Effects of Polyphenols in Tea (Camellia sinensis sp.) on the Modulation of Gut Microbiota in Human Trials and Animal Studies
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
- (Tea Polyphenol OR Tea Catechin) and (Intestinal Microbiota OR Intestinal Flora)
- (Caffeinated Tea OR Decaffeinated Tea) AND (Colon Microbiota OR Colon Microbial)
- (Green Tea OR Black Tea OR Oolong Tea) AND (Gut Bacteria OR Enteric Bacteria)
- (Tea OR Camellia sinensis sp.) AND (Gastrointestinal Microbiota OR Gastrointestinal Bacteria)
- (Tea) AND (Firmicutes OR Bacteroidetes OR Actinobacteria OR Proteobacteria)
- (Flavonoids OR Flavanols OR Flavonols OR Phenolic acids) AND (Gut Microbiota OR Gut Microbiome OR Gut Microflora
- (Catechin OR Gallocatechin OR Gallocatechin gallate OR Epicatechin OR Epicatechin gallate OR Epigallocatechin OR Epigallocatechin gallate) AND (Gut Microbiota OR Gut Microbiome OR Gut Microflora)
2.2. Study Selection
3. Results
3.1. Green Tea and Gut Microbiota
3.2. Oolong Tea and Gut Microbiota
3.3. Black Tea and Gut Microbiota
3.4. Pu-erh Tea and Gut Microbiota
3.5. Fuzhuan Tea and Gut Microbiota
3.6. Multiple Types of Tea and Gut Mmicrobiota
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kumar Singh, A.; Cabral, C.; Kumar, R.; Ganguly, R.; Kumar Rana, H.; Gupta, A.; Rosaria Lauro, M.; Carbone, C.; Reis, F.; Pandey, A.K. Beneficial Effects of Dietary Polyphenols on Gut Microbiota and Strategies to Improve Delivery Efficiency. Nutrients 2019, 11, 2216. [Google Scholar] [CrossRef]
- Cresci, G.A.; Bawden, E. Gut Microbiome: What We Do and Don’t Know: What We Do and Don’t Know. Nutr. Clin. Pract. 2015, 30, 734–746. [Google Scholar] [CrossRef] [PubMed]
- Rakoff-Nahoum, S.; Foster, K.R.; Comstock, L.E. The Evolution of Cooperation within the Gut Microbiota. Nature 2016, 533, 255–259. [Google Scholar] [CrossRef]
- Seo, D.-O.; Holtzman, D.M. Gut Microbiota: From the Forgotten Organ to a Potential Key Player in the Pathology of Alz-heimer’s Disease. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 1232–1241. [Google Scholar] [CrossRef]
- Salvucci, E. Microbiome, Holobiont and the Net of Life. Crit. Rev. Microbiol. 2016, 42, 485–494. [Google Scholar] [CrossRef]
- Khosravi, A.; Mazmanian, S.K. Disruption of the gut microbiome as a risk factor for microbial infections. Curr. Opin. Microbiol. 2013, 16, 221–227. [Google Scholar] [CrossRef]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef]
- Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef] [PubMed]
- D’Argenio, V.; Salvatore, F. The role of the gut microbiome in the healthy adult status. Clin. Chim. Acta 2015, 451, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Dimitrijević, M.; Aleksić, A.; Neffe-Skocińska, K.; Zielińska, D.; Kołożyn-Krajewska, D.; Sharifi-Rad, J.; Stojanović-Radić, Z.; Prabu, S.M.; Rodrigues, C.F.; et al. Human microbiome and homeostasis: Insights into the key role of prebiotics, probiotics, and symbiotics. Crit. Rev. Food Sci. Nutr. 2021, 61, 1415–1428. [Google Scholar] [CrossRef]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What Is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef]
- Rolhion, N.; Chassaing, B. When pathogenic bacteria meet the intestinal microbiota. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20150504. [Google Scholar] [CrossRef] [PubMed]
- Marques, F.Z.; Mackay, C.R.; Kaye, D.M. Beyond gut feelings: How the gut microbiota regulates blood pressure. Nat. Rev. Cardiol. 2018, 15, 20–32. [Google Scholar] [CrossRef]
- Mouzaki, M.; Comelli, E.M.; Arendt, B.M.; Bonengel, J.; Fung, S.K.; Fischer, S.E.; McGilvray, I.D.; Allard, J.P. Intestinal microbiota in patients with nonalcoholic fatty liver disease. Hepatology 2013, 58, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Schwabe, R.F.; Jobin, C. The microbiome and cancer. Nat. Rev. Cancer 2013, 13, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Shahi, S.K.; Freedman, S.N.; Mangalam, A.K. Gut microbiome in multiple sclerosis: The players involved and the roles they play. Gut Microbes 2017, 8, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Liu, L.; Ji, H.-F. Alzheimer’s Disease Histological and Behavioral Manifestations in Transgenic Mice Correlate with Specific Gut Microbiome State. J. Alzheimer’s Dis. 2017, 56, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Ley, R.E.; Volchkov, P.Y.; Stranges, P.B.; Avanesyan, L.; Stonebraker, A.C.; Hu, C.; Wong, F.S.; Szot, G.L.; Blue-stone, J.A.; et al. Innate Immunity and Intestinal Microbiota in the Development of Type 1 Diabetes. Nature 2008, 455, 1109–1113. [Google Scholar] [CrossRef]
