Clinical Features of Acute Ischemic Stroke Patients with Hypoesthesia as an Initial Symptom
Abstract
1. Introduction
2. Materials and Methods
2.1. Inclusion Criteria
2.2. Collected Data
2.3. Neuroimaging and the Cause of Ischemic Stroke
2.4. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Lesions and the Distribution of Hypoesthesia
3.3. Comparison of Patients with and without Hypoesthesia
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Takamatsu, K.; Fukushima, T.; Shimoe, Y.; Ohta, T.; Kuriyama, M. Stroke Databank 2015; Kobayashi, S., Ed.; Nakayama Shoten: Tokyo, Japan, 2015; pp. 26–27. [Google Scholar]
- Chen, W.H. Cheiro-oral syndrome: A clinical analysis and review of literature. Yonsei Med. J. 2009, 50, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Satpute, S.; Bergquist, J.; Cole, J.W. Cheiro-Oral syndrome secondary to thalamic infarction: A case report and literature review. Neurologist 2013, 19, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Ten Holter, J.; Tijsse, C. Cheiro-oral syndrome: Does it have a specific localizing value? Eur. Neurol. 1988, 28, 326–330. [Google Scholar] [CrossRef] [PubMed]
- An, J.Y.; Song, I.U.; Kim, S.H.; Kim, Y.I.; Lee, K.S.; Kim, J.S. Cheiro-oral syndrome in a patient from a cortical infarction in the midfrontal gyrus. Eur. Neurol. 2008, 59, 219–220. [Google Scholar] [CrossRef]
- Chen, W.H.; Li, T.H.; Chen, T.H.; Lin, H.S.; Hsu, M.C.; Chen, S.S.; Liu, J.S. Crossed cheiro-oral syndrome. Clin. Neurol. Neurosurg. 2008, 110, 1008–1011. [Google Scholar] [CrossRef]
- Sekine, T.; Kawabe, K.; Kawase, Y.; Iwamoto, K.; Ito, H.; Iguchi, H.; Ikeda, K.; Iwasaki, Y. Cheiro-oral syndrome associated with lateral medullary infarction. Neurol. Sci. 2011, 32, 321–322. [Google Scholar] [CrossRef]
- Chen, W.H.; Lin, H.S.; Chui, C.; Wu, S.S.; Lui, C.C.; Yin, H.L. Clinical analysis of cheiro-pedal syndrome. J. Clin. Neurosci. 2012, 19, 991–995. [Google Scholar] [CrossRef]
- Yasuda, Y.; Morita, T.; Okada, T.; Seko, S.; Akiguchi, I.; Kimura, J. Cheiro-oral-pedal syndrome. Eur. Neurol. 1992, 32, 106–108. [Google Scholar] [CrossRef]
- Igarashi, O.; Iguchi, H.; Ogura, N.; Ichikawa, Y.; Kiyozuka, T.; Kawabe, K.; Maruyama, M.; Fujioka, T.; Ikeda, K.; Iwasaki, Y. Cheiro-oral-pedal syndrome due to brainstem hemorrhage. Clin. Neurol. Neurosurg. 2006, 108, 507–510. [Google Scholar] [CrossRef]
- Kim, J.S. Restricted acral sensory syndrome following minor stroke. Further observation with special reference to differential severity of symptoms among individual digits. Stroke 1994, 25, 2497–2502. [Google Scholar] [CrossRef]
