1. Introduction
Staphylococcus aureus, though a commensal, has over the years evolved into a very important human pathogen, causing mild to severe infections, including folliculitis and furunculosis, meningitis, septicaemia, pneumonia, endocarditis, and osteomyelitis [
1,
2]. As a commensal, it is predominantly isolated from the anterior nares, but also co-colonizes the nasopharynx with other microbiota, such as
Haemophilus influenzae,
Moraxella catarrhalis, and the predominant nasopharyngeal colonizer—
Streptococcus pneumoniae [
3,
4,
5]. Of these colonizers,
S. aureus and
S. pneumoniae, which antagonize each other [
6,
7], appear to be the most clinically significant, owing to their greater invasive disease-causing capacity [
8,
9,
10,
11] and their propensity for multidrug resistance development [
12,
13,
14,
15,
16,
17,
18]. Colonization with these organisms is a precursor of their infections [
6,
19]. As
S. pneumoniae carriage is especially high in young children [
20,
21,
22], who bear the brunt of its high disease burden and case fatality [
23,
24,
25], pneumococcal conjugate vaccines (PCVs) were incorporated into paediatric vaccination programmes to protect the children against
S. pneumoniae carriage and infections [
23,
25,
26].
However, the nasopharyngeal microbiota of vaccinated individuals and their close contacts have been reportedly altered in response to PCV introduction [
27,
28]. Given the antagonism between
S. aureus and
S. pneumoniae, there are concerns that
S. aureus could potentially replace
S. pneumoniae vaccine serotypes and emerge as the dominant colonizer of the nasopharynx as coverage of PCVs increases [
6,
7,
29,
30,
31]. This raises legitimate concerns, as
S. aureus is frequently implicated in respiratory tract infections, and its presence in the nasopharynx significantly predicts subsequent invasive infections [
31,
32]. Moreover, some studies have demonstrated that among sickle cell disease (SCD) patients,
S. aureus holds more potential in causing invasive diseases than
S. pneumoniae [
33,
34,
35,
36,
37]. Besides, owing to the fact that antibiotics are frequently used among SCD patients as prophylaxes, due to their immunocompromised nature and consequent high predisposition to infections, selective antibiotic pressure could result, generating multidrug resistant microbes, such as methicillin-resistant
S. aureus (MRSA) [
38]. Infections with MRSA are often in tandem with extended hospital stays, increased healthcare costs, and high mortality rates [
39,
40,
41]. Moreover, a recent study has reported that MRSA is responsible for 74.1% of all healthcare associated
S. aureus infections and 30.1% of all community-associated infections [
42]. In Ghana, there have been several MRSA outbreaks since 2012 [
43]; that was the same year in which one of the PCVs—PCV-13—was introduced in the country. It is unknown whether the introduction of PCV-13 played a role in the insurgence of the MRSA outbreaks.
Most of the studies carried out on
S. aureus and MRSA carriage appear to have focused on the occurrence of the pathogen in its ecological niche, the anterior nares [
17,
18,
44,
45,
46,
47]. Little attention seems to have been given to the occurrence of the pathogen in the nasopharynx, and how vaccination with PCVs influences that. The few studies that have evaluated the dynamics of the pathogen in relation to the introduction of PCVs appear to have emanated from developed countries [
27,
28,
29,
30]. Moreover, it is unknown how the introduction of PCVs has influenced the genetic diversity and antimicrobial resistance patterns of
S. aureus. In addition, no such study seems to have been conducted among sickle cell disease patients in the country. Preliminary data to the current study, involving samples collected from healthy school children [
48], reported that the nasopharyngeal carriage prevalence of
S. aureus was high (23.2%). Evidently, the implications of an increased risk of occurrence of
S. aureus and MRSA, with increased levels of antimicrobial resistance, in PCV-vaccinated SCD patients cannot be overemphasized, and an evaluation of the role of PCVs in the dynamics of
S. aureus within this risk group would certainly contribute to addressing the MRSA menace in the country. This study thus aimed to investigate
S. aureus and MRSA nasopharyngeal carriage epidemiology in relation to other nasopharyngeal bacterial colonizers among SCD children about four years into PCV-13 introduction in Ghana, including
S. aureus antimicrobial susceptibility patterns, occurrence of the
LukF-PV (
pvl) and toxic shock syndrome-1 (
tsst-1) genes among cefoxitin-resistant
S. aureus, as well as identification of determinants of
S. aureus and MRSA carriage.
4. Discussion
This study investigated S. aureus nasopharyngeal carriage epidemiology in relation to other nasopharyngeal bacterial colonizers among SCD children about five years into PCV-13 introduction in Ghana. It is the first to conduct such an evaluation among SCD children post-PCV-13 introduction in the country, thus providing insights into dynamics of PCV- and SCD-induced S. aureus nasopharyngeal carriage epidemiology.