- Liu, Y.-C.; Li, X.-Y.; Shen, L. Modulation effect of tea consumption on gut microbiota. Appl. Microbiol. Biotechnol. 2019, 104, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Barko, P.; McMichael, M.; Swanson, K.; Williams, D. The Gastrointestinal Microbiome: A Review. J. Vet. Intern. Med. 2017, 32, 9–25. [Google Scholar] [CrossRef] [PubMed]
- Rothschild, D.; Weissbrod, O.; Barkan, E.; Kurilshikov, A.; Korem, T.; Zeevi, D.; Costea, P.I.; Godneva, A.; Kalka, I.N.; Bar, N.; et al. Environment dominates over host genetics in shaping human gut microbiota. Nat. Cell Biol. 2018, 555, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Zmora, N.; Suez, J.; Elinav, E. You are what you eat: Diet, health and the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 35–56. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.S.; Raes, J.; Bork, P. The Human Gut Microbiome: From Association to Modulation. Cell 2018, 172, 1198–1215. [Google Scholar] [CrossRef]
- Zhernakova, A.; Kurilshikov, A.; Bonder, M.J.; Tigchelaar, E.F.; Schirmer, M.; Vatanen, T.; Mujagic, Z.; Vila, A.V.; Falony, G.; Vieira-Silva, S.; et al. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Science 2016, 352, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Duda-Chodak, A.; Tarko, T.; Satora, P.; Sroka, P. Interaction of Dietary Compounds, Especially Polyphenols, with the Intes-tinal Microbiota: A Review. Eur. J. Nutr. 2015, 54, 325–341. [Google Scholar] [CrossRef] [PubMed]
- Malongane, F.; McGaw, L.J.; Mudau, F.N. The synergistic potential of various teas, herbs and therapeutic drugs in health improvement: A review. J. Sci. Food Agric. 2017, 97, 4679–4689. [Google Scholar] [CrossRef]
- Khan, N.; Mukhtar, H. Tea Polyphenols in Promotion of Human Health. Nutrients 2018, 11, 39. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jimenez, J.; Vos, F.; Crespy, V.; Du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, bap024. [Google Scholar] [CrossRef] [PubMed]
- Espín, J.C.; González-Sarrías, A.; Tomás-Barberán, F.A. The gut microbiota: A key factor in the therapeutic effects of (poly)phenols. Biochem. Pharmacol. 2017, 139, 82–93. [Google Scholar] [CrossRef]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The Gut Microbiota and Host Health: A New Clinical Frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Henning, S.M.; Choo, J.J.; Heber, D. Nongallated Compared with Gallated Flavan-3-ols in Green and Black Tea Are More Bioavailable. J. Nutr. 2008, 138, 1529S–1534S. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.F.; Dos Santos, C.N.; Ventura, M.R. Polyphenols, their Metabolites and Derivatives as Drug Leads. Curr. Pharm. Des. 2018, 24, 2188–2207. [Google Scholar] [CrossRef]
- Ozdal, T.; Sela, D.A.; Xiao, J.; Boyacioglu, D.; Chen, F.; Capanoglu, E. The Reciprocal Interactions between Polyphenols and Gut Microbiota and Effects on Bioaccessibility. Nutrients 2016, 8, 78. [Google Scholar] [CrossRef] [PubMed]
- Duda-Chodak, A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. 2012, 63, 497–503. [Google Scholar] [PubMed]
- Danneskiold-Samsøe, N.B.; Dias de Freitas Queiroz Barros, H.; Santos, R.; Bicas, J.L.; Cazarin, C.B.B.; Madsen, L.; Kristiansen, K.; Pastore, G.M.; Brix, S.; Maróstica Júnior, M.R. Interplay between Food and Gut Microbiota in Health and Disease. Food Res. Int. 2019, 115, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Ivey, K.L.; Chan, A.T.; Izard, J.; Cassidy, A.; Rogers, G.B.; Rimm, E.B. Role of Dietary Flavonoid Compounds in Driving Patterns of Microbial Community Assembly. mBio 2019, 10, e01205-19. [Google Scholar] [CrossRef]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of Polyphenols on Gut Microbiota and Implications in Human Health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef]
- Parkar, S.G.; Trower, T.M.; Stevenson, D.E. Fecal Microbial Metabolism of Polyphenols and Its Effects on Human Gut Microbiota. Anaerobe 2013, 23, 12–19. [Google Scholar] [CrossRef]
