Tinzaparin vs. Nadroparin Safety and Efficacy in Neurosurgery
Abstract
1. Introduction
2. Methods
2.1. Patient Selection
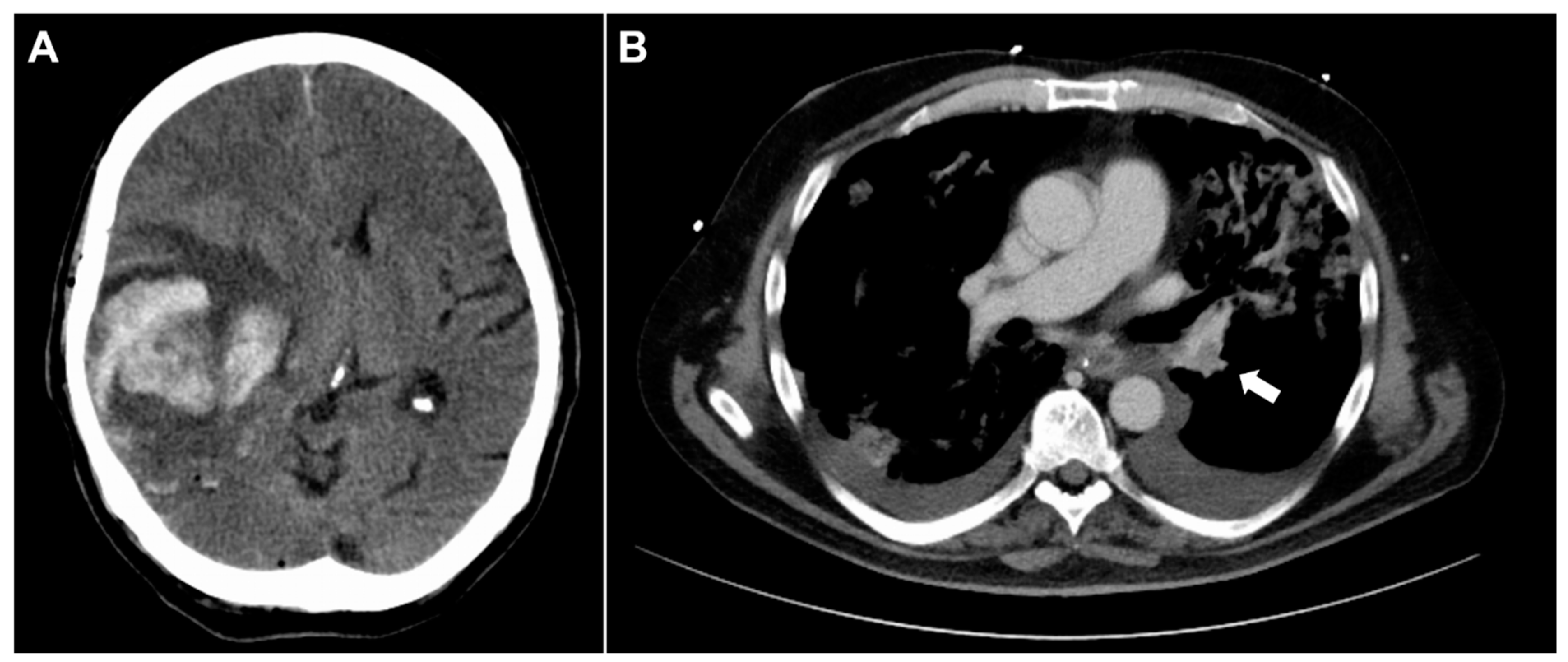
2.2. Diagnosis of Adverse Events
2.3. Anticoagulation Protocol for SAH Patients at Our Clinic
2.4. Assessed Data
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Encke, A.; Haas, S.; Kopp, I. Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (AWMF)-Ständige Kommission Leitlinien: S3-Leitlinie: Prophylaxe der Venösen Thromboembolie (VTE). 2015. Available online: https://www.awmf.org/uploads/tx_szleitlinien/003-001l_S3_VTE-Prophylaxe_2015-12.pdf (accessed on 8 November 2019).