- Fisher, C.M. Pure sensory stroke involving face, arm, and leg. Neurology 1965, 15, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Arboix, A.; García-Plata, C.; García-Eroles, L.; Massons, J.; Comes, E.; Oliveres, M.; Targa, C. Clinical study of 99 patients with pure sensory stroke. J. Neurol. 2005, 252, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Adams, H.P., Jr.; Bendixen, B.H.; Kappelle, L.J.; Biller, J.; Love, B.B.; Gordon, D.L.; Marsh, E.E., 3rd. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 1993, 24, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, J.L.; Torebjork, H.E. Paraesthesiae from ectopic impulse generation in human sensory nerves. Brain 1980, 103, 835–853. [Google Scholar] [CrossRef]
- Burke, D. Microneurography, impulse conduction, and paresthesias. Muscle Nerve 1993, 16, 1025–1032. [Google Scholar] [CrossRef]
- Mogyoros, I.; Bostock, H.; Burke, D. Mechanisms of paresthesias arising from healthy axons. Muscle Nerve 2000, 23, 310–320. [Google Scholar] [CrossRef]
- Bostock, H.; Burke, D.; Hales, J.P. Differences in behaviour of sensory and motor axons following release of ischaemia. Brain 1994, 117 Pt 2, 225–234. [Google Scholar] [CrossRef]
- Barolat, G.; Massaro, F.; He, J.; Zeme, S.; Ketcik, B. Mapping of sensory responses to epidural stimulation of the intraspinal neural structures in man. J. Neurosurg. 1993, 78, 233–239. [Google Scholar] [CrossRef]
- Altmann, M.; Thommessen, B.; Ronning, O.M.; Reichenbach, A.S.; Fure, B. Blood pressure differences between patients with lacunar and nonlacunar infarcts. Brain Behav. 2015, 5, e00353. [Google Scholar] [CrossRef]
- Hokkoku, K.; Sonoo, M.; Murashima, H.; Iwanami, T.; Nishiyama, K.; Kawamura, Y.; Arai, Y.; Tsukamoto, H.; Hatanaka, Y.; Shimizu, T. Clinical features of predominantly sensory stroke due to brainstem infarction. Rinsho Shinkeigaku 2011, 51, 248–254. [Google Scholar] [CrossRef]
- Bowsher, D.; Leijon, G.; Thuomas, K.A. Central poststroke pain: Correlation of MRI with clinical pain characteristics and sensory abnormalities. Neurology 1998, 51, 1352–1358. [Google Scholar] [CrossRef]
- Sprenger, T.; Seifert, C.L.; Valet, M.; Andreou, A.P.; Foerschler, A.; Zimmer, C.; Collins, D.L.; Goadsby, P.J.; Tölle, T.R.; Chakravarty, M.M. Assessing the risk of central post-stroke pain of thalamic origin by lesion mapping. Brain 2012, 135, 2536–2545. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.P.; Marcussen, N.S.; Klit, H.; Andersen, G.; Finnerup, N.B.; Jensen, T.S. Pain following stroke: A prospective study. Eur. J. Pain 2012, 16, 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- MacGowan, D.J.; Janal, M.N.; Clark, W.C.; Wharton, R.N.; Lazar, R.M.; Sacco, R.L.; Mohr, J.P. Central poststroke pain and Wallenberg’s lateral medullary infarction: Frequency, character, and determinants in 63 patients. Neurology 1997, 49, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, A.C.; Lindgren, I.; Hallström, B.; Norrving, B.; Lindgren, A. Prevalence and intensity of pain after stroke: A population based study focusing on patients’ perspectives. J. Neurol. Neurosurg. Psychiatry 2006, 77, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Widar, M.; Ahlström, G. Disability after a stroke and the influence of long-term pain on everyday life. Scand. J. Caring Sci. 2002, 16, 302–310. [Google Scholar] [CrossRef]