- Selma, M.V.; Espín, J.C.; Tomás-Barberán, F.A. Interaction between Phenolics and Gut Microbiota: Role in Human Health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, X.; Miao, Y.; Cao, J.; Wu, Z.; Weng, P. The modulatory effect of (-)-epigallocatechin 3-O-(3-O-methyl) gallate (EGCG3″Me) on intestinal microbiota of high fat diet-induced obesity mice model. Food Res. Int. 2017, 92, 9–16. [Google Scholar] [CrossRef]
- Chen, G.; Xie, M.; Wan, P.; Chen, D.; Dai, Z.; Ye, H.; Hu, B.; Zeng, X.; Liu, Z. Fuzhuan Brick Tea Polysaccharides Attenuate Metabolic Syndrome in High-Fat Diet Induced Mice in Association with Modulation in the Gut Microbiota. J. Agric. Food Chem. 2018, 66, 2783–2795. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, X.; Zhu, J.; Cheng, L.; Cao, J.; Wu, Z.; Weng, P.; Zheng, X. A metagenomics approach to the intestinal microbiome structure and function in high fat diet-induced obesity mice fed with oolong tea polyphenols. Food Funct. 2018, 9, 1079–1087. [Google Scholar] [CrossRef]
- Sun, H.; Chen, Y.; Cheng, M.; Zhang, X.; Zheng, X.; Zhang, Z. The modulatory effect of polyphenols from green tea, oolong tea and black tea on human intestinal microbiota in vitro. J. Food Sci. Technol. 2017, 55, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Cassotta, M.; Forbes-Hernández, T.Y.; Calderón Iglesias, R.; Ruiz, R.; Elexpuru Zabaleta, M.; Giampieri, F.; Battino, M. Links between nutrition, infectious diseases, and microbiota: Emerging technologies and opportunities for human-focused re-search. Nutrients 2020, 12, 1827. [Google Scholar] [CrossRef]
- Perlman, R.L. Mouse Models of Human Disease: An Evolutionary Perspective. Evol. Med. Public Health 2016, 2016, eow014-6. [Google Scholar] [CrossRef]
- Pham, V.; Mohajeri, M. The application of in vitro human intestinal models on the screening and development of pre- and probiotics. Benef. Microbes 2018, 9, 725–742. [Google Scholar] [CrossRef]
- Van Norman, G.A. Limitations of Animal Studies for Predicting Toxicity in Clinical Trials: Is It Time to Rethink Our Current Approach? JACC Basic Transl. Sci. 2019, 4, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Jadad, A.R.; Tugwell, P. Assessing the Quality of Randomized Controlled Trials: Current Issues and Future Di-rections. Int. J. Technol. Assess. Health Care 1996, 12, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhang, N.; Arikawa, A.Y.; Chen, C. Inhibitory Effects of Green Tea Polyphenols on Microbial Metabolism of Aro-matic Amino Acids in Humans Revealed by Metabolomic Analysis. Metabolites 2019, 9, 96. [Google Scholar] [CrossRef]
- Mai, V.; Katki, H.A.; Harmsen, H.; Gallaher, D.; Schatzkin, A.; Baer, D.J.; Clevidence, B. Effects of a Controlled Diet and Black Tea Drinking on the Fecal Microflora Composition and the Fecal Bile Acid Profile of Human Volunteers in a Double-Blinded Randomized Feeding Study. J. Nutr. 2004, 134, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Janssens, P.L.H.R.; Penders, J.; Hursel, R.; Budding, A.E.; Savelkoul, P.H.M.; Westerterp-Plantenga, M.S. Long-Term Green Tea Supplementation Does Not Change the Human Gut Microbiota. PLoS ONE 2016, 11, e0153134. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Long, Y.; Ji, Z.; Gao, J.; Fu, T.; Yan, M.; Zhang, L.; Su, H.; Zhang, W.; Wen, X.; et al. Green Tea Liquid Consumption Alters the Human Intestinal and Oral Microbiome. Mol. Nutr. Food Res. 2018, 62, e1800178. [Google Scholar] [CrossRef]
- Huang, F.; Zheng, X.; Ma, X.; Jiang, R.; Zhou, W.; Zhou, S.; Zhang, Y.; Lei, S.; Wang, S.; Kuang, J.; et al. The-abrownin from Pu-Erh Tea Attenuates Hypercholesterolemia via Modulation of Gut Microbiota and Bile Acid Metabolism. Nat. Commun. 2019, 10, 4971. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.-S.; Touyama, M.; Hisada, T.; Benno, Y. Effects of Green Tea Consumption on Human Fecal Microbiota with Special Reference to Bifidobacterium Species: Effects of Green Tea on Fecal Microbiota. Microbiol. Immunol. 2012, 56, 729–739. [Google Scholar] [CrossRef]
- Ahn, J.; Sinha, R.; Pei, Z.; Dominianni, C.; Wu, J.; Shi, J.; Goedert, J.J.; Hayes, R.B.; Yang, L. Human Gut Microbiome and Risk for Colorectal Cancer. J. Natl. Cancer Inst. 2013, 105, 1907–1911. [Google Scholar] [CrossRef]
