádiz, Spain | 2000–2001 | [40,44] | Continent |
| Galliformes | Callipepla californica | California quail | 89 | 21 | Oregon, USA | 1982–1987 | [68] | Continent |
| Galliformes | Callipepla californica | California quail | 256 | 26 | Oregon, USA | 1975–1978 | [67] | Continent |
| Galliformes | Colinus virginianus | Bobwhite quail | 2586 | 0–38.9 (mean 12.1) | Florida, Georgia, North Carolina, Tennessee, USA | 1978–1979 | [15] | Continent |
| Galliformes | Callipepla gambelii | Gambel’s Quail | 175 | 11–16 | Arizona, USA | 1963 | [60] | Continent |
| Columbiformes | Columba macroura | Morning Dove | 1795 | 2.1 | Arizona, USA | 1963 | [60] | Continent |
| Passeriformes | Haemorhous mexicanus | House Finch | 9195 | Up to 25 | California, USA | 1977–1987 | [51] | Continent |
| Passeriformes | Haemorhous mexicanus | House Finch | 174 | 5–30 | Arizona, USA | 2011 | [33] | Continent |
| Passeriformes | Corvus corone Corvus macrorhynchos | Carrion crow, Large-billed crow | 2036 | 17.6 | Hokkaido, Japan | 2007–2012 | [47] | Islands |
| Passeriformes | Nesomimus parvulus | Galapagos mockingbird | 417 | 18.9 | Galapagos, Ecuador | 1982–1983 | [49] | Islands |
| Passeriformes | Nesomimus parvulus | Galapagos mockingbird | 117 | 16 | Galapagos, Ecuador | 1979–1980 | [69] | Islands |
| Passeriformes | Anthus berthelotti | Berthelot´s pipit | 139 | 28 | Canary Islands, Spain | 2002–2003 | [48] | Islands |
| Passeriformes | Calandrella rufescens | Short-toed lark | 395 | 50 | Canary Islands, Spain | 2002–2003 | [48] | Islands |
| Passeriformes | Chasiempis sanswichensis | Hawaii ‘elepaio | 97 | Up to 40 | Hawaii, USA | 1994–1997 | [37] | Islands |
| Passeriformes | SEVERAL | Galapagos finches (7 spp.) | 545 | 4–9 | Galapagos, Ecuador | 2008 and 2009 | [70] | Islands |
| Procellariiformes | Phoebastria inmutabilis | Laysan albatross | 92 | Up to 88 | Hawaii, USA | 2004 and 2006 | [42] | Islands |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Williams, R.A.J.; Truchado, D.A.; Benitez, L. A Review on the Prevalence of Poxvirus Disease in Free-Living and Captive Wild Birds. Microbiol. Res. 2021, 12, 403-418. https://doi.org/10.3390/microbiolres12020028
Williams RAJ, Truchado DA, Benitez L. A Review on the Prevalence of Poxvirus Disease in Free-Living and Captive Wild Birds. Microbiology Research. 2021; 12(2):403-418. https://doi.org/10.3390/microbiolres12020028
Chicago/Turabian StyleWilliams, Richard A. J., Daniel A. Truchado, and Laura Benitez. 2021. "A Review on the Prevalence of Poxvirus Disease in Free-Living and Captive Wild Birds" Microbiology Research 12, no. 2: 403-418. https://doi.org/10.3390/microbiolres12020028
APA StyleWilliams, R. A. J., Truchado, D. A., & Benitez, L. (2021). A Review on the Prevalence of Poxvirus Disease in Free-Living and Captive Wild Birds. Microbiology Research, 12(2), 403-418. https://doi.org/10.3390/microbiolres12020028