- Jahngir, M.U.; Qureshi, A.I. Dejerine Roussy Syndrome. In StatPearls; StatPearls Publishing LLC.: Tampa, FL, USA, 2022. [Google Scholar]
- Jang, S.H.; Kim, J.; Lee, H.D. Delayed-onset central poststroke pain due to degeneration of the spinothalamic tract following thalamic hemorrhage: A case report. Medicine 2018, 97, e13533. [Google Scholar] [CrossRef]

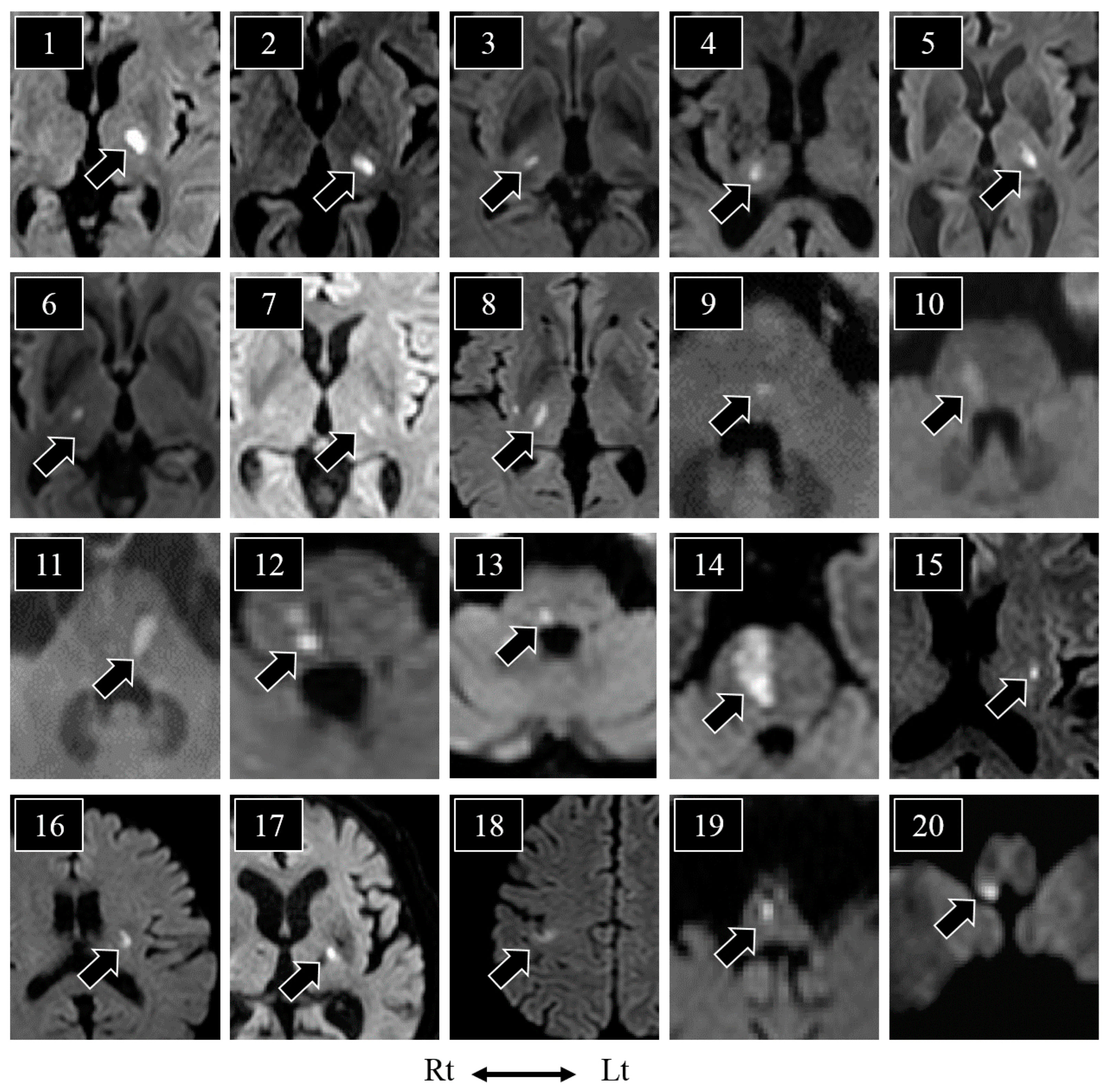
| Age/Sex | Hypoesthesia | Neurological Symptoms | MRI Lesion | Disease Type | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| FP | DA | Weakness | Ataxia | DSS | DVS | |||||
| 1 | 65/M | face | + | + | arm | arm/leg | − | − | thalamus | UD |
| 2 | 54/F | hemibody | − | − | − | arm/leg | − | − | thalamus | SVO |
| 3 | 67/M | lip/cheek/finger | − | − | arm | − | face/arm | NA | thalamus | SVO |
| 4 | 82/M | wrist-finger | − | + | arm | arm/leg | − | − | thalamus | SVO |
| 5 | 79/M | hemibody | − | − | arm/leg | − | face/arm/leg | − | thalamus | SVO |
| 6 | 71/M | cheek | − | − | − | − | face | NA | thalamus | SVO |
| 7 | 69/M | face/arm/leg | − | − | − | − | face/arm/leg | - | thalamus | SVO |
| 8 | 61/M | face/arm/leg | − | + | arm/leg | arm/leg | face/arm/leg | leg | thalamus | LAA |
| 9 | 70/M | hand/leg | − | − | − | arm | arm | − | PT | SVO |
| 10 | 72/M | palm/lower leg | − | + | − | arm/leg | − | − | PT | SVO |
| 11 | 66/M | hand/foot | − | − | − | arm | − | NA | PT | SVO |
| 12 | 49/M | forearm-finger/knee-toe | + | − | − | − | arm/leg | leg | PT | SVO |
| 13 | 65/M | hemibody | − | − | arm | - | face/arm/leg | NA | PT | SVO |
| 14 | 75/M | wrist-finger/sole | − | + | arm/leg | arm/leg | − | − | PB/PT | UD |
| 15 | 62/F | intraoral/throat/cheek | − | + | − | − | face | NA | putamen | UD |
| 16 | 62/F | forearm | − | − | arm | − | − | NA | corona radiata | SVO |
| 17 | 79/M | finger | + | + | − | − | − | NA | PLIC | SVO |
| 18 | 66/M | forearm-finger | − | − | arm | − | arm | NA | cerebral cortex | CE |
| 19 | 71/M | wrist-finger/knee-toe | + | + | − | arm/leg | − | − | MM | LAA |
| 20 | 64/M | hand * | − | − | arm * | arm/leg * | − | − | LM | SVO |
| Characteristics | Total (n = 176) | With Hypoesthesia (n = 20) | Without Hypoesthesia (n = 156) | p-Value |
|---|---|---|---|---|
| Physical examination | ||||
| Age (y.o.) | 71 (39–91) | 66.5 (49–82) | 71 (39–91) | 0.160 |
| Sex (Male), n (%) | 130 (73.8%) | 17 (85%) | 113 (72.4%) | 0.177 |
| SBP on admission (mmHg) | 158 (92–264) | 171.5 (128–230) | 156 (92–264) | * 0.031 |
| DBP on admission (mmHg) | 89 (33–132) | 99 (63–125) | 89 (33–132) | * 0.037 |