- Diringer, M.N.; Bleck, T.P.; Claude Hemphill, J.; Menon, D.; Shutter, L.; Vespa, P.; Bruder, N.; Connolly, E.S.; Citerio, G.; Gress, D.; et al. Critical care management of patients following aneurysmal subarachnoid hemorrhage: Recommendations from the Neurocritical Care Society’s Multidisciplinary Consensus Conference. Neurocritical Care 2011, 15, 211–240. [Google Scholar] [CrossRef] [PubMed]
- Faraoni, D.; Comes, R.F.; Geerts, W.; Matthew, D.W. European guidelines on perioperative venous thromboembolism prophylaxis: Neurosurgery. Eur. J. Anaesthesiol. 2018, 35, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Cote, D.J.; Smith, T.R. Venous thromboembolism in brain tumor patients. J. Clin. Neurosci. 2016, 25, 13–187. [Google Scholar] [CrossRef] [PubMed]
- Darkwah Oppong, M.; Buffen, K.; Pierscianek, D.; Herten, A.; Ahmadipour, Y.; Dammann, P.; Rauschenbach, L.; Forsting, M.; Sure, U.; Jabbarli, R. Secondary hemorrhagic complications in aneurysmal subarachnoid hemorrhage: When the impact hits hard. J. Neurosurg. 2019, 132, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Khaldi, A.; Helo, N.; Schneck, M.J.; Origitano, T.C. Venous thromboembolism: Deep venous thrombosis and pulmonary embolism in a neurosurgical population. J. Neurosurg. 2011, 114, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Rosovsky, R.P.; Barra, M.E.; Roberts, R.J.; Parmar, A.; Andonian, J.; Suh, L.; Algeri, S.; Biddinger, P.D. When Pigs Fly: A Multidisciplinary Approach to Navigating a Critical Heparin Shortage. Oncologist 2020, 25, 334–347. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.sueddeutsche.de/wissen/afrikanische-schweinepest-kloeckner-ansteckung-1.5027058 (accessed on 10 September 2020).
- Available online: https://www.reuters.com/article/us-germany-swinefever/germany-says-has-suspected-case-of-african-swine-fever-idUSKBN26033P (accessed on 10 September 2020).
- Wilhelmy, F.; Hantsche, A.; Wende, T.; Kasper, J.; Reuschel, V.; Frydrychowicz, C.; Rasche, S.; Lindner, D.; Meixensberger, J. Perioperative anticoagulation in patients with intracranial meningioma: No increased risk of intracranial hemorrhage? PLoS ONE 2020, 15, e0238387. [Google Scholar] [CrossRef] [PubMed]
- Nutescu, E.A.; Shapiro, N.L.; Feinstein, H.; Rivers, C.W. Tinzaparin: Considerations for Use in Clinical Practice. Ann. Pharmacother. 2003, 37, 1831–1840. [Google Scholar] [CrossRef] [PubMed]
- Marciniak, C.M.; Kaplan, J.; Welty, L.; Chen, D. Enoxaparin Versus Tinzaparin for Venous Thromboembolic Prophylaxis During Rehabilitation After Acute Spinal Cord Injury: A Retrospective Cohort Study Comparing Safety and Efficacy. PM&R 2012, 4, 11–17. [Google Scholar] [CrossRef]
- Nelson-Piercy, C.; Powrie, R.; Borg, J.-Y.; Rodger, M.; Talbot, D.J.; Stinson, J.; Greer, I.A. Tinzaparin use in pregnancy: An international, retrospective study of the safety and efficacy profile. Eur. J. Obstet. Gynecol. Reprod. Biol. 2011, 159, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Chibbaro, S.; Tacconi, L. Safety of deep venous thrombosis prophylaxis with low-molecular-weight heparin in brain surgery. Prospective study on 746 patients. Surg. Neurol. 