- Borges-Canha, M.; Portela-Cidade, J.P.; Dinis-Ribeiro, M.; Leite-Moreira, A.F.; Pimentel-Nunes, P. Role of Colonic Microbio-ta in Colorectal Carcinogenesis: A Systematic Review. Rev. Esp. Enferm. Dig. 2015, 107, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Hale, V.L.; Chen, J.; Johnson, S.; Harrington, S.C.; Yab, T.C.; Smyrk, T.C.; Nelson, H.; Boardman, L.A.; Druliner, B.R.; Levin, T.R.; et al. Shifts in the Fecal Microbiota Associated with Adenomatous Polyps. Cancer Epidemiol. Biomark. Prev. 2017, 26, 85–94. [Google Scholar] [CrossRef]
- Russo, E.; Bacci, G.; Chiellini, C.; Fagorzi, C.; Niccolai, E.; Taddei, A.; Ricci, F.; Ringressi, M.N.; Borrelli, R.; Melli, F.; et al. Preliminary Comparison of Oral and Intestinal Human Microbiota in Patients with Colorectal Cancer: A Pilot Study. Front. Microbiol. 2017, 8, 2699. [Google Scholar] [CrossRef]
- Zhang, H.-H.; Liu, J.; Lv, Y.-J.; Jiang, Y.-L.; Pan, J.-X.; Zhu, Y.-J.; Huang, M.-G.; Zhang, S.-K. Changes in Intestinal Microbio-ta of Type 2 Diabetes in Mice in Response to Dietary Supplementation with Instant Tea or Matcha. Can. J. Diabetes 2020, 44, 44–52. [Google Scholar] [CrossRef]
- Wang, L.; Zeng, B.; Liu, Z.; Liao, Z.; Zhong, Q.; Gu, L.; Wei, H.; Fang, X. Green Tea Polyphenols Modulate Colonic Microbiota Diversity and Lipid Metabolism in High-Fat Diet Treated HFA Mice. J. Food Sci. 2018, 83, 864–873. [Google Scholar] [CrossRef]
- Wang, L.; Zeng, B.; Zhang, X.; Liao, Z.; Gu, L.; Liu, Z.; Zhong, Q.; Wei, H.; Fang, X. The effect of green tea polyphenols on gut microbial diversity and fat deposition in C57BL/6J HFA mice. Food Funct. 2016, 7, 4956–4966. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.S.; Park, H.M.; Hyun, S.M.; Shon, J.C.; Singh, D.; Liu, K.-H.; Whon, T.W.; Bae, J.-W.; Hwang, J.S.; Lee, C.H. The green tea modulates large intestinal microbiome and exo/endogenous metabolome altered through chronic UVB-exposure. PLoS ONE 2017, 12, e0187154. [Google Scholar] [CrossRef]
- Seo, D.-B.; Jeong, H.W.; Cho, D.; Lee, B.J.; Lee, J.H.; Choi, J.Y.; Bae, I.-H.; Lee, S.-J. Fermented Green Tea Extract Alleviates Obesity and Related Complications and Alters Gut Microbiota Composition in Diet-Induced Obese Mice. J. Med. Food 2015, 18, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Liu, J.; Zhang, N.; Fu, Y.; Zhang, Z.; Li, Y.; Wang, W.; Li, Y.; Shen, P.; Cao, Y. Ripened Pu-erh Tea Extract Protects Mice from Obesity by Modulating Gut Microbiota Composition. J. Agric. Food Chem. 2019, 67, 6978–6994. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Tan, D.; Akbary, R.; Kong, J.; Seviour, R.; Kong, Y. Aqueous raw and ripe Pu-erh tea extracts alleviate obesity and alter cecal microbiota composition and function in diet-induced obese rats. Appl. Microbiol. Biotechnol. 2019, 103, 1823–1835. [Google Scholar] [CrossRef]
- Gao, X.; Xie, Q.; Kong, P.; Liu, L.; Sun, S.; Xiong, B.; Huang, B.; Yan, L.; Sheng, J.; Xiang, H. Polyphenol- and Caffeine-Rich Postfermented Pu-erh Tea Improves Diet-Induced Metabolic Syndrome by Remodeling Intestinal Homeostasis in Mice. Infect. Immun. 2017, 86, e00601-17. [Google Scholar] [CrossRef] [PubMed]
- Foster, M.T.; Gentile, C.L.; Cox-York, K.; Wei, Y.; Wang, D.; Estrada, A.L.; Reese, L.; Miller, T.; Pagliassotti, M.J.; Weir, T.L. Fuzhuan tea consumption imparts hepatoprotective effects and alters intestinal microbiota in high saturated fat diet-fed rats. Mol. Nutr. Food Res. 2016, 60, 1213–1220. [Google Scholar] [CrossRef]
- Henning, S.M.; Yang, J.; Hsu, M.; Lee, R.-P.; Grojean, E.M.; Ly, A.; Tseng, C.-H.; Heber, D.; Li, Z. Decaffeinated green and black tea polyphenols decrease weight gain and alter microbiome populations and function in diet-induced obese mice. Eur. J. Nutr. 2018, 57, 2759–2769. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, Z.; Guo, H.; He, D.; Zhao, H.; Wang, Z.; Zhang, W.; Liao, L.; Zhang, C.; Ni, L. The modulatory effect of infusions of green tea, oolong tea, and black tea on gut microbiota in high-fat-induced obese mice. Food Funct. 2016, 7, 4869–4879. [Google Scholar] [CrossRef]
- Engelhardt, U.H. Tea Chemistry—What Do and What Don’t We Know?—A Micro Review. Food Res. Int. 2020, 132, 109120. [Google Scholar] [CrossRef]