The isolated bacterial flora colonizing the nasopharynx of the SCD children belonged to both the Gram-positive and Gram-negative categories, comprising
S. aureus, CoNS,
Streptococcus pneumoniae, alpha haemolytic streptococci, Diphtheroids,
Enterococcus spp.,
Pseudomonas spp., and
Klebsiella oxytoca. While the other bacteria isolated were present at low numbers, MRSA and MSSA prevalence were 3.5% and 55.0%, respectively, translating to
S. aureus domination among the isolated nasopharyngeal colonizers at a prevalence of 57.9%. It is noted that Dayie et al. [
50] (the parent study) reported a significantly lower carriage prevalence of
S. pneumoniae (39.1%) compared to the carriage prevalence of
S. aureus observed in the current study (57.9%). Interestingly, the nasopharynx is the ecological niche of
S. pneumoniae, but not
S. aureus. Hence, the significantly higher carriage prevalence of
S. aureus relative to
S. pneumoniae among the participants, as well as the presence of the other nasopharyngeal colonizers in low numbers, suggest that the modification of the nasopharyngeal microbiota resulting from PCV-13 vaccination led to nasopharyngeal proliferation of
S. aureus as the dominant colonizer. The observations concurrently demonstrate that PCV-13 vaccination was effective in decolonizing the study participants of vaccine-captured serotypes of
S. pneumoniae.
The
S. aureus carriage prevalence recorded in the current study is higher than the post-PCV-13 vaccination anterior nasal carriage prevalence reported by Appiah et al. [
18] among a cohort of SCD children (33%) and the 44.9% nasal carriage prevalence reported by Donkor et al. [
19] among HIV-infected children, another immunologically challenged population, who were also studied during the post-PCV-13 vaccination era. The nasopharyngeal carriage prevalence observed in this study is higher than those reported in other countries, such as Ethiopia, The Gambia, Iran, and Portugal (10.3–30.6%) [
5,
56,
57,
58].
That
S. aureus carriage was higher in this study than it was in the anterior nares of the SCD children in Appiah et al.’s [
18] study and HIV-infected children in Donkor et al. [
17] study seems to confirm an insurgence of
S. aureus in the nasopharynx, as asserted above. This is because carriage is expected to be higher in the anterior nares (the anatomical site sampled by Appiah et al. [
18] and Donkor et al.’s [
17]), as it is the ecological niche of
S. aureus, rather than the nasopharynx. It is possible that slight variations between the study population of the current study and each of the other two populations in question may account for this disparity. Indeed, should information on concurrent nasal carriage prevalence be available alongside the reported nasopharyngeal carriage prevalence, a more conclusive deduction could have been made. That notwithstanding, the higher
S. aureus nasopharyngeal carriage prevalence of this study than the nasal carriage prevalence reported by Donkor et al. [
17] also supports the deduction that
S. aureus is becoming established in the nasopharynx. This claim is strengthened by the known proneness of HIV-infected persons to
S. aureus nasal carriage [
17,
59].
Appiah et al.’s [
18] study, although also conducted in the PCV-13 era, focused on nasal carriage of staphylococci, and hence it is not possible to glean from it the diversity of the colonizing bacteria for the purpose of comparing with the diversity observed in the current study. Another carriage study conducted in the study area [
60], entailing surveillance of an additional organism to
S. aureus among SCD patients, but done prior to the PCV-13 era, focused only on
S. pneumoniae carriage, and its study design was atypical—it was an amalgam of two sub-studies, one involving a haemoglobin SS-haemoglobin AA case-control nasal carriage study focusing on
S. aureus only, and the other involving a different cohort of SCD children who were studied for nasopharyngeal carriage of
S. pneumoniae exclusively. Therefore, that study also did not provide data with which appropriate comparisons could be made in the context of colonizing nasopharyngeal bacterial flora.