| Body mass index (kg/m2) | 23.1 (14.5–34.4) | 23 (14–33) | 24 (18–34) | 0.268 |
| NIHSS on admission | 2 (0–10) | 2 (1–5) | 2 (0–10) | 0.182 |
| Past medical history | ||||
| Hypertension, n (%) | 106 (61%) | 8 (42%) | 98 (63%) | 0.064 |
| Dyslipidemia, n (%) | 53 (30%) | 6 (30%) | 47 (30%) | 0.607 |
| Diabetes mellitus, n (%) | 52 (30%) | 6 (30%) | 46 (29%) | 0.573 |
| Hemodialysis, n (%) | 7 (4%) | 0 (0%) | 7 (4%) | 0.423 |
| Stroke (ischemic/hemorrhagic), n (%) | 38 (22%) | 3 (15%) | 35 (22%) | 0.332 |
| Laboratory data | ||||
| Blood sugar (mg/dL) | 119 (76–337) | 118 (76–332) | 127 (88–337) | 0.402 |
| HbA1c (%) | 6.1 (4.4–13.6) | 6.1 (4.4–13.6) | 6.2 (4.9–10.8) | 0.531 |
| LDL-Cho (mg/dL) | 122 (31–242) | 123 (49–242) | 110 (31–183) | 0.119 |
| Creatinine (mg/dL) ** | 0.78 (0.42–9.00) | 0.77 (0.42–9.00) | 0.87 (0.52–2.35) | 0.311 |
| CRP (mg/dL) | 0.15 (0.01–30.0) | 0.15 (0.01–30.0) | 0.11 (0.06–0.56) | 0.771 |
| NT-pro BNP (pg/mL) ** | 174 (10–11,222) | 174 (10–11,222) | 151 (10–8462) | 0.211 |
| Disease type | ||||
| Large-artery atherosclerosis, n (%) | 20 (11.3%) | 2 (10%) | 18 (11.5%) | 0.596 |
| Cardio-embolism, n (%) | 43 (24.4%) | 1 (5%) | 42 (26.9%) | * 0.021 |
| Small-vessel occlusion, n (%) | 47 (26.7%) | 14 (70%) | 33 (21.2%) | * <0.001 |
| Undetermined etiology, n (%) | 66 (37.5%) | 3 (15%) | 63 (40.4%) | * 0.021 |
| Prognosis | ||||
| mRS on discharge | 1 (0–6) | 1 (0–3) | 1 (0–6) | 0.319 |
| Duration of hospitalization (days) *** | 19 (8–104) | 15.5 (8–24) | 20 (9–104) | * 0.007 |
| Discharge to home | 127 (73%) | 15 (75%) | 112 (73%) | 0.534 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akimoto, T.; Ogawa, K.; Hara, M.; Ninomiya, S.; Ishihara, M.; Morita, A.; Kamei, S.; Nakajima, H. Clinical Features of Acute Ischemic Stroke Patients with Hypoesthesia as an Initial Symptom. Neurol. Int. 2023, 15, 508-517. https://doi.org/10.3390/neurolint15010030
Akimoto T, Ogawa K, Hara M, Ninomiya S, Ishihara M, Morita A, Kamei S, Nakajima H. Clinical Features of Acute Ischemic Stroke Patients with Hypoesthesia as an Initial Symptom. Neurology International. 2023; 15(1):508-517. https://doi.org/10.3390/neurolint15010030
Chicago/Turabian StyleAkimoto, Takayoshi, Katsuhiko Ogawa, Makoto Hara, Satoko Ninomiya, Masaki Ishihara, Akihiko Morita, Satoshi Kamei, and Hideto Nakajima. 2023. "Clinical Features of Acute Ischemic Stroke Patients with Hypoesthesia as an Initial Symptom" Neurology International 15, no. 1: 508-517. https://doi.org/10.3390/neurolint15010030
APA StyleAkimoto, T., Ogawa, K., Hara, M., Ninomiya, S., Ishihara, M., Morita, A., Kamei, S., & Nakajima, H. (2023). Clinical Features of Acute Ischemic Stroke Patients with Hypoesthesia as an Initial Symptom. Neurology International, 15(1), 508-517. https://doi.org/10.3390/neurolint15010030