- Yan, Z.; Zhong, Y.; Duan, Y.; Chen, Q.; Li, F. Antioxidant Mechanism of Tea Polyphenols and Its Impact on Health Benefits. Anim. Nutr. 2020, 6, 115–123. [Google Scholar] [CrossRef]
- Anandh Babu, P.; Liu, D. Green Tea Catechins and Cardiovascular Health: An Update. Curr. Med. Chem. 2008, 15, 1840–1850. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Liu, M.; Hu, Y.; Xue, Q.; Yao, F.; Sun, J.; Sun, L.; Liu, Y. Systemic Characteristics of Biomarkers and Differential Me-tabolites of Raw and Ripened Pu-Erh Teas by Chemical Methods Combined with a UPLC-QQQ-MS-Based Metabolomic Ap-proach. Lebenson. Wiss. Technol. 2021, 136, 110316. [Google Scholar] [CrossRef]
- Zhu, M.-Z.; Li, N.; Zhou, F.; Ouyang, J.; Lu, D.-M.; Xu, W.; Li, J.; Lin, H.-Y.; Zhang, Z.; Xiao, J.-B.; et al. Microbial bioconversion of the chemical components in dark tea. Food Chem. 2020, 312, 126043. [Google Scholar] [CrossRef]
- Ning, J.; Li, D.; Luo, X.; Ding, D.; Song, Y.; Zhang, Z.; Wan, X. Stepwise Identification of Six Tea (Camellia sinensis (L.)) Categories Based on Catechins, Caffeine, and Theanine Contents Combined with Fisher Discriminant Analysis. Food Anal. Methods 2016, 9, 3242–3250. [Google Scholar] [CrossRef]
- Yi, T.; Zhu, L.; Peng, W.-L.; He, X.-C.; Chen, H.-L.; Li, J.; Yu, T.; Liang, Z.-T.; Zhao, Z.-Z.; Chen, H.-B. Comparison of Ten Major Constituents in Seven Types of Processed Tea Using HPLC-DAD-MS Followed by Principal Component and Hierarchical Cluster Analysis. Lebenson. Wiss. Technol. 2015, 62, 194–201. [Google Scholar] [CrossRef]
- Li, Q.; Chai, S.; Li, Y.; Huang, J.; Luo, Y.; Xiao, L.; Liu, Z. Biochemical Components Associated with Microbial Community Shift During the Pile-Fermentation of Primary Dark Tea. Front. Microbiol. 2018, 9, 1509. [Google Scholar] [CrossRef]
- Li, Z.; Feng, C.; Luo, X.; Yao, H.; Zhang, D.; Zhang, T. Revealing the influence of microbiota on the quality of Pu-erh tea during fermentation process by shotgun metagenomic and metabolomic analysis. Food Microbiol. 2018, 76, 405–415. [Google Scholar] [CrossRef]
- Lyu, C.; Chen, C.; Ge, F.; Liu, D.; Zhao, S.; Chen, D. A Preliminary Metagenomic Study of Puer Tea during Pile Fermentation: Metagenomic Study of Puer Tea during Pile Fermentation. J. Sci. Food Agric. 2013, 93, 3165–3174. [Google Scholar] [CrossRef]
- Hasan, N.; Yang, H. Factors affecting the composition of the gut microbiota, and its modulation. PeerJ 2019, 7, e7502. [Google Scholar] [CrossRef]
- Magne, F.; Gotteland, M.; Gauthier, L.; Zazueta, A.; Pesoa, S.; Navarrete, P.; Balamurugan, R. The Firmicutes/Bacteroidetes Ratio: A Relevant Marker of Gut Dysbiosis in Obese Patients? Nutrients 2020, 12, 1474. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A.; Troufflard, S.; Serafini, M.; Crozier, A. Absorption, Metabolism and Excretion of Choladi Green Tea Fla-van-3-Ols by Humans. Mol. Nutr. Food Res. 2009, 53 (Suppl. 1), S44–S53. [Google Scholar] [CrossRef]
- Jalil, A.M.M.; Combet, E.; Edwards, C.A.; Garcia, A.L. Effect of β-Glucan and Black Tea in a Functional Bread on Short Chain Fatty Acid Production by the Gut Microbiota in a Gut Digestion/Fermentation Model. Int. J. Environ. Res. Public Health 2019, 16, 227. [Google Scholar] [CrossRef] [PubMed]
- Donohoe, D.R.; Garge, N.; Zhang, X.; Sun, W.; O’Connell, T.M.; Bunger, M.K.; Bultman, S.J. The Microbiome and Butyrate Regulate Energy Metabolism and Autophagy in the Mammalian Colon. Cell Metab. 2011, 13, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Corrêa-Oliveira, R.; Fachi, J.L.; Vieira, A.; Sato, F.T.; Vinolo, M.A.R. Regulation of immune cell function by short-chain fatty acids. Clin. Transl. Immunol. 2016, 5, e73. [Google Scholar] [CrossRef] [PubMed]
- Joossens, M.; Huys, G.; Cnockaert, M.; De Preter, V.; Verbeke, K.; Rutgeerts, P.; Vandamme, P.; Vermeire, S. Dysbiosis of the faecal microbiota in patients with Crohn’s disease and their unaffected relatives. Gut 2011, 60, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Kumari, R.; Ahuja, V.; Paul, J. Fluctuations in Butyrate-Producing Bacteria in Ulcerative Colitis Patients of North India. World J. Gastroenterol. 2013, 19, 3404–3414. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Ozaki, M.; Tsubosaka, M.; Kim, H.-K.; Sasaki, H.; Matsui, Y.; Hibi, M.; Osaki, N.; Miyashita, M.; Shibata, S. Effects of Timing of Acute and Consecutive Catechin Ingestion on Postprandial Glucose Metabolism in Mice and Hu-mans. Nutrients 2020, 12, 565. [Google Scholar] [CrossRef]