Only 20% of the cefoxitin-resistant
S. aureus isolates were confirmed to carry the
mecA gene, meaning that 80% of these isolates lacked the presence of the gene. This is of concern, given that in resource-poor settings, such as Ghana, clinical laboratories generally base the identification of MRSA on oxacillin or cefoxitin resistance. Even though it is possible that the cefoxitin-resistant,
mecA-negative,
S. aureus isolates may be harbouring other variants of the
mec gene, such as
mecC, a significant proportion (80%) lacked the gene. This could be an indictment on MRSA diagnosis that is exclusively based on phenotypic detection. Concerns similar to these have been raised elsewhere [
61]. It may however be argued that this apparent flaw in MRSA diagnosis in resource-poor settings probably has an invariably little impact in clinical settings, particularly, when such laboratory investigations are focused on guiding the selection of an antimicrobial agent with a contemporarily demonstrated effectiveness against an infecting pathogen for the purpose of achieving success in treatment. Hence, in that case, phenotypic demonstration of methicillin resistance would suffice in ruling out the use of certain antimicrobial regimens. The flaw, nonetheless, becomes relevant when such clinical data are submitted directly to epidemiological studies involving MRSA surveillance, or published to reflect MRSA burden in hospitals, in which cases the data may be deemed misleading. Accordingly, it is important to prioritize the upgrade of clinical laboratories in resource-poor settings so as to ensure the use of present-day techniques, such as molecular detection methods, in diagnosis.
The high proportion of
pvl carriage was not surprising, as immunosuppression is known to predispose individuals to a higher risk of
pvl-positive
S. aureus carriage [
62]. Besides this fact, Ghana, like other countries in Africa, is a
pvl-positive
S. aureus-endemic region, with the high occurrence observed even among MSSA isolates. For instance, Eibach et al. [
63] reported the
pvl gene to be present in 58% of the MSSA isolates recovered in their study conducted in Kumasi, Ghana. Devine et al. [
64] also reported a
pvl carriage proportion of 72.1% in Nigeria. That said, the high proportion of
pvl-positive nasopharyngeal
S. aureus colonizers is of immense clinical significance, as it indicates that this cohort of SCD patients, a known immunocompromised population, would have a higher risk of developing invasive infections with
S. aureus strains that could cause polymorphonuclear leucocyte lysis and tissue necrosis owing to their borne virulence determinant—
pvl [
65,
66,
67,
68].
Based on available literature published in peer-reviewed journals, this study is the first to evaluate
tsst-1 carriage among
S. aureus strains recovered from SCD patients in Ghana. A similar proportion—9.8%—was reported in Nigeria [
64]. The low carriage of the gene may mean that only a few of these SCD patients may be prone to developing the toxic shock syndrome.
It is quite interesting that having previously received PCV-13 vaccination appeared to protect the study participants against
S. aureus colonization. This is because PCV-13, like other PCVs, was designed to specifically curtail
S. pneumoniae carriage in the nasopharynx [
69,
70], and hence logically, it is feared that it would cause an insurgence of
S. aureus nasopharyngeal carriage, given the
S. aureus–
S. pneumoniae antagonism [
6,
7,
71]. Thus, the apparent unusual protective effects observed with regard to PCV-13 vaccination in this cohort is most likely not directly linked to the PCV-13 itself, but must have been mediated by secondary factors, such as the presence of CoNS, which was reported at a high rate in the current study. CoNS are also known to be antagonistic to
S. aureus, by virtue of the CoNS-produced autoinducing peptide [
72]. In fact, in the current study, CoNS colonization was observed to protect against
S. aureus colonization. Besides the potential contribution of CoNS, other members of the nasopharyngeal microbiome, which may have been missed by the culture methods relied on in the current study, may have a bearing on this seeming protectiveness of PCV-13 against
S. aureus carriage. Future studies could employ high-throughput sequencing, such as 16S ribosomal ribonucleic acid (rRNA) sequencing, to further investigate the dynamics introduced by CoNS colonization as well as other members of the nasopharyngeal microbiome to this indirect and atypical PCV-13-