2008, 70, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Pahatouridis, D.; Alexiou, G.A.; Zigouris, A.; Mihos, E.; Drosos, D.; Voulgaris, S. Coagulopathy in moderate head injury. The role of early administration of low molecular weight heparin. Brain Inj. 2010, 24, 1189–1192. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.-R.; Yao, P.-S.; Liu, C.-B.; Shang-Guan, H.-C.; Zheng, S.-F.; Yu, L.-H.; Lin, Y.-X.; Lin, Z.-Y.; Kang, D.-Z. Risk factors of acute coagulation dysfunction after aneurysmal subarachnoid hemorrhage. Chin. Neurosurg. J. 2018, 4, 1–6. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Magnus, N.; D’Asti, E.; Garnier, D.; Meehan, B.; Rak, J. Brain Neoplasms and Coagulation. Semin. Thromb. Hemost. 2013, 39, 881–895. [Google Scholar] [CrossRef] [PubMed]
| Patients’ Baseline Data (n = 814) | |||
|---|---|---|---|
| Demographic | Nadroparin (n = 398) | Tinzaparin (n = 416) | p-Value (t-Test/Fisher’s Exact) |
| Gender | 167 M (42.0%)231 F (58.0%) | 182 M (43.8%)234 F (56.2%) | 0.6205 |
| Age (average in years) | 59 ± 15.0 | 60 ± 14.6 | 0.3354 |
| BMI (average) | 26 ± 4.5 | 26 ±4.6 | 1.000 |
| Underlying disease | |||
| Malign brain tumor | 106 (26.6%) | 133 (32.0%)) | 0.0906 |
| Meningioma | 149 (37.4%) | 129 (31.0%) | 0.0550 |
| SAH | 143 (35.9%) | 154 (37.0%) | 0.7710 |
| Medical history | |||
| Pre-existing anticoagulation | 49 (12.3%) | 55 (13.2%) | 0.7530 |
| Smoker | 71 (17.8%) | 80 (19.2%) | 0.6523 |
| Steroid medication | 196 (49.2%) | 190 (45.7%) | 0.3259 |
| Coagulation disorder | 35 (8.8%) | 34 (8.2%) | 0.8018 |
| Adverse Events Combined (n = 814) | ||||
|---|---|---|---|---|
| Adverse Event | Nadroparin (n = 398) | Tinzaparin (n = 416) | p-Value | |
| Chi-Squared | Fisher’s Exact | |||
| Rebleeding conservative (overall) | 35 (8.8%) | 43 (10.3%) | 0.4996 | 0.4757 |
| Rebleeding operative | 13 (3.3%) | 11 (2.6%) | 0.7551 | 0.6818 |
| Thromboembolic event (overall) | 22 (5.5%) | 18 (4.3%) | 0.5287 | 0.517 |
| Deep vein thrombosis | 8 (2.0%) | 4 (0.96%) | 0.3513 | 0.2561 |
| Pulmonary embolism | 9 (2.3%) | 9 (2.2%) | 0.9417 | 1 |
| Systemic thromboembolism | 5 (1.3%) | 5 (1.20%) | 0.9568 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilhelmy, F.; Hantsche, A.; Gaier, M.; Kasper, J.; Fehrenbach, M.K.; Oesemann, R.; Meixensberger, J.; Lindner, D. Tinzaparin vs. Nadroparin Safety and Efficacy in Neurosurgery. Neurol. Int. 2021, 13, 202-206. https://doi.org/10.3390/neurolint13020021
Wilhelmy F, Hantsche A, Gaier M, Kasper J, Fehrenbach MK, Oesemann R, Meixensberger J, Lindner D. Tinzaparin vs. Nadroparin Safety and Efficacy in Neurosurgery. Neurology International. 2021; 13(2):202-206. https://doi.org/10.3390/neurolint13020021
Chicago/Turabian StyleWilhelmy, Florian, Annika Hantsche, Michael Gaier, Johannes Kasper, Michael Karl Fehrenbach, Rene Oesemann, Jürgen Meixensberger, and Dirk Lindner. 2021. "Tinzaparin vs. Nadroparin Safety and Efficacy in Neurosurgery" Neurology International 13, no. 2: 202-206. https://doi.org/10.3390/neurolint13020021
APA StyleWilhelmy, F., Hantsche, A., Gaier, M., Kasper, J., Fehrenbach, M. K., Oesemann, R., Meixensberger, J., & Lindner, D. (2021). Tinzaparin vs. Nadroparin Safety and Efficacy in Neurosurgery. Neurology International, 13(2), 202-206. https://doi.org/10.3390/neurolint13020021