- Wang, W.; Chen, L.; Zhou, R.; Wang, X.; Song, L.; Huang, S.; Wang, G.; Xia, B.; Forbes, B.A. Increased Proportions of Bifidobacterium and the Lactobacillus Group and Loss of Butyrate-Producing Bacteria in Inflammatory Bowel Disease. J. Clin. Microbiol. 2014, 52, 398–406. [Google Scholar] [CrossRef]
- Pascal, V.; Pozuelo, M.; Borruel, N.; Casellas, F.; Campos, D.; Santiago, A.; Martinez, X.; Varela, E.; Sarrabayrouse, G.; Machiels, K.; et al. A microbial signature for Crohn’s disease. Gut 2017, 66, 813–822. [Google Scholar] [CrossRef]
- Nie, P.; Li, Z.; Wang, Y.; Zhang, Y.; Zhao, M.; Luo, J.; Du, S.; Deng, Z.; Chen, J.; Wang, Y.; et al. Gut Microbiome Interventions in Human Health and Diseases. Med. Res. Rev. 2019, 39, 2286–2313. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, K.V.; Sherwin, E.; Schellekens, H.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Feeding the microbiota-gut-brain axis: Diet, microbiome, and neuropsychiatry. Transl. Res. 2017, 179, 223–244. [Google Scholar] [CrossRef]
- A David, L.; Materna, A.C.; Friedman, J.; I Campos-Baptista, M.; Blackburn, M.C.; Perrotta, A.; E Erdman, S.; Alm, E.J. Host lifestyle affects human microbiota on daily timescales. Genome Biol. 2014, 15, R89. [Google Scholar] [CrossRef] [PubMed]
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomás-Barberán, F.A. The effects of polyphenols and other bioactives on human health. Food Funct. 2019, 10, 514–528. [Google Scholar] [CrossRef]
- Hildebrandt, M.A.; Hoffmann, C.; Sherrill-Mix, S.A.; Keilbaugh, S.A.; Hamady, M.; Chen, Y.-Y.; Knight, R.; Ahima, R.S.; Bushman, F.; Wu, G.D. High-Fat Diet Determines the Composition of the Murine Gut Microbiome Independently of Obesity. Gastroenterology 2009, 137, 1716–1724. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial Ecology: Human Gut Microbes Associated with Obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef]
- De Wit, N.; Derrien, M.; Bosch-Vermeulen, H.; Oosterink, E.; Keshtkar, S.; Duval, C.; Bosch, J.D.V.-V.D.; Kleerebezem, M.; Müller, M.; Van Der Meer, R. Saturated fat stimulates obesity and hepatic steatosis and affects gut microbiota composition by an enhanced overflow of dietary fat to the distal intestine. Am. J. Physiol. Liver Physiol. 2012, 303, G589–G599. [Google Scholar] [CrossRef] [PubMed]
- De Bandt, J.-P.; Waligora-Dupriet, A.-J.; Butel, M.-J. Intestinal microbiota in inflammation and insulin resistance: Relevance to humans. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 334–340. [Google Scholar] [CrossRef]
- Zou, Y.; Ju, X.; Chen, W.; Yuan, J.; Wang, Z.; Aluko, R.E.; He, R. Rice bran attenuated obesity via alleviating dyslipidemia, browning of white adipocytes and modulating gut microbiota in high-fat diet-induced obese mice. Food Funct. 2020, 11, 2406–2417. [Google Scholar] [CrossRef]
- Tseng, C.-H.; Wu, C.-Y. The gut microbiome in obesity. J. Formos. Med. Assoc. 2019, 118, S3–S9. [Google Scholar] [CrossRef]
- Candela, M.; Biagi, E.; Maccaferri, S.; Turroni, S.; Brigidi, P. Intestinal Microbiota Is a Plastic Factor Responding to Environ-mental Changes. Trends Microbiol. 2012, 20, 385–391. [Google Scholar] [CrossRef]
- Derrien, M.; Alvarez, A.-S.; de Vos, W.M. The Gut Microbiota in the First Decade of Life. Trends Microbiol. 2019, 27, 997–1010. [Google Scholar] [CrossRef] [PubMed]
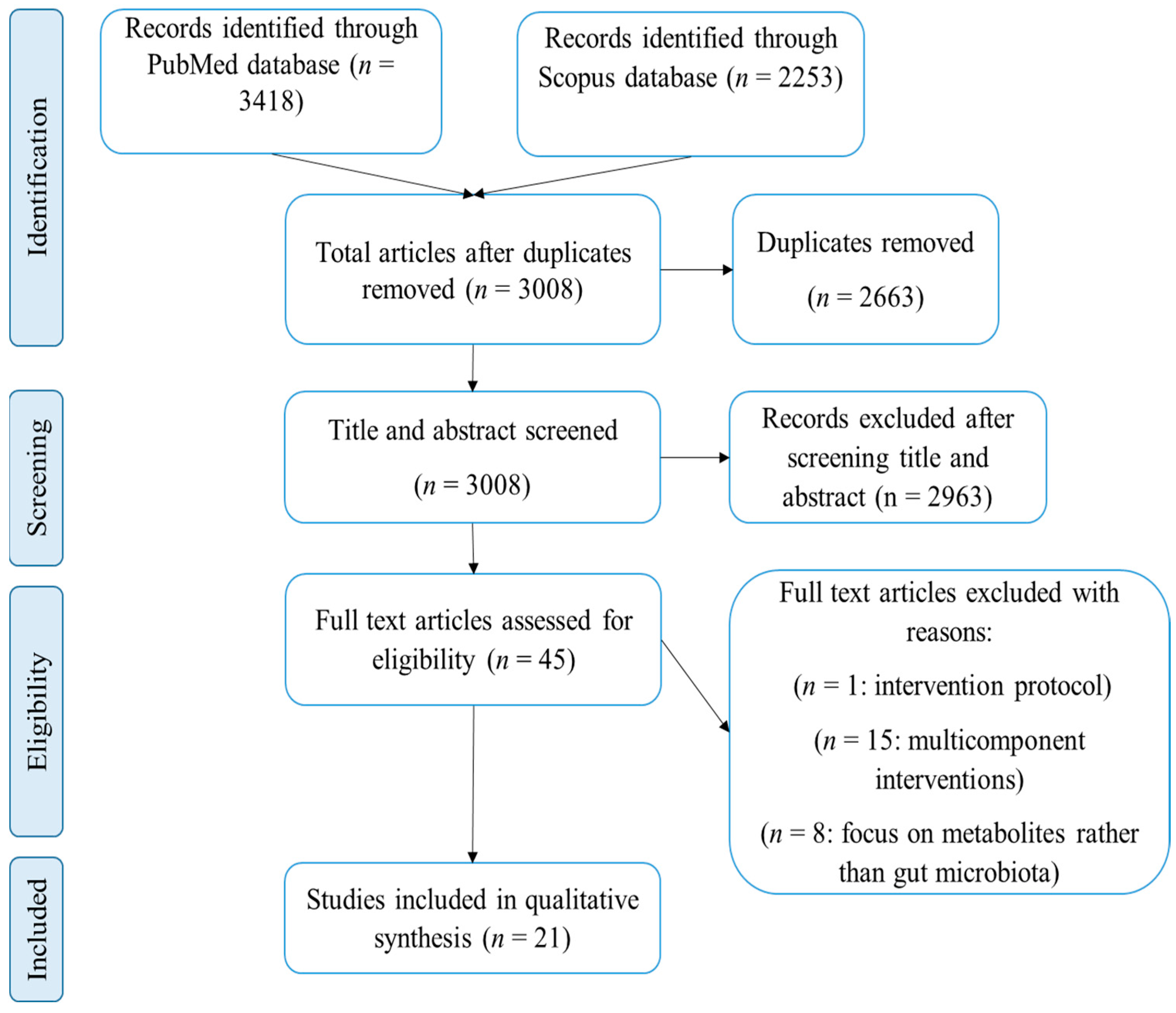
| Reference | Jaded Scores | Study Quality |
|---|---|---|
| Zhou et al., 2019 [50] | 5 | High |
| Mai et al., 2004 [51] | 5 | High |
| Janssens et al., 2016 [52] | 4 | High |
| Yuan et al., 2018 [53] | 0 | Low |
| Huang et al., 2019 [54] | 0 | Low |
| Jin et al., 2012 [55] | 0 | Low |
| Reference | Subjects | Dose, Duration | Alpha Diversity | Beta Diversity | Key Findings | ||
|---|---|---|---|---|---|---|---|
| Richness | Richness, Evenness | Relative Abundance | |||||
| Zhou et al., 2019 [50] Country: United States | 124 post-menopausal females | Four green tea pills/day (1315 ± 115.0 mg catechins) for 12 months | No change | No change | No changes in Firmicutes, Bacteroidetes, or Actinobacteria | No change | Green tea does not affect gut microbiota |