S. aureus relationship.
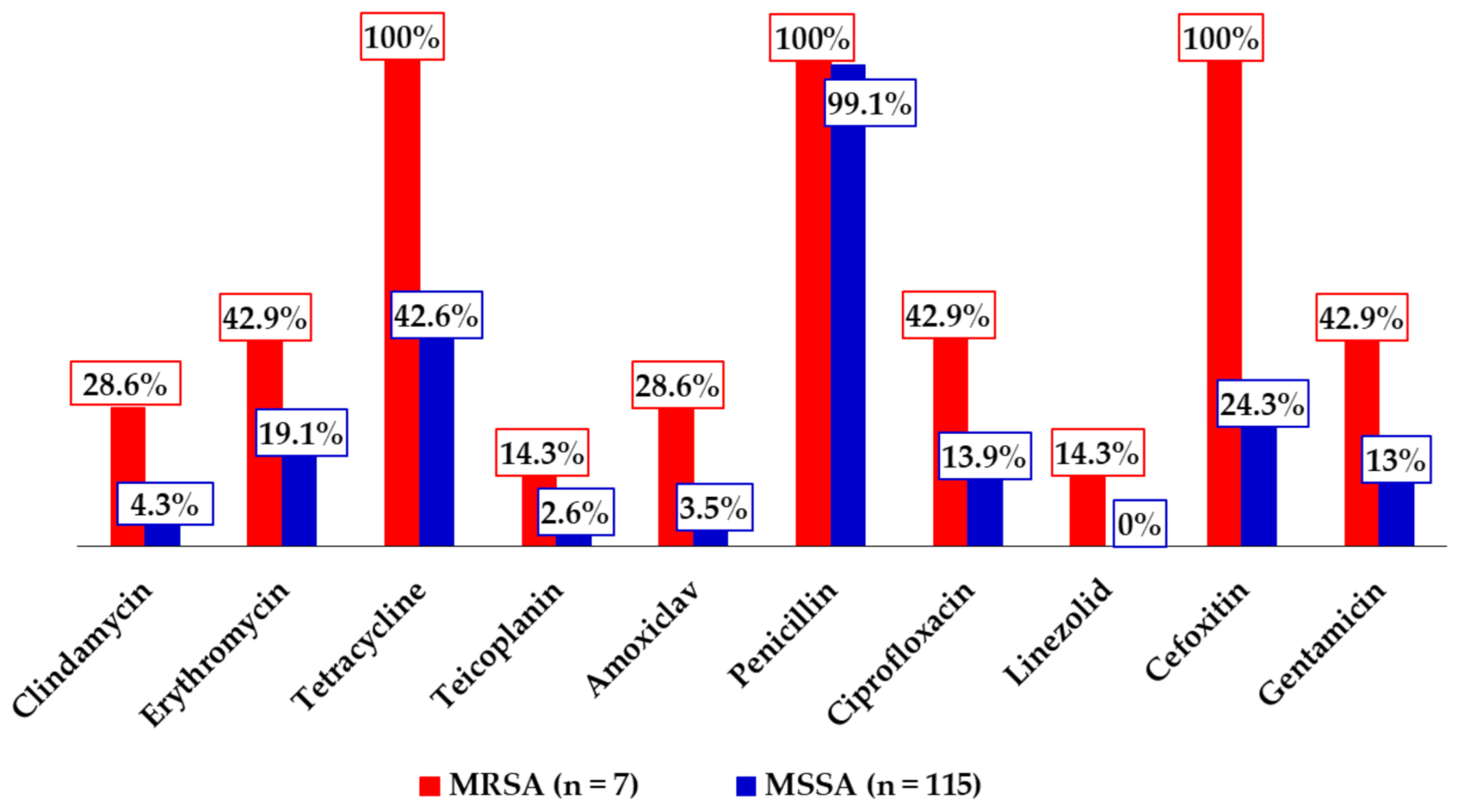
The highest antimicrobial resistance rate was recorded against penicillin, which was expected, as the antibiotic is routinely used among SCD individuals for prophylactic purposes against
S. pneumoniae infections, has been in wide circulation for years, and is easily accessible in the country. Moreover, other
S. aureus studies have reported similar resistance rates against it [
17,
18,
47,
73]. The low resistance rates recorded against linezolid was also expected, as the antibiotic does not have a wide coverage in the country, and previous studies conducted on
S. aureus have reported comparable rates [
17,
74]. The observed rate is however at odds with the 30% resistance rate recorded among anterior nasal
S. aureus isolates obtained in the above-cited recent study of Appiah et al. [
18] among SCD children. That notwithstanding, the low rate signals that linezolid can still be administered in the treatment of MRSA infections. The high rate of tetracycline resistance, although alarming, lacks clinical significance, as the drug is rarely used in clinical practice. Moreover, the high resistance rates demonstrated by the MRSA isolates against erythromycin, amoxiclav, ciprofloxacin, and gentamicin relative to the MSSA isolates is consistent with a key attribute of MRSA—resistance to several antibiotic groups in routine use [
12,
75,
76]. The 37.7% proportion of multidrug resistance observed in this study appears to be higher than those reported with regard to
S. aureus in some studies in the country (3.2–16.7%) [
16,
48,
73], whereas strikingly lower than others (62.3–100%) [
17,
18], and although it may reflect some beneficial effects of campaigns against irrational antimicrobial use, it synchronously underscores the need to intensify these campaigns, particularly among risk groups for
S. aureus carriage and infections, such as SCD patients.
This study was limited by a few factors. First, the absence of information on concurrent S. aureus nasal carriage prevalence alongside the reported nasopharyngeal carriage prevalence makes the assertion of S. aureus insurgence of the nasopharynx a bit less conclusive. Furthermore, screening for virulent genes was restricted to the cefoxitin-resistant S. aureus isolates due to financial constraints, and hence, the study could not provide information on the proportion of S. aureus isolates that harboured the screened virulent genes.