| Yuan et al., 2018 [53] Country: China * | 12 healthy normal and overweight males and females | 400 mL/day (100.2 μg GAE/mL of total polyphenols) for 2 weeks | Increased | Increased | ↓Bacteroidetes, ↑Firmicutes, ↑Actinobacteria ↑FIR:BAC, ↑Lachinospiraceae, ↑Ruminococcaceae, ↑Dorea, ↑Roseburia, ↑Feacalibacterium, ↑Eubacterium, ↑Blautia, and ↑Coprococcus | Changed | Green tea significantly increased microbial diversity |
| Janssens et al., 2016 [52] Country: United States | 58 Caucasian normal to overweight males and females | Nine green tea pills/day (0.56 g of EGCG) for 12 weeks | Not measured | No change | No changes in Firmicutes, Bacteroidetes, Actinobacteria, Fusobacteria, or Verrucomicrobia | No change | Green tea does not affect gut microbiota |
| Jin et al., 2012 [55] Country: Japan * | 10 nonhabitual male and female tea drinkers | 1000 mL green tea/day (unknown amount of polyphenols) for 10 days | Not measured | Not measured | ↑Bifidobacteria | Not measured | Green tea increased probiotic Bifidobacteria |
| Mai et al., 2004 [51] Country: United States | 8 hypercholesterolemia subjects | Black tea infusion (unknown dose and polyphenol amount) for 6 weeks | Not measured | Not measured | No changes in Bacteroides, Prevotella Faecalibacterium, Bifidobacteria, Atopobium, Clostridium, or Ruminococci. ↓Unknown species | Not measured | Black tea does not affect gut microbiota |
| Huang et al., 2019 [54] Country: China * | 13 normal and overweight males | 50 mg/kg/day of instant Pu-erh tea infusion (52.75% theabrownin) for 4 weeks | Not measured | Not measured | ↓Bacilli, ↓Clostridia, ↓Lactobacillus, ↓Bacillus, ↓Streptococcus, and ↓Lactococcus | Changed | Pu-erh tea reduced diversity of hyper-cholesterol-enriching bacteria |
| Reference | Dose, Duration | Alpha Diversity | Beta Diversity | Key Findings | ||
|---|---|---|---|---|---|---|
| Richness | Richness, Evenness | Relative Abundance | ||||
| Zhang et al., 2019 [60] | 0.1 g of matcha powder (14% tea polyphenols and 4.5% EGCG) or instant green tea (22.7% tea polyphenols and 8.4% EGCG) per 100 g of diet for 30 days | Increased | Increased | ↓Firmicutes, ↑Bacteroidetes ↑Actinobacteria, ↓Proteobacteria, ↑Coriobacteriaceae, ↑Lactobacillaceae, ↑Bifidobacteriaceae, ↑Prevotellaceae, ↓Bacteroidaceae, ↓Ruminococcaceae, ↓Helicobacteraceae, and ↓Enterobacteriaceae | Changed | Tea increased diversity of microbiota, reversing the changes caused by diabetes |
| Wang et al., 2018 [61] | 0.05, 0.2, and 0.8 g of green tea extract per 100 mL of water (contains 804 mg/g of total catechins and 455 mg/g EGCG) in HFD for 8 weeks | Not measured | Increased | ↓Firmicutes, ↑Bacteroidetes, ↓FIR:BAC, ↑Bacteroides, ↑Turicibacter, ↑Lachnospira, and ↓Clostridium | Changed | Tea increased diversity of microbiota, reversing the changes caused by obesity |
| Wang et al., 2016 [62] | 0.05, 0.2, and 0.8 g of green tea polyphenol compound per 100 g of HFD for 8 weeks | Not measured | Increased | ↑Lactobacillus | Changed | Tea increased beneficial Lactobacillus |
| Jung et al., 2017 [63] | 1 g extract/100 g diet (contains 50% of total catechins) for 10 weeks | Not measured | Increased | ↓Firmicutes, ↑Bacteroidetes, and ↑Proteobacteria | Not measured | Tea increased diversity of microbiota, reversing the changes caused by chronic ultraviolet exposure |
| Seo et al., 2015 [64] | 500 mg of fermented green tea extract/kg in HFSD (contains 7.85% catechins) for 8 weeks | Not measured | Not measured | ↓FIR:BAC and ↓BAC:PREV | Not measured | Tea reduced biomarkers of obesity and insulin resistance |
| Cheng et al., 2018 [43] | 0.1 g of oolong tea polyphenols per 100 g of HFD (contains 43.55 ± 3.77 µg/g of EGCG) for 4 weeks | Not measured | Increased | ↓Firmicutes, ↑Bacteroidetes, ↓FIR:BAC, and ↑Proteobacteria | Changed | Tea increased diversity of microbiota, reversing the changes caused by obesity |
| Cheng et al., 2017 [41] | 0.1 g oolong tea polyphenols (EGCG) per 100 g of HFD for 8 weeks | Not measured | Increased | ↓Firmicutes, ↑Bacteroidetes, ↓FIR:BAC, and ↑Proteobacteria | Changed | Tea increased the diversity of microbiota, reversing the changes caused by obesity |
| Huang et al., 2019 [54] | 450 mg/kg/day of ripe Pu-erh tea extracts in HFD (containing 52.75% theabrownin) for 26 weeks | Not measured | Not measured | ↓Bacilli, ↓Lactobacillus, ↓Bacillus, ↓Enterococcus, ↓Lactococcus, and ↓Streptococcus | Changed | Pu-erh tea reduced the diversity of hyper-cholesterol-enriching bacteria |
| Lu et al., 2019 [65] | 0.1, 0.2, and 0.4 g of ripe Pu-erh tea extract per 100 mL of water in HFD (contains 4156.63 mg/kg of epicatechins) for 8 weeks | Increased | Increased | ↓FIR:BAC, ↑Anaerotruncus, ↑Alistipes, ↑Odoribacter, ↑Akkermansia, ↑Blautia, ↑Roseburia, ↑Bacteroides, ↑Parabacteroides, ↓Bilophila, ↓Leuconostoc, and ↓Allobaculum | Changed | Tea increased the diversity of microbiota, reversing the changes caused by obesity |
| Xia et al., 2019 [66] | 0.15 and 0.4 g extracts/kg body-weight of raw Pu-erh tea (2.73 ± 0.28% of catechin) and ripe Pu-erh tea (contains 0.56 ± 0.07% of catechin) in HFD for 5 weeks | Increased | Increased | ↑Firmicutes, ↓Bacteroidetes, and ↑Actinobacteria | Not measured | Tea increased the diversity of microbiota, reversing the changes caused by obesity |
| Gao et al., 2017 [67] | 750 mg/kg of ripe Pu-erh tea extract and 250 mg/kg of Pu-erh tea polyphenol and oxidized tea polyphenol in HFD for 12 weeks | Not measured | Not measured | ↑Firmicutes, ↓Bacteroidetes, ↑FIR:BAC, ↑Eubacterium rectale, ↑Clostridiumcoccoides, ↑Faecalibacterium prausnitzii, ↑Akkermansia muciniphila, ↑Bifidobacterium, ↑Lactobacillus, and ↑Roseburia | Not measured | Tea increased the diversity of microbiota, reversing the changes caused by obesity |
| Chen et al., 2018 [42] | 400 mg/kg/day of Fuzhuan tea extract (contains 26.05 ± 1.15% polyphenols) in HFD for 8 weeks | Not measured | Increased | ↓Firmicutes. ↑Bacteroidetes, ↓FIR:BAC, ↑Proteobacteria, and ↑Bifidobacteriaceae | Changed | Tea increased the diversity of gut microbiota and beneficial bacteria, reversing the changes caused by obesity |
| Foster et al., 2016 [68] | 1400 mg/kg/week of Fuzhuan tea extract (unknown polyphenol amount) in HFD for 8 weeks | Not measured | Not measured | ↓Firmicutes, ↑Bacteroidetes, and ↑Lactobacillus | Not measured | Tea increased the diversity of gut microbiota, reversing the changes caused by obesity |
| Henning et al., 2017 [69] | 0.5 g of decaffeinated green tea extract (contains 565 ± 24 GAE mg/g) or black tea extract (contains 532 ± 25 GAE mg/g) per 100 g of HFD for 4 weeks | Not measured | Not measured | ↑Bacteroidetes, ↓Firmicutes, ↓Actinobacteria, ↓FIR:BAC, ↑Parabacteroides, ↑Clostridium, ↑Coprococcus, and ↑Pseudobutyrivibrio | Changed | Tea increased the diversity of gut microbiota, reversing the changes caused by obesity |
| Liu et al., 2016 [70] | 100 mL tea infusion of green tea (contains 3332.35 ± 70.91 mg/L of total polyphenols), oolong tea (contains 2911.52 ± 51.51 mg/L of total polyphenols), and black tea (contains 2732.11 ± 23.64 mg/L total polyphenols) in HFD for 13 weeks | Increased | Increased | ↑Alistipes, ↑Rikenella, ↑Lachnospiraceae, ↑Akkermansia, ↓Bacteroides, and ↓Parabacteroides | Changed | Tea increased the diversity of gut microbiota, reversing the changes caused by obesity |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khairudin, M.A.S.; Mhd Jalil, A.M.; Hussin, N. Effects of Polyphenols in Tea (Camellia sinensis sp.) on the Modulation of Gut Microbiota in Human Trials and Animal Studies. Gastroenterol. Insights 2021, 12, 202-216. https://doi.org/10.3390/gastroent12020018
Khairudin MAS, Mhd Jalil AM, Hussin N. Effects of Polyphenols in Tea (Camellia sinensis sp.) on the Modulation of Gut Microbiota in Human Trials and Animal Studies. Gastroenterology Insights. 2021; 12(2):202-216. https://doi.org/10.3390/gastroent12020018
Chicago/Turabian StyleKhairudin, Mus Azza Suhana, Abbe Maleyki Mhd Jalil, and Napisah Hussin. 2021. "Effects of Polyphenols in Tea (Camellia sinensis sp.) on the Modulation of Gut Microbiota in Human Trials and Animal Studies" Gastroenterology Insights 12, no. 2: 202-216. https://doi.org/10.3390/gastroent12020018
APA StyleKhairudin, M. A. S., Mhd Jalil, A. M., & Hussin, N. (2021). Effects of Polyphenols in Tea (Camellia sinensis sp.) on the Modulation of Gut Microbiota in Human Trials and Animal Studies. Gastroenterology Insights, 12(2), 202-216. https://doi.org/10.3390/gastroent12020018






